Abstract
There is a strong push towards standardisation of treatment approaches, care processes and documentation of clinical practice. However, confusion persists regarding terminology and description of many clinical care process specifications which this research seeks to resolve by developing a taxonomic characterisation of clinical care process specifications. Literature on clinical care process specifications was analysed, creating the starting point for identifying common characteristics and how each is constructed and used in the clinical setting. A taxonomy for clinical care process specifications is presented. The De Bleser approach to limited clinical care process specifications characterisation was extended and each clinical care process specification is successfully characterised in terms of purpose, core elements and relationship to the other clinical care process specification types. A case study on the diagnosis and treatment of Type 2 Diabetes in the United Kingdom was used to evaluate the taxonomy and demonstrate how the characterisation framework applies. Standardising clinical care process specifications ensures that the format and content are consistent with expectations, can be read more quickly and high-quality information can be recorded about the patient. Standardisation also enables computer interpretability, which is important in integrating Learning Health Systems into the modern clinical environment. The approach presented allows terminologies for clinical care process specifications that were widely used interchangeably to be easily distinguished, thus, eliminating the existing confusion.
Keywords
Introduction
Care process specification, also described as care process modelling, arose from a need to evaluate the quality of healthcare delivery.1,2 Documentation of care processes is said to result in healthcare quality improvement as it helps those involved in patient care to develop a shared understanding of care to be provided, and a reference point for identifying areas for future improvement.3–6 The intention is to ensure that services being provided to patients deliver the desired health outcomes consistent with current clinical knowledge.
2
Deviation from those quality standards established in the care process specification (described as
A wide range of terms exist for what we describe collectively here as
The British Medical Association and Royal College of Nursing developed a joint guidance stating that use of standardised forms is beneficial in reducing variation in healthcare practice. 26 The use of different versions of the same clinical care specification in different units within the same care facility, and between different care facilities, may not previously have been seen as such an important issue. However, in this increasingly digital healthcare environment we are seeing greater amounts of data being generated and captured daily, including from diagnostic devices used, or sensors worn, by the patient while in the community. Any differences in the documentation approach or data recording method results in fragmented data, complicates the integration of data about the same patient from different sources and inhibits health information exchange (HIE).27,28
Even though standardisation of healthcare practice, procedure and tools can improve healthcare quality, outcomes and accountability, there remains significant resistance to its implementation.16,29,30 This is not surprising, as the issue of distinguishing these clinical tools persists.
16
We have sought to resolve the disagreement in the distinction between these terms using extensive literature review and input from a wide range of clinical experts to identify a taxonomy and characterisation, with specific attention focused on identifying differences and similarities for a range of common clinical document types. We address the problem of a lack of standardisation of
Produce a taxonomy based on all of the terminologies that are used to represent CCPS documents, to aid in resolving the confusion observed in the literature regarding whether these terms are synonymous, similar or representation of distinct clinical specifications.
Characterise each CCPS, so that readers can identify that if a document has these properties, then it must be this type of CCPS. Specific questions associated with this objective are: What are they composed of? What are the common and unique elements? Can we develop a descriptive terminology, definition and demonstrate the structure of each based on our review of the literature, their individual domains and overall use case?
Evaluate the
Background, context and problem
Why clinical documents were developed?
Many of the clinical documents, or tools, reviewed in this research were developed and refined during the 1980s and 1990s in response to a number of key needs, including the need to control costs and to improve the quality of patient care.31–33 Using project management (PM) and total quality management (TQM) tools more common to industry, hospital managers sought to reengineer the processes of hospital care to reduce clinical resources and error rates, and improve patient outcomes.20,33–35 However, clinical costs continued to increase, and error rates persisted with
The move to standardisation
In the early 2000s, the focus of hospital management and clinical literature shifted towards the theme of
Methodology and approach
A search was conducted using Scopia, Science Direct, PubMed, EBSCOhost, DOAJ and Elsevier for literature discussing the definition, development or use of a range of clinical document types including: caremaps (
Mapping methodologies to deliverables and objectives.

Content and thematic analyses
Concept analysis (CA) and thematic analysis (TA) are separate but interrelated qualitative approaches for descriptive data analysis with low levels of interpretation.
47
CA is an accepted systematic coding and categorisation method for investigating texts and resolving quantitative description of the features.47,48 CA first establishes categories and then records the instances in which that category is evident or can be inferred from within the collected texts being analysed.
