Abstract
As the vision of mobile health (mHealth) is beginning to be realized, rigorous intervention development protocols are needed in order to draw optimal efficacy and effectiveness to support patient-centered oncology care. The purpose of the current study was to conduct a review of published articles that describe the development process of mHealth interventions for patients’ cancer care self-management. The review search yielded 11 interventions, reported by 14 manuscripts. The following trends emerged: importance of stakeholder engagement during the development process, addressing the unique needs and experiences of cancer patients and care providers, ensuring user satisfaction with the system, and identifying perceived benefits and limitations of the system. This review provides practical suggestions for mHealth intervention development. Assessments of user perceptions should be both qualitative and quantitative, and researchers should follow an established framework when developing a randomized controlled trial employing mHealth.
Introduction
Cancer patients undergoing treatment often suffer many psychological and physical adverse effects as a result of their diagnosis and treatment.1,2 Therapeutic advances have contributed to improved 5-year survival rates, resulting in a growing number of cancer survivors. 3 Despite prolonging survival, these treatments also result in both short- and long-term symptoms that negatively impact health-related quality of life.4–7 Many live with ongoing concerns about recurrence and related surveillance issues for years after initial treatment.8–10 Therefore, having effective strategies for promoting self-care is integral to the adaptive functioning of patients at all stages of the cancer continuum.11,12
Mobile health (mHealth), a broad term typically used to describe the use of mobile telecommunication technologies for the delivery of health care and in support of wellness, has received substantial enthusiasm and attention in recent years. 13 Within oncology, mHealth has the potential to provide a seamless platform for patients becoming more committed to their self-care management and cancer prevention lifestyles. For example, mobile text messaging can be used by cancer care specialists to communicate with their patients without creating a significant burden that an in-person visit or a phone call might create.14,15 Tracking of symptoms such as pain as a result of cancer treatment can easily be done via mobile phone in patients’ natural settings, as most mobile devices are convenient to carry and use, and generally would not disrupt patients’ daily lives.16,17 Despite the potential utility of mHealth applications for supporting cancer care, there are relatively few interventions in this area, compared to the emerging mHealth interventions for diabetes management,18,19 smoking cessation, 20 and physical activity adherence.21,22
Although randomized controlled trials (RCTs) utilizing mHealth for cancer patients are scarce, several initiatives are currently in development. Given the rapid advancement of mobile and wireless technologies and increasing capabilities of these devices, interventions utilizing mHealth should incorporate a number of qualitative and quantitative methods during development to ensure that users find the intervention satisfying and useful. Whittaker et al., 23 for example, described their use of a process for developing and evaluating mobile health interventions, which entails a process of intervention conceptualization, formative research (such as use of focus groups and surveys), pretesting (assessing acceptability of content), and a pilot test prior to implementation of an RCT. Although this process is not new, it is only beginning to be applied to development of interventions employing mobile technology.24,25 A model meant to inform development of behavioral intervention technologies has also recently been conceptualized by Mohr et al. 26 This model posits that use of technology for behavior change includes both theoretical (i.e. clinical aims and how these should be addressed) and concrete (i.e. specific elements and characteristics) components. Models such as these provide developers with a framework that allows them to keep in mind clinically related goals, while also keeping in mind system development needs and available resources.
Lessons can also be drawn from development of Internet (i.e. eHealth) interventions. For example, a review of methodological issues associated with web-based interventions recommended utilizing a stage of research paradigm that includes formative intervention evaluation, as adapted by the United States Department of Health and Human Services (USDHHS) Science Panel on Interactive Communications and Health. 27 Activities involved in formative evaluation may include, but are not limited to, reviewing the relevant scientific literature and developing and pretesting communication strategies and content, as well as system features and user interface. Process evaluation may also be used to more fully assess usability and user satisfaction with a developed intervention prototype prior to carrying out an RCT. A theoretical framework, such as the behavior change model developed by Ritterband et al., 28 could also be taken into consideration when developing a web- or mobile-based intervention. This model proposes that such an intervention should consider user characteristics (e.g. demographics, beliefs and attitudes associated with the disease or clinical problem of interest), the user’s environment (e.g. worksite, health-care system), web/mobile phone use, the system itself and the most optimal components to include in the system, hypothesized mechanisms of change (e.g. self-efficacy for engaging in a target behavior), and treatment/behavior maintenance.
