Abstract
Background and aims:
Androgen deprivation therapy (ADT), either by surgical or chemical castration, is the standard treatment for metastatic prostate cancer (PCa). ADT induces osteoporosis and increases the risk of osteoporotic fractures, such as hip fractures. However, the effect of castration on overall survival (OS) after hip fracture is unclear. The aim of this study was to evaluate the association between ADT and OS among PCa patients with surgically treated low-energy hip fractures.
Methods:
Male patients with surgically treated low-energy hip fractures (ICD-10 codes S72.0, S72.1, and S72.2) were retrospectively identified from the databases of Pirkanmaa Hospital District during the years 2002–2018. Patients with pathological hip fractures were excluded from the analysis. Patients with prior PCa were identified (n = 323) and categorized into three groups according to therapy that affects testosterone levels: no hormonal therapy (n = 94), ADT (n = 212), and nonsteroidal antiandrogen (NSAA) (n = 17) at the time of the fracture. The primary outcome was OS after hip fracture surgery. Postoperative OS was analyzed using the Kaplan–Meier method, and the difference in OS between groups was analyzed using Cox regression in univariable, age-adjusted, and multivariable-adjusted models.
Results:
Kaplan–Meier survival analysis suggested that patients under ADT had shorter OS after hip fracture surgery (p = 0.006). After adjustment for potential confounders in Cox regression models, no clear association was observed between ADT (hazard ratio (HR) 1.19, 95% confidence interval (CI) 0.90–1.58) or NSAA (HR 1.02 (0.59–1.78)) and OS when compared to patients with PCa not undergoing hormonal therapy. Higher age and comorbidities, rather than ADT, appeared to be associated with poorer OS.
Conclusions:
In the cohort of patients with PCa, who underwent surgery for low-energy hip fracture, ADT or NSAA utilization was not associated with OS. Wide CIs allow for both modest benefit and potential harm, and the findings should therefore be considered inconclusive.
Context and Relevance
Androgen deprivation therapy (ADT) induces osteoporosis and increases the risk of osteoporotic fractures, such as hip fractures. However, the effect of ADT on overall survival (OS) after hip fracture is unclear. The aim of this study was to evaluate the association between ADT and OS among prostate cancer (PCa) patients with surgically treated low-energy hip fractures. In our retrospective cohort study of 323 PCa patients with surgically treated low-energy hip fractures, there was no association between ADT and OS. Our study suggests that poorer OS after hip fracture surgery may be more closely associated with comorbidities and advanced age rather than ADT utilization.
Introduction
Hip fractures are associated with high mortality, morbidity, and prolonged hospitalization.1,2 Survival rates after hip fracture are lower in men than in women, even though men are generally younger at the time of fracture. 3 In addition, men appear more likely to suffer from medical complications following a hip fracture.4,5 The role of androgens influencing mortality among men and whether the level of sex hormones explains the gender differences remains unclear. Despite extensive research on factors affecting mortality following hip fractures, the impact of androgens on mortality remains uncertain.
Prostate cancer (PCa) is the most common malignancy and the second leading cause of cancer death among Finnish men, with the highest incidence in those over 70 years. 6 At diagnosis, about 82% of patients with PCa have localized or regional disease, with a nearly 100% five-year survival rate, whereas metastatic PCa has a five-year survival of 36.6%. 7 Androgen deprivation therapy (ADT) is the standard treatment for metastatic PCa. 8 Despite guidelines, studies show that nearly half of patients with low-risk localized PCa have received ADT as primary therapy.9–11 ADT includes both surgical castration and chemical castration with luteinizing hormone-release hormone (LHRH) agonists/antagonists. 8 Both treatments effectively decrease testosterone to castrate levels (testosterone below 50 ng/dL). 12 In advanced but nonmetastatic PCa, nonsteroidal antiandrogen (NSAA) as monotherapy presents an alternative therapeutic approach to inhibit cancer growth while maintaining, or even raising testosterone levels. 13 ADT is known to cause osteoporosis, and thereby increasing the risk of osteoporotic hip fractures, which are associated with higher mortality rates.14–16 In addition, ADT is associated with overall morbidity, including sarcopenia and cardiovascular events, and may therefore contribute to overall survival (OS)17,18 Given this, it is crucial to determine whether ADT also adversely affects postfracture prognosis.
The aim of this study was to assess the impact of ADT on OS after hip fracture surgery. Our primary hypothesis posited a correlation between ADT and poorer OS compared to PCa patients not undergoing hormonal therapy at the time of fracture. To test this hypothesis, we examined the influence of ADT on OS after surgically treating hip fractures in patients with PCa. In addition, we segregated patients with PCa receiving NSAA as monotherapy into a distinct group, as NSAA does not inhibit testosterone production.
