Abstract
Kidney transplantation is the treatment of choice for patients with end-stage renal disease, and leads to everyday self-management of this chronic condition. This article aims to provide documentation for a participatory design study of a telehealth solution to improve the kidney transplantation process, and to identify the impact from the different participants in the participatory design study. Through a participatory design approach, a smartphone application (app) was developed for the entire kidney transplantation process together with a workflow for post-transplantation follow-up. A core element in participatory design is user involvement. By way of workshops and laboratory tests, the telehealth solution was developed in close cooperation with patients, their families, healthcare professionals, kidney association representatives, and Information Technology designers. The participatory design approach means that the telehealth solution was designed to be functional in a clinical setting, address patients’ needs, and support their self-management.
Background
Kidney transplantation is the optimal treatment whenever feasible for patients with end-stage renal disease. Transplantation offers survival benefit, better quality of life, reduced medication intake, and fewer restrictions.1,2 Lifelong self-management skills are essential for adaptation to a changed everyday life and prevention of graft loss. Patients also have to manage increased risk of comorbidities, such as skin cancer and infections. 3 As is the case for patients with other chronic diseases, kidney transplantation requires self-management, by taking an active part in managing one’s health condition and reacting to symptoms. Patient involvement in treatment and care is important if self-management is to be successful, and it requires knowledge and confidence. 4 A study exploring self-management experienced by kidney transplant recipients found that it implied managing treatment, forming a long-lasting relationship with the healthcare professionals (HCPs), adjusting daily life activities, improving self-image and dealing with social and emotional issues. 5 Kidney recipients need HCPs’ support to develop self-management skills. 5 Telehealth could be a way to support kidney recipients’ self-management in everyday life.
The use of telehealth has increased over the last decade, and seems to be continuously evolving with increasing potential to improve patients’ health, and change the way healthcare is provided. 6 Telehealth has proved promising in its potential to improve health outcomes, such as enhanced symptom control, improved physical functioning, medical adherence, and improved communication between patients with chronic disease and HCPs.7,8 Overall, it appears that telehealth is leading to increased self-management. An investigation of solid organ transplantation recipients found that they are familiar with information technology (IT). 9 Similarly, others have found a positive attitude towards telehealth among kidney recipients, and the use of smartphones and apps appears to be widespread.10,11
Despite a high symptom burden, wait-listed kidney recipients are interested in telehealth as a way to improve their condition before transplantation. Patients have participated in telephone-adapted, mindfulness-based stress reduction which improved mental health. However, it did not reduce anxiety as expected. 12 Another study found that patients were receptive to home-based physical training support provided via telehealth. 13 Similarly, telehealth for follow-up post-transplantation has been shown to have potential.14–16 A study found that a majority of recipients experienced the same standard during telehealth consultations as in face-to-face appointments, and, in addition, recipients achieved a better understanding of their condition. 16 Likewise, another study found that the number of follow-up appointments could be reduced by using home-based self-monitoring of blood creatinine and blood pressure. Home-based self-monitoring was well received by recipients. 14 It has been found to increase patient satisfaction and can improve detection of acute rejection and hypertension by more frequent measurements. 14 Research have found telehealth to be an acceptable approach to improving medication adherence, that recipients are receptive to its use, and that it can effectively support the interaction between patients and HCPs.17–20