As the vision of mHealth is beginning to be realized, we need to address the importance of rigorous intervention development protocols and identify their efficacy and effectiveness to support patient-centered oncology care. Therefore, the purpose of the current study was to conduct a review of published articles that describe the development process of interventions for cancer patients that employ mobile technology. This review focused on the descriptions of the development process and how development was carried out, specifically methods used (i.e. samples, qualitative or quantitative methods utilized) and conclusions drawn by researchers as a result of the development process. We identified some limitations and provided recommendations for development of interventions utilizing mobile technology, taking into account the unique needs and experiences of cancer patients and care providers.
Methods
Search method
A review of the literature concerning mHealth interventions was conducted using PubMed, Web of Science, PsycINFO via Ovid, ProQuest Nursing & Allied Health, CINAHL, Google Scholar, and the Cochrane Library with no restrictions placed on country or publication date. Search terms included the following: (app, apps, device, devices, intervention, interventions, management, mhealth, mobile, phone, phones, smartphone, smartphones, survivorship, telephone, OR telephones) AND cancer. Relevant articles were also found by scanning the references of found articles (backward search) and locating newer articles that included the original cited paper (forward search).
Criteria
Four main inclusion criteria were utilized: the article was written in the English language, the study focused on cancer treatment self-care management, the article focused on development of the system, and the study used mobile phone technologies for the intervention (i.e. smartphones (mobile phones with Internet capabilities), mobile phones with no Internet capabilities, personal digital assistants (PDAs), and tablet PC).
Studies were excluded if they did not focus specifically on mobile technology, were not focused on cancer treatment self-care (i.e. system should be primarily for the patient’s use), and did not focus on development of a system. Manuscripts describing the results of an RCT were excluded, as were protocol or editorial articles.
Information extraction
Information concerning intervention components, development, and study design characteristics was extracted from the articles. Information gathered for each study primarily concerned the presence and type of intervention goal and design, target population, study design and execution, and study findings, including participant demographics, intervention length, and intervention outcomes (i.e. retention rate, participant usage, satisfaction, and impact on health behavior outcomes if reported). We also noted conclusions drawn by the investigators. The two authors independently reviewed each of the identified articles to determine eligibility and extract study information. Any disagreements between the two authors resulted in a discussion that led to a consensus. Tables of study descriptions and outcomes were developed by both authors for completeness and accuracy. The authors counted various features among the manuscripts’ findings and conclusions (e.g. importance of training for system use) and, below, have summarized trends, features, and conclusions made in multiple manuscripts.
Results
Interventions included in the review
Figure 1 shows the number of studies identified, screened, and included or excluded at each stage of study selection. A total of 11 interventions, reported by 14 manuscripts, were included in the review. Table 1 describes the intervention goals and components and the population for which it was intended. Seven interventions were designed for use with a general adult cancer patient population,31,33–36,38,39 with two interventions intended for a specific cancer patient population (lung and colorectal cancers),32,37 and two interventions intended for adolescent cancer patients.40,41 All of the interventions were designed to help patients keep track of their symptoms and manage their care information, and enhance communication with their health-care team. Active components (i.e. components that required action by the patients) included in the interventions are as follows: electronic patient-reported symptom data collection (n = 11), availability of online peer community (n = 2), and self-care calendar (n = 1). Passive components (i.e. did not require action by the patients) included in the interventions are as follows: library of information and care advice (n = 9), feedback from health-care professionals (n = 5), and gamification/reward system (n = 1).