Methods
The present study cohort included all male hip fractures (defined by ICD-10 codes S72.0-S72.2) diagnosed in the Pirkanmaa Hospital District from 2002 to 2018. The cohort was retrospectively collected through a computer-based search of electronic medical record databases (MRDs). Patient records were subsequently manually reviewed. Patients with PCa who underwent surgery for low-energy hip fractures during the research period were included in the final study cohort. Low-energy fracture was defined as falling from a standing height or less.
We excluded 14 PCa cases where the hip fracture was due to a malignant bone lesion in the proximal femur, identified clinically or, when available, by histology. These pathological fractures were omitted, as they are known to carry significantly higher mortality than osteoporotic fractures. 19 In addition, we excluded hip fracture patients with PCa who died prior hip fracture operation or were inoperable (n = 5), had conservative treatment of fracture (n = 1), high energy trauma (n = 7), underwent Girdlestone procedure (n = 2) or had insufficient information in patient databases either on fracture or treatment method (n = 1). In the case of a second hip fracture during follow-up time, only the first one was included in the analysis. In five patients, PCa was found between hip fractures, and these patients were not included in the study cohort as patients with PCa.
The prescribed medications prior hip fracture was collected from the MRD. To analyze the effect of testosterone levels, we divided PCa patients into three groups: (1) patients not undergoing hormonal therapy, (2) patients under ADT, and (3) patients under NSAA monotherapy at the time of fracture. ADT contained surgical castration or chemical castration with receiving LHRH agonist/antagonist as monotherapy or together with antiandrogen (combined androgen blockade). Among patients under ADT, it was required that the last dose of LHRH agonist/antagonist had been administered no more than one year before the fracture. Patients with PCa receiving NSAA as monotherapy were analyzed separately since NSAA does not lower testosterone levels. 20 All patients in the hormonal therapy groups had received treatment for more than one month prior to the occurrence of the fracture.
The information on comorbidities at the time of fracture was gathered and recorded according to the Charlson comorbidity index (CCI). We categorized CCI as 0, 1, 2, 3, and 4+ points. We excluded PCa information from the CCI to emphasize the impact of other comorbidities among patients with PCa. All other cancer types were included in CCI. The American Society of Anesthesiologists (ASA) score was extracted from the MRD based on prior information on the patient’s overall health. In this study cohort, preoperative ASA scores ranged from 2 to 4. In addition, we collected information on date of birth, prior living situation (categorized as either home or nursing home), mobility aids or equipment prior hip fracture, previous comorbidities (not part of CCI), prior medications, hospital admission date for hip fracture, operation date of hip fracture, date of exclusion from the operation hospital, energy of trauma, subtype of hip fracture (S72.0, S72.1, and S72.2), surgical method, information on second hip fracture during follow-up time, and date of death in case of death before 31 December 2022. The follow-up time started from the first treatment contact due to hip fracture and ended until the date of death or on 31 December 2022, whichever came first. The information on the death date was reliably available from the MRD.
Risk category for PCa were divided into low-, intermediate-, and high-risk groups according to TNM classification, World Health Organization (WHO) grade, Gleason grade, or prostate-specific antigen (PSA) at the time of PCa diagnosis: low-risk PCa: T1–2, WHO grade I–II, Gleason grade sum less than 7 or PSA less than 20 ng/mL; intermediate-risk PCa: T3, WHO grade III, Gleason grade sum 7 or PSA between 20 and 50 ng/mL; and high-risk PCa: T4 or M1, Gleason grade sum greater than 7 or PSA greater than 50 mg/mL. Cases with missing data on risk categories were analyzed as a separate group. We also gathered data on whether patients had bone metastases at the time of the fracture.
After the data collection, the comparisons between the study characteristics of each group were analyzed by using ANOVA for continuous variables and the Chi-square test for categorical variables. The main exposure in our study was ADT, and the primary outcome was the difference in OS after hip fracture surgery between groups. OS was calculated by using Kaplan–Meier and continued with Cox regression in univariable, age-adjusted, and multivariable-adjusted analysis to determine the impacts of ADT with potential confounders for risk of mortality after hip fracture surgery. Potential confounders in multivariable-adjusted analysis included age at hip fracture diagnosis, prior living situation categorized as either home or nursing home, preoperative ASA score, presence of bone metastases, and risk category for PCa. Confounders for the multivariable model were selected based on a directed acyclic graph (Supplemental Fig. 1) and significance in univariable and age-adjusted analyses (Supplemental Table 1). Variables were included to adjust for bias, not to infer independent causality. The ASA score was retained over CCI due to better model performance. Coronary artery disease was excluded as a potential mediator. Although the PCa risk category was not statistically significant, it was included based on its known prognostic value.
