Abstract
Introduction:
Up to 20% of patients with traumatic brain injury (TBI) develop acute respiratory distress syndrome (ARDS), which is associated with increased odds of mortality. Guideline-based treatment for ARDS includes “lung protective” ventilation strategies, some of which are in opposition to “brain protective” strategies used for ventilation with patients with TBI. We conducted a scoping review of ventilation management strategies with clinical outcomes among patients with TBI and ARDS.
Methods:
We searched three databases (MEDLINE, Embase, Web of Science) using a systematic search strategy. We included any studies of patients with TBI and ARDS with ventilation strategies including PEEP, oxygenation, prone positioning, recruitment maneuvers, pulmonary vasodilators (e.g., nitric oxide), high frequency oscillatory ventilation (HFOV), and extracorporeal membrane oxygenation (ECMO). All clinical outcomes were included. Extracted data included details about sample (age, gender), study design, inclusion/exclusion criteria, intervention details, and outcomes.
Results:
The search returned 10,514 articles, 35 of which met final inclusion criteria. Interventions studied included ECMO (n = 13 articles), HFOV (n = 4), PEEP interventions (n = 3), prone positioning (n = 3), vasodilators (n = 4), and other lung recruitment maneuvers (n = 9). No randomized controlled trials were identified; studies were mostly case reports (n = 18/35, 51%) and series (n = 7/35, 20%), with some cohort studies (n = 5/35, 14%) and non-randomized experimental trials (n = 5/35, 14%), all at single institutions. Outcomes included physiologic changes (e.g., change in cerebrodynamics or hemodynamics with intervention) and clinical outcomes such as mortality, complications, or neurologic recovery. Five studies (14%) included pediatric patients.
Discussion:
In this scoping review of ventilatory strategies for patients with concurrent TBI and ARDS, we found variation in heterogeneity of study design, interventions, and outcomes. Studies were mostly case report/series and observational studies, seriously limiting our ability to draw conclusions about effectiveness of interventions. Targeted areas of further research are discussed.
Keywords
Introduction
Traumatic brain injury (TBI) is a significant cause of morbidity and mortality globally. 1 TBI is defined as an external force to the brain—termed the primary injury—which is thought to lead to catecholamine surges, systemic inflammation, and hemodynamic instability leading to downstream consequences, termed the secondary injury. 2 When treating TBI, focus is primarily given to preventing and treating this secondary injury.
Multi-organ dysfunction following TBI is independently linked to higher mortality rates and—in cases of severe TBI—two-thirds of patient deaths involve extracranial organ dysfunction.3,4 Pulmonary dysfunction following TBI comes in many forms, the most severe of which is acute respiratory distress syndrome (ARDS). A previous study using over 38,000 patients from the National Trauma Databank suggests about 8% of patients with isolated, severe TBI will develop ARDS, 5 though other estimates have been higher than 20%.6,7 Further, patients with TBI and ARDS have increased odds of mortality and, among those who survive their hospitalization, poorer neurological outcomes compared to TBI patients without ARDS.7,8
The mainstay of treatment for ARDS involves mechanical ventilation strategies that are “lung protective.”2,9 These strategies include limiting tidal volumes and inspiratory pressures, prone positioning, higher positive end-expiratory pressure (PEEP) values, and recruitment maneuvers (Table 1). These strategies are directed as promoting lung recruitment, avoiding atelectotrauma, and minimizing oxygen toxicity to lungs. Additionally, allowing for higher levels of CO2 (permissive hypercapnia) in patients with ARDS is often tolerated to reduce tidal volume and alveolar stretch. 10 In contrast, in TBI, a high normal PaCO2 target can promote vasoconstriction and thereby help reduce intracerebral blood volume. ARDS-based strategies such as hypercapnia can cause cerebral vasodilation, intracranial hypertension, and subsequent increase in intracranial pressure, which is typically at odds with goals of TBI therapy. 11 Frequently used TBI ventilation strategies (“brain protective”) include goals of normocapnia and normotension with the goals of avoiding hypoxia and maintaining good cerebral perfusion pressure (CPP). 11 Tight CO2 and CPP control can mean greater tidal volumes and other ventilation strategies that strain the lungs, as opposed to lung protective ventilation. 10 Other conflicting strategies include sedation (which is recommended early in ARDS, but can impede an accurate neurological exam in TBI) and fluid balance (conservative fluid resuscitation in ARDS, indeterminant data in TBI).11,12
Guideline based care for mechanical ventilation in isolated acute respiratory distress syndrome or traumatic brain injury.