48
TA is a more qualitative method used to identify, analyse and report patterns, or themes, that emerge as being important within the material being analysed.47,48 TA provides the systematic element characteristic of CA, and can also combine analysis of frequency with analysis of
The literature review resulted in spreadsheets with the details and a brief description of each article. CA was then used to data mine the literature to inductively develop a list of concepts that were used either in discussion of the particular CCPS document, or which were a structural component of examples provided or included by the authors. These concepts were used to populate the spreadsheets in the format of formal CA charts.51–53 TA extended these with context, collecting together examples of where different authors represented the same general idea in a variety of ways or with differing language, and thus making each concept of interest identified in the literature more generic by describing it based on its underlying theme.
To identify the scope of author positions on whether CCPS documents were the same or distinct, we collected data on how authors described and discussed them in this context as shown in Appendix 1. Where authors described a document type as also known by, or was the same as another, these were indicated with an unfilled square. Where they provided narrative that differentiated or distinguished different types, this is indicated with a filled circle. Separate rows are used to record subsequent instances identified in the same text being reviewed. In our primary spreadsheet we also collected any definitions authors provided for each document type, as well as descriptions of input materials, goals, uses and whether they provided an exemplar document for the reader.
The expanded De Bleser approach
As part of our methodology, we viewed CCPS documents as consisting of elements that can be represented along continuums describing either their (1) degree of prescriptiveness or (2) input to healthcare quality improvement. De Bleser et al.
54
identified both the continuum and their elements which they applied in development of a characterisation framework for
Drawing on De Bleser et al’s framework, we extended De Bleser’s coordinate system approach for use in describing CCPS documents’ evidential and practical application, that is, the addition of an hierarchy of CCPS based on the strength of systematically developed evidence contained within the document along a virtual
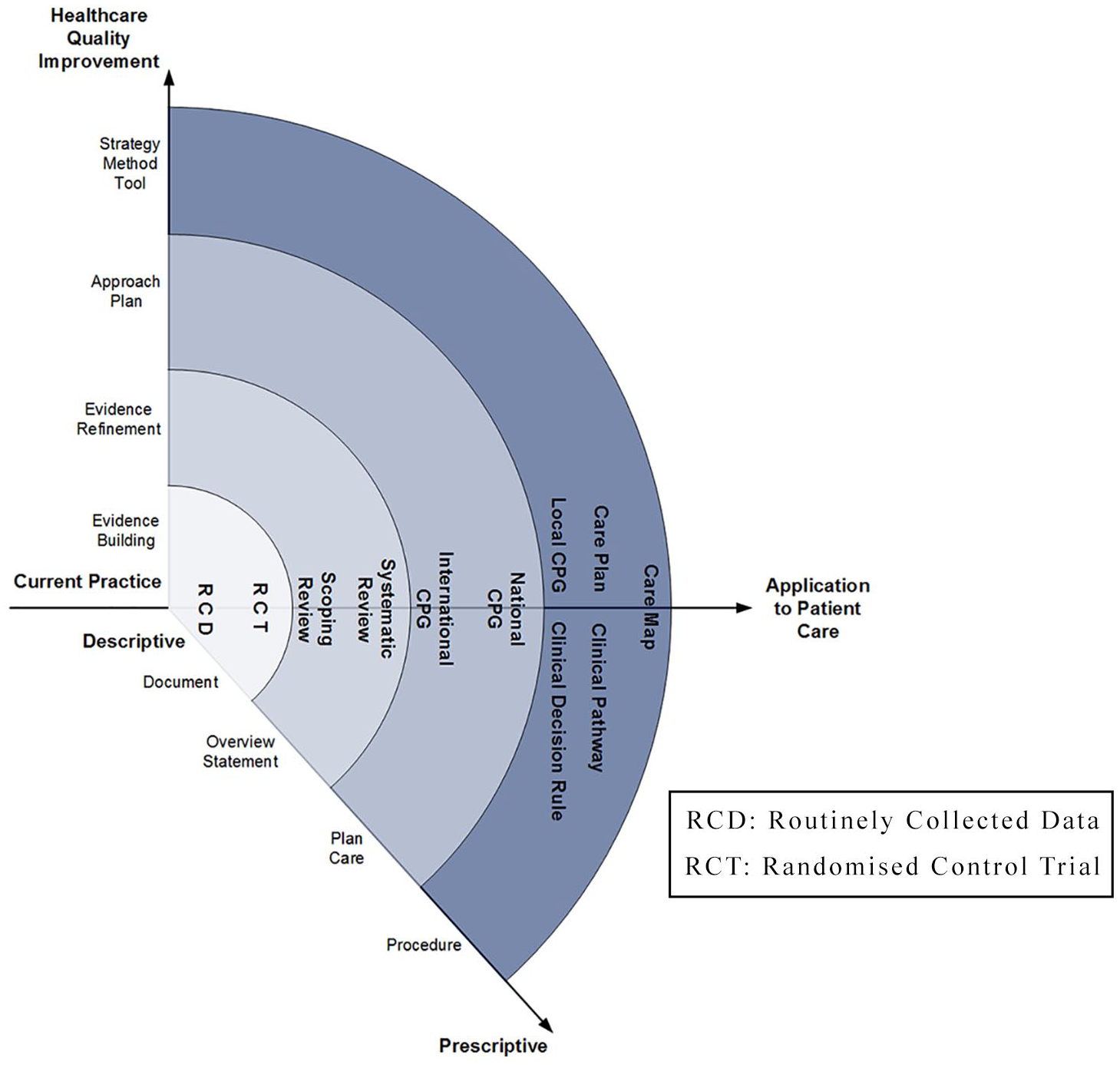
The expanded De Bleser approach.
Evaluative case study
The taxonomy and CCPS document characteristics through a
Results
The taxonomy for evidence-based clinical care processes
In developing the taxonomy shown in Figure 2, we considered all instances identified in the table in Appendix 1, along with authors’ definitions and descriptions of each document type and input from our clinical experts. The taxonomy identifies the categories of clinical documents we determined from our review as being unique or distinct, as well as a hierarchy for how authors described each as being derived from, dependant on, or acting as a component of another. A solid connecting line, or arc, represents where a strong and direct, almost, parent–child relationship was resolved from the literature. A dashed connecting arc represents those instances where an indirect relationship was inferred from authors’ narratives. Each document type and their described interrelationships are discussed in the following section.
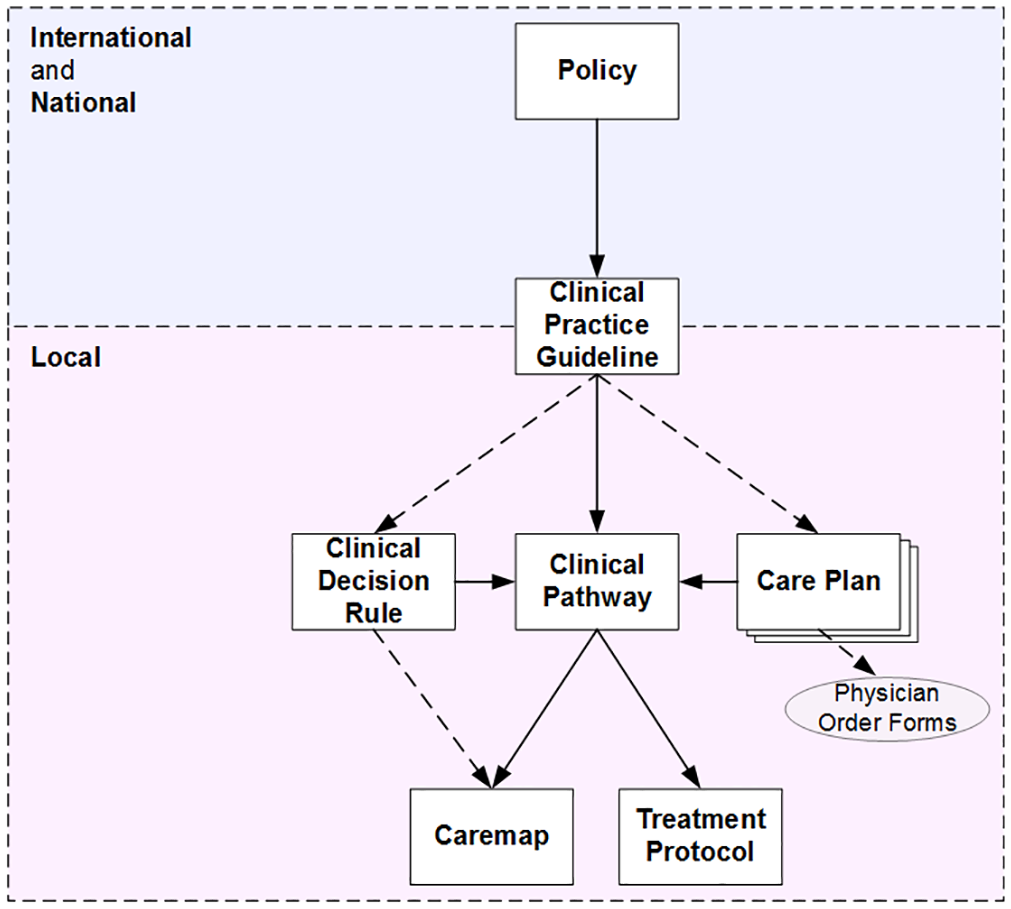
Taxonomy and hierarchy for clinical care process specification documents.