Cox regression analyses were performed using IBM SPSS Statistics, version 27 (IBM Corp., Armonk, NY, USA). A p-value less than 0.05 was considered statistically significant. Kaplan–Meier survival curves were generated using Stata version 18.5 (StataCorp, College Station, TX, USA). The proportional hazards assumption for the Cox regression models was assessed using Schoenfeld residuals with the estat phtest command in Stata. No violations of the assumption were detected.
Results
Overall, 2672 male patients suffered hip fractures, of which 2365 were low-energy in nature and surgically treated in Pirkanmaa Hospital District between 2002 and 2018. Out of these 2365 patients, we identified 323 patients, who had a prior diagnosis of PCa and met the inclusion criteria. During the study period, 30 patients with PCa suffered a second hip fracture. Patients with PCa were categorized into three groups based on their current hormonal therapy status at the time of fracture: 94 patients not undergoing hormonal therapy, 212 patients under ADT, and 17 patients under NSAA. The baseline characteristics and comorbidities of the study population are presented in Tables 1 and 2, respectively.
Basic characteristics of the study population, which covers 323 patients with prostate cancer (PCa), who had low-energy hip fractures, and were surgically treated in the Pirkanmaa district hospital region between 2002 and 2018.
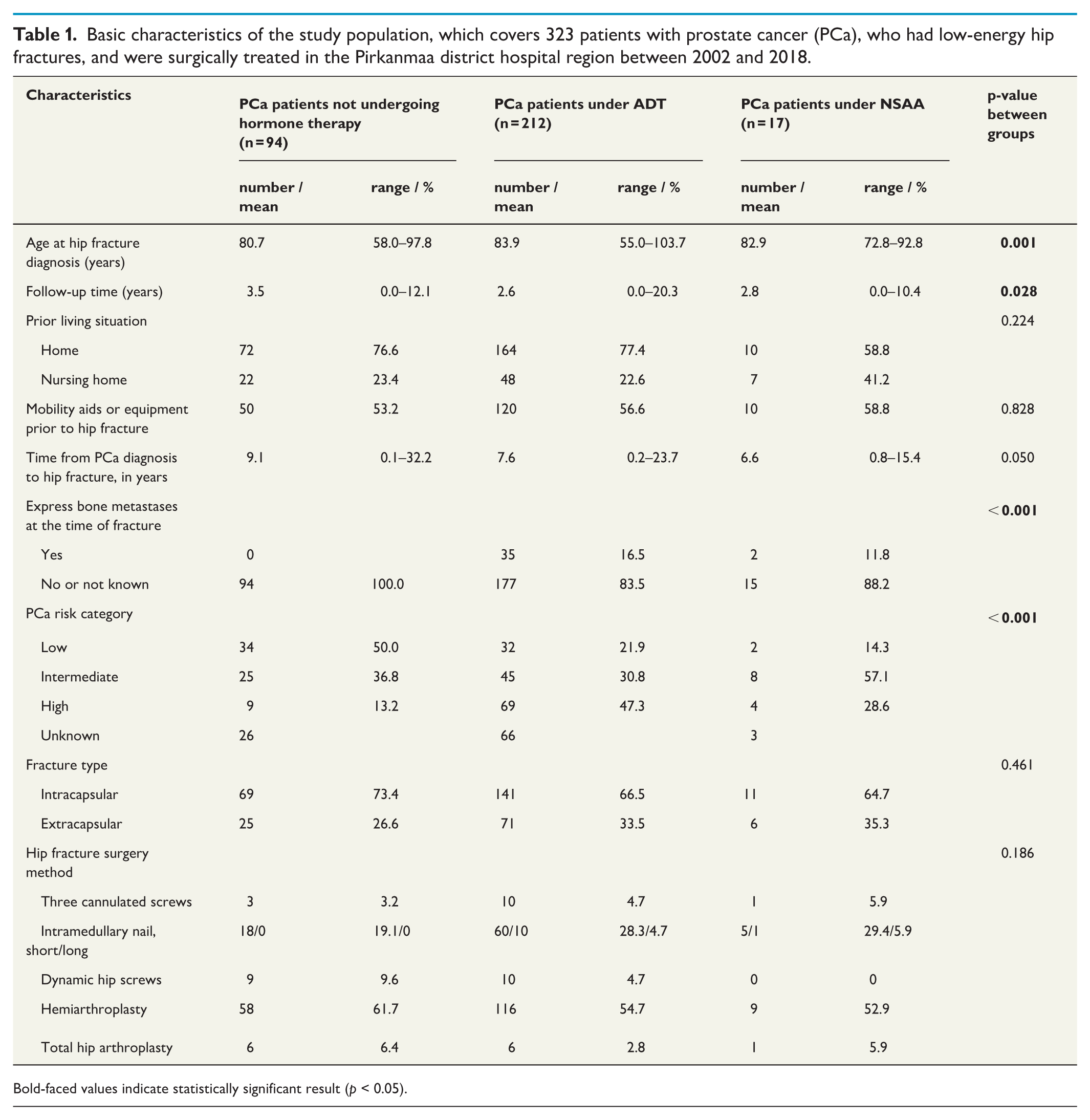
Bold-faced values indicate statistically significant result (p < 0.05).