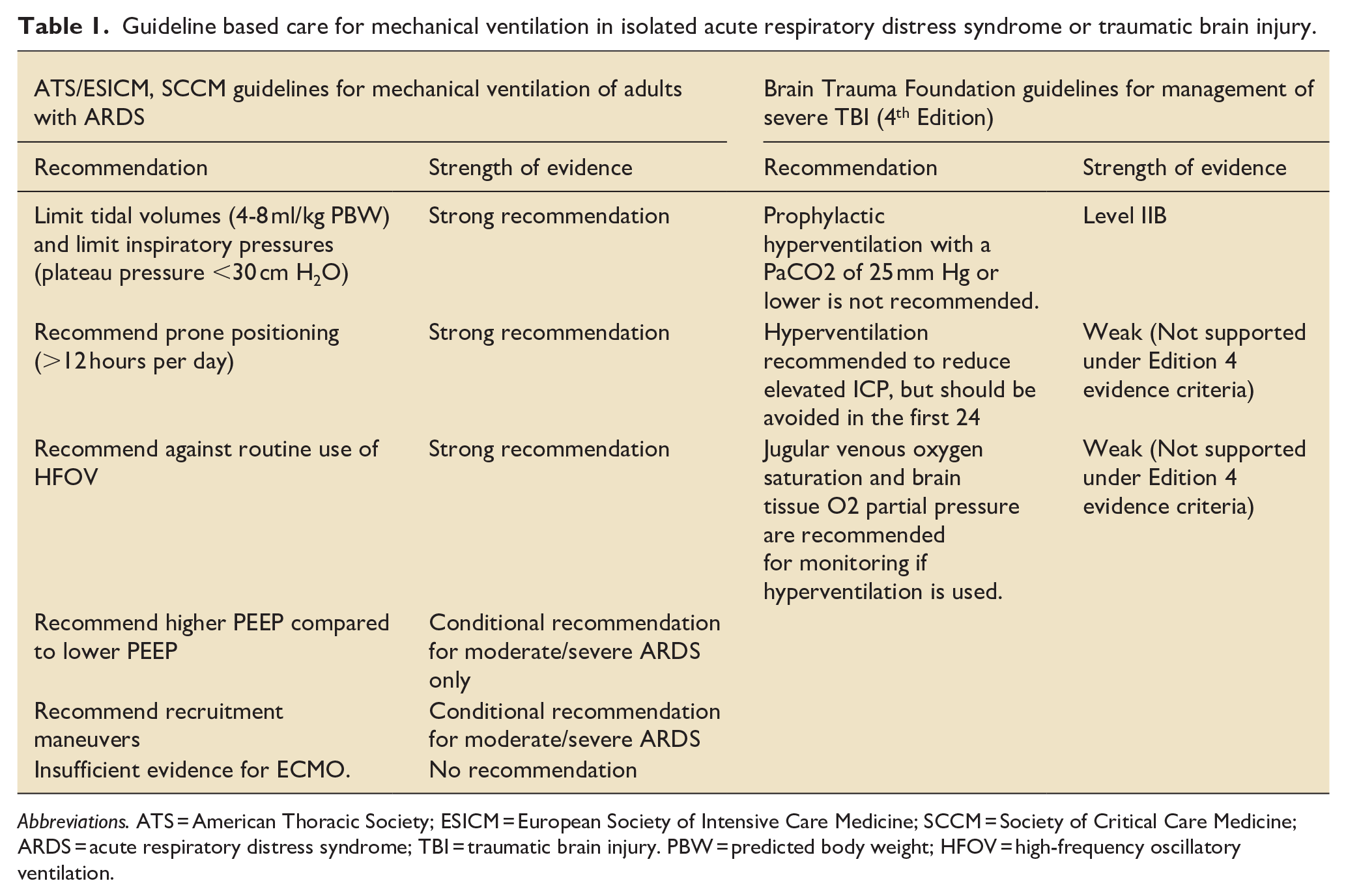
Abbreviations. ATS = American Thoracic Society; ESICM = European Society of Intensive Care Medicine; SCCM = Society of Critical Care Medicine; ARDS = acute respiratory distress syndrome; TBI = traumatic brain injury. PBW = predicted body weight; HFOV = high-frequency oscillatory ventilation.
