Abstract
Many Clinical Document Architecture (CDA) referrals and reply documents have been accumulated for patients since the deployment of the Health Information Exchange System (HIES) in Korea. Clinical data were scattered in many CDA documents and this took too much time for physicians to read. Physicians in Korea spend only limited time per patient as insurances in Korea follow a fee-for-service model. Therefore, physicians were not allowed sufficient time for making medical decisions, and follow-up care service was hindered. To address this, we developed CDA Integration Template (CIT) and CDA Integration System (CIS) for the HIES. The clinical items included in CIT were defined reflecting the Korean Standard for CDA Referral and Reply Letters and requests by physicians. CIS integrates CDA documents of a specified patient into a single CDA document following the format of CIT. Finally, physicians were surveyed after CIT/CIS adoption, and they indicated overall satisfaction.
Keywords
Introduction
When integrated patient data are offered to physicians, practice of evidence-based medicine can be made very efficient. Physicians’ time is saved and quality of care improved. Exchanging and integrating clinical data from different hospitals is achieved via standardization of documents for interoperability. 1
In June 2008, Korea’s government launched the National Health Information Exchange pilot project.2,3 The designated project site was Seoul National University Bundang Hospital (SNUBH), which is a tertiary hospital with 1200 beds along with 33 affiliated physician clinics. In the core of the project, Clinical Document Architecture (CDA) and Integrating the Healthcare Enterprise Cross-Enterprise Document Sharing.b (IHE-XDS.b) were incorporated. CDA, developed by Health Level 7, is a document markup standard based on XML that specifies the structure and semantics of “clinical documents” for the purpose of exchange and it is a widely recognized standard in the field of health informatics. 4 Through the first and second versions released in 2001 and 2005 in sequence, 5 various projects have adopted CDA for clinical settings.6–8 IHE-XDS.b facilitates archival, maintenance, and exchange of clinical documents. 9
A few years after the deployment of the Health Information Exchange System (HIES) in Korea, however, it became apparent that the increasing amount of per-patient CDA documents was causing several problems.
On the HIES, patient care data were scattered in various CDA documents, causing delay in physician’s comprehension of patient’s medical history. Hospitals in Korea stay financially viable by treating more patients per day because the insurance payment model in Korea is fee-for-service. Thus, physician’s time spent with patients per visit is short in Korea. 10 Hence, physicians were not allowed sufficient time for making medical decisions. Follow-up care service was also hindered because insufficient time was allowed. For follow-ups, noticing of abnormal diagnostic test results is critical. 11 This is an important issue because patients with several chronic diseases may see up to 16 different physicians in a year. 12 Furthermore, referred patients were not likely to be given effective care because a large amount of CDA documents are generated each time a patient was referred and reply made.
Therefore, it is imperative that CDA documents be integrated for quick review of patient care history to be presented to physicians to ensure quality of care. Unless patient treatment history per clinical item is integrated and classified by chronological order, effective care of patients becomes difficult to achieve.
This article describes the CDA Integration Template (CIT), and CDA Integration System (CIS), which we developed with the goal of integrating CDA documents produced at different hospitals. CIT was developed to reflect what physicians thought important to show patient’s medical history at a glance. CIS was designed to construct CDA documents that integrate various scattered CDA documents per patient in the format of CIT. CIS integrates patient’s data either by following CIT or by including data selected by physician user.
CIS provides a web service based on a loosely coupled structure so that it would be easy to apply to a wide variety of platforms. It also minimizes modification by letting the hospitals keep their existing information system. CIT and CIS were adopted into the HIE National Pilot Project as an upgrade in June 2010. The final section of this article describes the result of CIT/CIS adoption, user satisfaction survey, and concludes with suggestion for future work.
Methods
In this section, we present the related technical aspects of CDA Integration Template (CIT) and CDA Integration System (CIS).
CIT
CIT integrates multiple CDA documents generated at different hospitals per patient into a single CDA document consisting of necessary sections to show consolidated care history to physicians. CIT is based on the Korean Standard for CDA Referral and Reply Letters (preliminary version), which defines the required and optional items to generate referral and reply letters in the CDA format, offering guidelines for hospitals and affiliated physicians’ clinics. The definition of CDA and specifications at the levels of document, header, and body, are included in the Korean Standard for CDA Referral and Reply Letters. It also includes a sample of CDA document. The document-level specification contains definitions of referral and reply letters; items included in the body level, LOINC code (Logical Observation Identifiers Names and Codes) for each item, and cardinality, which indicates whether the referred item is required or optional. The header-level specification contains implementation guidelines for document information, patient information, healthcare provider, and a header sample. The body-level specification includes implementation guidelines for each item and their samples. Table 1 shows the items included in the body-level of the referral and reply letter CDA documents.
Sections in the CIT body reflecting Korean standard for CDA referral and reply letters and physician’s requests.