Comorbidities of study population (n = 323).
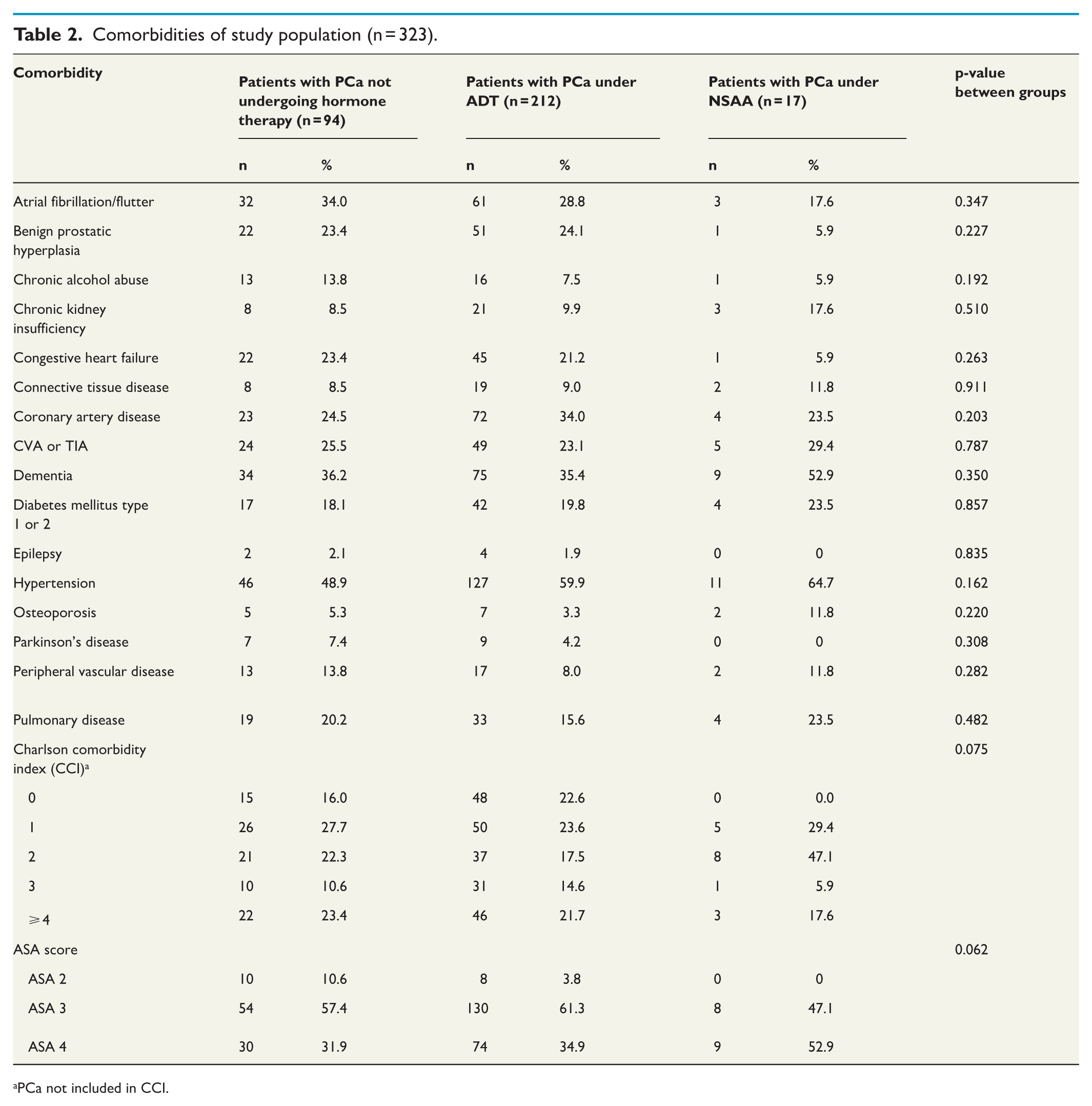
PCa not included in CCI.