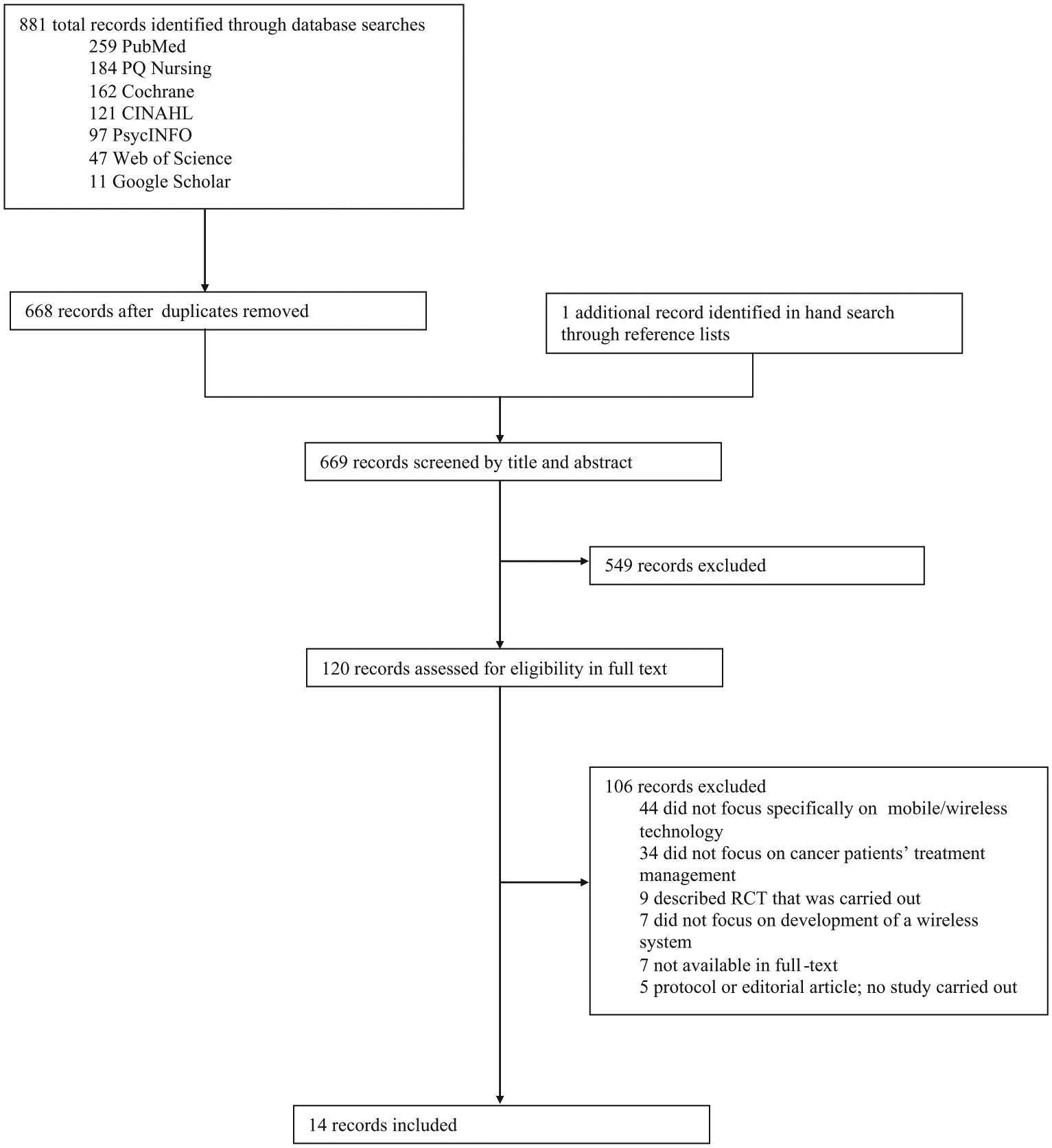
Flow diagram of the review process.
Interventions included in the review.