Given the conflicting treatment recommendations for “lung protective” versus “brain protective” management strategies, patients with TBI who subsequently develop ARDS are challenging to manage safely and effectively. While several management strategies have been proposed, these strategies are limited to expert opinion and reviews without systematic search strategies.11 –14 These strategies are based comparing existing TBI and ARDS guidelines and trials—such as the ARDS Network trial and extrapolating for patients who have both TBI and ARDS.11 –15
The purpose of a scoping review is to systematically synthesize knowledge on a topic and identify gaps in the literature, particularly in areas where evidence is still emerging and there is insufficient data to conduct a systematic review or meta-analysis. 16 While systematic reviews aim to answer one question with clear clinical implications, a scoping review can help to identify the type of evidence available in an area of practice, but are limited in their ability to summarize information statistically (compared to a meta-analysis). 16 We set out to characterize the evidence on caring for patients with TBI and ARDS. Given there is not robust research on this population, we conducted a scoping review of ventilation management strategies with clinical outcomes among patients with concurrent TBI and ARDS. The aim of this review is to describe published evidence for ventilatory management of these patients and highlight gaps in the existing literature. To our knowledge, there has not been a scoping review on this topic.
Methods
We documented our review according to the Preferred Reporting Items for Systematic Reviews and Meta-Analyses standards (PRISMA) guidelines (Appendix 1). 17
Search strategy and data sources
A medical librarian with expertise in scoping reviews (S.K.) composed a search utilizing subject headings and keywords to represent the concepts of traumatic brain injury and adult respiratory distress syndrome (For search terms, see Appendix 2). The search was peer-reviewed by an additional medical librarian using a modified PRESS checklist. 18 The databases, MEDLINE via PubMed, Embase via Elsevier, and the Web of Science Core Collection via Clarivate, were searched from inception to November 7, 2022. When possible, non-human studies and conference abstracts were removed.
Selection criteria
The population of interest was patients of any age with TBI and ARDS, with plans to stratify the data by pediatric and adult populations. We included any study where the authors identified the patients having ARDS and collected their chosen definition when stated (Berlin criteria for ARDS; 1994 American European Consensus Conference definition), intentionally casting a wide net for inclusion.19,20 Similarly, we recorded reported TBI severity when included (e.g., Glasgow Coma Scale (GCS)). 21 There was no required severity of TBI for inclusion in the study.
Interventions of interest included ventilation strategies, PEEP, oxygenation, prone positioning, recruitment maneuvers, pulmonary vasodilators (e.g., nitric oxide), high frequency oscillatory ventilation (HFOV), and extracorporeal membrane oxygenation (ECMO). These interventions were generated through a preliminary literature review and expert opinion from our team. There was no comparison group specified or required.
We included any clinical outcomes including oxygenation, intracranial pressure (ICP) or cerebral perfusion pressure (CPP), mortality, and functional recovery. Any article type could be included so long as it presented original data (e.g., reviews excluded). Given the paucity of research in this area, we elected to include case reports and case series. Exclusion criteria included animal studies, non-English articles, and articles where we could not obtain the full text. Articles that included patients with brain injury (both traumatic and non-traumatic) were included only if they reported data for the subgroup with TBI separately. Articles looking at risk factors for developing ARDS after TBI were excluded.
All results were compiled in EndNote and imported into Covidence to remove duplicate and conduct screening. Each reference was screened at the title/abstract and the full-text level by two, independent screeners (M.K.H., S.L., A.L.S., A.M.B., H.J.H., or E.O.). Conflicts were resolved by a third member of the team with neurocritical care expertise (J.K. or K.C.).