In summary, kidney recipients’ self-management is significant to a positive transplantation outcome, and is dependent on support from HCPs. Research indicates that telehealth could be an effective way to support and improve self-management in kidney recipients.
Aim
The aims of this article are as follows:
To provide detailed exemplary documentation for a participatory design (PD) study of a telehealth solution.
To identify the impact from the different participants in a PD study.
Hence, it leads to the following research question: How can a PD approach contribute to research within health science?
Design
This article reports from the second phase of a PD study. 6 Having roots in action research, PD is a practice and research methodology that was developed within computer science and various branches of social sciences and the humanities.21,22 Action research focuses on changes towards better practice in close collaboration between researchers and the people who are subject to the changes.21,22 Similarly, the aim in PD is direct involvement of people. As for example development of information technology in close cooperation with the users that will be affected by the technology, to make sure the design fits the actual needs in their work practice. 21
A core element is user involvement, and the intention is to equalise the power between designers and users, using participation to facilitate mutual learning. The IT designers need to have knowledge of users’ needs and daily lives, whereas users need to have knowledge of technological options. Through this mutual learning, a shared understanding between IT designers and users can be obtained.21,23 This approach gives the users a voice in designing and developing a technology which will affect their daily lives and enables the IT designers to develop solutions adapted to the users’ needs and their practice.21,23
PD has been applied in health science for more than a decade. Patients and HCPs have been involved in research and have changed clinical practice, which has, in turn, led to a significant impact on patients’ everyday lives.24–28 In health science, PD is conducted in three phases. 6 Phase 1 is identification of needs, through an exploration of experiences. Ethnographic methods are employed to explore everyday life and clinical practice through patients’ and HCPs’ experiences. Phase 2 is the design and development of a telehealth solution, to support the needs identified in the findings from phase 1. Finally, in phase 3, the telehealth solution is tested in clinical practice and in patients’ everyday life, and subsequently evaluated. The study is conducted, by reflecting on the results from the previous phase, and consists of iterative processes. 6 The three phases in our PD study are illustrated in Figure 1. The ethnographic work from phase 1 has been reported in a previous paper. 29
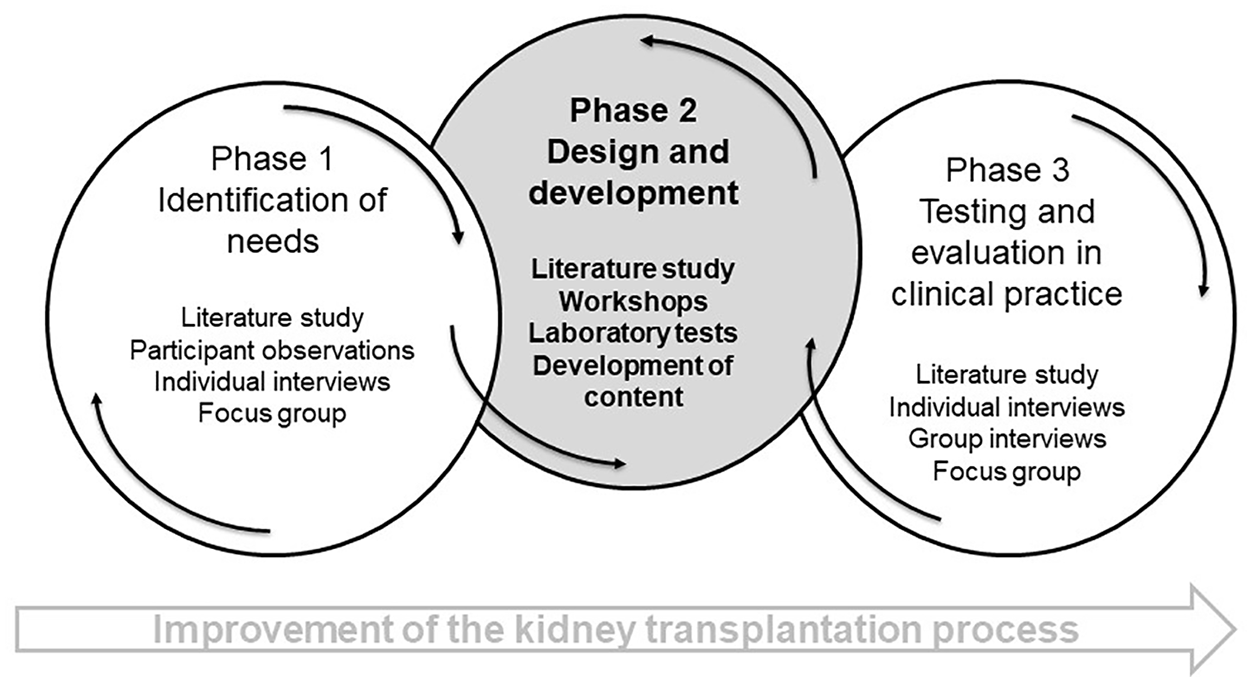
The three phases in the PD study, with a focus on phase 2.
Material and methods
Setting
The study concerns the kidney transplantation process in a Danish transplant centre from time of acceptance until 4 months post-transplantation. Acceptance for kidney transplantation includes a consultation with a nephrologist after a number of clinical examinations and physical tests. Subsequently, transplantation is planned if there is a living donor, or the patients are referred to a waiting list of unknown duration for a deceased donor. After transplantation, the patients are hospitalised for approximately 1 week. They have to visit the outpatients clinic frequently for evaluation of their clinical condition and renal status. In the first 4 weeks, the patients attend the outpatient clinic twice a week. Over time, the consultations become less frequent but continue as long as the kidney graft is functioning.