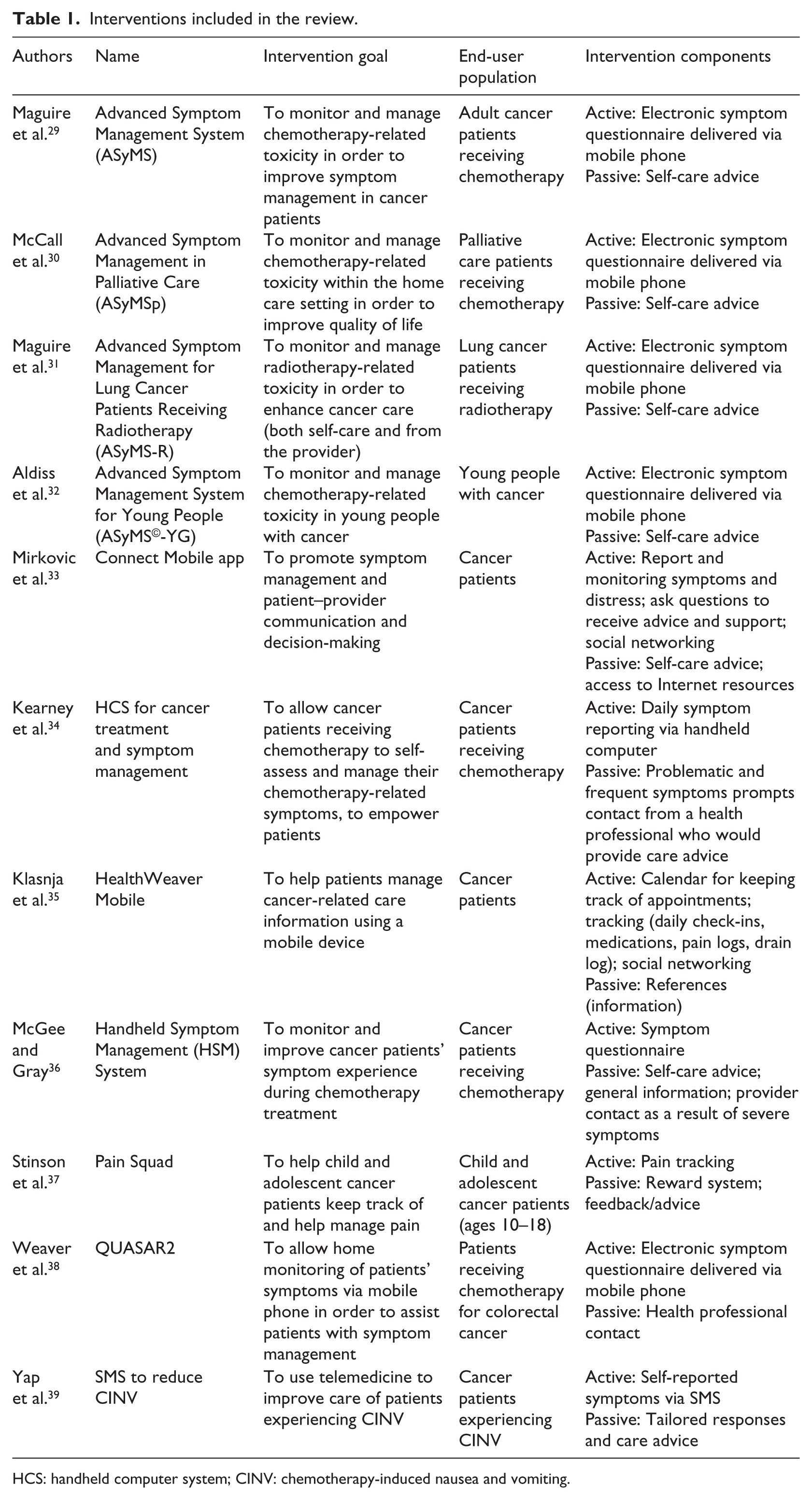
HCS: handheld computer system; CINV: chemotherapy-induced nausea and vomiting.
Design and characteristics of development studies
A total of 14 manuscripts describing the development of the 11 interventions were reviewed (see Table 2). Formative testing of the interventions was described in 1 manuscript each, except the ASyMS©-YG intervention, in which development testing was described in 3 separate manuscripts, 40 and Pain Squad, which was described in 2 manuscripts. 41
Formative testing studies and results.