Data extraction
For the included articles, two members of the research team independently extracted information from each article using the Covidence extraction form, with discussion to reach consensus for conflicts. Data collected included age of patients (adults or pediatrics; descriptive statistics of age of sample), country where study was conducted, study design (case report, case series, cohort study, case control, non-randomized experimental study, etc.), site information (single versus multi-institutional site), gender of sample, inclusion/exclusion criteria, total number of participants, definition of TBI (e.g., Glasgow Coma Scale ⩽ 8 21 ), definition of ARDS (2012 Berlin definition 19 or American-European Consensus Conference 20 ), other characteristics of sample (e.g., Injury Severity Score), intervention, primary and secondary outcomes and associated results, and comparison group if present (Appendix 2). This list of extraction variables was generated iteratively by the research team. Adults were considered those greater than 14 years-old, consistent with the trauma literature. The data extraction form was built in Covidence by the research team and downloaded as an Excel file after consensus was reached. We also reviewed the references of included articles for studies that may be missing from our search.
Approach to summarizing data
Article were summarized in tables. As there were multiple study types included in this review, there was no meta-analysis conducted. Due to the scarcity of studies with pediatric patients only, they were included in the final summary tables with the adult patients.
Results
Study selection
Our search identified 10,514 articles, 94 of which were screened at the full-text level, and a final 31 of which met final inclusion criteria (Figure 1). Articles that included patients with ARDS and multiple types of brain injury (e.g., TBI, subarachnoid hemorrhage, hemorrhagic stroke) and did not stratify their data by brain injury type were excluded.10,22 –26 We also excluded a case report of a patient with TBI, ARDS, and acute kidney injury as the intervention described was related to using citrate anticoagulation for dialysis, and not a ventilation strategy. 27 We reviewed the references of the 31 included articles and identified 4 additional studies to include, bringing the final sample to 35 studies.28 –31
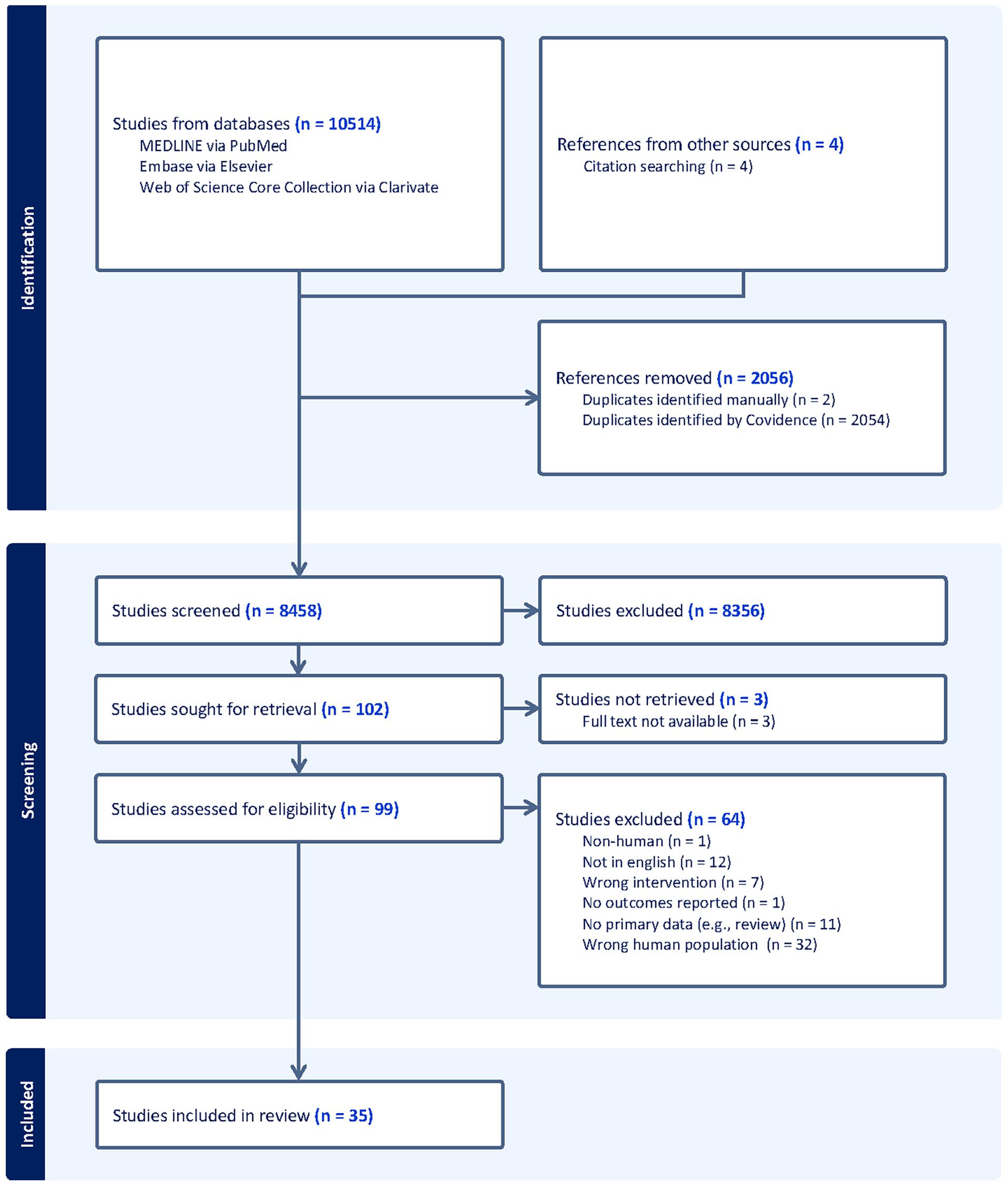
PRISMA article inclusion flowchart.
Study characteristics
Of the 35 articles, interventions studied included ECMO (n = 13 articles), HFOV (n = 4), PEEP interventions (n = 3), prone positioning (n = 3), vasodilators (n = 4, one of which also looked at prone positioning), and other lung recruitment maneuvers (n = 9) (Table 2). All studies included were reports from single institutions and most focused on adults; five articles included pediatric patients.32 –35
Data extracted from 35 studies looking at clinical outcomes for patients with ARDS and TBI with ventilatory interventions.
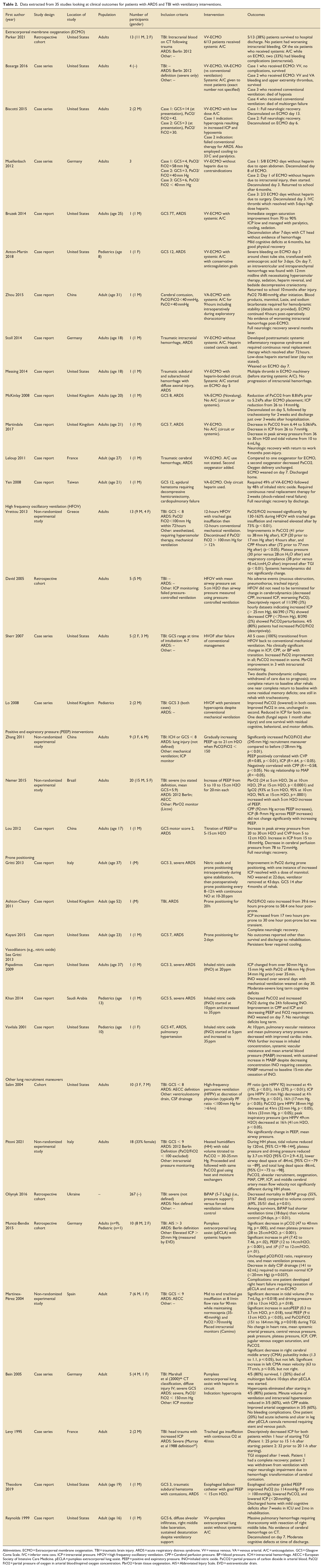
Abbreviations. ECMO = Extracorporeal membrane oxygenation. TBI = traumatic brain injury. ARDS = acute respiratory distress syndrome. VV = venous venous. VA = venous arterial. A/C = anticoagulation. GCS = Glasgow Coma Scale. IVC = inferior vena cava. ICP = intracranial pressure. HFOV = high frequency oscillatory ventilation. CPP = Cerebral perfusion pressure. BP = blood pressure. ICH = intracranial hemorrhage. AECC = European Society of Intensive Care Medicine. pECLA = pumpless extracorporeal lung assist. PEEP = positive end expiratory pressure. INO=inhaled nitric oxide. PaCO2 = partial pressure of carbon dioxide in arterial blood. PaO2/FiO2 = partial pressure of oxygen in arterial blood/inspired oxygen concentration. PbrO2 = brain tissue oxygenation. AIS = Abbreviated Injury Scale. EVD = extraventicular drain.