Participants
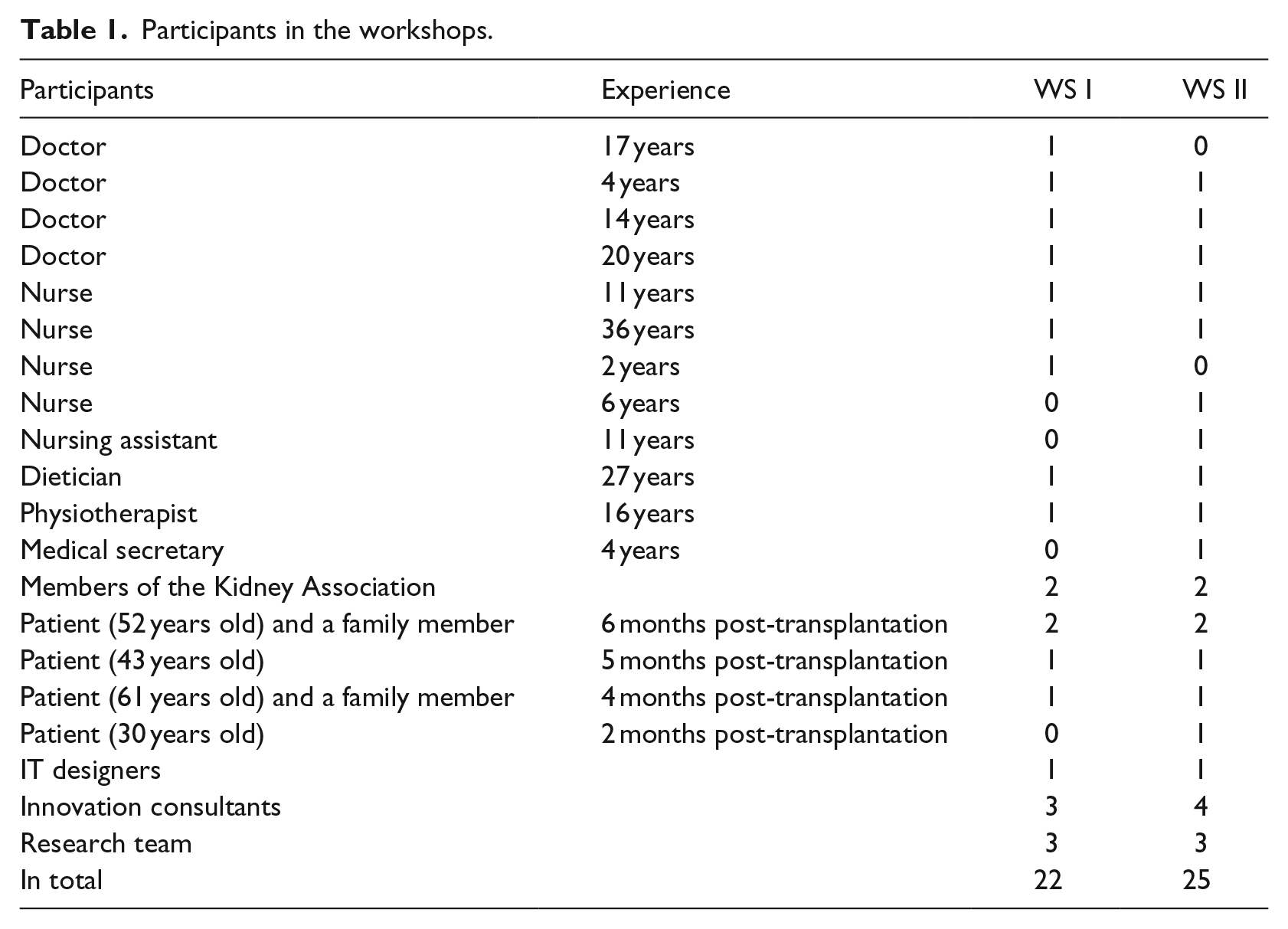
In total, 14 patients were invited, of which 10 declined participation due to lack of physical and mental resources. Four patients, who were in the post-transplantation period, participated, two of them together with a family member. In addition, two patients representing the Danish Kidney Association participated, both of whom had had transplantation years earlier. To ensure changes to suit clinical practice and ensure engagement, it was decided to involve a broad representation of healthcare professions in the design and development process. Therefore, the various professions involved in the transplantation process, that is, doctors, nurses, nursing assistants, medical secretaries, physiotherapists, and dieticians, were represented. In addition, IT designers and innovation consultants participated, together with the research team consisting of two senior researchers within nephrology, a senior researcher within PD, and the first author who was the project leader.
User activities
We used various activities to design and develop a telehealth solution in phase 2 of the PD study. This is illustrated in Figure 2.

An overview of the user activities in phase 2.
Workshops
Two workshops were conducted in May 2017 at the Health Innovation Centre of Southern Denmark, a neutral setting for both patients and HCPs. Both workshops lasted 3 h.
The participants, as characterised in Table 1, were mainly the same in both workshops.
Participants in the workshops.
Creative tools and techniques such as posters (Picture 1), Post-it labels and building of creative models were used at the workshops to facilitate reflections on everyday life, and creative activities were conducted to facilitate mutual learning and ideas for possible telehealth solutions.6,21

A working station with posters.
At the first workshop, the aim was to explore the identified needs, from the ethnographic investigations in phase 1 of the study, and to come up with possible solutions to meet these needs. The participants were divided into three groups consisting of representatives of various healthcare professions and patients, family members, or representatives of the kidney association. Each group joined a working station facilitated by both a researcher and an innovation consultant. Their role as facilitators was to initiate the discussions and take part in the emerged mutual learning process in the groups.
At the working stations there were posters on the wall with headings that had emerged from the ethnographic investigations. The aim was to facilitate group discussions of patients’ needs in the transplantation process. The findings from the ethnographic investigations are presented in Table 2. The participants exchanged points of view and reflections related to the headings, which were added to the posters on Post-it labels.
After discussing and exploring the needs at the stations, each group made a creative model of their proposed solution to accommodate the needs. The creative process was documented by notes and pictures.
At the second workshop the aim became to qualify a solution, which was developed based on the results of the first workshop. The solution was an app to accommodate the identified needs through the entire transplantation process. In addition, a new way to structure follow-up after transplantation supported by the app was developed, as frequent follow-up was identified by the patients as a prominent challenge in the transplantation process. Again, the participants were divided into three groups and alternately visited three stations. The stations represented the various features in the app, and the group discussions qualified the content of the app using posters and Post-it notes similar to the first workshop. Also one station represented follow-up after transplantation, to discuss and provide knowledge to structure and improve the follow-up in a new way for both patients and HCPs.
