Abstract
Background and aims:
To compare the clinical and oncological results of anatomical resection of primary non-small cell lung cancers performed by resident and specialist surgeons as the lead surgeon in a medium-volume, mixed-practice hospital.
Methods:
We retrospectively collected individual patient record data. Between 1 January 2000 and 31 December 2020, a total of 959 primary lung cancer cases underwent intention-to-treat surgical resection at the Oulu University Hospital. Of these surgeries, 108 were performed by a resident surgeon as the lead surgeon. Propensity score matching was used to find similar unique resident-led cases to compare with unique specialist-led cases in a 1:2 ratio.
Results:
After propensity score matching, 65 resident-led cases were eligible for comparison to 130 specialist-led cases. Intra-operative complications were similar in both groups; resident-led cases had a complication rate of 12.3%, whereas specialist-led cases had a complication rate of 8.5% (p = 0.445). The incidence of major Clavien-Dindo complications (>IIIa) was 12.3% and 15.4% (p = 0.668), respectively. In the Kaplan–Meier analysis, the overall survival rate at 1, 3, and 5 years was 90.0%, 71.3%, and 65.3%, respectively, in resident-led cases and 88.2%, 66.6%, and 54.5%, respectively, in specialist-led cases (p = 0.389). Disease-specific survival at 1, 3, and 5 years was 90.0%, 77.6%, and 71.1%, respectively, in resident-led cases and 91.4%, 76.3%, and 76.3%, respectively, in specialist-led cases (p = 0.931).
Conclusion:
There was no difference in intra- and post-operative complication rate based on surgeon expertise. Both short-term and long-term results were comparable between resident- and specialist-led surgeries.
Keywords
Context and relevance
Lung cancer remains the second most common type of cancer worldwide and the leading cause of cancer-related mortality with poor prognosis. An earlier nationwide study from Finland reported an improvement in the long-term surgical outcomes for lung cancer patients between 2004 and 2014. A concern has been raised about the ethics of teaching surgical skills in real-life patients, with speculation around surgical outcomes. This study aimed to assess the results of intention-to-treat lung cancer surgeries at the Oulu University Hospital from 2000 to 2020 between resident- and specialist-led operations. Our main finding suggests that supervised resident-led operations do not compromise outcomes.
Introduction
Lung cancer remains the second most common type of cancer worldwide and the leading cause of cancer-related mortality. 1 Despite efforts to combat mortality rates, survival rates remain poor, especially in advanced stages of the disease. 2 The best survival outcome for patients is associated with operable non-small cell lung cancer (NSCLC) and radical surgery. 3 In 2021, in Finland, the overall incidence of new lung or trachea cancer was 2856 cases, and the 5-year relative survival rates were 22% for females and 14% for males. 4 Approximately 600 lung cancer operations are performed annually in Finland, mainly at five university hospitals, which are also responsible for the education and training of thoracic surgery residents. 5 Operation volume, mixed-practice, surgeon specialty, and experience are associated with mortality outcomes, which leads to discussions of centralizing lung cancer surgery. 6
Historically, surgery has been taught via lectures to build the foundation for learning anatomy and surgical skills. More experienced practitioners have been the instructors and supervisors during the operations. Using cadavers is a safe way to evaluate and train surgical approaches, although ethical and resource issues hinder the practicality. 7 Skills acquired by simulation have been proven to be transferable to real-life situations and improve surgical residents’ performance, especially with modern technologies, such as robotic surgery.7,8 Operating room experience is a key factor in teaching resident surgeons on their way to becoming independent practitioners. Different kinds of supervisory approaches can be used, from a high level of autonomy to a very minimalist role. 7 However, patient-driven outcomes and public consensus seem to support lessening the independence of resident surgeons in the operating room. 9 The involvement of inexperienced surgical residents in procedures raises concerns about patient safety. However, an earlier study did not find resident involvement to have a negative influence on surgical outcomes. 10 Concerns about a resident’s lack of ownership, autonomy, psychomotor ability, and academic interests have increased with current residency programs. 11
Teaching a new generation of thoracic surgeons to safely and effectively carry out lung cancer surgery is vital to guaranteeing good oncological results in the treatment of surgically operable lung cancer. In Finland, after finishing 6 years of medical school, it takes another approximately 6 years of surgical training to become a board-certified cardiothoracic surgeon, involving roughly 100 cardiac and 100 thoracic operations and a written exam. 5
The aim of the present study was to assess the clinical and oncological results of primary NSCLC surgeries performed by resident surgeons under supervision in a propensity-matched cohort in Northern Finland.