ECMO
We found one retrospective cohort study, three case series, and nine case reports of use of ECMO in patients with TBI and concomitant ARDS.31,32,36 –46 Generally articles reported use of venous-venous ECMO (VV-ECMO), though there were four cases using venous-arterial ECMO (VA-ECMO).31,37,41,43 The retrospective cohort study of 13 adults who received VV-ECMO only reported descriptive outcomes which included survival (38% (5/13) patients survived) and bleeding complications (15% (2/13) patients). In terms of complications for all included ECMO articles, three patients developed thrombi; two of these patients were not given anti-coagulation, and for the third anticoagulation use was not specified.37,39,42 Three patients developed bleeding complications.32,36,37 There was one report of a patient who developed posttraumatic systemic inflammatory response after starting VV-ECMO that required continuous renal replacement therapy (CRRT) for 7 days. 42 Another case also required CRRT for two weeks after VA-ECMO due to shock related renal failure. 31
HFOV
We found one retrospective cohort study, one non-randomized experimental study, and two case series reporting on use of HFOV in patients with concomitant TBI and ARDS.35,47 –49 Vrettou et al prospectively collected physiologic data during HFOV and conventional mechanical ventilation and found that PaO2/FiO2 increased significantly during HFOV use, which was sustained after returning to conventional ventilation. 47 David et al retrospectively reviewed patients who received HFOV and descriptively reported PaO2/FiO2 increased in four of five of the patients, with infrequent occurrences of increased ICP, decreased CPP, and PaCO2 changes that did not require termination of HFOV. 48 The remainder of the articles reported either no clinically significant changes in ICP or decreased ICP following initiation of HFOV.35,47,49 Several deaths were reported that were not attributed to HFOV.
PEEP interventions
Two non-randomized experimental studies and one case report described PEEP interventions, all of which involved up titrating PEEP.50 –52 Both Nemer et al and Zhang et al (non-randomized experimental studies) found that oxygenation improved with increasing PEEP.50,51 ICP was found to be positively correlated with PEEP in one study, 50 descriptively increased in a case report, 52 and not significantly changed with increasing PEEP in the third. 51
Prone positioning
Three case reports described use of prone positioning in patients with TBI and ARDS.53 –55 Two of the case reports described improved oxygenation with prone positioning, with associated increases in ICP, one of which required treatment with mannitol.53,54 The third case study did not provide outcomes beyond survival. 55
Vasodilators
Four case reports described use of inhaled nitric oxide (range of dosing: 5-35 ppm).30,33,53,56 The cases described by Papadimos et al and Khan et al both descriptively report improvements in ICP and oxygenation following initiation of inhaled nitric oxide, in an adult and pediatric patient respectively.33,56 Gritti et al mentioned that use of inhaled nitric oxide of lab values without further elaboration (case report was focused on use of prone positioning). 53 Vavilala et al describe the use of inhaled nitric oxide in a pediatric patient with TBI, ARDS, and pulmonary hypertension which initially resulted in improvement in pulmonary vascular resistance, mean pulmonary artery pressure, and cardiac index, but increasing the concentration of inhaled nitric oxide resulted in increased systemic vascular resistance and mean arterial blood pressure requiring cessation of therapy, which resolved the hemodynamic disturbances. 30
Other lung recruitment maneuvers