Laboratory tests
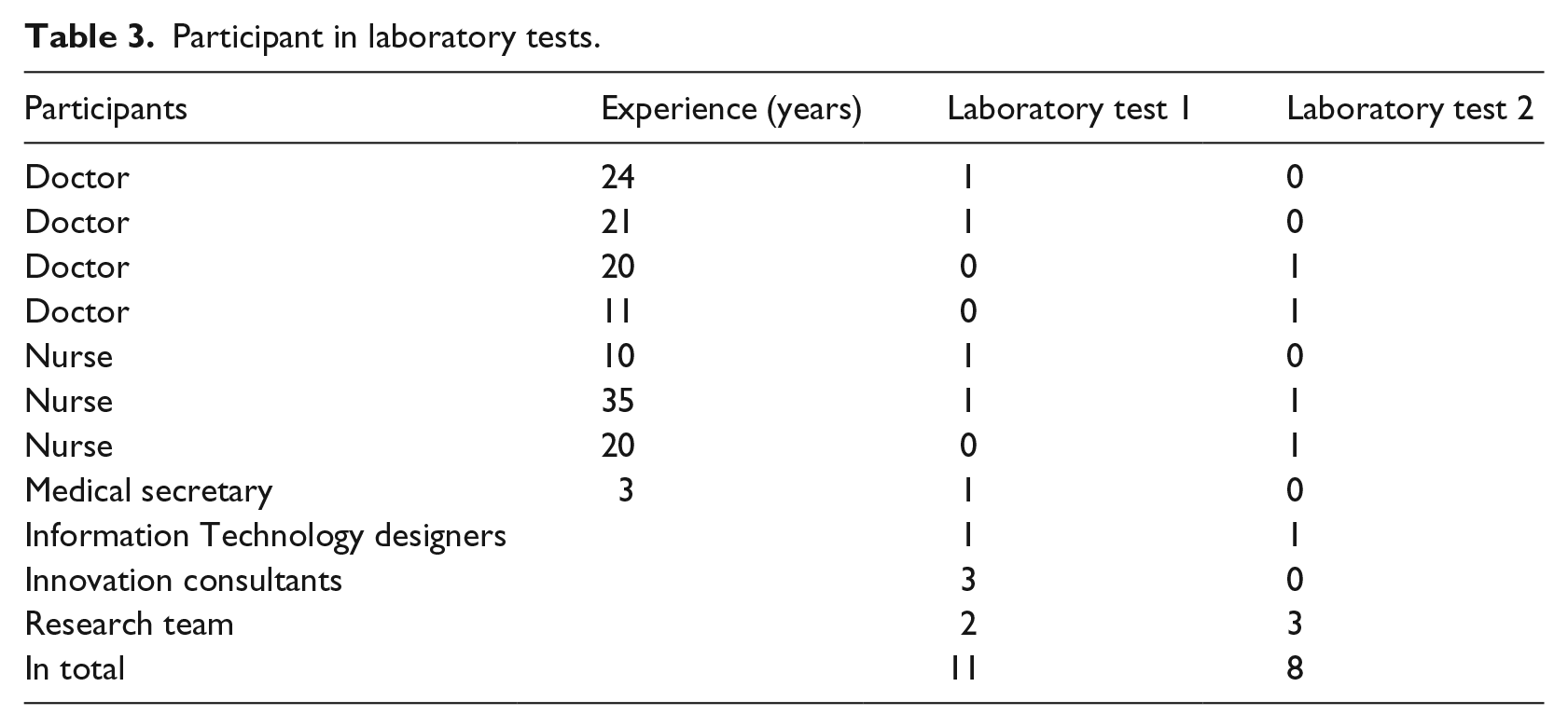
A prototype app and a suggestion for a workflow for follow-up were developed based on the knowledge from the two workshops. The workflow needed testing in a laboratory test, and the aim was to develop a final workflow for follow-up after kidney transplantation. The first laboratory test was conducted at the Health Innovation Centre of Southern Denmark in June 2017, and the second test at the university hospital in January 2018. Both tests lasted 3 h. The participants were nurses and doctors, members of the research team, and IT designers. In addition, innovation consultants and a medical secretary participated in the first laboratory test. The patient perspective was represented by data from both the ethnographic investigations and the workshops. The participants in the laboratory tests are presented in Table 3. The participants shared and reflected on different points of view and experiences, facilitated by the first author. Sample screenshots were used to illustrate and discuss how the technology could support the workflow, as the workflow was reviewed step by step. The screenshots were supported by headings on paper, and comments from the participants were added on Post-it labels to facilitate further comments.
Participant in laboratory tests.
Development of prototype and member check
The workshops led to the development of a prototype app. The content was developed in collaboration with HCPs and innovation consultants, and guided by the patients’ voice from the ethnographic findings from phase 1 and the workshops. Videos, text manuscripts, visual statistics, and a questionnaire were developed. Patients and HCPs gave feedback on the layout and text in the questionnaire, and HCPs gave feedback on videos, statistics, and text manuscripts.
Analysis
The analysis of the material from the workshops and laboratory tests was inspired by a PD approach. The steps plan, act, observe, and reflect, were taken in iterative processes analysing the data material. 6 Each workshop and laboratory test in the study was planned one activity at a time, as it was uncertain how many iterations would be required to develop a final prototype. The group – consisting of the research team, IT designers, and innovation consultants – planned each activity. They participated as facilitators and observers, and made shared reflections after each activity, as part of the data analysis. Based on that, the next step in the design and development process was planned. Each workshop or laboratory test began with a presentation of results from the previous activity to give the users opportunity to comment and contribute with their perspectives on the data analysis. The iterative processes are illustrated in Figure 3.
