Abstract
The aim of this analysis is to investigate reasons why patients with chronic obstructive pulmonary disease decline to participate in a controlled trial of telemedicine. Patients with previous chronic obstructive pulmonary disease exacerbations were invited to participate in a 6-month randomized telemedicine trial. For eligible patients, reasons for refusal were registered. Of 560 eligible patients, 279 (50%) declined to participate in the trial, 257 (92%) reported a reason: 53 (20.6%) technical concerns, 164 (63.8%) personal reasons, 17 (6.6%) preferred outpatient clinic visits, and 23 (8.9%) did not want to participate in clinical research. Compared to consenting patients, subjects declining participation were significantly older, more often female, had higher lung function (%predicted), lower body mass index, higher admission-rate for chronic obstructive pulmonary disease in the previous year, and were more often diagnosed with osteoporosis. Many eligible patients decline participating in a controlled tele-healthcare trial and, furthermore, a tailored approach for recruiting females and elderly patients appears appropriate.
Keywords
Introduction
Telemedicine (TM) has in recent years gathered a lot of attention as a way to improve monitoring of patients’ condition. Particularly within the area of chronic obstructive pulmonary disease (COPD), home monitoring has been seen as a means to prevent hospital admissions and thus to improve the efficiency in treatment and time spent on transportation. 1 The research in this area, however, has not yet reached conclusive results; some studies have shown reductions in admission2–5 whereas others have found no effect.6–9 The effects of TM are insufficiently clarified, and currently there are more than 30 randomized studies reported to www.clinicalTrials.gov. Despite mixed evidence of effectiveness and cost-effectiveness, a political decision has been made to implement TM to patients with COPD in Denmark. Thus, recruitment issues are important to explore.
Patient recruitment to TM trials has also been reported as difficult. However, more detailed studies into the reasons for refusal of participation, bias in selection, and drop-out are lacking. 10 A recent review found a mean refusal rate of 33 per cent (4%–71%) in patients with heart failure or COPD, the most common reason for refusal being ‘not interested and/or believing TM to be unnecessary’. 10 Studies into the rates and reasons for refusal among patients with COPD – as the primary outcome of interest – have not yet been reported. However, based on the available studies the rate of non-participation seems to vary from 22 to 80 per cent.5,6,8,11–14 In future studies and implementation of TM, it is important to be aware of barriers for participation.
The aim of this study was to investigate the relationship between reasons given for refusal and the characteristics of the patients. This was done in a randomized TM trial with patients with COPD.
Materials and methods
Inclusion and exclusion criteria for the NetKOL trial (NetCOPD trial)
We recruited COPD patients with severe, but stable, COPD at high risk of exacerbations and hospital admissions.
Patients eligible for the study had to fulfil the following inclusion criteria: (1) COPD defined according to the GOLD (Global Initiative for Chronic Obstructive Lung Disease) criteria, 15 (2) post-bronchodilator FEV1 < 60 per cent of predicted value, (3) hospital admission for COPD exacerbation within the previous 36 months and/or on long-term oxygen therapy (LTOT) for at least 3 months, and (4) regular scheduled visits to the respiratory outpatient clinic. Exclusion criteria were as follows: (1) an exacerbation of COPD within 3 weeks prior to enrolment requiring a change in medical treatment; (2) planned vacation or other stay outside the catchment area, for 2 weeks or more during the study period; (3) unable to participate due to language barrier or cognitive disorders; and (4) not possible to establish a working telephone line.
Patient selection
Patients were recruited from respiratory wards from four hospitals in the area of Copenhagen. Between November 2013 and April 2014, 860 patients were identified in the hospital files and subsequently screened, with 560 patients meeting the inclusion criteria and no exclusion criteria.
All eligible patients were informed about the trial by letter. This was followed up within a week with further information given over the phone about the trial objective, procedures, required computer skills, and randomization (TM vs usual care 1:1). This information given over the phone also included an introduction to the TM equipment that would be installed in their homes. During the phone call, eligible patients were asked to attend an appointment to give their informed consent. If patients expressed interest, an informed consent form was mailed, giving them the opportunity to read it at home and to discuss participation with their relatives and/or general practitioner. In most cases, informed consent was signed at the outpatient clinic, but a small group of patients with poor mobility signed the consent during a visit to their home.
