Abstract
Introduction
Primary blast lung injury (PBLI) is the most common and fatal of all primary blast injuries. The majority of those with PBLI will require early intubation and mechanical ventilation, and thus, ventilation strategy forms a crucial part of any management plan.
Results
A PBLI ventilation strategy must strive to minimise all four of ventilator-associated lung injury (VALI), volutrauma, barotrauma and biotrauma. The three main ventilation strategies available are conventional low tidal volume (LTV) ventilation, airway pressure release ventilation (APRV) and high frequency oscillatory ventilation (HFOV). Conventional LTV ventilation together with a variable positive end-expiratory pressure (PEEP) and permissive hypercapnia has demonstrated reduced inflammation and mortality with a greater number of ventilator-free days. APRV has the potential to reduce dynamic strain, PaO2/FiO2 ratios, levels of applied mechanical power and extravascular lung water while encouraging spontaneous breathing. HFOV is able to effectively avoid VALI while curbing inflammation and histological lung injury, though not necessarily mortality.
Keywords
Background
The multi-system life-threatening injury patterns that ensue after explosions present unique management challenges to the physician. 1 These injuries can be classified into five mechanisms: primary from the overpressure wave, secondary from fragmentation, tertiary from the body’s physical displacement, quaternary from other means such as thermal and quinary from post-detonation environmental contaminants and immunosuppression-driven reactivation of latent disease.2,3 Injury patterns will differ depending on whether the explosive was high-order (such as TNT, C-4 and Semtex) or low-order (such as pipe bombs, pure petroleum bombs and gunpowder). 1 Primary blast injuries are unique to high-order explosives and the most common and fatal of all is primary blast lung injury (PBLI). 1 PBLI can be defined as an ‘acute lung injury within 12 h of blast exposure, which is not due to penetrating or blunt injury’.4,5 Overall, 76% of those with PBLI will require early intubation and mechanical ventilation; thus, ventilation strategy forms a crucial part of any management plan. 6 Though the concept of a ‘lung-protective ventilation’ strategy has been coined, discussions over how best to achieve this evolve as new technologies and considerations emerge. This narrative review aims to explore the ventilation strategies available for PBLI and provide an educational resource for clinicians managing this injury.
Methods
A literature search was conducted on the 4th May 2021 across PubMed and Google Scholar databases in order to identify articles that contribute to our current understanding of ventilation strategies in PBLI. The search terms used were as follows: ‘Primary blast lung injury’ AND (‘Low tidal volume ventilation’ OR ‘airway pressure release ventilation’ OR ‘High frequency oscillatory ventilation’). No publication date limitations were set. Titles and abstracts were screened, followed by a thorough assessment of full-text articles for inclusion. Included articles had their reference lists screened for additional articles of relevance to our aim. This enabled the identification of widely accepted and recurring theories that were collated and discussed in this narrative review, producing an overview of PBLI and the ventilation strategies that modern clinicians may wish to consider.