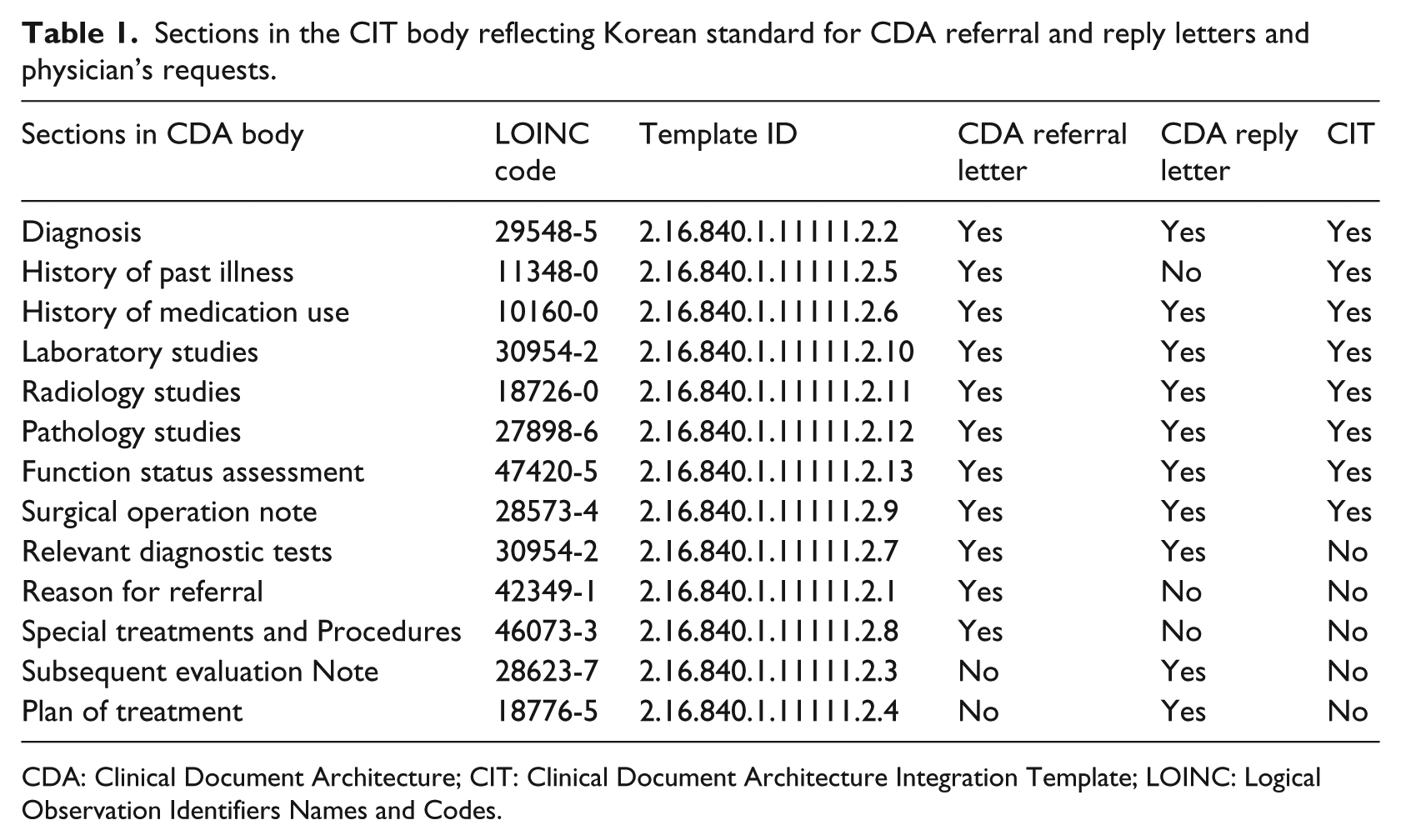
CDA: Clinical Document Architecture; CIT: Clinical Document Architecture Integration Template; LOINC: Logical Observation Identifiers Names and Codes.
CIT is essentially a CDA file and has a header and a body. The header contains information of physician, hospital, and patient. In the header of the CIT, the physician information indicates the physician who initiated integration of related CDA documents; hospital information shows the hospital that integrated the CDA document; patient’s personal information contains the name, date of birth, gender, and phone number of the patient. The names of the hospitals or the physicians who generated CDA documents are excluded because the priority is comprehensively reading the clinical data of a specific patient when a physician diagnoses a patient, not who previously looked into the case of the patient or at what hospital. However, each CDA document does not disappear, even after a document is integrated through CIT. It is still possible for the physician to access the original CDA documents which have data on the physician and the hospital where the documents are originated from.
The body of the CIT has important clinical data grouped into sections. The body in the CIT contains the following sections: diagnosis, history of past illness, initial assessment note, history of medication use, laboratory studies, radiology studies, pathology studies, functional status assessment, and surgical operation note. Each data item in sections is marked with a timestamp of the recorded time to display the data in time order. The sections in the CIT body were defined based on the existing CDA referral and reply letters as well as requests by physicians. As the purpose of CIT is to enhance quality of care by showing patient’s essential clinical data, the committee decided to include only the indispensable sections in the National HIE Pilot Project. Table 1 lists the sections in detail.