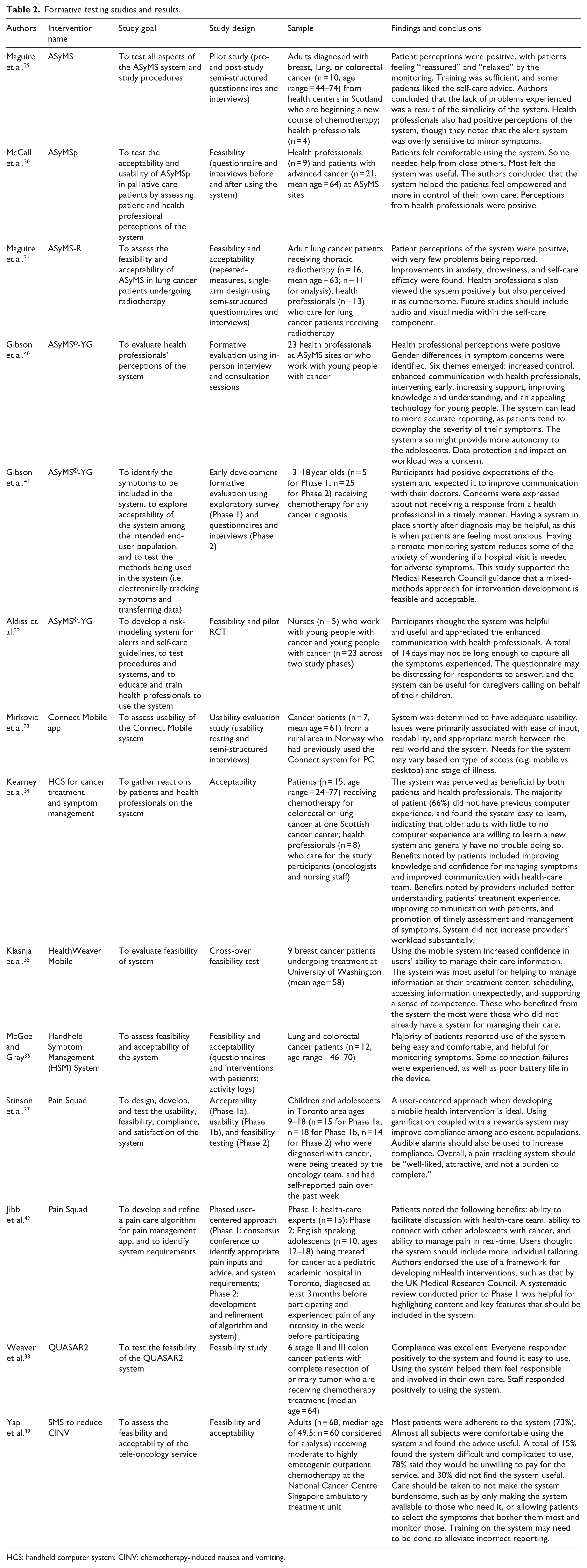
HCS: handheld computer system; CINV: chemotherapy-induced nausea and vomiting.
The studies reviewed referred to their testing procedures using the following terms: feasibility study (n = 8), acceptability study (n = 5), pilot study (n = 2), usability testing (n = 2), formative evaluation (n = 2), and phased user-centered approach (n = 1). Most of the studies used self-report and semi-structured questionnaires and interviews to identify user perceptions of the system of interest and assess technology use and demographics. Goals of the testing described varied, with some studies describing system development, 41 and others describing functioning of and acceptance and reactions toward a developed system prior to carrying out the RCT.30,35 The study sample size depended on the purpose of the testing, with an average sample size for patient samples being 17.3 (standard deviation (SD) = 15.3). Health professional perceptions were assessed in seven studies, with the average sample size being 11.5 (SD = 7.1).
Trends found in development studies
Addresses needs of cancer patients
The development studies reviewed broached the importance of systems such as these addressing the unique needs and experiences of cancer patients. Based on the studies reviewed, this most often refers to symptoms associated with cancer treatment. The ASyMS©-YG 32 intervention collected feedback from the end-user population to determine which symptoms should be included in the electronic symptom questionnaire; specifically, nausea, mouth problems, vomiting, diarrhea, weight loss, fatigue, pain (in head, back, and stomach), taste changes, and constipation. 40 Ensuring that interventions address cancer patients’ problems and concerns, such as physical symptoms experienced, can be done by involving them in the development process as stakeholders (see below). Developers of systems designed to be used by cancer patients should utilize feedback by them in order to anticipate potential barriers of use, and efforts should be made to help users overcome these barriers.
Importance of engaged stakeholders
Two studies addressed the importance of stakeholder involvement when developing the system. The ASyMS©-YG intervention 32 solicited the opinions of both health professionals who work with young adults 40 and the target end-user group (i.e. young people with cancer) 41 when developing the intervention. Developers of this intervention highlighted the need for involvement of the target end-user group, as they are the ones who are most aware of their own symptoms. 40 Developers of the Pain Squad app began their process with a consensus conference made up of clinical and technology experts. 42 Eliciting the expertise of a diverse group provides a greater probability that the system will be comprehensive. Patient caregivers could also potentially be included in the development process.30,32
Benefits of systems
Some themes emerged regarding perceived benefits of the systems, through both questionnaires and in-person interviews. Most frequently, satisfaction and perceived usefulness were assessed, with satisfaction referring to how much users like the system, and usefulness referring to ease of use, convenience, and helpfulness.