There is a notable relationship between this taxonomy and the expanded De Bleser model in Figure 1, especially in the hierarchy of systematically developed evidence described on the
Policy
Policy was the most fundamental and formative document identified. Because policymaking can be driven or impeded by highly charged and competitive political ideologies and value systems, the resulting policies are not always based on clear and convincing scientific evidence.62,63 International and national organisations can also give rise to health policy.64–66 Policy can be applied at both a national and local level, with local policies often being representative of aspects of national or regional policy made relevant to specific local populations, whether based on ethnicity, age or disease criteria.66,67 Policy can directly determine a population’s health and health outcomes, especially where it is used to guide regulation of the distribution of specific types of health service in terms of human and material resources, how those healthcare services are funded, and the accessibility of those services for patients.66,68
CPG
It is generally accepted that CPG are
Global guidelines are
In combination with local synthesis of evidence, global guidelines are regularly used in the production of
Clinical pathway
Although CPGs and
Our work revealed that clinical pathways were the term most often described as being
There was very limited evidence from which to infer the existence of minor operational differences between the three. In our analysis, the term
Care plan
Care plans are an organised multidisciplinary day-by-day lists of care activities with intermediate outcome-based goals that healthcare providers will undertake to support identified patient problems.84,89 A clinical unit may have a care plan specified for patients with particular medical problem, for example, caesarean section, 84 community-acquired pneumonia 90 or stroke. 86 However, as care plans are specific to the patient they can only become prescriptive once the nurse or clinician has assessed that patient’s particular deficits and needs. 88 An implied relationship exists whereby the care plan may draw knowledge of the condition from the CPG, as indicated in Figure 1 by the dashed line. However, in practice care plans include no supporting literature or knowledge gained from retrospective chart or patient record reviews that would justify the practices used as part of the plan. 90 Care plans are a constituent component of the clinical pathway,84,89,91 and due to their prescriptive and scheduled nature, are credited as the component used to achieve reduced LOS. 90
Clinical decision rule
CDR help clinicians in their decision-making role by providing a link between published and clinical evidence, best practice and the diagnosis or clinical outcome under consideration.62,92,93 CDRs draw on evidence from guidelines to provide operationalised and efficient approaches to assessing probabilities for diagnostic, treatment and prognostic decisions integrated into the clinical pathway.69,92–94 The rules help to establish pretest probability, provide simple screening tests for common problems and estimate risk.62,95
Caremap
Treatment protocol
Known variously as
Characterisation of evidence-based clinical care processes
Table 2 provides a characterisation for CCPS based on their presentation, structure, the population they effect and their clinical intention. It also positions each document with reference to De Bleser et al.’s continuums or axes: the
Characterisation framework of clinical care process specification documents.