From the CDA referral letter, the following sections were included: diagnosis, history of past illness, history of medication, surgical operation note, laboratory studies, radiology studies, pathology studies, functional status assessment. However, the following sections were not included: reason for referral, relevant diagnostic tests, special treatments and procedures. From the CDA reply letter, the following sections were included: diagnosis, operation note, laboratory studies, radiology studies, pathology studies, and functional status assessment. On the contrary, the following sections were not included: subsequent evaluation note, plan of treatment, and relevant diagnostic tests.
CIS
CIS is a system designed to integrate multiple CDA referral and reply letters into one document in the format of CIT. The architecture of Korea’s HIES was developed based on the profile of IHE-XDS.b. The overall architecture of CIS coupled with IHE-XDS.b-based HIES in Korea is shown below in Figure 1.
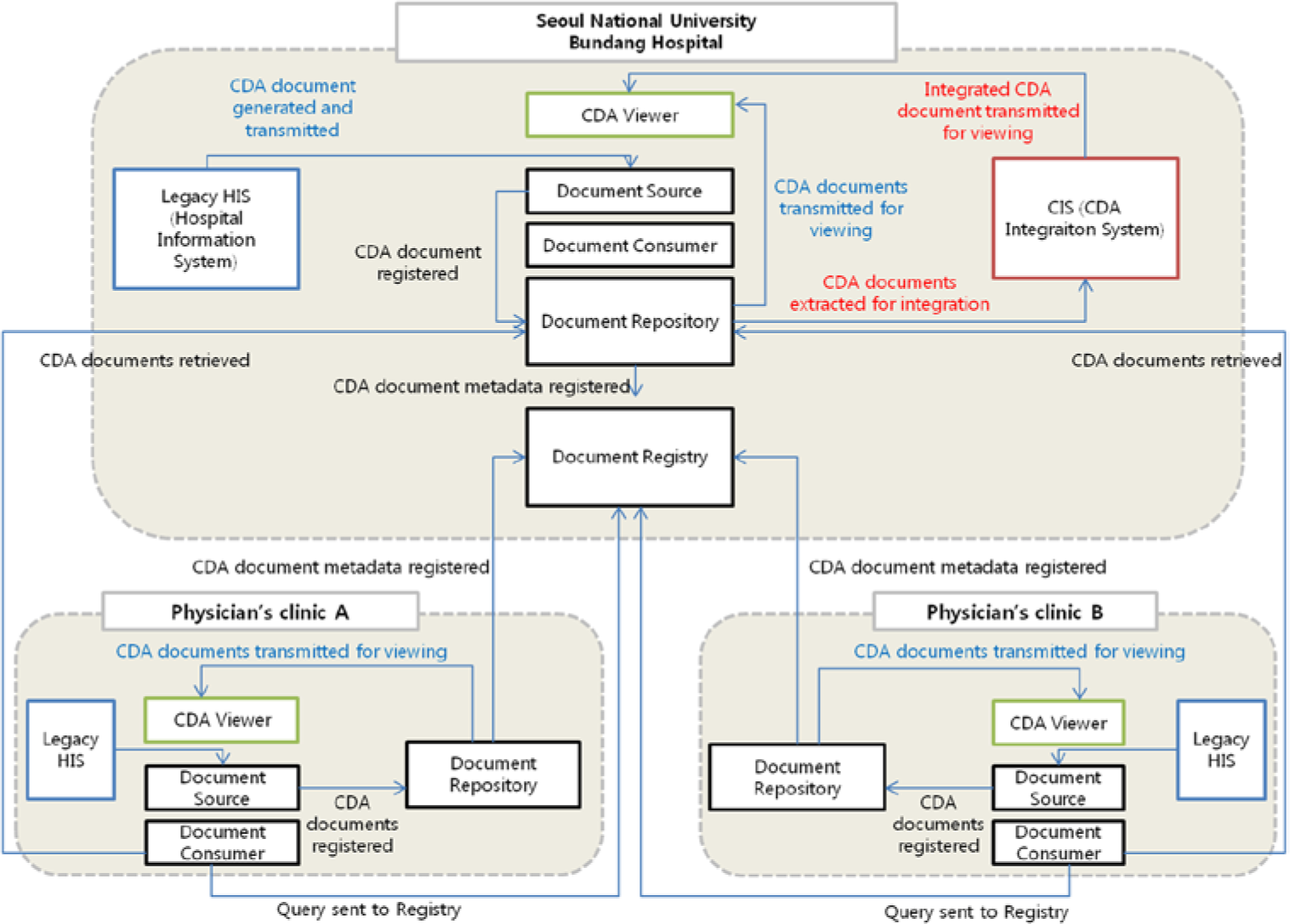
An overview of architecture of CIS coupled with IHE-XDS.b–based HIES in Korea.
CDA documents are integrated in the CIS with the following procedure. A CDA document is first generated at the Hospital Information System (HIS) of a hospital and stored at the Document Repository via Document Source.