Patients and methods
Study design
This study was a retrospective cohort study carried out at the Oulu University Hospital. The study hospital is a mixed-practice unit with thoracic and cardiac surgeries performed by the same surgeons. Patients were identified from electronic patient records, which were used to collect the majority of the data to form the cohort. All 1114 patients who underwent intention-to-treat surgery for lung cancer in our cardiothoracic department between the 1st of January 2000 and the 31st of December 2020 were included. The data are considered nearly population-based as, during the study period, elective lung surgery was centralized into the Oulu University Hospital in Northern Finland. Of 959 primary lung cancer cases, 828 were NSCLC. The study was approved by the Oulu University Hospital and the Northern Ostrobothnia Ethics Committee. Due to the retrospective nature of the study, the individual patient consent requirement was waived by the Ethics Committee.
Data collection
Primary data collection took place between March 2020 and March 2021. All intention-to-treat lung cancer patients were included in the data collection, but only histologically confirmed primary lung cancer cases were included in the final draft and propensity score pairing and analysis. Individual hospital patient records were used to collect data on patient demographics, operation-specific records, and primary survival after surgery. The database of histological samples from the pathology department was used to crosslink with the hospital archives to achieve a 100% identification rate. All patients and their clinical (cTNM) and pathological (pTNM) tumor data were re-assessed by the study authors and recoded to the TNM Eighth Classification to maintain comparability across a long time frame. Short- and long-term survival data were obtained from the Finnish National Institute for Health and Welfare Registry, which has a 100% collection rate for causes of death in Finland.
Study groups
Surgeries performed by residents of cardiothoracic surgery were identified from the electronic patient records. Cases in which the primary operating surgeon could not be identified with certainty were excluded from the analysis. Two surgeons participated in all cases, and, in all resident-led operations, the assistant surgeon was a specialist in cardiothoracic surgery.
In primary lung cancer cases, the lead surgeon was a resident 108 times and specialist 846 times, whereas 5 cases did not have clear enough recording to determine the primary surgeon, as shown in the patient selection flow chart (online supplementary material). After defining the study groups to only contain anatomically resected cases, 85 resident-led cases were left to be considered in the propensity matching. Propensity score matching was performed in a 1:2 ratio. Propensity matching by patient age, sex, Charlson’s comorbidity index, 12 pre-operative forced expiratory volume in the first second of expiration (FEV1) and diffusing capacity of the lungs for carbon monoxide (DLCO) divided by 60% of age-related maximum into two groups, the year the surgery was performed, tumor histology (adeno, squamous, or other NSCLC), TNM Eighth Edition pathological stage, and surgical approach (video-assisted thoracoscopy or open thoracotomy) resulted in 65 resident-led cases and 130 specialist-led cases. Only anatomical resection cases were included in the propensity matching, and pneumonectomies and wedge resections were excluded from the study. Two patients did not receive curative surgery because tumor spread was revealed intra-operatively to be more extensive than anticipated. However, by intention-to-treat analysis, these cases were included in the final analysis.
Surgery-related details
For the purpose of this article, the lead surgeon was defined as the surgeon who was responsible for the clear majority of the intra-operative phases, including the approach to the operative field, dissection of vital structures, resection of vascular structures and bronchi, including lymphadenectomy, and final closure of the wound(s). The intra-operative decision-making responsibility was a part of the surgical team dynamics.
In resident-led cases, similar technical arrangements were in place, but the decision-making process for problem solving and acute complications was largely the responsibility of the specialist surgeon. To analyze intra-operative issues and the specialist surgeon’s pedagogical role, all 195 surgical charts were assessed for intra-operative complications. Intra-operative complications were situations in a resident-led case in which the specialist surgeon was likely to have intervened and temporarily taken on the role of primary surgeon to solve a critical issue, even if the patient record did not indicate a clear transfer of responsibility. Such events included non-intentional pulmonary vein or artery lesions, airway perforation or leakage, major lung parenchyma lesions, and any other major bleeding from thoracic vessels. We also included a case in which a surgical sponge was closed inside the thoracic cavity. Data on intra-operative complications were also collected from specialist-led cases to perform a comparison.
Extended resection was defined as local metastasis or local growth exceeding the anatomical borders of the lung parenchyma to the walls of the thoracic cavity, into costal bones, the spine, pericardium, carinal area, or bronchial walls with sleeve or similar resection being needed to achieve intra-operative R0 eradication.
The European Society of Thoracic Surgeons (ESTS) guidelines for lymph node staging in NSCLC were used to evaluate areas involved in lymph node sampling. 13 Limited N2 lymph node yield meant that only one or two N2 lymph node areas were removed for histological evaluation. Systematic lymph node yield meant that three or more N2 lymph node areas were removed for further evaluation.