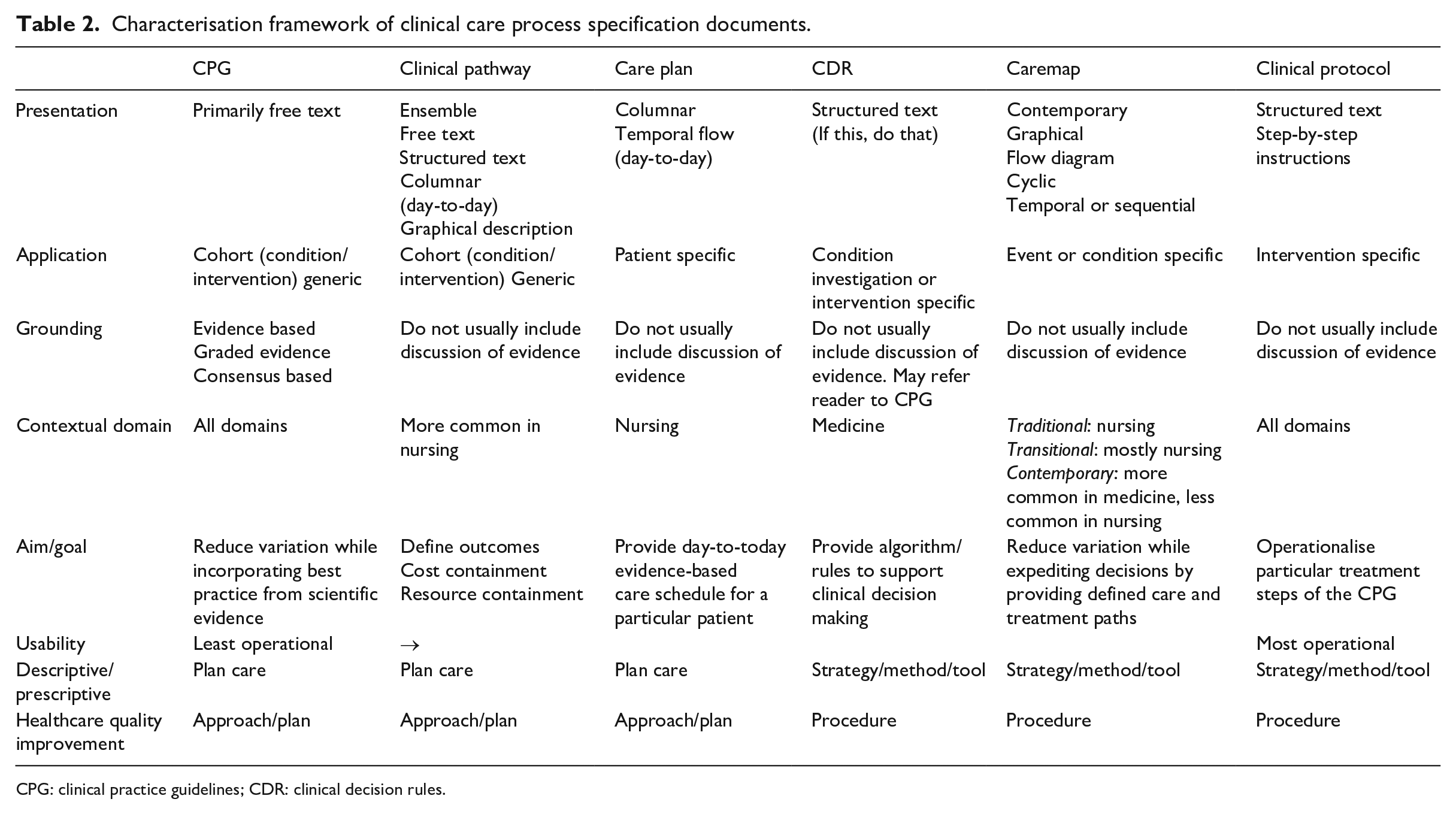
CPG: clinical practice guidelines; CDR: clinical decision rules.
This characterisation is robust in the sense that in addition to De Blesser’s elements it also incorporates aspects that allow CCPS to be analysed more comprehensively in terms of their content, structure and target patient cohort. This table would be useful in tasks seeking to identify or design particular CCPS documentation and ensure that it covers all of the aspects incorporated in Table 2.
Case study: Type 2 Diabetes and diabetes in pregnancy CCPSs in England
This section applies the taxonomy and characterisation to clinical documentation from England on the topic of Type 2 Diabetes (T2D) and diabetes in pregnancy. In each of the following, any reference to a section identifies a section so labelled within the particular clinical care specification document being described. Each of the specifications cited includes a URL in the reference so that the reader may access the document.
Policy
We identified three related documents that describe policy regarding diabetes, and more specifically T2D, namely:

Government policies generally open by expounding on the scope and scale of a public health issue. This information, which centres around the community and resource impact of the medical condition tends to be followed by messages to the lay public that are over-simplified such that they lose the proper meaning and efficacy they are intended to convey. In effect, they miss their goal and become more like promotional material intended for the media, as seen in the example of Figure 3, with claims based on a very small subset of cherry-picked and highly motivated volunteers.