While generating a CDA document, its document ID is created in the object identifier (OID) structure, which is a globally unique identifier of a tree structure with non-negative integers. 13 Figure 2 shows the structure of OID used for CDA documents in Korea’s HIE Pilot Project.
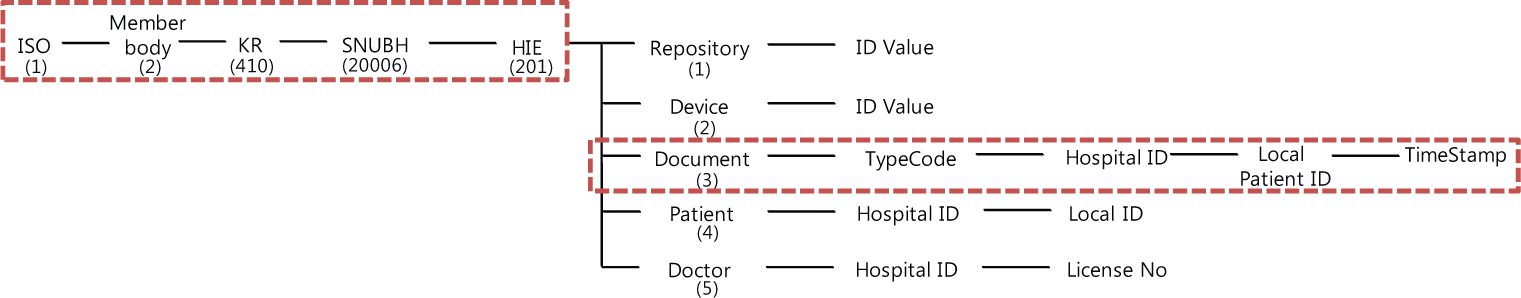
The structure of OID in the CDA document in Korea’s HIE Pilot Project.
The ID of a CDA document consists of 10 numbers, separated by dots. Therefore, the ID 1.2.410.20006 signifies International Organization for Standardization (ISO) number, member body, Republic of Korea, and SNUBH in sequence. Usages for the numbers from the 5th up to the 10th have been defined by SNUBH. The fifth number “201” signifies that the object is involved in the HIE. The sixth number signifies the type of the object, which could be one of the following: a clinical document, a repository, a device, a patient, or a doctor. For instance, a clinical document is signified with “3.” The seventh number signifies the type of the clinical document; the number “1” here indicates that the document is a CDA referral letter, the number “2” as a CDA reply letter, and the number “5” as an integrated CDA document. The eighth number indicates the hospital ID and the ninth implies the local patient ID at each hospital. The 10th number is the timestamp when the clinical document was generated.
The Document Repository extracts metadata from the document and registers it in the Document Registry. Document metadata are representative data of a given clinical document, and are used as parameters for document browsing and extraction. Then, the Document Consumer may make requests to the Document Registry for patient clinical documents stored in other hospitals using metadata. When a request is received, the Document Registry returns the locations of Document Repositories that have the documents, according to the requested metadata. Afterward, the Document Consumer is allowed to access the requested documents in the Document Repository by referring to the retrieved location data. The physician then can read the retrieved CDA document with a CDA document viewer.
CIS retrieves the CDA documents only relevant to a specified patient out of the data from the Document Repository and integrates each item to suit the specification defined for CIT in chronological order. Figure 3 shows the detailed flow of CDA document integration via CIS in coordination with HIES. The Document Extractor verifies the patient’s Social Security Number (SSN) and the timestamp when the document was generated. With these data, a query is sent to Document Registry to retrieve the locations of CDA documents generated within the last 6 months, and the patient’s CDA documents stored in the Document Repository are retrieved using the locations. The Document Integrator then integrates the patient’s multiple CDA documents in accordance with the CIT format. The template IDs for the sections in the CDA document body are parsed during CDA document integration, and the sections in the body with the same template IDs are put together. If the corresponding section or template ID does not exist in the CDA documents, the section or template ID are appended in the integrated document. Each piece of clinical data is shown from the latest to the oldest indicated by the timestamp. The integrated CDA document can be read with the CDA Viewer of HIES.