Kaplan–Meier survival analysis suggested that patients under ADT had shorter OS (p = 0.006) (Fig. 1). In univariable Cox regression analysis, patients with PCa under ADT had shorter OS than patients with PCa not undergoing hormonal therapy (hazard ratio (HR) 1.53, 95% confidence Interval (CI) 1.18–1.99). However, after considering potential confounders in multivariable Cox regression analysis, ADT was not clearly associated with OS (HR = 1.19 (0.90–1.58)). Similarly, NSAA did not show a clear association with OS either in univariable, age-adjusted, or multivariable-adjusted analyses (HR = 1.47 (0.87–2.49); HR = 1.47 (0.87–2.49); HR = 1.02 (0.59–1.78), respectively) (Table 3). Higher age, higher ASA score, and living in a nursing home were associated with poorer OS in multivariable-adjusted analysis. None of the PCa risk category groups showed a clear association with OS after hip fracture in adjusted Cox regression analysis. (Supplementary Table 1)
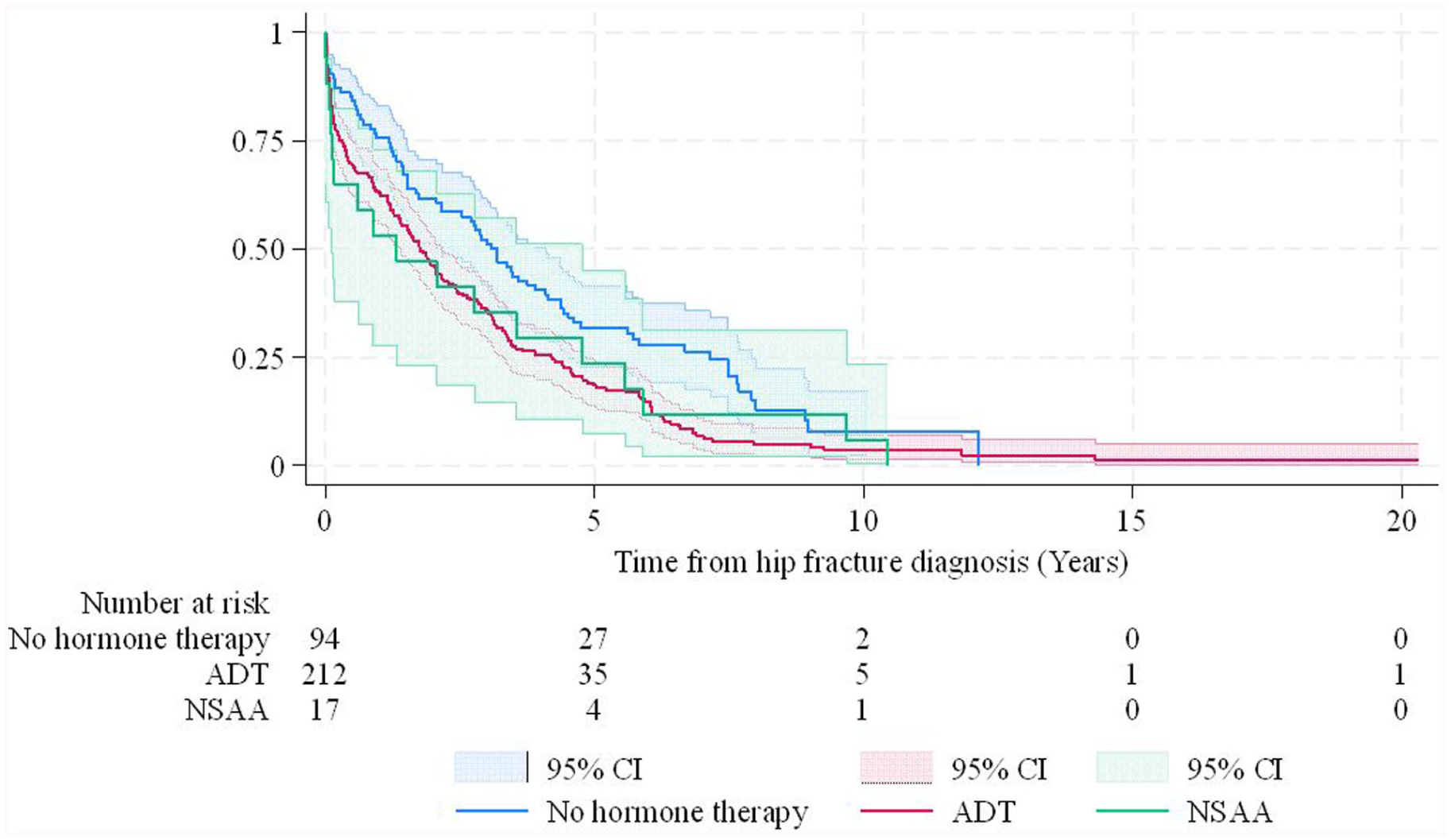
Kaplan–Meier OS curves for surgically treated low-energy hip fracture patients with prior PCa diagnosis.