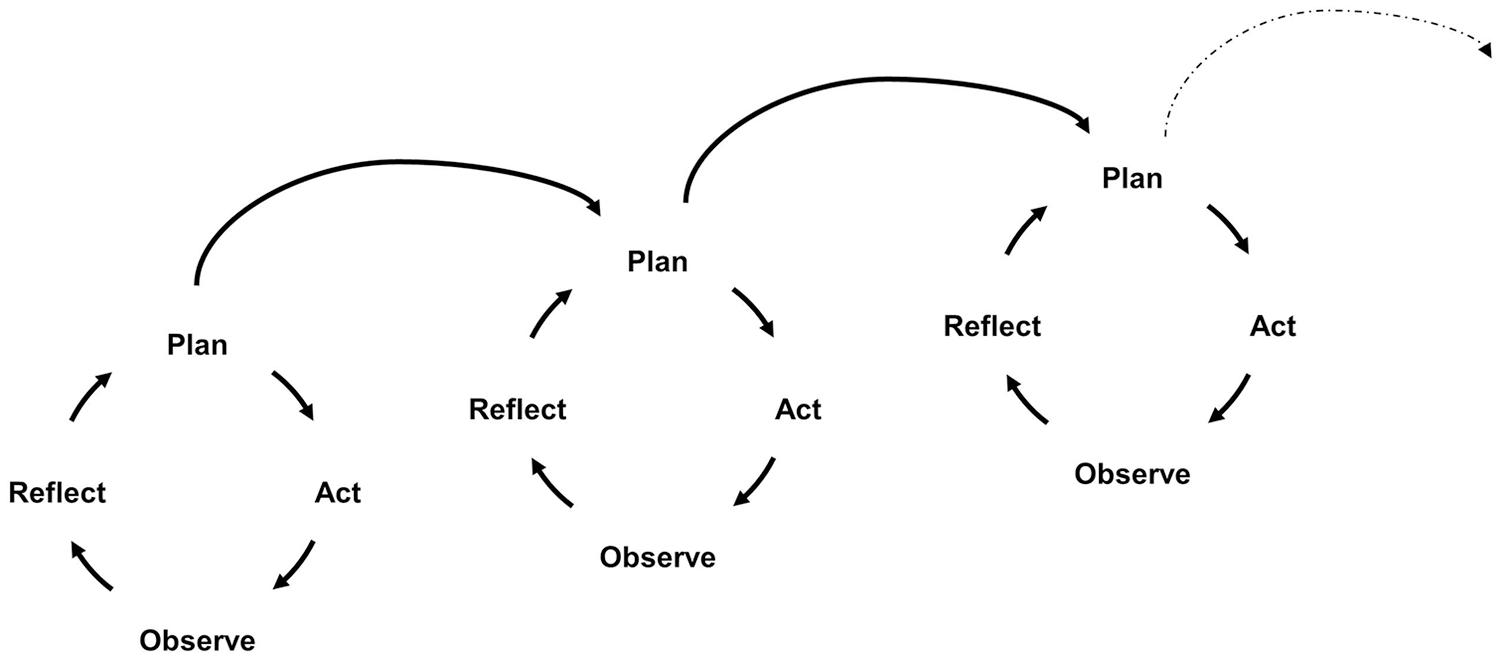
Iterations of plan, act, observe, and reflect inspired by Clemensen et al. 6
Ethics
Participants were informed orally and in writing about the study, in accordance with applicable ethical rules, 31 and also gave their written informed consent. The study was approved by the Danish Data Protection Agency, journal number: 15/48886.
Results
Posters with Post-it labels and notes from workshops and laboratory tests were transcribed by the first author and discussed with the group after each workshop or laboratory test, and a report was drawn up, including pictures of the activities. In the following, the content from across the entire data set is presented and supplemented with illustrating quotations.
Workshop 1
At the first workshop, the needs that had emerged during the study of the kidney transplantation process were explored, and possible solutions to meet the needs were developed. The participants reflected on various aspects concerning the kidney transplantation process, and the dialogue between the participants at the working stations provided a deeper understanding, and facilitated mutual learning. The needs that emerged from the first workshop are presented below.
Before transplantation there was a need for information about transplantation activity and waiting list figures to support the patients’ belief that the transplantation would succeed one day. As an example, one group wrote in keywords: ‘Openness about the waiting list. It [the waiting list] seems secret. So one can see that something is happening’. This period was described as follows: ‘vulnerable, the worst period – much was unknown’.
Both prior to and during the time of the transplantation, knowledge related to kidney transplantation in general and concerning complications was needed. A number of suggestions for information were listed, for example recommendations on activity, nutrition and fluid intake, and knowledge about known complications and treatment. One patient expressed, ‘You can eat normally [post-transplantation], but what does that mean when one has lived a life of restrictions?’. Furthermore, there was a need for preparation before engaging with the healthcare system, as expressed by one group: ‘To be prepared for how the process would transpire’. Patients were not fully prepared for the consultations, the admission for transplantation or possible complications concerning kidney biopsy that might occur in the early stage after transplantation.