Outcomes
The primary outcomes of this study were overall and disease-specific mortality. Secondary outcomes were resection type, extended resection, lymph node yield (lymph node yield, area, count), intensive care stay, hospital stay, discharge place (home or rehabilitation center), Clavien-Dindo classification, 14 complications, and intra-operative complications.
Statistical analysis
Cross-tabulation was used to compare demographics and clinical and oncological outcomes. The chi-square was used to calculate p-values for class variables, Student’s t-test for normally distributed continuous variables, and the Mann–Whitney test for skewed continuous variables. The Kaplan–Meier survival curves were used to visualize the overall and disease-specific survival of patients. The log-rank test (Mantel–Cox) was used to determine the equality of the survival distributions. p < 0.05 was considered significant.
Logistic regression was used to calculate propensity scores to compare the case similarities. The variables were selected to mitigate the possible impact of patient-related attributes and reduce the risk of pre-operative selection bias favoring more streamlined cases for residents. The covariables were sex (male or female), age (as a continuous variable), Charlson comorbidity index (0, 1, 2, 3, or more), the year of the operation (2000–2004, 2005–2009, 2010–2014, 2015–2020), histology of the cancer (squamous cell cancer, adenocarcinoma, or other NSCLC), FEV1 and DLCO (calculated as a binary variable, >60% of age-related maximum), surgical approach (as categorical covariate, thoracoscopy, or thoracotomy), and the pathological stage (divided into four corresponding TNM Eighth Edition stage groups). Matching was performed using a non-replacing method, in which a specialist surgeon’s case could only be paired once to a single resident entry. A propensity score of 0.05 was used as a cutoff value to determine cases that were similar enough; the mean propensity score was 0.84 with a standard deviation of 0.10.