The development studies reviewed examined system satisfaction in both patient and health professional samples, as applicable. Overall, both patient and health professional respondents expressed satisfaction with the system in development in all studies where satisfaction was assessed. Satisfaction was assessed using both questionnaires30,37,41 and in-person interviews. 35 For example, 86 percent of adolescents evaluating a prototype of the Pain Squad app indicated in an evaluation questionnaire that they “very much liked it” or “liked it okay.” 37 On the other hand, investigators associated with HealthWeaver Mobile, who employed in-person interviews to gather reactions to the developed systems, inferred satisfaction and liking based on the transcribed interviews. 35
The systems designed for patients’ symptom management were overall perceived as useful and beneficial for care by both patients and health-care professionals.30,32,35,37 Measurement of usefulness varied, with some investigators utilizing self-reported questionnaires, and others utilizing in-person interviews. For example, investigators of ASyMS©-YG utilized questionnaires which users filled out and returned on their own, while investigators of the HealthWeaver Mobile system inferred perceived usefulness based on in-person interviews. 35 Several of the systems were also perceived to improve and enhance communication with health-care professionals32,34,39,42 and helped foster autonomy by providing a sense of competence and agency over one’s own care.30,31,34,35,38
Limitations of systems
Perceptions of the developed systems were overall positive. However, the studies addressed some potential limitations of the systems. First, cancer patients are largely an older population that may either not be familiar with or may not feel comfortable using the technology for which the system was developed. Developers should ensure that the system is easy to use and the technology appropriate for the intended end users.29,31,33,37,39 Training should be carried out if the system is not intuitive. For example, some errors in reporting occurred during testing of the SMS to reduce CINV (chemotherapy-induced nausea and vomiting) system, which resulted in intervention calls being erroneously made. 39 The authors reasoned that training would help alleviate this problem. Training was provided for the ASyMS pilot study, for example, which was evaluated to be helpful for using the system. 29
Two studies addressed concerns regarding creation of an additional burden on health-care providers, if providers are to be involved in use of the system.38,40 One of the ASyMS©-YG development manuscripts was the only study to discuss concerns regarding data protection and privacy. 40 Developers should ensure that participant information uploaded or input into a system is secure, and data security information should be communicated to users.
Discussion
The purpose of the current study was to review the development process used by researchers creating mobile technology interventions for cancer patients. The intent of this review was to identify gaps in the conduct and reporting of the development of interventions utilizing mobile technology, and to provide recommendations regarding the development process, taking into account the unique needs and experiences of cancer patients and care providers. The following trends emerged from our review: importance of stakeholder engagement during the development process, addressing the unique needs and experiences within cancer treatment, ensuring user satisfaction with the system and optimal usability prior to implementing an RCT, and the importance of identifying perceived benefits and limitations of the system. Overall, these interventions were perceived as beneficial for improving communication with care providers, as well as being viewed as a mechanism for fostering autonomy by providing a sense of competence and agency over patients’ own care. Any aspects to be used by care providers should not create a significant burden beyond their normal workload. Developers of mobile health systems should be mindful of concerns regarding data protection and privacy, though only one manuscript addressed this issue. We have documented the best practices for mHealth development for cancer patients in Table 3.
Best practices for development of mobile health interventions for cancer patients’ self-management.

Development testing should be inclusive in the variables chosen to be assessed. Specifically, researchers should assess compliance, confidence with use, and satisfaction with the system. Although researchers should assess what users like about the system and what they find most useful, it is also crucial to identify concerns, potential limitations, and “bugs.” Doing so may require multiple phases of testing (i.e. multiple rounds of developing and editing a draft followed by user testing). It may also be beneficial to assess user perceptions before and after using the system, such as first assessing expectations of the system prior to using it. User perceptions should be assessed using both qualitative (e.g. focus groups and interviews) and quantitative (e.g. surveys, biological data) methods. This “mixed-methods” approach is recommended for development of interventions that target health44,45 and was utilized in most of the interventions for which development manuscripts were reviewed. On the other hand, it is also important to consider uniform measures of acceptability and usability, such as by using the System Usability Scale 43 or by measuring and reporting satisfaction and perceived usefulness on a numeric scale, for ease of interpretation.