Overall survival after hip fracture surgery in patients with PCa (n = 323) in univariable-, age-adjusted, and multivariable-adjusted analyses.
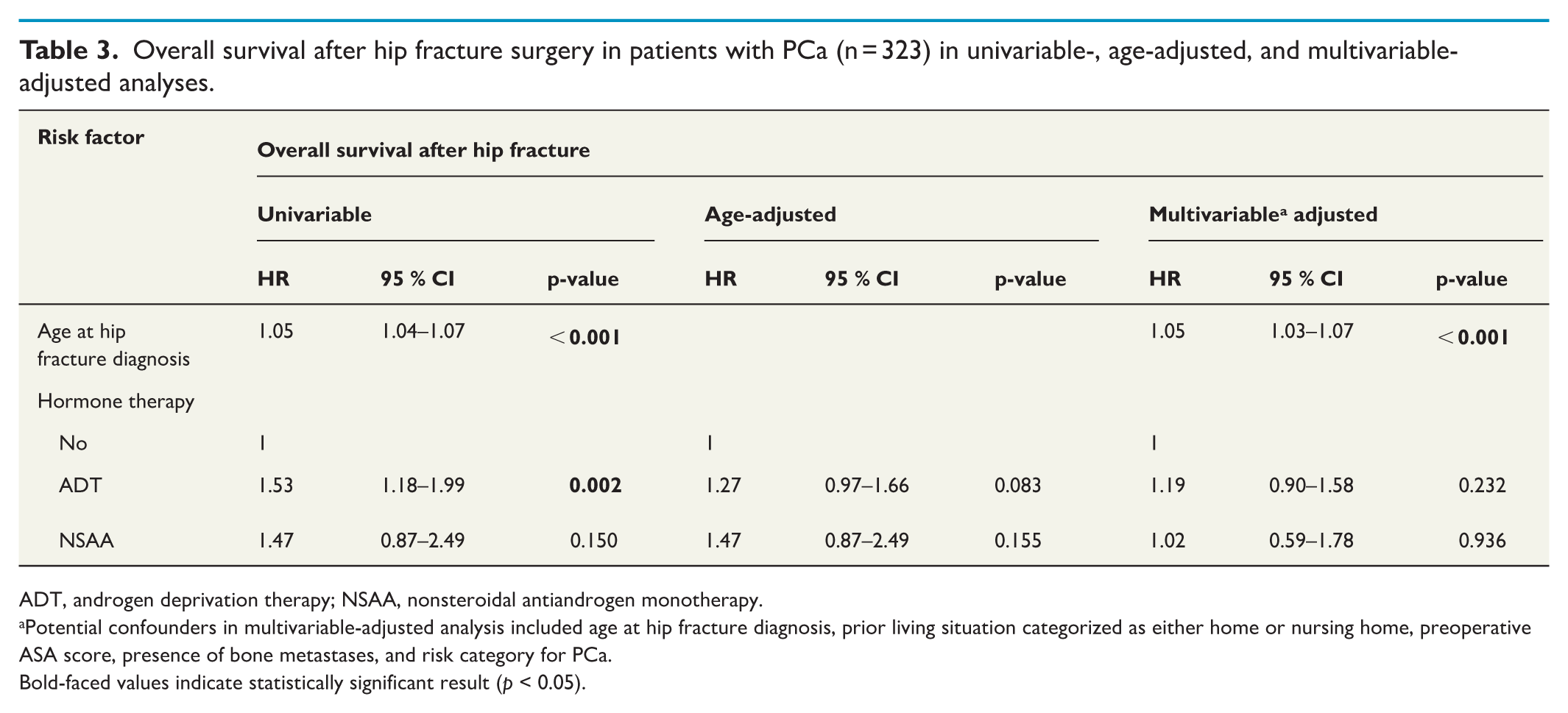
ADT, androgen deprivation therapy; NSAA, nonsteroidal antiandrogen monotherapy.
Potential confounders in multivariable-adjusted analysis included age at hip fracture diagnosis, prior living situation categorized as either home or nursing home, preoperative ASA score, presence of bone metastases, and risk category for PCa.
Bold-faced values indicate statistically significant result (p < 0.05).