All statistical analyses were performed in IBM SPSS version 28.0.0.0. For missing data, multiple imputation was performed in the original unmatched cohort. Imputed values included FEV1 (n = 129 patients) and DLCO (n = 299 patients). Smoking was used as a predictor for both FEV1 and DLCO. If both values were missing, no imputation was performed.
Results
A total of 769 lung cancer surgeries were anatomical resection of primary NSCLC. The main surgeon was a resident in 85 cases and a specialist in 681 cases. The mean age in both groups was 67 years (resident: 67 ± 8 years, specialist: 67 ± 10 years). Male patients were over-represented, comprising 71.8% of the resident-led cases and 65.3% of the specialist-led cases. A substantial proportion (70.6%) of the resident-led cases occurred in the last 6 years of the study period (1st of January 2015 to 31st December 2020), whereas only nine patients underwent anatomical resection by residents in 2005–2014. Adenocarcinoma was the most common histological cancer type, comprising 57.9% of resident-led operations and 55.0% of specialist-led operations. Baseline characteristics are presented in Table 1. After propensity score matching, no significant differences in the baseline parameters were observed between the groups (Table 1).
Baseline characteristics of patients who underwent surgery for non-small cell lung cancer in the Oulu University in 2000–2020.
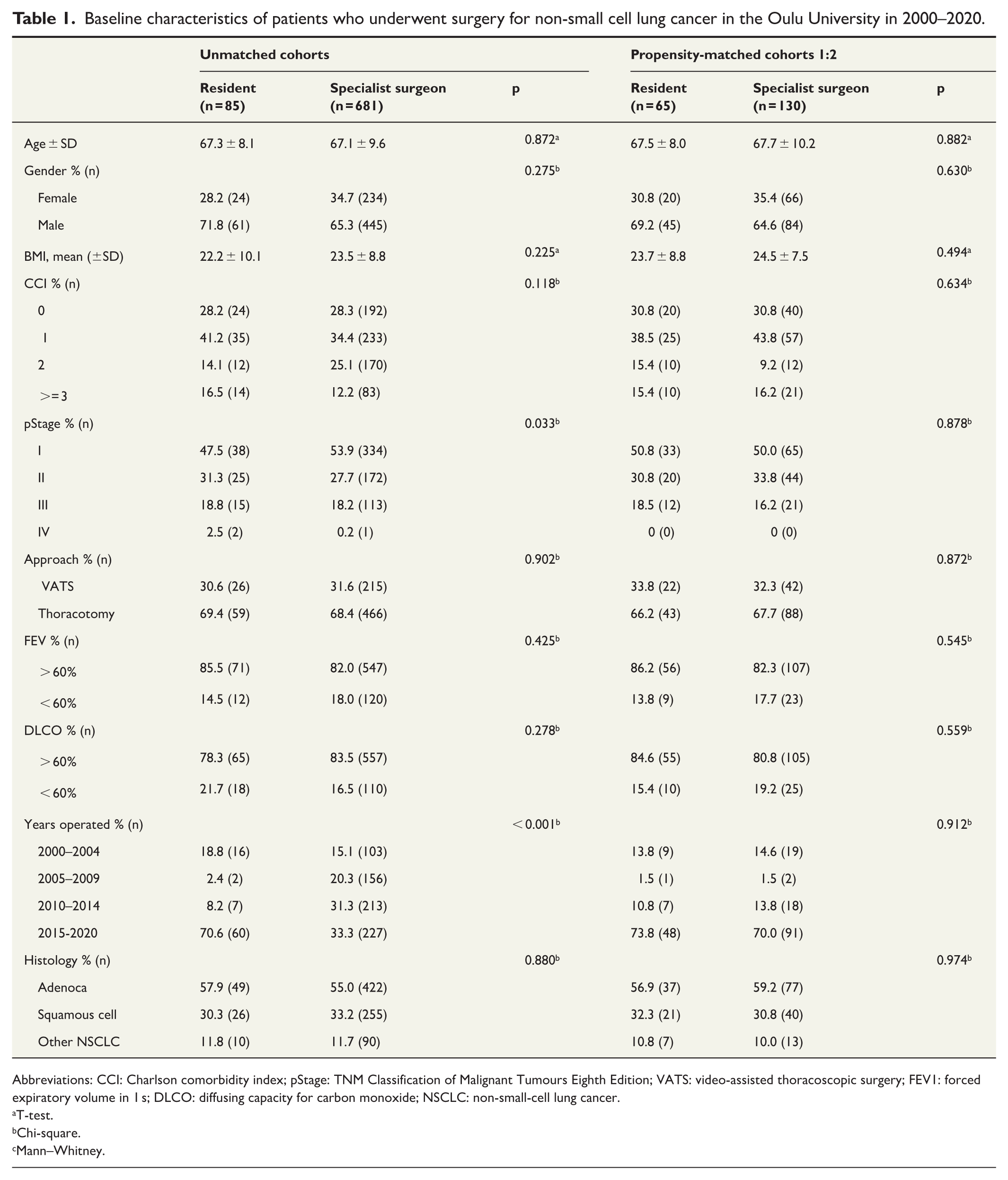
Abbreviations: CCI: Charlson comorbidity index; pStage: TNM Classification of Malignant Tumours Eighth Edition; VATS: video-assisted thoracoscopic surgery; FEV1: forced expiratory volume in 1 s; DLCO: diffusing capacity for carbon monoxide; NSCLC: non-small-cell lung cancer.
T-test.
Chi-square.
Mann–Whitney.
Propensity-matched analysis
After propensity matching, 65 resident-led cases could be compared to 130 specialist-led cases. The clinical parameters did not differ between resident- and specialist-led operations (Table 2). However, all segmentectomies were performed by specialist surgeons.
Clinical and oncological outcomes in the propensity-matched cohort.