Clinical practice guidelines for Type 2 Diabetes. 101
One component of this same policy which has become enshrined in UK taxation legislation, is the laudable challenge to remove 20 per cent of the caloric content comprised by sugar from all products produced across the food and drinks industry. While put in holistic context, this caloric-reduction policy carries over in Section 4 of the identified National Health Policy. However, this policy approach has resulted in manufacturers finding it necessary to replace sugar primarily with artificial sweeteners like aspartame and saccharin. Both these non-nutritive sweeteners have received sufficient research to suggest that the health dangers of frequent or daily use may also be harmful.104–106 This example demonstrates our claims regarding policy that it is: (1) politically motivated, (2) often not based on established scientific evidence and (3) it tends towards having been constructed by politicians from many small
CPG
A selection of CPG relevant to T2D and diabetes in pregnancy are shown in Table 3.
Clinical practice guidelines for Type 2 Diabetes.

CPG: clinical practice guidelines; IDF: International Diabetes Federation; ADA: American Diabetes Association; EASD: European Association for the Study of Diabetes; NHS: National Health Services.
Similar to the government policy which claimed it had, or would, deliver weight loss, the international guideline at Section 1.2 recommends weight loss as the key tool for preventing T2D.
The international guideline at Section 1.3 also provides for a regimen of testing and thresholds to be used in diagnosing the diabetic patient. In this case the International Diabetes Federation (IDF) guideline follows a diagnostic standard from another international guideline, that of the WHO. Similar diagnostic testing and threshold recommendations carry through in the national CPG at Section 1.6 and local CPG at Section 4.3.
At each level, the recommendations in CPGs become more applicable to patient care. The international and national guidelines present the perfect scenario or perfect model for treatment, while local guidelines recognise that there are non-compliant patients and not everything will go according to plan.
Clinical decision rule
In many cases, CDR can be identified and isolated from within the content of local CPGs. For example, from within the Barts Health National Health Services (NHS) Trust local CPG described above, comes a diagnostic threshold rule for glycaemic control in pregnancy for diabetic mothers who were treated only with diet control: 10.1 Monitor blood glucose 4 hourly. If blood glucose >7.0 mmol/L on 2 consecutive occasions, start IV insulin/glucose infusion. If the woman requires a Caesarean section and the blood glucose >7.0 mmol/L, start IV insulin/glucose regime.
Clinical pathway
Two English diabetes clinical pathways are:
Each presents an approach to screening and patient assessment, care planning, patient support and education that includes for lifestyle changes in diet, weight, exercise and insulin/glucose control. Each also makes reference to, and recommendations regarding, potential complications that may arise in future for these patients and advises the signs and symptoms that clinicians and patients should be mindful of. These pathways present as a generic blueprint for care of patients with diabetes.
Care plan
Two relevant care plans are:
My personal diabetes health care plan and record 115
My personal diabetes handheld record and care plan 117
Each contains information and contact details for services the patient may require. More importantly however, each provides space and tables for recording the day-to-day condition, treatment and care recommendations specific to that patient.
Caremaps
Caremaps provide a visual roadmap of care for a specific condition or treatment process. Caremaps can aid in treatment selection, understanding of disease progression, and can also provide the base knowledge that together with clinical expertise can be used in health research and development of Learning Health System (LHS). The PamBayesian project 118 has developed the following caremaps for diabetes in pregnancy as part of a project to develop clinical decision support LHS for use by clinicians and patients:
Midwifery Booking Visit GDM Booking Visit
Diagnostic Caremap GDM Diagnostic Decisions 2.0
Management Caremap GDM Management Decisions 2.0
Standardisation before computerisation
CCPS computerisation
Various types of CCPS such as caremaps, CPG and clinical protocols are increasingly becoming the subject of computerization. 119 For computerisation to be easily achievable, CCPS need to be formalised to make them computer-interpretable. 120 Formalisation of CCPS refers to specification using precise and unambiguous terms and structures, usually using computational or mathematical formalisms, making the resulting CCPS as computer interpretable.