Several other articles were included that describe other lung recruitment maneuvers.28,29,34,57 –62 Salim et al describe a cohort of 10 patients who received high-frequency percussive ventilation which significantly improved oxygenation status but also increased ICP. 57 Pitoni et al reported use of heated humidifiers in a non-randomized experimental trial of 18 patients and found heated humidifier use reduced required tidal volume and dead space without increasing ICP. 58 Oliynyk et al looked at corticosteroid usage for patients with severe TBI, sepsis and ARDS using a retrospective cohort study; however, in addition to comparing those exposed to corticosteroids and those not, they also compared two ventilation strategies: BiPAP (i.e., pressure support) and forced ventilation volume control and descriptively found decreased mortality in patients who received pressure support (in the groups who did not receive corticosteroids). 59 Three articles reported use of pumpless extracorporeal lung assist (pECLA). Munoz-Bendix et al reported significantly improved hypercapnia, but one of ten patients in the retrospective cohort study developed right heart failure necessitating VV-ECMO; Bein et al also reported improved hypercapnia in four of five patients in their case series, with one patient developing ischemia and an ulcer related to removal of the pECLA cannula, and one death attributed to multiorgan failure.34,61 Reynolds et al reported a case of VV-pECLA in a 16 year old who survived, but had massive pulmonary hemorrhage necessitating surgery. 29 Martínex-Pérez et al prospectively looked at the use of tracheal gas insufflation in seven patients and found a significant increase in PaO2/FiO2 without change in cerebral oxygenation or pressure after initiation. 60 Levy et al reported two cases of patients with increased ICP which dropped to normal within one hour of starting tracheal gas insufflation. 29 Finally, Theodore et al described a case of the use of an esophageal balloon catheter to guide PEEP titration and descriptively found improved oxygenation and decreased ICP. 62
Discussion
We conducted a scoping review of clinical outcomes associated with ventilatory strategies (ECMO, HFOV, PEEP interventions, prone positioning, vasodilators, and other lung recruitment maneuvers) of patients with concurrent TBI and ARDS. Overall, there was a very weak evidence base (few studies, mostly limited to cohort studies, and case reports and series) for each of the interventions with varying outcomes and lack of control groups, limiting our ability to compare studies or draw conclusions about effectiveness. Outcomes varied and included both physiologic changes (e.g., change in cerebrodynamics or hemodynamics with intervention) and clinical outcomes such as mortality, complications, or neurologic recovery. Studies were all conducted at single institutions, most were with adult populations, and none were randomized controlled trials.
ECMO (primarily venous-venous) was the most studied intervention with reports of survival and some neurologic recovery following its use, though there was only one cohort study which lacked a control group; the remainder were case series or case reports. Both bleeding and thrombotic-related complications were reported amalgamated across the reports; there was inconsistent use of systemic anticoagulation reported.
Two tentatively promising areas of further investigation were use of HFOV and inhaled nitric oxide. HFOV was studied in four studies with reports for some patients of sustained improvements in PaO2/FiO2 with reports of both improved and unchanged ICP compared to prior to starting HFOV. HFOV—as the name suggests—uses high respiratory rate and low tidal volumes while holding mean airway pressure constant, preventing alveoli from undergoing the expansion/collapse cycle which can cause trauma. 63 Evidence is generally equivocal for HFOV use in adult patients with ARDS compared to conventional ventilation. The ATS/ESICM/SCCM guidelines recommend against the routine use of HFOV for patients with ARDS, though they recognize there is inconclusive evidence on HFOV’s role as a rescue therapy (e.g., for those who fail conventional ventilation).9,64,65 To this point, in our review most studies on HFOV reported its use in patients who failed conventional management. In addition, three case studies reported successful use of nitric oxide to decrease intracranial pressure. Nitric oxide is a known as a vasodilator, but there is also speculation based on animal models that it could provide some anti-inflammatory effects in the acute period.56,66 While tentatively promising, case studies are one of the lowest levels of the evidence pyramid and use of NO needs further study.