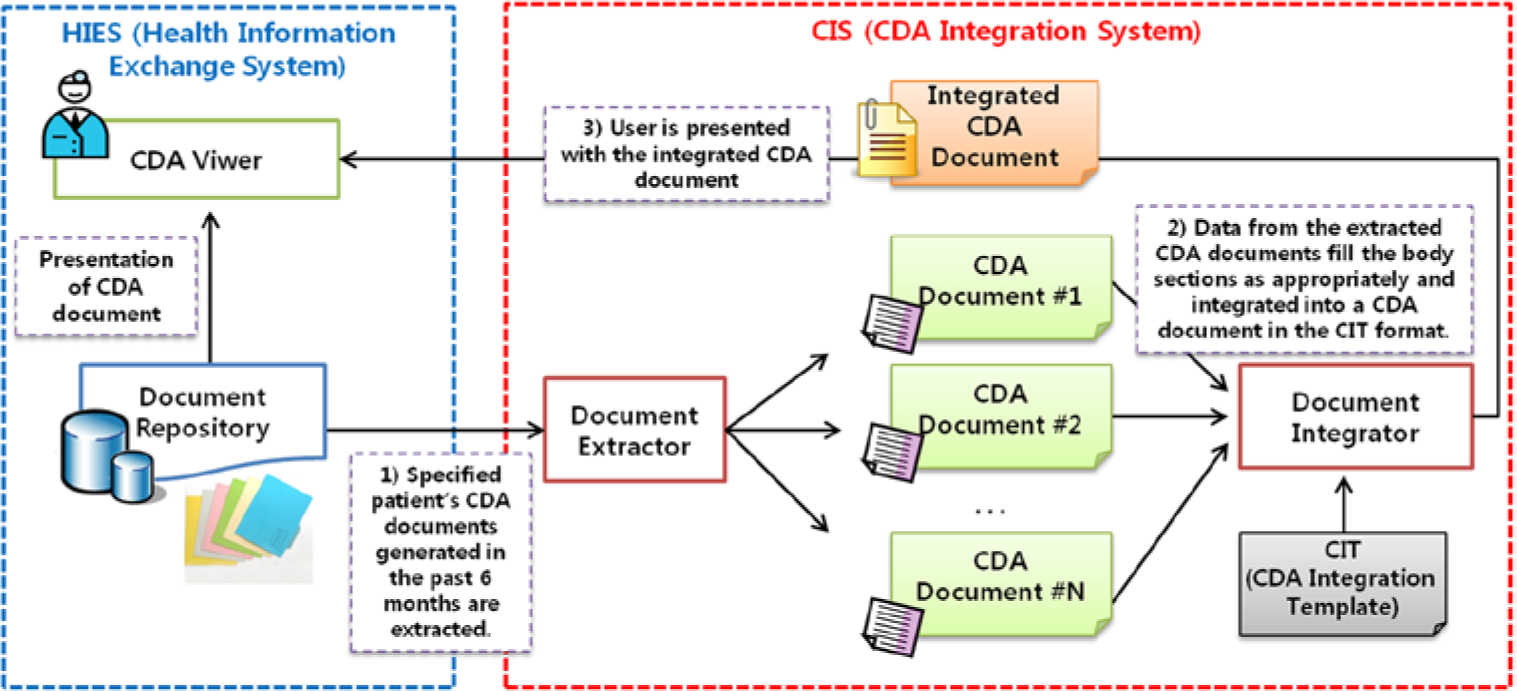
CIS (CDA Integration System) architecture.
CIS was developed as a web service based on a loosely coupled structure to be easy to apply to various platforms and minimize modification for existing hospital information systems. We also developed the web service Application Programming Interface (API) for CDA document integration. Hence, a hospital that chooses to utilize CDA document integration feature may synchronize its system with the CIS web service by using this API to integrate CDA documents in the CIT format.
Results
This section describes adoption of CIT and CIS into Korea’s HIES, usage rate, and user responses to the updated system.
A screen from the HIES following adoption of CIT and CIS is portrayed in Figure 4. The upper left quadrant shows the list of CDA referral letters sent from each affiliated physician’s clinics to SNUBH; the lower left quadrant shows the list of CDA reply letters sent from SNUBH to the affiliated physician’s clinics. When the “Integrate patient’s CDA documents” button is clicked, a single document summary of CDA referral and reply letters within the past 6 months is compiled and displayed on the right half of the screen. The user may customize the summary in a single document by selecting which sections to include among the following: diagnosis, history of medication use, laboratory studies, special treatments, and procedures.