Standardisation as a prerequisite to computerization
CCPS computerisation is enabled by CCPS formalisation which in turn is enabled by CCPS standardisation. Consequently, computerising CCPS that have not first been standardised creates more problems than it solves, or as one group of authors put it, that
Role of data and information standardisation in CCPS computerization
Data standardisation is needed to resolve the
The benefits of standardisation in the context of CCPS computerization
Standardisation of clinical documents has been observed to improve data collection, data quality, and to enable automated coding and analysis.124,125 Standardisation of CCPS using approaches that include: (1) defined data definitions and (2) attention to
Discussion and related works
The confusion arising out of the absence of standardised definitions and nomenclature for clinical documents has been known since at least 2006.18,54,127,128 As a result of this
Authors have sought to define or present approaches to develop different CCPS during the last two decades, yet significant variance in the
Standardised approaches, especially in clinical documentation, ensure that each time a healthcare provider approaches that type of document, the format and content are consistent with expectation. 16 Standardised documentation ensures sufficient higher quality information is recorded and reported, enabling documents to be read quicker and content within to be better retained, all with the effect of improving overall patient safety and outcomes.16,131,137,138
Summary and conclusion
Since the early 2000s there has been a significant shift towards standardisation of healthcare and healthcare practice. Efforts to standardise healthcare can be held back by many things, one of which may be the lack of consistent nomenclature and definitions for CCPS documents, many of which are used every day in hospitals and general practice clinics. We have addressed the challenge by: (1) presenting a four-layer taxonomy for CCPS documents that begins with those documents that are least-able to be applied in clinical care and moves towards those which are most capable of use in the clinical setting and (2) drawing upon previous efforts to standardise the definition and structure of one type of CCPS document, expanding that method to apply to all documents included in the taxonomy. We evaluated our approach through a case study looking at a range of CCPS documents from the United Kingdom concerning diagnosis and treatment of patients with T2D and diabetes in pregnancy. We hope that the results and discussion will stimulate those in healthcare and healthcare service management to consider the potential benefits of standardising clinical documentation in the same way that evidence-based medicine aims to standardise healthcare. Standardising the way patient care is documented and reported will reduce significant data integration and interoperability issues as national health services continue to move towards implementing centralised electronic shared care records.
Footnotes
Appendix

| References | Policy | Clinical decision rule | Case management plan | Clinical practice guideline | Caremap | Clinical pathway | Care pathway | Critical pathway | Clinical/treatment protocol | Care plan | Careflow |
|---|---|---|---|---|---|---|---|---|---|---|---|
| Goode 84 | □ | □ | □ | ||||||||
| Goode 84 | • | • | |||||||||
| Goode 84 | □ | □ | |||||||||
| Goode 84 | • | • | |||||||||
| Giffin and Giffin 139 | • | • | |||||||||
| Giffin and Giffin 139 | □ | □ | |||||||||
| Seaberg and Jackson 140 | • | • | |||||||||
| Hydo 86 | □ | □ | |||||||||
| Hydo 86 | • | • | |||||||||
| Hydo 86 | • | • | |||||||||
| Stiell et al. 141 | □ | □ | |||||||||
| Gordon 142 | □ | □ | |||||||||
| Gordon 142 | • | • | |||||||||
| Gordon 142 | • | • | |||||||||
| Holocek and Sellards 143 | □ | □ | □ | □ | |||||||
| Morreale 90 | □ | □ | □ | ||||||||
| Morreale 90 | • | • | |||||||||
| Morreale 90 | □ | □ | |||||||||
| Hill 144 | □ | □ | |||||||||
| Chu and Cesnik 34 | • | • | |||||||||
| Chu and Cesnik 34 | • | • | |||||||||