Technology and service offered in NetKOL
Patients received a tablet computer with a web camera, a microphone, and measurement equipment (spirometer, pulse oximeter, and bathroom scale). Besides, patients reported changes in dyspnoea and sputum. These observations were transferred to a call centre, which were open weekdays between 9 a.m. and 3 p.m. In case of ‘alarming’ measurements, the patient was contacted by a respiratory nurse. Patients could also contact the call centre by phone. Video consultations with spirometry were taken regularly and as needed. For further details, see Ringbaek et al. 7
Reason for refusing participation in NetKOL
Patients who declined participation were asked the reasons for refusal. The main reason for declining participation was registered by the interviewing nurse among the following four categories: (1) technical concerns (e.g. not wanting computer in their home); (2) personal concerns (e.g. not having enough energy due to illness, comorbidities, taking care of a sick spouse, lacking time, not feeling ill enough, or feeling too old to take in new technology); (3) wishing to continue usual care, that is, regular visits at the outpatient clinic (wishing to meet with clinical staff in person); and (4) not wishing to participate in a clinical trial (randomization procedure or completing several questionnaires). There are no established categories for refusal and therefore the four categories were chosen inspired by Gorst et al., 10 Mair et al., 12 and Sanders et al. 17
Some patients gave more than one reason for declining participation but were asked to state the main reason. Only the main reason was included in the present analysis.
Patients’ characteristics
From the patients’ hospital file, we retrieved information on body mass index (BMI), FEV1% predicted, The Medical Research Council (MRC) dyspnoea score (scale 1–5), 16 smoking history, and LTOT.
Data on the following were retrieved from the Danish National Registry of Patients and the Danish National Registry of Deaths: selected comorbidities (cardiovascular diseases, diabetes mellitus, skeletal/locomotion problems, neoplastic diseases, depression, or osteoporosis), hospital admissions (all-cause), and vital status during the 6-month study period.
This study has been approved by the Danish Data Protection Agency (j.nr. 2007-58-0006) and Danish Health and Medicine Authority.
Statistics
Data were analysed with the statistical package SPSS version 19.0 (IBM SPSS Statistics). The chi-square, two-sample t-tests and Mann–Whitney U tests were used, as appropriate, to compare groups of interest. Data were further analysed by logistic regression analysis, where covariates (confounders) with a p-value <0.05 in the univariate analysis were subsequently included into a multivariate analysis: gender, BMI (continuous variable), FEV1% predicted value (continuous variable), age (continuous variable), osteoporosis diagnosed (yes or no), hospital admission for COPD exacerbation in the year prior to being invited to participate in the trial (the estimated coefficients for one, two, and at least three admissions were equal and therefore analysed as yes or no). Assumption of linearity was assessed by categorizing the variable into multiple dichotomous variables of equal units on the variable’s scale. The estimated coefficients of each dichotomous variable were compared. Kaplan–Meier analysis with log-rank statistic was applied to estimate a model for time to death. A two-sided p-value of <0.05 was considered significant.
Results
Number and reasons for declining to participate
Of the 560 eligible patients, 279 (49.8%) patients declined to participate in the NetKOL trial (Figure 1). ‘Personal reasons’ were the most frequently stated reason for declining given by 164 patients (61.0%). Out of these 164 patients, 37 (22.5%) believed that weekly monitoring was unnecessary because they did not feel sick enough. In addition to ‘personal reasons’, ‘technical concerns’ were given by 55 (19.0%), ‘wishing to continue usual care at the outpatient clinic’ were given by 17 (6.3%), and ‘not wanting to participate in a clinical trial’ were given by 23 (8.6%) (Figure 1).