The mean age at the time of hip fracture among all patients with PCa was 82.9 years (range: 55.0–103.7). Within the specific groups, the mean age was 80.7 years for patients not undergoing hormonal therapy, 83.9 years for those under ADT, and 82.9 years for those under NSAA. The mean follow-up time was 3.5 years (range: 0.0–12.1) for nonhormonally treated patients, 2.6 years (range: 0.0–20.3) for those under ADT, and 2.8 years (range: 0.0–10.4) for those under NSAA. Patients with PCa under ADT were significantly older and had a shorter follow-up time than patients not undergoing hormonal therapy. Overall, 91.3% of patients with PCa died during the follow-up time. Patient characteristics are presented in Table 1. The prior living situation and need for mobility aids or equipment before hip fracture did not differ between the groups. In terms of different comorbidities, preoperative ASA score, CCI, hip fracture subtype, or surgery method, no differences were observed between groups. Bone metastases and high-risk PCa were more prevalent among patients under ADT or NSAA (Tables 1 and 2). The risk category for PCa was available for 70.6% of all PCa patients. Cases with missing data were analyzed separately. Furthermore, these patients with missing information on the risk category were older and had their PCa diagnosed earlier. The mean utilization time of ADT or NSAA before hip fracture was 6.9 and 5.3 years, respectively.
Discussion
In our retrospective cohort study of 323 patients with PCa undergoing surgery for low-energy hip fracture, no clear association was observed between ADT and OS. Furthermore, NSAA use was also not clearly association with OS. Our study suggests that poorer OS after hip fracture surgery may be more closely associated with comorbidities and advanced age rather than ADT utilization. It has been widely shown that preoperative indicators for both short- and long-term mortality following hip fracture surgery include advanced age, male gender, higher ASA score, residing in a nursing home, presence of multiple comorbidities, poor preoperative walking capacity, and malignancy disease.21,22 In our study cohort, advanced age, living in a nursing home, and higher ASA score were also associated with a poorer prognosis following hip fracture surgery.
Unlike the findings in our study, Hemelrijck et al. previously documented the adverse effects of ADT on survivorship after hip fracture in a Swedish nationwide population-register study. They established that PCa men on ADT who experienced a hip fracture were 2.44 times more likely to die compared to PCa men not on ADT. 23 Surprisingly, they found that the difference in mortality was most pronounced in the youngest age cohort (50–74 years old), diminishing with advancing age. This finding suggests that the mortality difference might be due to advanced PCa rather than ADT. To the best of our knowledge, there is a lack of other studies comparing the impact of ADT on mortality after osteoporotic fracture.
While studies on the effects of ADT on mortality after hip fracture are limited, the influence of ADT on mortality has recently been investigated in the context of coronavirus disease 2019 (COVID-19). Similar to hip fractures, male gender has also been identified as a risk factor influencing mortality and other medical complications in COVID-19 compared to women.24,25 Men seem to be more susceptible than women to pulmonary embolisms, arrhythmias, delirium, as well as more serious infections after hip fracture surgery.4,5 In addition, male gender is associated with poor clinical outcomes of COVID-19, including a greater likelihood of requiring intensive care unit admission or facing a higher risk of death. 25 Dhindsa et al. 26 found in their study that men suffering pre-COVID-19 hypogonadism were 2.4 times more likely to be hospitalized if they contracted COVID-19 disease compared to those with normal testosterone levels. Thus, ADT may have a negative effect on mortality and other health outcomes in men with COVID-19 disease. However, current evidence does not support a causal association. Schmidt et al. 27 found no difference in the 30-day mortality rate for COVID-19 in PCa men on ADT compared to PCa men not on ADT. In a case-control study by Gedeborg et al. 28 observed that increased mortality from Covid-19 was mainly associated with advanced age, comorbidities, and more advanced PCa rather than ADT among patients with PCa.
Different hormonal therapies for PCa may have different adverse effects on cardiovascular and metabolic health. A meta-analysis of 19 trials found that ADT increased cardiovascular risk, unlike NSAA. 29 ADT is also linked to sarcopenia, osteoporosis, frailty, metabolic syndrome, cognitive decline, and body composition changes.30,31 These factors may affect outcomes through complex pathways not fully accounted for in our model. As this is an observational study, causal inference is limited and residual confounding remains possible. Notably, osteoporotic hip fractures in elderly are themselves associated with declining health and are often considered indicators of impending mortality, 32 potentially overshadowing the effects of specific treatments such as ADT.