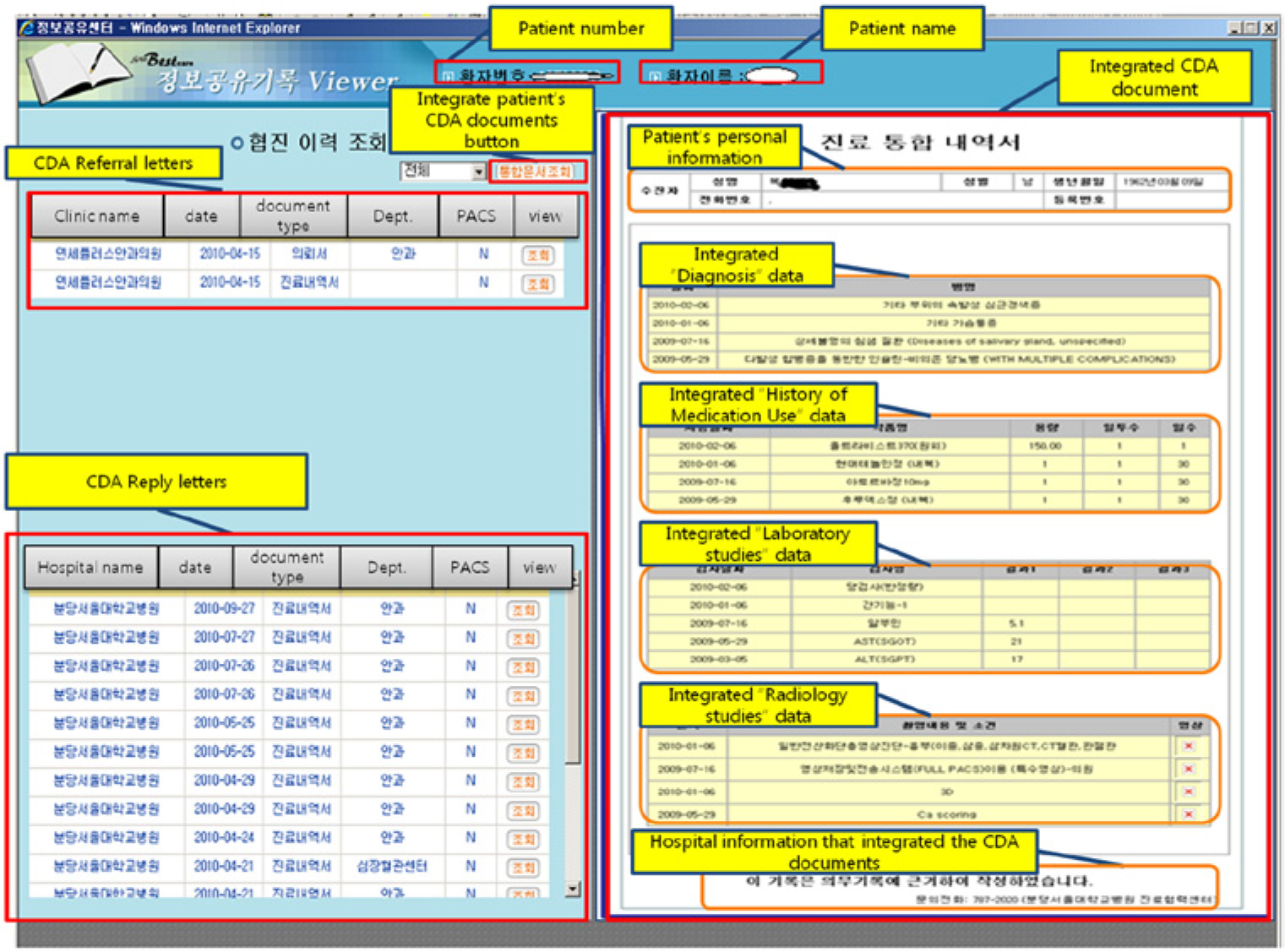
Example screen of a HIE system that CIS has adopted.
Six months after the adoption of CIT and CIS into Korea’s HIES, the system was being used to integrate 23 percent of CDA referral and reply letters, as shown in Table 2.
CDA Referral and reply letters, and instances of CDA document integration within six months after the adoption of CIT and CIS into Korea’s HIES.

CDA: Clinical Document Architecture.
Physicians using the system were surveyed for user satisfaction. Survey replies came from 12 respondents: four otolaryngologists, two family medicine doctors, one ophthalmologist, one rehabilitation medicine doctor, one cardiologist, one endocrinologist, one surgeon, and one rheumatologist. User satisfaction was scored on a five-point Likert scale and the results are shown in Table 3.
User satisfaction survey result.
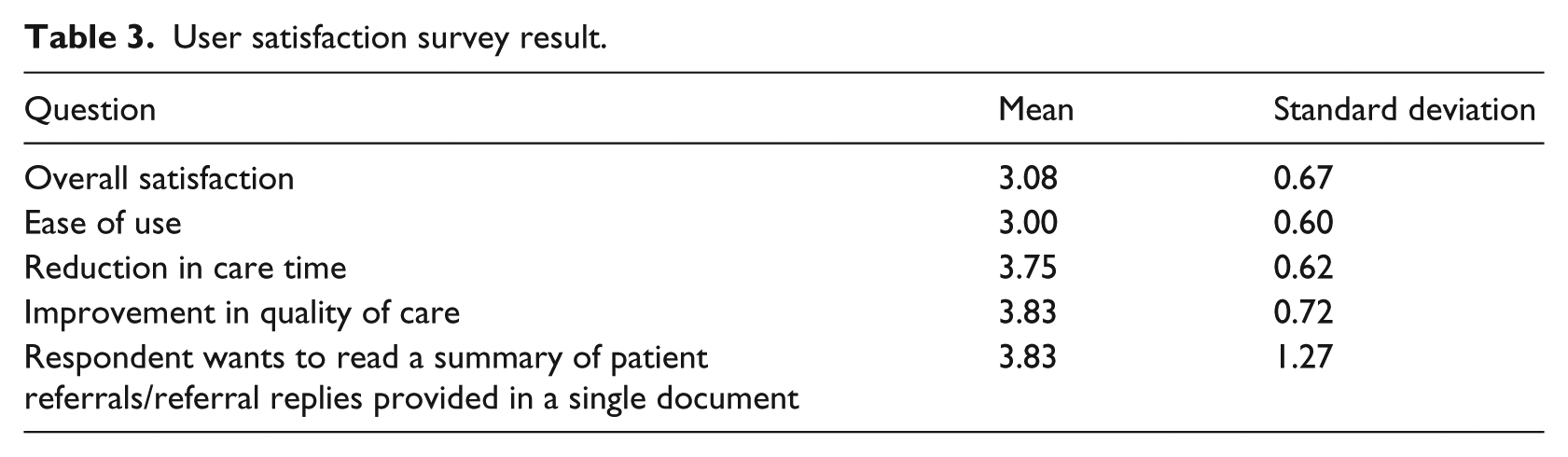
The participants in the survey indicated that they were satisfied with CIS overall and it was convenient to use as well. Many replied that CIS helps save time for patient care. Respondents especially gave high scores to improvement in quality of care and desire to read a single summary document, implying that viewing integrated data of CDA referral and reply letters significantly contributes to improve the quality of care.