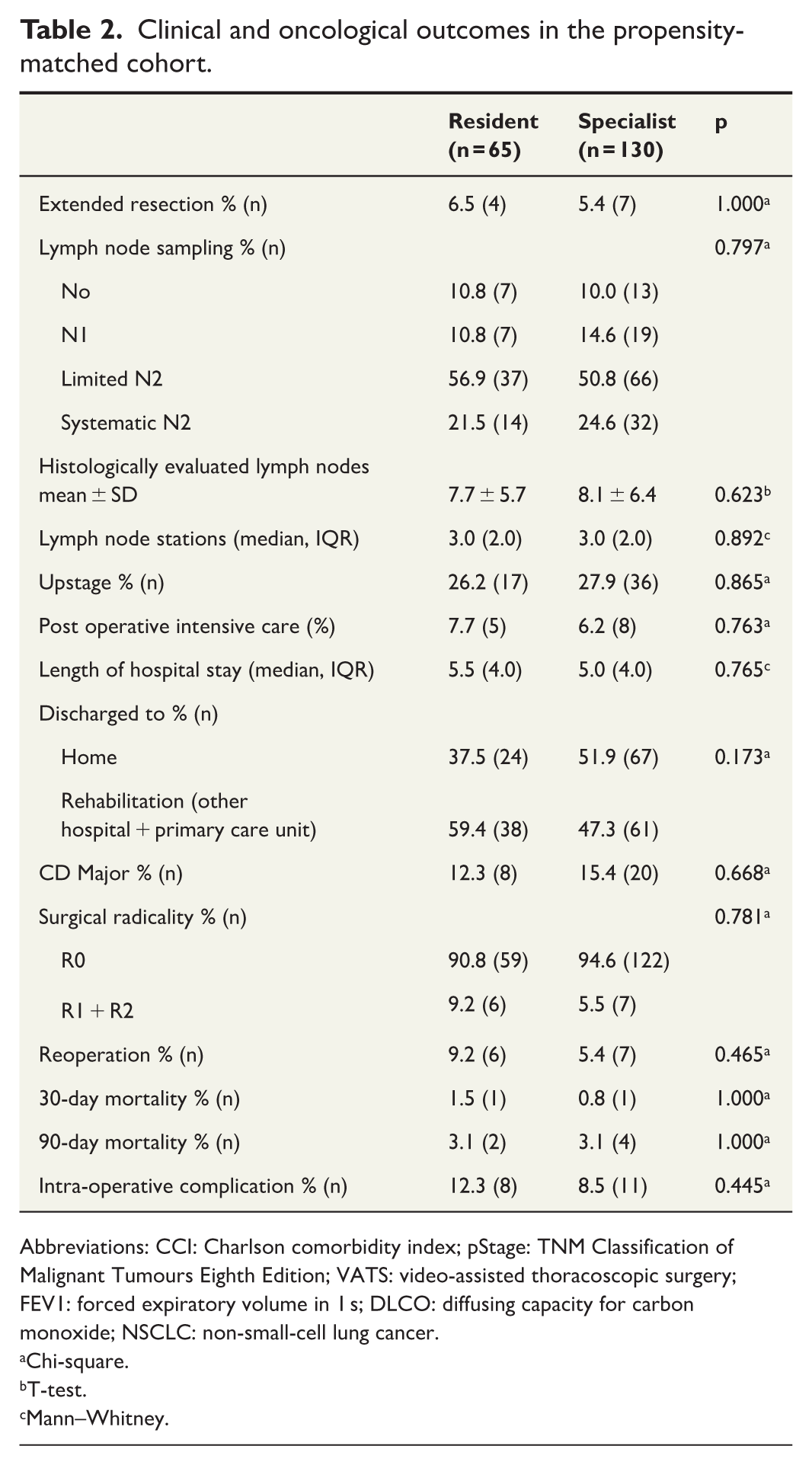
Abbreviations: CCI: Charlson comorbidity index; pStage: TNM Classification of Malignant Tumours Eighth Edition; VATS: video-assisted thoracoscopic surgery; FEV1: forced expiratory volume in 1 s; DLCO: diffusing capacity for carbon monoxide; NSCLC: non-small-cell lung cancer.
Chi-square.
T-test.
Mann–Whitney.
The 30-day mortality rate after surgical resection was 1.5% in the resident-led cases and 0.8% in the specialist-led cases (1/65 versus 1/130, p = 1.000); 90-day mortality was 3.1% versus 3.1%, respectively (2/65 versus 4/130, p = 1.000). The intra-operative complication rates were also similar in both groups; resident-led cases had a rate of 12.3%, whereas specialist-led cases had a rate of 8.5% (8/65 versus 11/130, p = 0.445). Major complication rates, classified by a Clavien-Dindo score of IIIa or more, were 12.3% and 15.4%, respectively (8/65 versus 20/130, p = 0.668).
Systematic lymph node sampling was achieved 21.5% of the time in resident-led cases and 24.6% of the time in specialist-led cases (p = 0.797). Comparing harvested lymph node stations, resident-led cases had a median of 3.0 (interquartile range (IQR) = 2.0) and specialist-led cases a median of 3.0 (IQR = 2.0, p = 0.892). Radical resection was achieved in 90.8% (59/65) of resident-led cases and 94.6% (122/135) of specialist-led cases (p = 0.781). Both groups had one individual R2 case with inoperable disease progression. The median stay in the operating hospital was 5.5 (IQR = 4.0) days in resident-led cases and 5.0 (IQR = 4.0) days (p = 0.765) in specialist-led cases.
Long-term outcomes
For the Kaplan–Meier survival estimates, the 1-, 3-, and 5-year overall survival was 90.0%, 71.3%, and 65.3%, respectively, in resident-led cases and 88.2%, 66.6%, and 54.5%, respectively, in specialist-led cases (p = 0.389, Fig. 1). Disease-specific survival rates at 1, 3, and 5 years were 90.0%, 77.6%, and 71.1%, respectively, in resident-led cases and 91.4%, 76.3%, and 76.3%, respectively, in specialist-led cases (p = 0.931, Fig. 2).