The blast lung
An appreciation of PBLI pathophysiology is crucial to understanding the goals of any ventilation strategy. Prevailing consensus is that the high-order explosive overpressure blast wave is dissipated within the lungs via low velocity shear and supersonic stress waves, resulting in low and high frequency responses of the torso, respectively.7–9 The overpressure blast wave itself constitutes a shock wave (extremely high pressures in surrounding air supersonically radiating away from the source) and blast wind (a body of gas discharging away from the source at high velocity). 9 The low frequency response of the torso sees inertial tearing damage of the lung parenchyma as tissues of various densities move erratically about fixed points. 9 The high frequency response of the torso sees stress waves undergoing spallation to cause diffuse implosive damage, alveolar over-distension and rupture as trapped air bubbles rapidly compress and expand.9–11 This damage is amplified as stress waves meet at the mediastinum and again as they are reflected back to form tension waves, exerting additional stretching forces. 9 This results in a combination of surface haemorrhage on tissue facing the explosion and an array of pathological air-filled spaces throughout the parenchyma, pleural space and vasculature. 9 Alveolar-venous fistulae and their resulting localised haemorrhages are of particular concern as macroscopic bleeding into airways and rapid respiratory compromise can follow. 9 Extravasated blood can also precipitate perivascular oedema as a cascade of free-radical mediated inflammatory processes is prompted, evolving over the first 24–56 h post-PBLI. 9 The size, nature of and proximity to the explosive as well as whether it was detonated in an enclosed space will all influence the incidence and severity of PBLI.6,9 Clinically, patients present with a characteristic ‘blast triad’ of bradycardia and apnoea following the immediate vago-vagal reflex but also hypotension from a reduction in cardiac output, myocardial impairment and nitric oxide (NO) release.2,12 Respiratory distress, hypoxia and ‘butterfly’ pulmonary infiltrates will classically give the blast lung syndrome diagnosis. 6 The extent of PBLI can be examined in further detail with computerised tomography. 5 These sequelae can be long-lasting and will significantly influence resuscitation. 2 Ventilation strategy must strive to minimise all four of ventilator-associated lung injury (VALI), volutrauma, barotrauma and biotrauma. 2
Conventional low tidal volume ventilation
Following a landmark study by the Acute Respiratory Distress Syndrome (ARDS) Network, a lung-protective ventilation strategy using a conventional ventilator set to volume-assist-control mode with a low tidal volume (LTV) is currently the most widely advocated.2,5,9,13 It should be noted that whilst the civilian and military incidence of post-traumatic ARDS in victims of explosions is unclear, they are believed to be a high-risk group owing to the associations with injury severity and blood products. 5 The ARDS Network study demonstrated that a reduced mortality and greater number of ventilator-free days could be achieved in patients with acute lung injury and ARDS by following this strategy. 13 These results could be a reflection of both reduced lung inflammation and a dampened systemic inflammatory response as the lungs are more protected from excessive stretch than with traditional tidal volumes.13,14 This is further supported by a greater observed reduction in levels of plasma interleukin-6 with LTV compared to traditional tidal volumes. 13 A significant fall in lung compliance is generally required for stretch-induced lung injury to occur, yet these LTV benefits appear to be independent of lung compliance. 13 To prevent oxygen toxicity, the lowest tolerable fraction of inspired oxygen (FiO2) should be used. 13 However, it should be noted that delivering a higher oxygen concentration (≥80%) for the first 24 h may have a role in dissolving gas emboli. 15 Another variable that must be adjusted in accordance with FiO2 and maintenance of oxygenation is the positive end-expiratory pressure (PEEP).5,13 PEEP may need to be increased in order to maintain adequate oxygenation, though this does oppose the logic of limiting PEEP when pneumatoceles and broncho-pleural fistulae are present in order to avoid precipitating air emboli. 9 A higher PEEP may nonetheless limit the cyclic opening and closing of minor airways and undue stress where aerated and atelectatic parenchyma meet. 16 Finally, while hypoventilation and subsequent respiratory acidosis are probable, a permissive hypercapnia should be employed when required, prioritising the prevention of pulmonary over-distention.2,5,17 This LTV approach in combination with a relatively high but variable PEEP and permissive hypercapnia has become globally recognised for its ability to minimise mechanical volutrauma and atelectasis. 18 It therefore remains the first-choice ventilation strategy for the majority of PBLI patients.2,5,9
Airway pressure release ventilation
Though a universal definition of airway pressure release ventilation (APRV) has not been devised, its utility in PBLI is promising.18,19 APRV is an alternative, pressure-controlled mode available on most conventional mechanical ventilators that aims to reduce dynamic strain by operating within a ‘safe window’, avoiding the zones of injury that would otherwise be exacerbated by the fluctuations of conventional ventilation (Figure 1).18–20 It achieves ‘open-lung’ ventilation without the need to determine optimal PEEP by applying a nearly continuous, positive airway pressure interspersed with scheduled low-pressure releases and spontaneous breaths.18–20 This improves oxygenation by maximising recruitment of lung tissue while encouraging both carbon dioxide clearance and spontaneous breathing.18,19 APRV should therefore be considered in PBLI patients when conventional LTV fails and parameters deteriorate.
