Abstract
Objective
Lung cancer is a malignancy with high a mortality rate that threatens human health. This study is aimed to explore the correlation among the triglyceride/high-density lipoprotein ratio (TG/HDL-C), non-high-density lipoprotein/high-density lipoprotein ratio (non-HDL-C/HDL-C) and survival of patients with non-small cell lung cancer (NSCLC) undergoing video-associated thoracic surgery (VATS).
Methods
This retrospective study analyzed 284 patients with NSCLC who underwent VATS at Hebei General Hospital, Shijiazhuang, China. The time-dependent receiver operating characteristic curve was used to determine the optimal cutoff value and evaluate the area under the curve. Kaplan–Meier and Cox regression analyses were performed to determine the prognostic effect.
Results
The median overall survival (OS) was 46 months. Patients with low TG/HDL-C and low non-HDL-C/HDL-C had a longer OS. The low non-HDL-C/HDL-C group showed a longer mean survival time (59.00 vs. 52.35 months). Multivariate analysis revealed that TG/HDL-C and non-HDL-C/HDL-C were significantly correlated with OS.
Conclusions
TG/HDL-C and non-HDL-C/HDL-C are associated with the prognosis of patients with NSCLC who received VATS. Preoperative serum TG/HDL-C and non-HDL-C/HDL-C may be effective independent prognostic factors for predicting the outcomes of patients with NSCLC.
Keywords
Introduction
Lung cancer (LC) is a malignancy with high morbidity and mortality. According to global cancer statistics, LC is the leading cause of cancer-related death (18%) and the second most common cancer in incidence (11.4%). 1 The most common pathological type of LC is non-small cell lung cancer (NSCLC), accounting for 83% of cases. At present, surgery is the main treatment for patients with NSCLC at early stages or partial resection for those with local progression. 2 Despite many advances in the surgical treatment of patients with NSCLC in recent years, the improvement in patient survival remains unsatisfactory. 3 Therefore, it is necessary to search for biomarkers that predict the clinical outcomes of patients with NSCLC to guide perioperative and long-term treatment.
Lipoproteins and blood lipids have been reported to be associated with the morbidity and mortality of cardiovascular diseases and cancer. 4 In addition, serum lipoproteins are reportedly related to the incidence of LC. 5 However, there are few studies on the relationship between blood lipids and lipoproteins and cancer prognosis. Preoperative serum triglyceride (TG) and high-density lipoprotein cholesterol (HDL-C) were reported to have a prognostic impact on patients with NSCLC. 6 In addition, some clinical indicators are associated with the prognosis of various malignancies, including LC. These include age, sex, smoking status, apolipoprotein A (Apo-A), apolipoprotein B (Apo-B) and total cholesterol (TC).7,8 Non-HDL-C, calculated as TC minus HDL-C, includes Apo-A, intermediate-density lipoprotein, low-density lipoprotein cholesterol (LDL-C) and very-low-density lipoprotein cholesterol (VLDL-C). The TG/HDL-C ratio and non-HDL-C/HDL-C ratio have greater predictive value for lipid changes than individual lipid markers.9,10 This study aims to investigate the effect of non-HDL-C/HDL-C and TG/HDL-C on the prognosis of patients with NSCLC following video-associated thoracic surgery (VATS).
Patients and methods
Study design
This retrospective study collected clinical and laboratory data of all patients with NSCLC who underwent VATS at the Department of Thoracic Surgery of Hebei General Hospital from July 2016 to January 2019. All patients were ≥18 years old at the time of surgery and were diagnosed with NSCLC at stage I to IIIA. Preoperative clinical and laboratory data were complete. Patients were excluded if they meet the following criteria: inoperable NSCLC or stage IIIB to IV, use of any drug that affects lipid metabolism prior to serum collection, non-primary lung tumor or blood transfusion within 3 months before blood collection. Patients underwent surgical procedures, including anatomical pneumonectomy, sublobar resection, segmentectomy or wedge resection, in accordance with national guidelines (NCCN Guidelines Chinese version).
The endpoint was overall survival (OS), which referred to the time from operation to death from any cause. For subjects who missed follow-up visits prior to death, the time of the last follow-up was counted as the time of death. Patient follow-up followed the 8th edition of the LC criteria (UICC/AJCC-8, 2017) and mainly involved hospital records or telephone calls. Follow-ups were performed every 3 months for 2 years and every 6 months for the next 3 to 5 years until 1 February 2022, death or loss to follow-up.
This retrospective study was approved by the Ethics Committee of Hebei General Hospital (No. 2022038, 17 March 2022). The need for informed consent was waived owing to the retrospective nature of the study. We confirm that data confidentiality was maintained and complied with the Declaration of Helsinki. We de-identified all patient details to protect their identity. The reporting of this study conforms to REMARK guidelines. 11
Clinical and laboratory parameters
The laboratory results and clinical parameters were obtained from the hospital’s electronic medical record database. Thirteen parameters were collected, including sex, age, smoking history, stage, histological type, body mass index (BMI), Eastern Cooperative Oncology Group performance status (ECOG PS), Charlson comorbidity index (CCI), surgical method, postoperative adjuvant therapy and serum lipid indexes. For patients with multiple serum collections during hospitalization, the last fasting blood sample within 7 days before surgery was chosen. Serum lipid indexes included TC, TG, Apo-A, Apo-B, HDL-C, LDL-C and VLDL-C.
Statistical analysis
R4.1.2 (www.r-project.org) and IBM SPSS Statistics for Windows, Version 23.0 (IBM Corp., Armonk, NY, USA) were used for statistical analysis. The time-dependent receiver operating characteristic (ROC) curve and Youden Index were used to determine the cutoff values of non-HDL-C/HDL-C, TG/HDL-C, LDL-C, VLDL-C, HDL-C, TC, Apo-A and Apo-B. The area under the curve (AUC) was calculated. Survival and subgroup analyses were performed using the Kaplan–Meier model and log-rank test. BMI and CCI subgroups were analyzed using the continuous chi-square test, and all other subgroups were evaluated using Pearson’s chi-square test. The Cox proportional hazards model with the hazard ratio and 95% confidence interval was applied for univariate and multivariate analyses to identify independent prognostic factors for NSCLC. The test level was set at both sides α = 0.05, and P < 0.05 was considered statistically significant.
Results
Baseline characteristics
ROC curves of non-HDL-C/HDL-C and TG/HDL-C were shown in Figure 1a and 1b, and the results of ROC analyses were shown in Table 1. The AUCs of TG/HDL-C and non-HDL-C/HDL-C were higher than those for other biomarkers.
Results of ROC curve analyses.