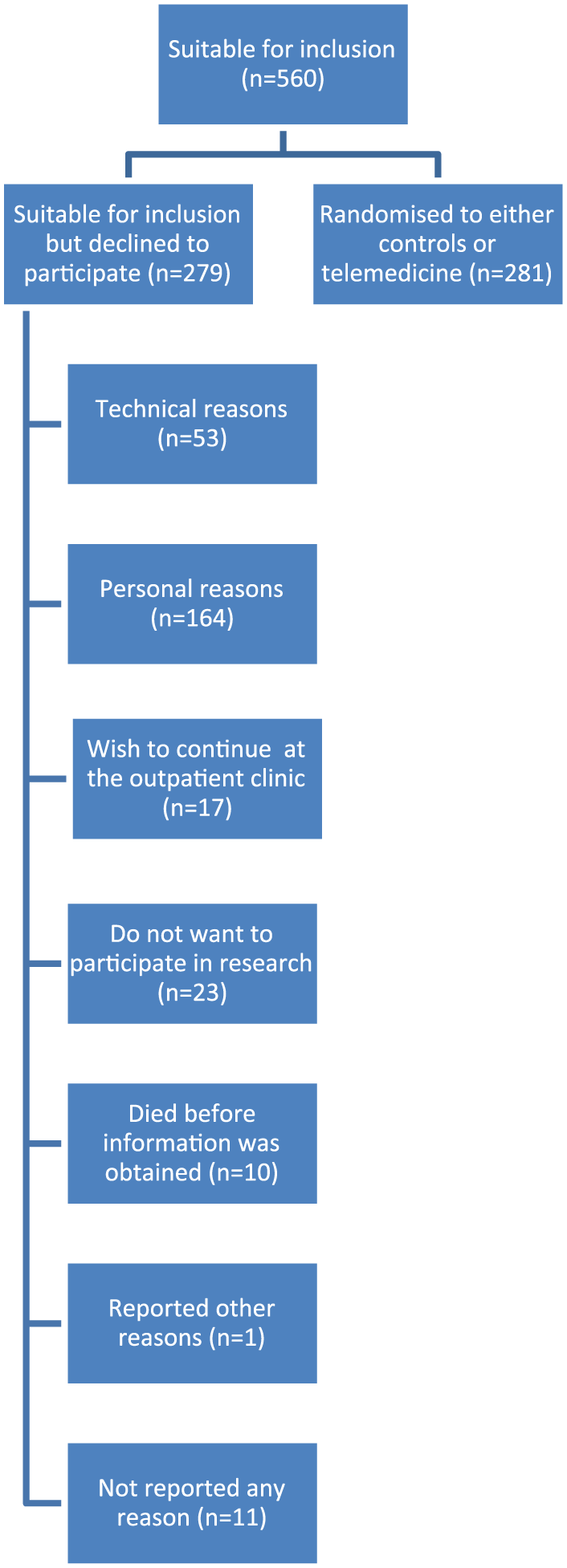
Patients’ reasons for declining participation.
Characteristics of patients who declined to participate in the NetKOL trial
Compared to patients consenting to participate, patients declining participation in the TM trial were older, more often female, had a higher FEV1%, had a lower BMI, were more frequently diagnosed with osteoporosis, and had more hospital admissions due to COPD exacerbation the year prior to the inclusion (Table 1).
Characteristics of the eligible COPD patients (n = 538) eligible for inclusion in a 6-month controlled trial of Tele Healthcare divided according to whether they wanted to participate or refuse.
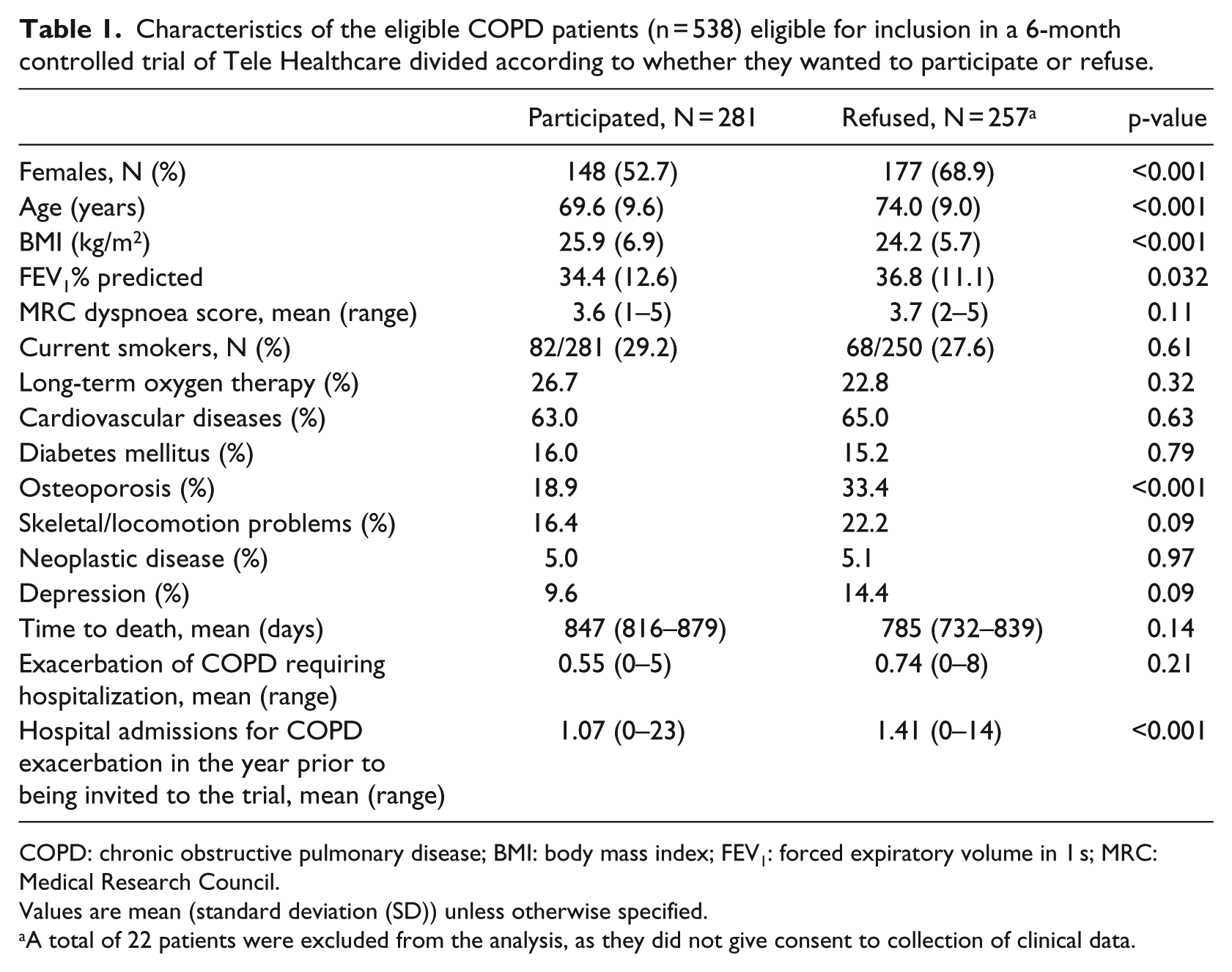
COPD: chronic obstructive pulmonary disease; BMI: body mass index; FEV1: forced expiratory volume in 1 s; MRC: Medical Research Council.
Values are mean (standard deviation (SD)) unless otherwise specified.
A total of 22 patients were excluded from the analysis, as they did not give consent to collection of clinical data.
The multivariate logistic regression analysis revealed that lower BMI was not independently associated with declining to participate in the trial. Female gender, higher age, concomitant osteoporosis, and previous hospital admission for COPD exacerbation were independently associated with declining to participate in the tele-healthcare trial (Table 2).
The likelihood (odds ratio) for patients with COPD to decline participation in a 6-month controlled tele-health trial (NetKOL).
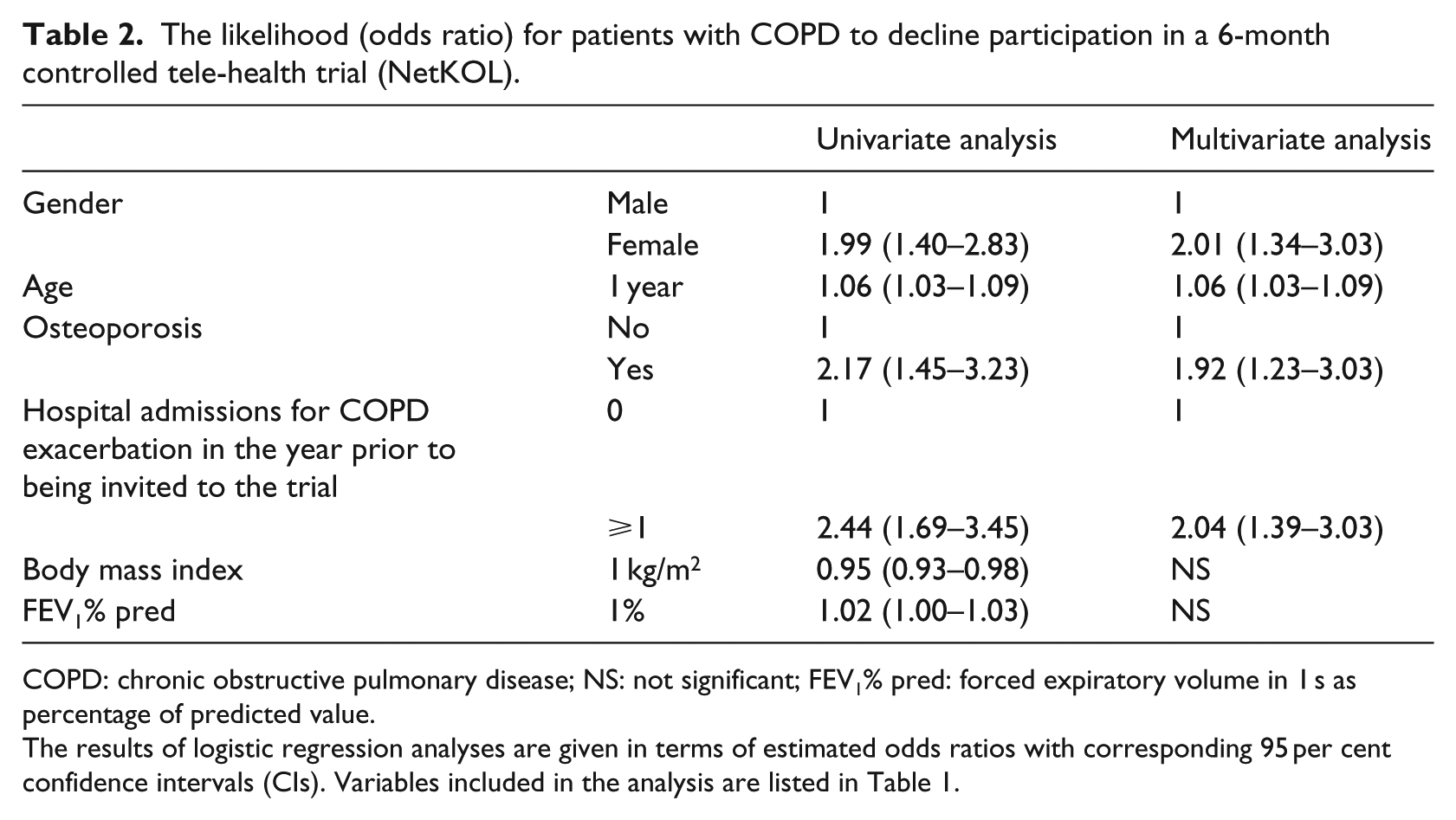