18
Though they both strive for homogenous lung inflation, an advantage of APRV over high frequency oscillatory ventilation (HFOV) is the spontaneous breathing of the patient, acting to assist gas exchange, augment lung tissue recruitment, facilitate venous return and mitigate respiratory muscle wasting.
19
This may also lessen some of the classical negative cardiovascular effects of mechanical ventilation as sedative and vasoactive drug doses can be lowered, improving overall haemodynamic state.21,22 However, some argue spontaneous breathing poses additional risks of ‘patient self-inflicted lung injury’ at T-high (time spent at highest pressure) by inducing raised local transpulmonary pressures and tachypnoea.
19
Others believe that APRV should be used with restraint and that the improved oxygenation is a trade-off for increased air trapping and alveolar over-distension.
5
Predicting the cardiovascular response to APRV in a patient can prove difficult due to these complex interactions.
19
Authors have subsequently identified seven contraindications and hazards to the use of APRV.
19
Unfortunately, an overall lack of high quality human randomised controlled trials and inconsistent application of APRV has rendered this technique controversial, despite increasing in popularity in the UK.19,21 Yet, failures to consider APRV may mean PBLI patients miss out on significant mortality-related benefits such as reduced PaO2/FiO2 ratios, reduced levels of applied mechanical power (a useful index for risk of VALI) and less extravascular lung water.
18
Graphical illustration comparing the ‘safe window’ exploited by both high frequency oscillatory ventilation and airway pressure release ventilation on a pressure-volume curve, avoiding volutrauma and atelectrauma. Figure adapted from Ref. 20.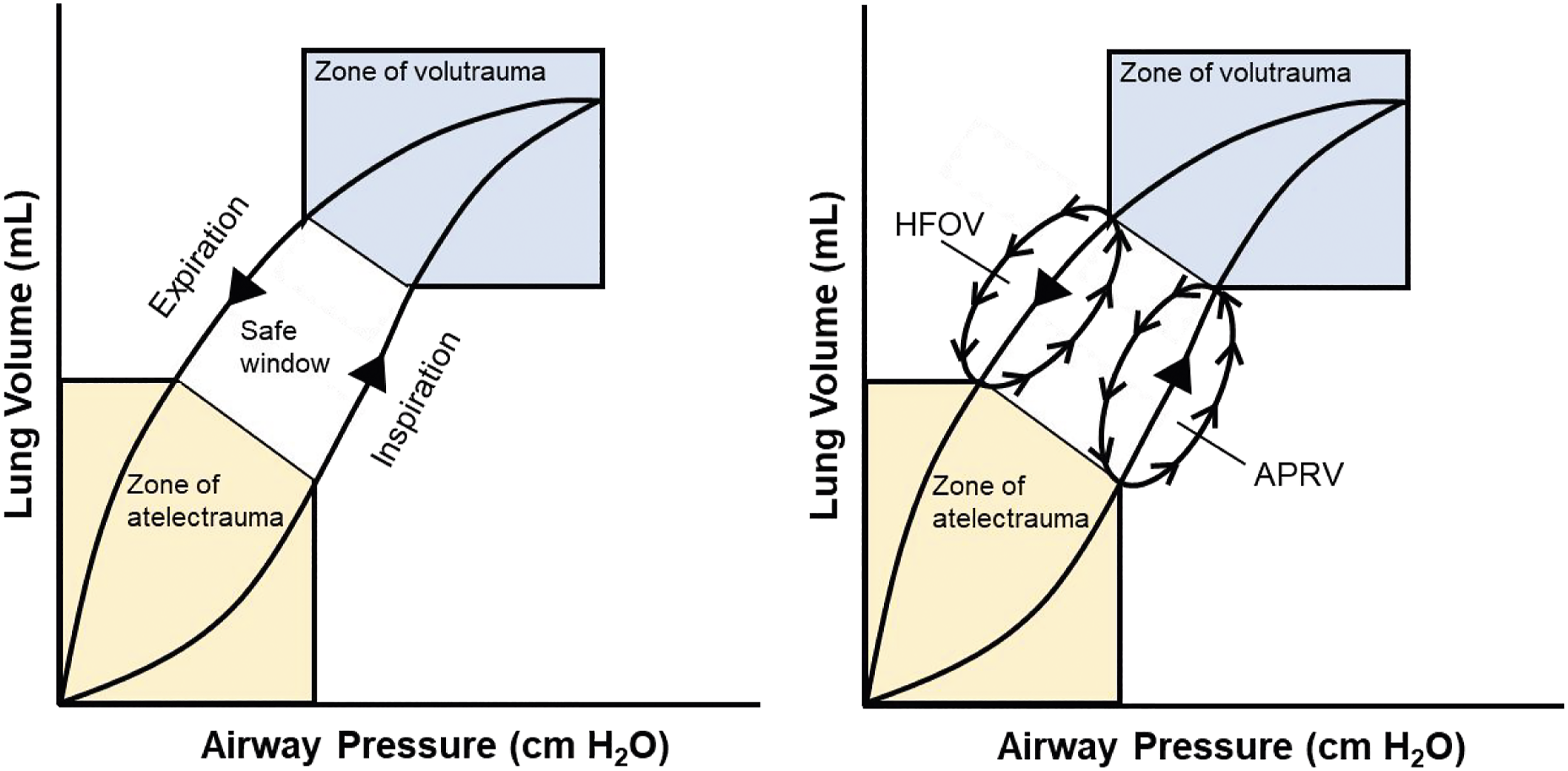
High frequency oscillatory ventilation
A more novel lung-protective strategy is high frequency oscillatory ventilation (HFOV). Though also an LTV technique, HFOV utilises a sinusoidal flow oscillation to deliver enhanced oxygenation, digressing from the traumatic ‘inflate-deflate’ cycle by providing a more constant airway pressure for alveolar inflation. 23 This mitigates VALI by again homogenously aerating the lungs within a ‘safe window’ (Figure 1).20,23 This does however require more specific types of ventilators, such as the Drager Babylog VN500, Care Fusion Oscillator 3100A or 3100B. 24 It may also lead to longer hospital stays for the patient due to a requirement for more sedation and neuromuscular blockade. 23 In practice, authors have identified 15 possible contraindications and hazards to the use of HFOV. 23 Nevertheless, a 2009 study concluded that HFOV achieves reduced inflammation, histological lung injury and mortality. 25 Therefore, when conventional ventilation fails for a minority of PBLI patients, many clinicians propose that HFOV should be indicated as a rescue strategy within four hours of refractory hypoxaemia.5,26 A permissive hypercapnia should again be used and challenges of raised CO2 can largely be managed with active cooling. 26 The efficacy of HFOV can be maintained despite regular patient transfers and when combined with continuous intracranial pressure monitoring it can even be used in traumatic brain injury. 