The key clinical implication of our study is that we did not observe an association between ADT and increased mortality after hip fracture surgery in patients with PCa. However, ADT is well known to increase the risk of osteoporosis and, consequently, hip fractures—events that are widely recognized as sentinel events associated with increased morbidity and mortality. Therefore, we emphasize the importance of fracture prevention in patients receiving ADT, including the potential role of bone-protective medications, as enrollment in an osteoporosis management program has been shown to reduce hip fracture risk by more than 70% in patients with PCa undergoing ADT. 33 This is particularly relevant in the current treatment landscape, where doublet therapies (ADT plus second-generation antiandrogens) have become standard in the management of metastatic PCa. These therapies have significantly extended OS, meaning that patients are now living longer and consequently spending more time at risk for osteoporotic hip fractures.34–36
The following strengths of this study should be highlighted. Our study cohort was manually gathered from the MRD, ensuring comprehensive data on fractures, prefracture comorbidities, and the medical treatment of PCa at the time of the fracture. While population-register studies are larger, they often lack the depth of individual patient data achieved in this investigation. Furthermore, our study population can be considered homogeneous based on the treatment received for PCa. Since our study cohort consisted solely of patients with PCa residing in a single area, it is presumed that they received uniform treatment for their cancer. Although patients under ADT were significantly older and had more aggressive PCa disease than patients without hormone therapy, otherwise, no differences were observed between groups in terms of comorbidities and basic characteristics. This uniformity in the groups enhances the comparability of our findings and strengthens the validity of our results.
Our study has also limitations. First, one important limitation of this retrospective study concerns statistical power. Although CI for the adjusted HR crosses both sides of one, the relatively wide range suggests that the study may lack sufficient power to detect a clinically meaningful effect. While formal pre-study power calculations are not typically feasible in retrospective designs, the limited number of events in some subgroups may affect the robustness of the results. Consequently, we cannot fully exclude the possibility that ADT influences mortality after hip fracture. Second, the data for this study were retrospectively collected, which comes with inherent limitations. Our study focused on ADT, and preoperative testosterone levels were unavailable due to the retrospective nature of the study. Therefore, it is possible that some PCa patients who did not receive hormonal therapy at the time of fracture may still had undiagnosed hypogonadism. Some PCa cases were diagnosed decades ago, leading to unavailable data on the cancer’s histology and risk classification in the MRD. Consequently, we categorized the cases with missing risk classifications into a separate group. We included risk classification in the multivariable model due to its significance in predicting PCa outcomes. Despite this, in our cohort, none of the risk categories demonstrated a significant impact on OS after hip fracture surgery. Third, different ADT treatments, such as surgical or medical castration, were not examined separately due to the small sample sizes, a limitation shared by many other studies. Fourth, the NSAA subgroup was very small (n = 17), resulting in imprecise estimates and substantial uncertainty, with wide CI allowing for both potential benefit and harm.
In conclusion, in this retrospective cohort of prostate cancer patients undergoing surgery for low-energy hip fracture, no association was observed between ADT use and OS. However, the wide CI around the hazard estimate allows for both a modest protective effect and a potentially clinically relevant increase in mortality risk. These findings should therefore be interpreted as inconclusive rather than indicative of no effect. Further studies with larger cohorts are needed to clarify the potential impact of ADT on survival following hip fracture in this population. Given the overall high mortality risk, fracture prevention and comprehensive postoperative care remain essential in patients receiving ADT.
Supplemental Material
sj-docx-1-sjs-10.1177_14574969251387489 – Supplemental material for Androgen deprivation therapy and overall survival after hip fracture surgery in patients with prostate cancer
Supplemental material, sj-docx-1-sjs-10.1177_14574969251387489 for Androgen deprivation therapy and overall survival after hip fracture surgery in patients with prostate cancer by Silja Vuorlaakso, Minna K. Laitinen, Heini Huhtala, Antti Kaipia and Jussi Kosola in Scandinavian Journal of Surgery
Footnotes
Acknowledgements
Not applicable.
Author contributions
SV: Conceptualization, Data curation, Formal analysis, Investigation, Methodology, Resources, Validation, Visualization, Writing—original draft, Writing—review & editing. ML: Conceptualization, Data curation, Investigation, Methodology, Validation, Writing—review & editing. HH: Data curation, Investigation, Formal analysis, Methodology, Visualization and Writing—review & editing. AK: Conceptualization, Data curation, Investigation, Formal analysis, Methodology, Project administration, Resources, Supervision, Validation, Visualization, Writing—original draft and Writing—review & editing. JK: Conceptualization, Data curation, Investigation, Formal analysis, Methodology, Project administration, Resources, Supervision, Validation, Visualization, Writing—original draft and Writing—review & editing.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Ethics approval and patient consent
The study was approved by Tampere University Hospital’s institutional review board (ETL-code R19502). Due to the retrospective nature of this study, individual consent was not required.
Availability of data and materials
The data that support the findings of this study are available on request from the corresponding author. The data are not publicly available due to institutional reasons.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