Discussion
The method of clinical data exchange can be either message-based or document-based. 14 Messages are usually used to trigger events. Message-based data exchange is primarily used for transmitting device-related data such as laboratory results, and HL7 V2.x Message is a representative standard. 15 The purpose of document-based data exchanges is data storage and transmission, and documents have to be readable by human users; the applications are mostly for clinical data summary exchange, and HL7 CDA is a representative standard. 16
Recent practices in the United States are such that diagnosis summaries are exchanged in CDA documents, while laboratory results, surveillance reporting, submission to immunization registries are transmitted in HL7 V2.x message. Office of the National Coordinator for Health Information Technology (ONC) announced the requirements for Electronic Health Record (EHR) certificates, and these include verification of whether the standards are correctly adhered to. 17 Those requirements contain a list of standards which have to be used in order for EHR system to achieve its qualification.
The method of data exchange may vary depending on the purpose of data transmission. In Korea, document-based method has been chosen for HIE in the national level, and the National HIE Pilot Project based on CDA and IHE-XDS was launched in June 2008. In other countries, a lot of research has been carried out focusing on exchange of CDA documents. Germany launched an HIE project based on XML. In the first stage of this project, CDA referral and reply letters were generated according to CDA Release 1 Standard, 18 and in the second stage, a method to exchange the documents was suggested. 19 In Italy, research was conducted to create a hierarchical structure for home healthcare in XML documents, and the developed model was designed to include CDA documents. 20 A CDA generation method for HISs was researched in Taiwan, and the Electronic Medical Record (EMR) template in CDA to guarantee semantic interoperability at the national level was developed.21,22 Currently, transferring health information in the CDA format on removable storages such as USB is under development. 23 In the case of Croatia, a project is in progress to consolidate HIS and Image Information Systems based on HL7 CDA and Digital Imaging and Communications in Medicine (DICOM) standards. 24
Other countries are supporting or launching HIE projects to summarize various clinical data to offer to physicians, which are listed in Table 4. The method proposed in this article has the following strengths over existing clinical data integration methods. First, our method guarantees interoperability between hospitals and seamless data transmission and integration because documents are integrated based on the CDA format, which is an international standard. Next, independence of HIS data is established because CIS uses the CDA documents registered at HIES rather than accessing a hospital database. Therefore, hospitals can minimize impact on existing HIS when a new system is adopted. Furthermore, integration is offered via the web, thus the burden on an existing HIS is marginal, while it is easy to support a wide variety of platforms.
Comparison of health information exchange projects.
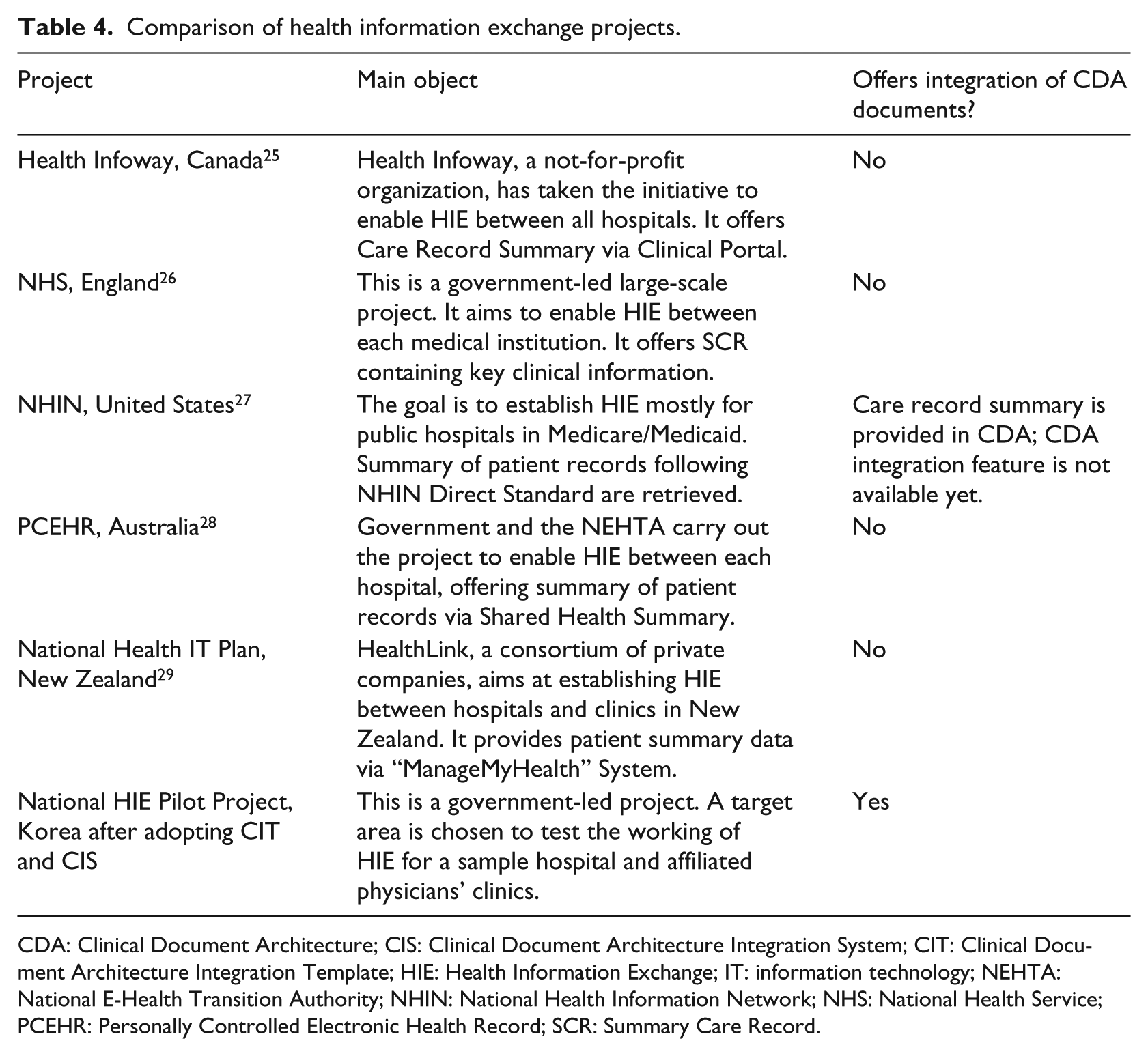