Overall, the studies included in this review are limited by study design, included populations, and outcomes. Most studies were retrospective and prone to selection bias due to study design, especially the case reports and series. Outcomes were often reported descriptively, without statistical tests of comparison which severely limits our ability to draw conclusions about effectiveness or superiority of interventions. Finally, some studies only included patients who failed conventional management most likely due to the experimental nature of the interventions; this limits generalizability to all hospitalized patients with ARDS and TBI. Given the landscape of evidence in this area, it is difficult to make evidence-based recommendations for clinical care of patients with ARDS and TBI, but clinicians can at least be aware this area of practice does not have a robust evidence-base and continues to be guided based on biological and physiological rationale, extrapolated data from other disease states, individual guidelines from ARDS and TBI care, and local practice and culture. Finally, few studies included pediatric patients; little is known about how to best care for pediatric patients with concurrent ARDS and TBI. 67
There are several limitations to our review process to note. We included all study designs and outcomes in our review, including case studies, as we suspected evidence for each intervention in this population would be limited. While this allowed us to broadly describe the landscape of evidence, it limited our ability to perform any meta-analysis across these study types and outcomes. As a result, our review cannot draw conclusions about the efficacy of studied interventions. We recognize our inability to perform a meta-analysis is a major limitation; a future meta-analysis would be useful to the field as the research surrounding care for TBI and ARDS patients becomes more robust with standardized interventions and outcomes. As included studies were at high risk of bias, particularly case studies and series which fall prey to selection bias, our speculations about promising interventions could tend towards overstating benefits. We also had broad inclusion criteria for the study populations. We did not standardize our definitions of TBI and ARDS, and included populations of all ages, illness severity, and both isolated TBI and polytrauma patients to broadly describe the existing evidence, though this also limited our ability to compare across studies.
In this scoping review of ventilatory strategies for patients with concurrent TBI and ARDS, we found a heterogeneous group of studies with high risk of bias due to study design. The heterogeneity of studies limits our ability to draw conclusions about efficacy of studied interventions which included ECMO, HFOV, PEEP interventions, prone positioning, vasodilators, and other lung recruitment maneuvers. This review highlights the limited data that exists in ventilatory strategies for patients with ARDS and TBI. Future research should focus on robust, prospective studies with patient-centered outcomes (e.g., neurologic and functional recovery) including randomized controlled trials, and may consider looking at subgroups of this heterogeneous patient population of TBI and ARDS (e.g., isolated versus polytrauma, age, severity, comorbidities). Prospective studies with comparison groups could help to establish clear, evidence-based guidelines on caring for patients with ARDS and TBI.
Supplemental Material
sj-docx-1-inc-10.1177_17511437241311398 – Supplemental material for Management of traumatic brain injury and acute respiratory distress syndrome—What evidence exists? A scoping review
Supplemental material, sj-docx-1-inc-10.1177_17511437241311398 for Management of traumatic brain injury and acute respiratory distress syndrome—What evidence exists? A scoping review by Margot Kelly-Hedrick, Sunny Liu, Jordan Hatfield, Alexandria L. Soto, Alyssa M. Bartlett, Helen J. Heo, Ellen O’Callaghan, Evangeline Arulraja, Samantha Kaplan, Tetsu Ohnuma, Vijay Krishnamoorthy, Katherine Colton and Jordan Komisarow in Journal of the Intensive Care Society
Supplemental Material
sj-docx-2-inc-10.1177_17511437241311398 – Supplemental material for Management of traumatic brain injury and acute respiratory distress syndrome—What evidence exists? A scoping review
Supplemental material, sj-docx-2-inc-10.1177_17511437241311398 for Management of traumatic brain injury and acute respiratory distress syndrome—What evidence exists? A scoping review by Margot Kelly-Hedrick, Sunny Liu, Jordan Hatfield, Alexandria L. Soto, Alyssa M. Bartlett, Helen J. Heo, Ellen O’Callaghan, Evangeline Arulraja, Samantha Kaplan, Tetsu Ohnuma, Vijay Krishnamoorthy, Katherine Colton and Jordan Komisarow in Journal of the Intensive Care Society
Footnotes
Acknowledgements
None.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: R01NS130832 (Krishnamoorthy)
Prior abstract publication/presentation
None.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