COPD: chronic obstructive pulmonary disease; NS: not significant; FEV1% pred: forced expiratory volume in 1 s as percentage of predicted value.
The results of logistic regression analyses are given in terms of estimated odds ratios with corresponding 95 per cent confidence intervals (CIs). Variables included in the analysis are listed in Table 1.
Discussion
This study shows that a significant proportion of the patients who were invited to participate in the NetKOL TM trial refused because of personal reasons and concerns regarding the use of the TM technology. More females than males refused to participate and the average age was higher.
When planning larger controlled TM trials, it should be taken into account that a large proportion of the screened patients are not likely to be eligible for the study and that the rates of refusal and drop-out are often considerable. Similar to other studies, we had to screen approximately three times as many patients as required for completing the trial5,6,8,12 and half of our eligible patients declined participation. Other similar studies have reported a refusal rate from 22 to 80 per cent with the highest rate found among patients with acute exacerbations in COPD.5,6,8,11–14 This supports our data, where the group of non-participants had higher rate of hospitalization prior to the trial than patients enrolled. COPD and osteoporosis are strongly associated because of common risk factors such as age, smoking, treatment with corticosteroids, and inactivity. Non-participants are characterized by having more severe COPD with more frequent hospitalization due to exacerbation in COPD. These patients are more inactive and often treated with corticosteroids which increase the risk of osteoporosis. They are less stable than the enrolled patients.
So far, only limited evidence has been published for the characteristics of patients declining participation in TM trials. In line with our findings, Mair et al. 12 showed that non-participants were older, but also that they more often than patients enrolled had inhaled corticosteroids prescribed. In our study, we unfortunately lack information on prescribed medication, but prior to the assessment of eligibility, patients declining participation had a higher rate of hospitalization caused by acute exacerbation of COPD. Since treatment is in accordance with the GOLD strategy document, 15 the majority of these patients are likely to be prescribed inhaled corticosteroids, thus supporting Mair et al. 12 Similar to our study, Jakobsen et al. 14 found that there were more females represented in the group of non-participants. One theory could be that females offered TM had less experience with computers and technique compared to males. Unfortunately, we do not have any data on experience with computer or Internet in the group of decliners, but among those who participated in NetKOL, 50 per cent of the males answered that they often used a computer, 24 per cent never, and 26 per cent ‘now and then’. The same figures for females were 38, 31, and 31 per cent, respectively. Supporting these data, a survey from ‘Danmarks Statistik’ in 2014 showed that among 65- to 89-year-old Danish people, 60 per cent of males and 45 per cent of females use Internet daily.