CDA: Clinical Document Architecture; CIS: Clinical Document Architecture Integration System; CIT: Clinical Document Architecture Integration Template; HIE: Health Information Exchange; IT: information technology; NEHTA: National E-Health Transition Authority; NHIN: National Health Information Network; NHS: National Health Service; PCEHR: Personally Controlled Electronic Health Record; SCR: Summary Care Record.
CIS would be a good case for other countries that have plans to implement HIE in the future or are currently in the process of implementing one. As the number of CDA documents increases, a physician will complain about having to read multiple CDA documents per patient during patient care. Adoption of CIS provides only the items that a physician wants to read conveniently in an integrated CDA document.
CDA document format is used when integrating a patient’s medical history for the following reason. Legal requirements have not been firmly established for electronic exchange of clinical documents between hospitals. Patient referral letters only allow “viewing” of documents for the purpose of referring patients for care, not “owning” of documents. Thus, storing patient CDA referral and reply letters from another hospital may be a legally controversial act. In the HIES pilot project, the Korean Ministry of Health and Welfare separated the exchanged documents from those at EMR system or databases of other hospitals, and made an exception for storing data in HIES, and allowed exchange of documents exclusively in the CDA format. Therefore, generation of integrated clinical document requires using the CDA documents stored in HIES for integration. In addition, integration of a patient’s multiple clinical documents over multiple episodes makes it easy to exchange the summarized health information when the patient is referred to another hospital.
The survey to measure physician satisfaction for CIS developed in this article indicated that the majority of the respondents were highly satisfied, and especially, they gave highest scores for contribution to improvement in quality of care, which implies that CIS is helpful for physicians in practicing evidence-based medicine.
CIS can be utilized as a foundation for patient-centric Personal Health Record document generation. 30 That is, only essential data are extracted from the clinical documents generated at each hospital and the summary data for the specified patient can be offered to the patient. The number of patients who manage various health data has been increasing. Hence, document guidelines are under development for patient-generated health data of various vital signs measured at home. 31 CIS can also be utilized to integrate the health data recorded at home and the patient’s clinical documents managed at hospitals.
The CDA for Korea’s HIE Pilot Project does not offer support down to the data entry level because a terminology code system has yet to be agreed by each stakeholder. Further research would upgrade CIT, reflecting the HL7 Consolidated CDA Template, 32 which defines codes down to the entry level for most sections, once agreement has been reached by stakeholders. In this HIE Pilot Project, there is only one document registry because the project has been conducted in only one region, Bundang-gu of Gyeonggi-do province of South Korea. Future studies will need to consider federated HIE with XCA (Cross-Community Access) 33 which connects multiple regions after extending HIE to other regions.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was supported by Kyungpook National University Research Fund, 2014 and the Korea Healthcare Technology R&D project (A112067), Ministry for Health, Welfare & Family Affairs, Republic of Korea.