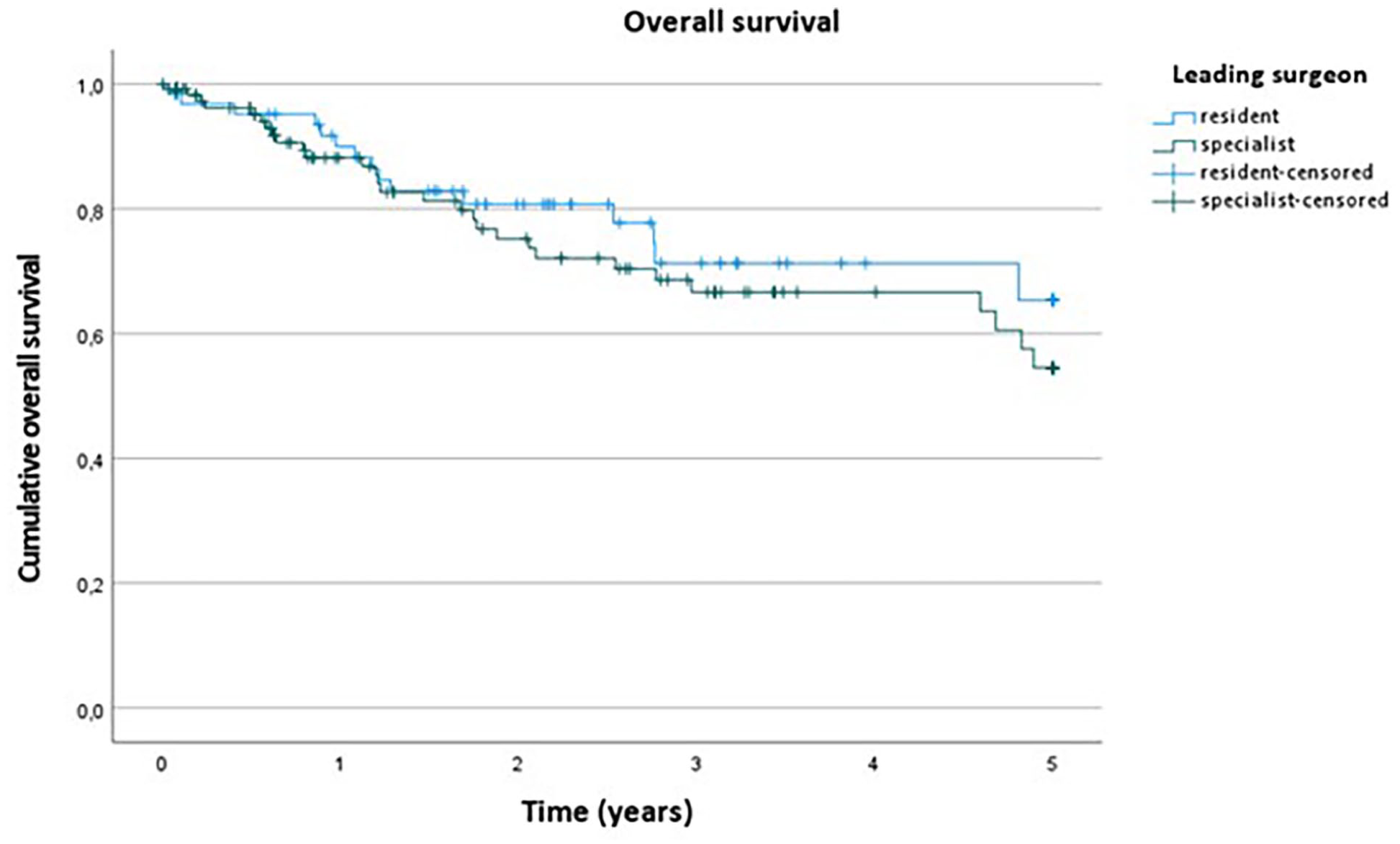
Overall survival stratified between study groups.