New ways to structure follow-up after transplantation were needed. The patients requested fewer meetings at the outpatient clinic and individual consultations – both in relation to planning, as expressed by one group: ‘It can be challenging to remember and find the resources for follow-up’, and the aspects addressed in the consultations were important, as a group wrote: ‘You have to enquire about a person’s life – what do you want to talk about’. A need for communication with the HCPs without having to disturb them with telephone calls was suggested, as it could provide a free space for less urgent questions.
The final activity was to build a model. There were proposals for an app that covered the entire kidney transplantation process, with necessary knowledge and tools integrated in one solution (Picture 2). One group presented a solution for individual follow-up, to give patients the opportunity to have consultations by phone or video (Picture 3).

Model of a proposed app.

Model of follow-up routine after transplantation.
Workshop 2
Based on the first workshop, it was decided to develop an app for the entire kidney transplantation process because it could meet the patients’ needs. At the second workshop, features and their possible content were discussed, based on the findings from the first workshop. Subsequent to the workshop, the features were developed for the app: ‘The Kidney Transplantation Process’. An overview of the identified needs and the related features in the app are shown in Table 4, together with the participants’ contributions and the potential impact of the features.
The identified needs, the related features in the app, and the potential impact.
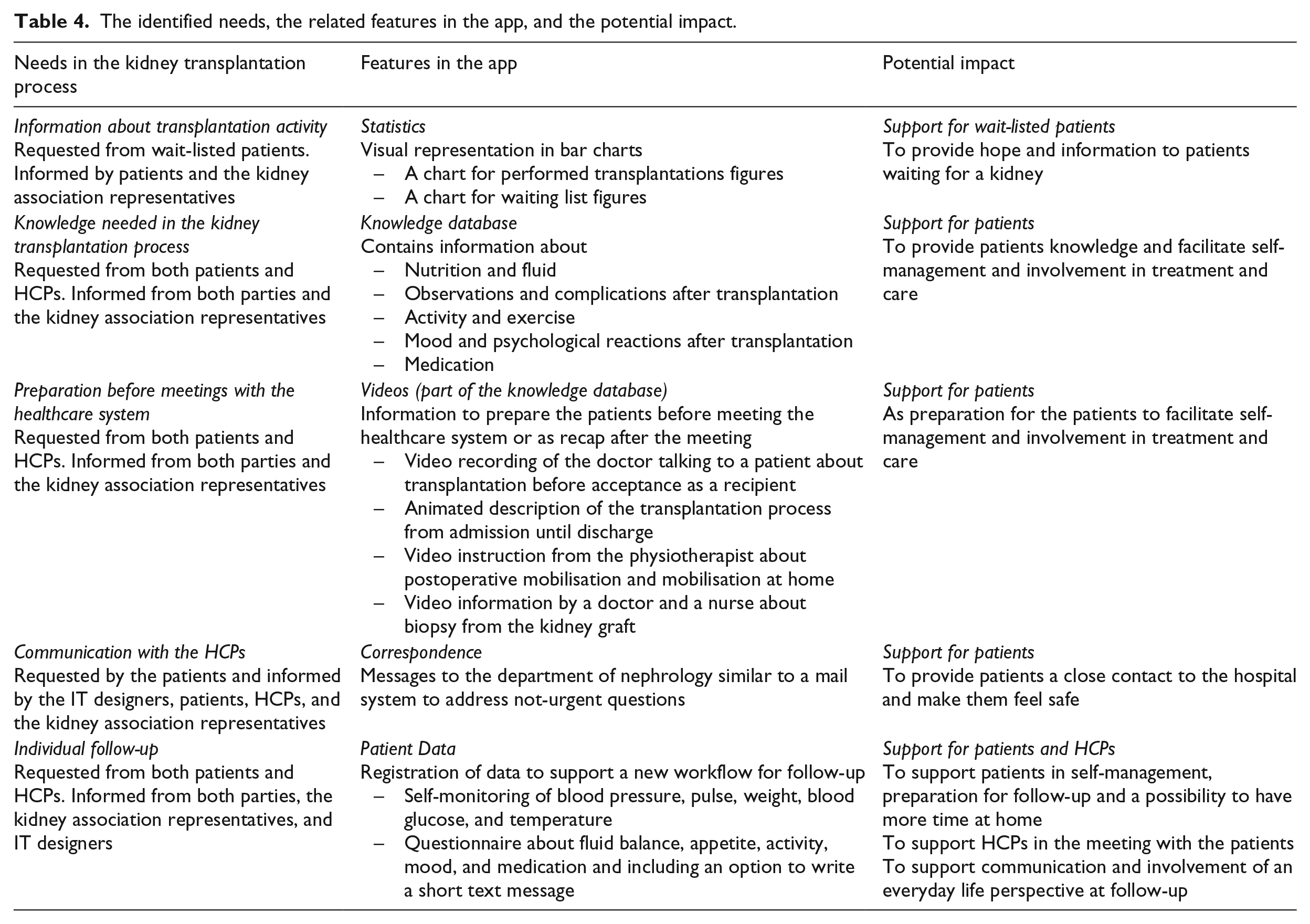
It was also decided to develop a new workflow for follow-up after transplantation. Elements for the workflow were discussed, and features to support the workflow were developed to be contained in the app. For example, a questionnaire was discussed where patients said, ‘Some departments use questionnaires that are too long, it should not be three pages’ and the doctors said, ‘It must be easy to read’. The workflow was further qualified at the following laboratory tests.