5 Yet, its efficacy is limited in patients with diseases of increased airway resistance where it can result in air trapping, hyperinflation and barotrauma. 23 It may additionally reduce venous return and cardiac output while increasing intrathoracic pressures and risks of intraventricular haemorrhage. 23 Interestingly, a recent paper documenting the UK military experience of PBLI noted no requirement for HFOV across even the most severe cases, though the sample size was admittedly small. 4 The aforementioned 2009 study has since been superseded by the 2013 OSCAR and OSCILLATE trials, respectively.27,28 Their new evidence suggests that mortality for mechanically ventilated ARDS patients may be unaffected or even increased by HFOV compared with conventional ventilation, rendering this a controversial routine ventilation strategy.27,28 Still, HFOV may offer utility as a rescue strategy in the small proportion of cases where conventional LTV ventilation fails or approaches harmful parameters.23,25,26
Additional considerations
A central decision between conventional LTV, APRV and HFOV is necessary, yet there are some additional adjunctive components to ventilation that warrant consideration. While patients are typically ventilated in a semi-upright position, prone positioning is a well-established alternative utilised to improve oxygenation and reduce VALI. 29 Long sessions of prone positioning have been shown to substantially reduce mortality when applied early in severe ARDS and enhanced gas exchange has been noted with PBLI patients.4,29 Some believe that all ARDS patients with a PaO2/FiO2 ratio <150mmHg should undergo prone positioning as standard, before commencing alternative strategies such as APRV. 19 Proper execution of prone positioning does however require an experienced, coordinated team to overcome the technical challenges of turning critically ill patients. 29 Another adjunctive therapy to maximise the effectiveness of a strategy is NO. This vasodilatory gas acts locally within the pulmonary circulation and is able to tackle aspects of ARDS such as ventilation perfusion mismatch, arterial hypoxaemia and pulmonary hypertension. 30 Inhaled NO has therefore been advocated alongside HFOV as a method of managing early and severe hypoxaemic respiratory failure in PBLI and should be considered once PEEP levels exceed 15 cm H2O, provided there are means for delivery.5,6 Overproduction of endogenous NO can also be monitored on exhalation and may be a useful tool in both PBLI diagnosis and early disease monitoring.31,32 Lastly, extracorporeal membrane oxygenation (ECMO) has the potential to form a component of your strategy. This functions to temporarily support the cardiopulmonary system with gas exchange and perfusion in cases of refractory cardiac and/or pulmonary failure. 33 ECMO can therefore be used to supplement HFOV in severe PBLI, though it may be complicated by persistent pulmonary haemorrhage.5,33,34 A study looking at University Hospital Birmingham’s blast lung combat casualties concluded that while ECMO was not required across their case series, it should form a part of any contingency plans to counter challenges of hypercapnia. 26