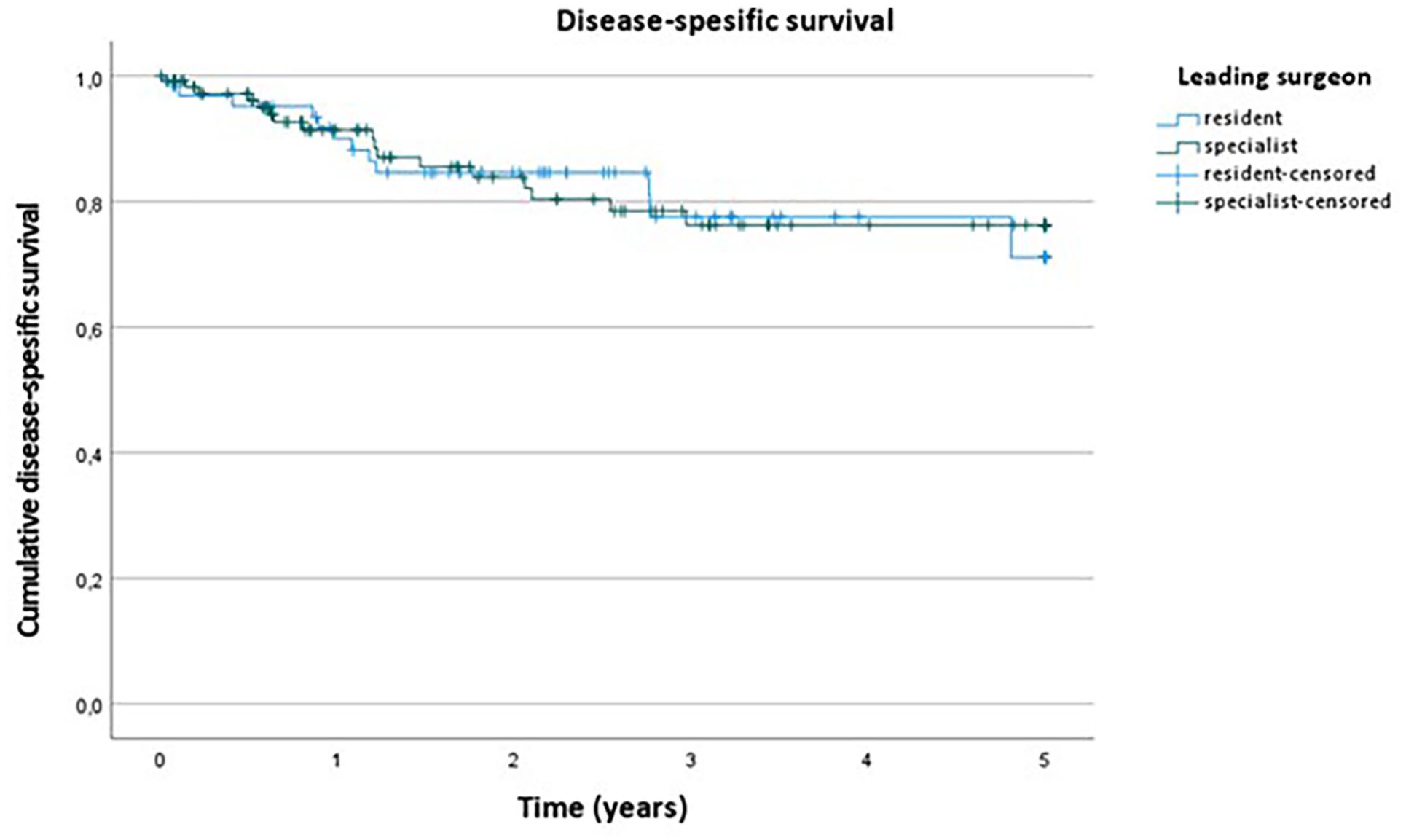
Disease-specific survival stratified between study groups.
Discussion
This study shows that residents performing lung cancer surgery under supervision do not compromise the clinical or oncological results of the treatment, even in a medium-volume mixed-practice hospital. Significant differences were not found between the study groups in regard to intra-operative complications or short- or long-term oncological results. Propensity score matching was used to mitigate selection bias between the study groups.
Teaching hospitals with residency programs generally have favorable surgical outcomes.15–17 More specifically, our findings on the overall complications and oncological results are similar to those of earlier studies. The reported 30-day mortality rate in a lobectomy-focused study from Cincinnati was 3%. 18 From our 195 patients in the propensity-matched group, the calculated 30-day mortality was 1.0% (2/195). A more recent Italian study consisting of 2613 lung cancer patients who underwent surgery reported 1-year and 5-year overall survival of 90% and 63%, respectively. 19 In our study, the 1- and 5-year overall survival was 90% and 65.3%, respectively, in the resident-led cases and 88.2% and 54.5%, respectively, in the specialist-led cases.
Our findings are also in line with earlier studies comparing residents to specialists. Rosenfeld et al. 20 reported a 30-day combined morbidity and mortality rate of 16.0% in resident and fellow involved cases, whereas cases in which only attending surgeons (specialists) were involved had a rate of 17.1% (p = 0.400). No significant differences were found between the groups in their study. The study had limitations as it only consisted of video-assisted thoracoscopic surgery (VATS) operations and the role of the residents was described as “involved.” A recent study, conducted in a setting similar to ours, compared the general resident surgeons’ participation in lung cancer surgery and found no significant differences in overall survival or disease-free survival. 21 Compatible results can be found in other fields of surgery. A Spanish study reports that resident involvement as the first surgeon had no effect on overall survival or disease-free survival in laparoscopic colorectal surgery. 22
A European multicenter study reported an incidence rate of 1.5% for major intra-operative complications. 23 By their definition, major intra-operative complications were only life-threatening situations, wrong transections of bronchovascular structures, injuries to other organs, and major reoperation to repair surgery-related, non-oncological intra-operative events. In our study, every unnecessary sudden bleed, erroneous transection, and non-protocol sudden onset that required active intervention from the surgeons were considered intra-operative complications. We used this approach to perform a more meaningful comparison between the resident- and specialist-led cases. Some or most intra-operative complications do not necessarily translate directly into a complication according to the Clavien-Dindo classification, and the rest of the recovery period can be uneventful. The same European multicenter study indicated that a surgeon’s experience with 10 or more lung operations significantly reduces the non-oncological conversion rate, but not the major intra-operative complications rate. Our study had rather high conversion rates in propensity-matched data. Also, non-propensity matched, both groups and all vats operations included, vats conversion rate was 22.7% during the study period. We did not witness any statistically significance improvement over the study years. One reason for the rather high conversion rates may be the low overall volume of operations. In addition, the overall main reason for the conversion was failure to progress, rather than bleeding. Adhesions, ventilation issues, and technical difficulties consisted 79.4% of the conversions. Similar to earlier studies, surgeon experience did not affect the rate of intra-operative complications.