Laboratory test 1 + 2
The research team presented a workflow for follow-up, based on the results from the workshop, at the first laboratory test. However, the suggested workflow was immediately dismissed by the HCPs, as it was too complicated to implement in a clinical setting. Instead, the HCPs developed another workflow based on the same principles, to give the patients a voice in the consultations and the opportunity of having consultations by phone. The new workflow was suitable in clinical practice, as it was developed by those who were going to use it. At the final laboratory test, the workflow was adjusted and completed, so it was ready to be tested in clinical practice.
The workflow was based on the feature Patient Data in the app. Before discharge, the patients attended education at the outpatient clinic, the aim of which was to understand how to complete the patient data, and learn how to report data in the app. After discharge, the patients received a questionnaire in the app before their follow-up, and self-monitoring data could be registered on an ongoing basis. At the consultations, the doctor could open the Patient Data feature, and read the answers and measurements reported by the patients, in the electronic medical record. This would be the procedure before every consultation, regardless of whether it was at the clinic or by phone. By reporting patient data, consultations could be held in person or by phone, on an alternating basis. The new workflow had a focus on patients’ self-management through patient education and the use of patient data, as illustrated in Figure 4.
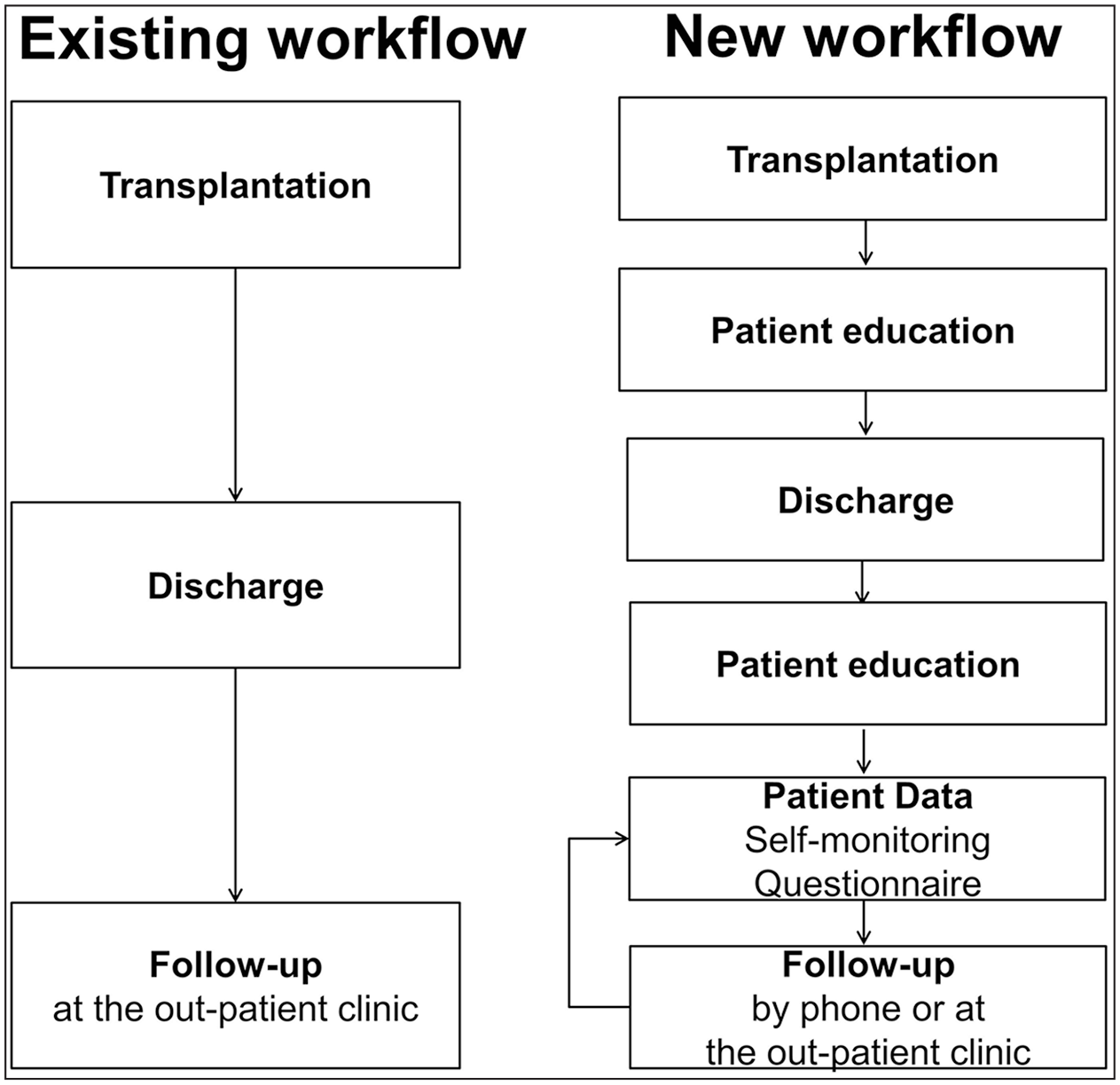
The existing workflow compared to the new workflow for follow-up after transplantation.