The current review showed that some researchers fail to utilize an established process for developing and evaluating mobile health interventions, such as that used by Whittaker et al. 23 There are several potential reasons for failing to put an intervention through multiple phases of development testing, such as a lack of time and resources and a strong focus in the scientific community on conducting RCTs. Some researchers only assess usability prior to the RCT, while others conduct a pilot test without any prior phases of development testing. This variation among testing procedures may partly be due to confusion among how the different types of testing are defined. The framework used by Whittaker et al., 23 for example, defines the following testing procedures: formative research to inform intervention content, pretesting to assess user acceptability, and a pilot study to test intervention procedures prior to carrying out an RCT. Usability testing (i.e. testing system functionality) may also be done during pretesting. 46 Stakeholders should be engaged throughout the entire development process. An extension to CONSORT guidelines specifically for e- and mHealth interventions has been proposed and could be applied to development testing as well as RCTs. 47 This extension includes the recommendation to attend to issues associated with computer- and Internet-related literacy, human–computer interaction, and being transparent and clear regarding the development process. It is also likely that many researchers do conduct appropriate development testing but simply do not publish their results. Information on development testing that is carried out should be made public, such as by documenting methods that were used in peer-reviewed publications.
While there is mounting enthusiasm that mHealth interventions have potential to improve patient-centered care, we found that the application of mobile technologies is very limited in the context of oncology patient care support across the cancer continuum. For example, we need to address the role of mHealth in the context of routine emergency care. Also, mobile text messages can be used to improve cancer screening rates and patient surveillance adherence. Treatment decision-making aids can be adopted as a template or mobile app to facilitate effective communication and informed decision-making between cancer patients and oncologists. Caregivers could also be targeted in these interventions. In addition to wireless symptom management applications, as found in the majority of studies in this review, cancer patients undergoing treatments would also benefit greatly from seamless connection to their personalized cancer information or other patients like them through creative applications of mobile technologies. 48 Furthermore, we also need to invest our efforts in utilizing the potential of mobile technologies for tertiary prevention, including smoking cessation, reducing alcohol intake, and increasing levels of physical activities for both cancer patients undergoing treatment and after treatment completion. To increase the likelihood of successful implementation in the real world, we should address issues of provider workflow, post-discharge quality of oncology care, and patient uptake when designing mHealth interventions.
A strength of the current review is its innovation; this is the first study of its kind to review development of interventions that utilize mobile technology for cancer patients. In addition, our search for eligible studies spanned a variety of sources and databases. However, we included published papers only, therefore introducing a publication bias. It is likely that many researchers do conduct thorough pretesting prior to carrying out RCTs, without publishing their process and results. Journals should be encouraged to publish a description of development testing that is carried out and the conclusions that can be drawn from testing. An alternative to publishing development testing procedures and findings in peer-reviewed journals would be the creation of a database or web repository for meta-data from the development process. The National Cancer Institute’s (NCI) Cancer Control PLANET, a database of resources for researchers and professionals in identifying evidence-based cancer control interventions, would be a potential example for such a repository.
A study limitation is that we did not conduct inferential quantitative analyses on available data and did not include a measure of quality scoring. Most of the data collected in the reviewed studies were qualitative and, thus, do not lend itself to typical quantitative analyses that may be done in a systematic review. In addition, the purpose and methods of the reviewed studies were so diverse that a quality scoring system could not be devised. As more mobile health RCTs for this population are developed, and if these studies follow an identical development framework, it will become possible to utilize a quality scoring system for these types of studies.
Conclusion
Mobile technology provides many opportunities for cancer patients to gain information and self-care skills on improving their communication with providers and managing their treatment. This review of development testing for mobile technology interventions for cancer patients highlighted the importance of not only the development of mHealth interventions for cancer care, but also for development of mHealth interventions more broadly. For example, the review confirmed the need for psychometrically robust measures of user and stakeholder perceptions throughout the development process. As the interventions using mobile technologies to change health behaviors and improve cancer outcomes are likely to be viable due to the growing wide access to mobile phones in the nation, system development must allow for the involvement of consumers and stakeholders to ensure an essential foundational basis for rigorous translational research in this area with a view to developing meaningful interventions that can be evaluated for effectiveness in clinical settings.49,50 Future research should consider multi-disciplinary collaborations integrating human factors engineering and user-centered design principles to develop interventions that will support cancer patients’ changing needs across the cancer continuum.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by T32CA009035 (SD) and P30CA006927 (Cancer Center Grant).