As mentioned, personal reasons were the main reason stated for declining participation in our study. Despite the fact that all eligible patients suffered from severe COPD and had had previous hospitalizations due to exacerbation in COPD, a high figure of 13 per cent of the patients did not feel sick enough to participate, and moreover, they believed that weekly monitoring was unnecessary. When their disease was in the stable phase, these patients wanted their lives to remain normal with as little focus on their COPD as possible. Similar findings have been reported from the Whole System Demonstrator, where 19 patients declined participation because they saw it as a threat to their identity. 17 They regarded telehealth as being associated with a high degree of dependency and poor health. One patient expressed, ‘I’d feel more crippled’, and another said, ‘I would be more stuck inside the house’, ‘Am I that ill – I do not need to be reminded every day of my illness. Give it to somebody who really needs it’. 17
Technical concerns are another common barrier for TM. 10 For our patients, previous experience with computers had positive impact on completing the NetKOL trial. But this is by no means prerequisite since 30 out of 35 (85.7%) patients, who had no computer experience, completed the trial (data not shown). Unfortunately, we do not have information on the level of experience with computer among patients refusing participation in TM. Another limitation of our study is that only the main reason for non-participation was included in the analysis. This might blur or simplify a more complex constellation of underlying reasons.
As half of our patients declined to participate in NetKOL, it is worth asking if better information for the patients might have increased the participation rate. Patients were initially contacted by phone. This is probably not the ideal media for describing the technical aspects of a TM trial or to raise interest for participation among patients with generally limited computer experience. The TM equipment used in this study was not shown to the patients until after the patients had consented to participation. Declining due to technological concerns is therefore most likely based on the individual patient’s expectations or presumptions. Furthermore, a quiet setting and dedicated time is essential for a successful communication of benefits of participation and for the demonstration of TM equipment. We therefore think that the participation rate might have been higher if information about the study and the actual demonstration of the equipment had been given at the same time, preferably when patient was seen at the outpatient clinic. Moreover, the health professionals did not have any special training in instructing the patient in the TM equipment and had little experience in using the equipment at the time of recruitment. We believe that in some cases, if the quality of information was not optimal, it could influence the patient’s decision to participate or not. This has not been analysed in this study and requires other studies investigating the influence and need of the skills of health professionals instructing patients in TM.
Also, exploring the patients’ personal needs and expectations will enable the health professional to better address the patients’ concerns. In our study and other randomized clinical trials with TM, individual adjustments to the technical setup have been limited, for example, the patients did not have an option to choose between different types of equipment. One could speculate that offering the patients a choice between different types of equipment or giving the staff an opportunity to tailor the TM to the patient’s needs would address some of their technical concerns.
Having said that, far from all barriers to participate due to personal concerns can be overcome by training of health professionals and thus providing higher quality of information for the patient. The patient’s decision not to participate should always be acknowledged and respected.
A minority of our patients declined participation on the ground that they wished to continue regular follow-up at the outpatient clinic. All the participants lived relatively close to the hospital, so it is possible that more patients would have preferred TM, had the distances been greater. However, whether distance and longer journeys affect participation in TM has, to our knowledge, not yet been investigated.
Factors influencing the acceptance of new technologies among elderly people (>65 years old) seem to be complex, and perhaps we as respiratory healthcare providers have to accept that we still have a long way to go in understanding the complexity of our patient’s lives and decision making.18,19 Also, we should acknowledge that not all barriers to participate in TM, technical as well as personal, should be overcome. The aim is to increase recruitment of the right patient for the right treatment, and TM is not right for all patients at all times.
Conclusion
We found that about half of the eligible patients declined to participate in a 6-month trial of TM. They were more often females and representing the group of unstable patients with frequent admission to hospital with COPD exacerbation. They most frequently stated ‘personal reasons’ and ‘technical concerns’ as the reasons for refusal.
Footnotes
Acknowledgements
The authors wish to thank the NetKOL group (Pia Andersen, Lisbeth Østergaard, Christine Lilliedahl, Torben Lage Frandsen, Jo-Ann Ramsrud Jensen, Lene Nissen, Marie-Louise Pagh Søndberg, Tanja S Hansen, and Zofia Mikolaczyk) for data collecting and for their enthusiastic collaboration throughout the study period.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