| Dentonet al. 145 | □ | □ | □ | □ | □ | ||||||
| Dickinson and Noud 146 | • | • | |||||||||
| Dickinson and Noud 146 | • | • | |||||||||
| Philie 147 | • | • | |||||||||
| Philie 147 | □ | □ | |||||||||
| Quaglini et al. 148 | • | • | |||||||||
| Lanzieri et al. 149 | □ | □ | |||||||||
| Trowbridge and Weingarten 32 | • | • | |||||||||
| Micik and Borbasi 150 | □ | □ | |||||||||
| Panzarasa et al. 151 | • | • | • | ||||||||
| Powell 152 | • | • | |||||||||
| Jones 87 | • | • | |||||||||
| Jones 87 | • | • | |||||||||
| Mingmei 153 | • | • | |||||||||
| Mingmei 153 | □ | □ | □ | ||||||||
| Beaupre 38 | □ | □ | |||||||||
| Beaupre 38 | • | • | |||||||||
| Van Herck et al. 154 | □ | □ | □ | □ | |||||||
| Agrawal and Kosowsky 62 | • | • | • | • | • | • | |||||
| Agrawal and Kosowsky 62 | • | • | |||||||||
| Brehaut et al. 155 | • | • | |||||||||
| Repasky 156 | □ | □ | |||||||||
| Ducharme 69 | • | • | • | • | • | ||||||
| Ducharme 69 | • | • | |||||||||
| Dunning et al. 157 | • | • | |||||||||
| De Bleser et al. 54 | □ | □ | □ | ||||||||
| Vanhaecht et al. 158 | □ | □ | □ | □ | |||||||
| Tay et al. 159 | □ | □ | □ | ||||||||
| Gaddis et al. 160 | • | • | • | ||||||||
| Sternberg 91 | • | • | |||||||||
| Sternberg 91 | □ | □ | |||||||||
| Mackey et al. 161 | • | • | |||||||||
| Kwan 162 | □ | □ | □ | □ | |||||||
| Richman et al. 163 | □ | □ | |||||||||
| Rotter et al. 164 | □ | □ | □ | □ | □ | ||||||
| Rotter et al. 164 | • | • | |||||||||
| Rotter et al. 164 | • | • | |||||||||
| Lougheed et al. 165 | • | • | |||||||||
| Lougheed (2008) | • | • | |||||||||
| Gemmel et al. 35 | □ | □ | □ | ||||||||
| Isern and Moreno 166 | • | • | |||||||||
| Ebell 92 | • | • | |||||||||
| Nakanishi et al. 167 | • | • | |||||||||
| Kinsman et al. 127 | □ | □ | □ | ||||||||
| Kinsman et al. 127 | • | • | |||||||||
| Kinsman et al. 127 | • | • | |||||||||
| Kinsman et al. 127 | • | • | |||||||||
| Tastan et al. 89 | • | • | |||||||||
| Lodewijckx et al. 168 | • | • | |||||||||
| Tastan et al. 89 | • | • | |||||||||
| Huang et al. 88 | • | • | |||||||||
| Huang et al. 88 | □ | □ | |||||||||
| Huang et al. 88 | • | • | |||||||||
| Lockie et al. 93 | • | • | |||||||||
| Jayaraj et al. 169 | • | • | |||||||||
| Jabbour et al. 170 | • | • | |||||||||
| Jabbour et al. 170 | • | • | |||||||||
| Morrow et al. 171 | □ | □ | |||||||||
| Yu 172 | • | • | |||||||||
| Yu 172 | • | • | |||||||||
| Li et al. 173 | • | • | |||||||||
| Royall et al. 174 | • | • | |||||||||
| Carter et al. 175 | • | • | |||||||||
| Carter et al. 175 | □ | □ | |||||||||
| Wang et al. 94 | • | • | |||||||||
| Wang et al. 94 | • | • | |||||||||
| Roopsawang and Belza 176 | □ | □ | □ | ||||||||
| Buchert and Buttler 128 | □ | □ | □ | □ | □ | □ | |||||
| Buchert and Buttler 128 | • | • | |||||||||
| Dagliati et al. 85 | • | • | • | ||||||||
| Dagliati et al. 85 | • | • | |||||||||
| Gopalakrishna et al. 177 | • | • | |||||||||
| Gopalakrishna et al. 177 | □ | □ | |||||||||
| Solsky et al. 19 | • | • | |||||||||
| Alonso-Coello et al. 178 | • | • | |||||||||
| NHS: London Clinical Networks 114 | • | • |
• = distinct/different items; □ = same or synonymous items.
Acknowledgements
The authors thank Anokunda, Danika, Thomas, Liam and James.
Authors Note
The ORCID for the co-authors are as follow, Norman E Fenton: 0000-0003-2924-0510, Kudakwashe Dube: 0000-0002-2829-8481, Evangelia Kyrimi: 0000-0001-6727-2279, Graham A Hitman: 0000-0002-6637-9004
Contributors
S.M. performed the primary research and prepared the first draft. S.M. proposed the basis for the taxonomy and expanded De Bleser approach, which was refined by E.K. and K.D. J.S. and G.H. provided clinical input and review. K.D. and N.F. supervised the research. All authors contributed, commented and approved the final draft.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship and/or publication of this article: S.M., N.F., G.H. and E.K. acknowledge support from the EPSRC under project EP/P009964/1: PAMBAYESIAN: patient managed decision-support using Bayes Networks. K.D. acknowledges financial support from Massey University for his study sabbatical with the PamBayesian team.