Limitations
It could be argued that the narrative nature of this review lacks the methodological robustness that could be expected of a systematic review. However, this review intended to provide a broad overview of a topic area for educational purposes and it was felt that a narrative style allowed greater methodological flexibility to achieve this with more potential for individual insight.
This review also applies some evidence from non-traumatic ARDS studies to explore the viability of ventilation strategies in PBLI. Non-traumatic systemic causes such as sepsis typically result in more widespread, severe pathology than that seen in PBLI-induced ARDS. 35 Furthermore, PBLI-induced ARDS most often afflicts young otherwise healthy individuals in military conflict whereby expectant recovery may be greater simply by virtue of milder lung pathology. 4 Conclusions regarding the viability of any one strategy in this review may therefore be limited. Nevertheless, consideration of such evidence was deemed necessary owing to the difficulties associated with and distinct lack of PBLI research.
Conclusion
While invasive mechanical ventilation is a necessity for the majority of PBLI patients, the optimal strategy remains somewhat elusive. Clinicians must contend with complex pathophysiology involving haemorrhage, air-filled spaces and inflammation amid the ‘blast triad’ presentation of bradycardia, apnoea and hypotension. Ultimately, any strategy must address VALI, volutrauma, barotrauma and biotrauma. PBLI worldwide is principally managed with conventional LTV ventilation in light of the ARDS Network’s findings. This approach alongside a variable PEEP and permissive hypercapnia protects the lungs from excessive stretch and has demonstrated reduced inflammation and mortality with a greater number of ventilator-free days. Where conventional LTV ventilation fails or parameters deteriorate, clinicians may resort to APRV or HFOV as alternative rescue strategies. APRV can be achieved on most conventional ventilators with the potential to reduce dynamic strain, PaO2/FiO2 ratios, levels of applied mechanical power and extravascular lung water while encouraging spontaneous breathing. HFOV relies on more specialised equipment but is able to effectively avoid VALI while curbing inflammation and histological lung injury, though not necessarily mortality. Clinicians should additionally consider adjunctive therapies such as prone positioning, inhaled NO and ECMO that may further reduce mortality and combat more severe respiratory and/or cardiac failure. Though far from comprehensive, this review has highlighted some of the key options available to the clinician faced with ventilating the blast lung.
Footnotes
Acknowledgements
The authors thank Lt. Col. Andy Johnston, consultant in respiratory and critical care medicine, Queen Elizabeth Hospital, for commenting on a draft of this manuscript.
Author Contributions
JT conceived and designed the study, carried out literature searches, data interpretation and writing. ZA contributed to writing and editing and figure drawing. All authors read and approved the final manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Guarantor
JT.