The aim of the Patient Data feature was to provide doctors with information they normally would get by meeting the patient face to face and also to make the patients reflect on and assess their own condition, with a view to improving their self-management at home. In addition, the patient data would also bring other perspectives to the consultations and give the patients opportunities to ask questions in advance, by using the option to write a short text message in the questionnaire. Hence, the app is both intended for patients to register data and to facilitate patient–HCP collaboration.
Discussion
An app was developed for patients going through the kidney transplantation process and a workflow for follow-up after kidney transplantation supported by a feature in the app. The development of these telehealth solutions was conducted in close collaboration with patients, their families, kidney association representatives, HCPs, and IT designers, through a PD process.
User involvement is the core of PD and has been essential to our study. Valuable mutual learning emerged between the participants throughout the development process. The significance of user involvement is emphasised by Kanstrup et al., 32 who found that, as key actors, patients can provide original designs that combine everyday living with medical technology in a way designers could not have done by themselves. This was similar in our study; the features and the content of the app and the workflow were developed in cooperation with patients and HCPs, which has been important for the usability in clinical practice. The connections between the features in the app and the needs and impact of the users are illustrated in Table 4. When a suitable workflow was developed by the HCPs after having dismissed the one suggested by the research team, the impact from the different participants was illustrated. Another example was the development of the questionnaire in the app. The HCPs suggested several issues to address; however, it was important for the patients to only have a maximum of five questions. This led to a questionnaire contained in the app that both patients and HCPs agreed on.
Recruitment of participants is important, and in research involving users as active partners, users constitute a varied group of representatives with interest in the research field, to provide a broad perspective that reaches further than one’s own. 33 In our study, user involvement was based on a broad representation of HCPs, IT designers, and the patient association. However, it was challenging to recruit patients and families, so they were represented by four patients and two family members, 2–6 months after kidney transplantation. The patients contributed with a retrospective perspective, and their distance to the time of transplantation could make it easier for them to take a broad perspective on the transplantation process. Von Hippel has investigated the value of user involvement, and he recommends ‘lead users’ as valuable participants in the innovation process. ‘Lead users’ are described as being familiar with conditions that lie in the future for most others, which means that they are valuable to the innovation process. 34 The patients and their families in our study could be seen as ‘lead users’ in the transplantation process, as they had gone through the process before they participated at the workshops. Patients from the phases before and during the transplantation were represented at the workshops by the ethnographic findings from the first phase of our study. The workshops were planned on the basis of the ethnographic findings, which were taken into account when reflecting on the outcome of the workshops. The difficulties including patients before and at the time close to transplantation highlighted that this was a challenging time for the patients. This is an example of circumstances that can make it difficult to accomplish user involvement, according to Kylberg et al. 33 In a study of elderly patients with hip fractures, Jensen et al. 35 accommodated the challenging circumstances by conducting face-to-face workshops with the patients in their homes. In our study, we accommodated this challenging situation by giving the patients a voice through the ethnographic investigations.
A PD study can be resource-demanding. User involvement was a time-consuming process, both for the participants and the research team, and it was challenging to recruit participants – both in terms of patients undergoing a course of treatment with a chronic condition and HCPs who were busy at work. However, the user involvement provided valuable knowledge and was essential for the adaptation of features in the app and the workflow to clinical practice. Furthermore, the involvement of the users from clinical practice, where the telehealth solution will be based, was important. User involvement through the PD process had a significant impact on the participants and changed the mindset of the participants. At the beginning of the study, the HCPs were against the idea of reducing the frequency of follow-ups at the outpatient clinic but ended up developing a workflow where it was possible to replace every second meeting with follow-up by phone. Likewise, the research team and the HCPs learned how patients had resources and motivation to be involved in treatment and care, and patients learned about the HCPs professional considerations. The research team needed the HCPs to learn about clinical practice. The various contributions from the participants were essential for developing a functional solution that met the users’ needs, and created potential significant impact on the users.
Our study was a single-centre study connected to a clinical setting, which raises a note of caution in regard to transferability. However, the development was based on findings from our ethnographic investigation, which echoes previous research into kidney recipients’ experiences.
Conclusion
Results from workshops and laboratory tests, based on findings from ethnographic investigations in phase 1, led to the design and development of an app for patients in the kidney transplantation process and a workflow for post-transplantation follow-up. Using a PD approach, the telehealth solution was designed in close cooperation with patients, their families, kidney association representatives, health professionals, and IT designers. Thus, the participants had significant impact on the telehealth solution, as it was based on the users’ needs and customised to the clinical setting, to improve care and treatment in the kidney transplantation process.
Implications
The testing of the telehealth solution has been conducted over approximately 1 year from May 2018, and the experiences of both patients and HCPs will be evaluated through qualitative interviews and focus groups. The outlined areas of impact that the telehealth solution potentially might provide can be used to guide the evaluation process. Based on the evaluation, it has to be decided if the telehealth solution should be fully implemented in clinical practice.
Footnotes
Acknowledgements
The authors wish to thank the patients, their families, kidney association representatives, and the HCPs who participated in the study, together with the designers from the IT company MedWare and the Health Innovation Centre of Southern Denmark. The study was approved by the Danish Data Protection Agency, journal number: 15/48886.
Declaration of conflicting interests
The author(s) declare no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclose receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the EWII Group, the Danish Kidney Association, the Region of Southern Denmark, Odense University Hospital and the Danish Council of Nurses.