An earlier study found a significant difference in operation time between residents and specialists, with the latter being shorter. 24 However, other studies reported similar operation times and blood loss. 25 In our study, we focused more on the learning aspects, intra-operative safety, and oncological results of resident-led cases under the supervision of a specialist surgeon. Gathering comprehensive survival data, we compared residents to specialists in a way that matters the most.
Study limitations include the possibility of missing minor post-operative complications after discharge from the hospital. Any major complications were referred to the study hospital and survival data are retrievable through the compulsory National Cause of Death registry. The 21-year study period may also be a confounding factor due to changes to guidelines and working habits, although this impact might be mitigated by the fact that the majority (70.6%) of matched cases were from 2015 to 2020. The design of the study limited the reliable collection of intra-operative variables. As a result, potentially valuable data, such as operation time and bleeding, are missing. This highlights the need for further research on these aspects. As a medium-volume, mixed-practice hospital, deep specialization in lung cancer surgery and teaching has not been feasible. This limitation could impact both the learning process and patient outcomes.
The use of hospital records for patient data is a major strength of this study. Every patient entry was collected by hand and data were not as restricted as in register studies. This results in a more accurate and unified patient data set, especially regarding demographics, risk factors, surgical data, and complications. Our data are also considered nearly population-based because Northern Finland’s elective lung surgeries were centralized into the Oulu University Hospital during the study period. The Finnish National Institute for Health and Welfare Registry was used to define causes and time of death. This registry has a 100% collection rate and provides complete survival data.
Conclusion
This study shows that lung cancer operations performed by resident surgeons under supervision are safe. Clinical and oncological outcomes of resident-led surgeries are comparable to specialist-led cases.
Supplemental Material
sj-docx-1-sjs-10.1177_14574969251359866 – Supplemental material for The impact of surgeon experience on lung cancer operations: A retrospective propensity-matched cohort study
Supplemental material, sj-docx-1-sjs-10.1177_14574969251359866 for The impact of surgeon experience on lung cancer operations: A retrospective propensity-matched cohort study by Olli E. Mustonen, Anne K. Niskakangas, Topias H. Karjula, Iiris L. Puro, Olli Helminen and Fredrik Yannopoulos in Scandinavian Journal of Surgery
Footnotes
Author contributions
O.E.M.: Data acquisition, Conceptualization, Methodology, Formal analysis, Investigation, Writing the original draft, and Review and Editing. A.K.N.: Data acquisition, Investigation, and Review and Editing. T.H.K.: Data acquisition, Investigation, and Review and Editing. I.L.P.: Data acquisition and Investigation. O.H.: Conceptualization, Methodology, Formal analysis, Investigation, Writing the original draft, and Review and Editing, Visualization, Supervision, Project administration, and Funding acquisition. F.Y.: Conceptualization, Methodology, Formal analysis, Investigation, Writing the original draft, Review and Editing, Visualization, Supervision, and Project administration.
Data availability statement
On reasonable request. Data sharing will require additional Ethics Committee approval.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Instrumentarium Science Foundation (O.H.), Mary and Georg C. Ehrnrooth Foundation (O.H.), and Finnish State Research Funding (O.H. and F.Y.).
Research ethics
This study was approved by the local hospital district and the Northern Ostrobothnia Ethics Committee (EETTMK 81/2008).
Patient consent
The study was approved by the Oulu University Hospital and the Northern Ostrobothnia Ethics Committee. Due to the retrospective nature of the study, the individual patient consent requirement was waived by approval from the Ethics Committee.
Clinical trial registration
Not applicable (retrospective cohort).
Supplemental material
Supplemental material for this article is available online.
Twitter handle
Comparing survival results of surgically operated lung cancer patients between residents and specialists in mixed-practice hospital. A two-decade retrospective study.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