ROC, receiver operating characteristic; AUC, area under the curve; TG, triglyceride; HDL-C, high-density lipoprotein cholesterol; TC, total cholesterol; LDL-C, low-density lipoprotein cholesterol; VLDL-C, very-low-density lipoprotein cholesterol; Apo-A1, apolipoprotein A1; Apo-B, apolipoprotein B.
The baseline characteristics are shown in Table 2. A total of 284 patients aged 31 to 81 years with a median age of 61.5 years were enrolled. Among them, 238 cases (83.8%) were diagnosed as lung adenocarcinoma, and 200 cases (70.4%) were at stage I. Eighty patients (28.2%) underwent sublobar lung resection, which referred to wedge resection and segmentectomy. The median OS was 45 months (ranged 8 to 67 months). Eighty-four patients (29.6%) died, including patients lost to follow-up.
Baseline characteristics and comparison between groups.

BMI, body mass index; ECOG PS, Eastern Cooperative Oncology Group performance status; CCI, Charlson comorbidity index; TG, triglyceride; HDL-C, high-density lipoprotein cholesterol. Note: The death group includes deceased patients and patients lost to follow-up. For subgroups of BMI and CCI with sample sizes less than 5, the continuous corrected chi-square test was chosen. Pearson’s chi-square test was chosen for the remaining subgroups.
Comparison between groups
According to the cutoff value of TG/HDL-C, patients were divided into two groups: group 1 with a low ratio (TG/HDL-C < 1.379, n = 201) and group 2 with a high ratio (TG/HDL-C≥ 1.379, n = 83). Similarly, patients were divided into two groups according to the cutoff value of non-HDL-C/HDL-C: group 3 with a low ratio (non-HDL-C/HDL-C<2.530, n = 91) and group 4 with a high ratio (non-HDL-C/HDL-C ≥ 2.530, n = 193). Comparisons of baseline characteristics between groups are shown in Table 2. In addition, Figure 2a showed the survival curves of groups 1 and 2, and Figure 2b showed the survival curves of groups 3 and 4. Patients with low TG/HDL-C (56.61 vs. 46.81 months) and low non-HDL-C/HDL-C (59.00 vs. 52.35 months) had a longer mean survival time and higher survival rate than those in the high-ratio groups (P < 0.05). Compared with groups 2 and 4, patients with low TG/HDL-C and low non-HDL-C/HDL-C had milder clinical and laboratory characteristics. Patients with low TG/HDL-C and low non-HDL-C/HDL-C had a lower death rate, more normal BMI and lower CCI (P < 0.05). In addition, patients in group 1 received fewer postoperative treatments than those in group 2 (P < 0.05).

Optimal cutoff values for TG/HDL-C and non-TG/HDL-C applied to time-dependent ROC curves. (a) Time-dependent ROC curve for TG/HDL-C with an AUC of 0.651, corresponding to an optimal cutoff value of 1.38 and (b) Time-dependent ROC curve for non-HDL-C/HDL-C with an AUC of 0.634 and an optimal cutoff value of 2.53.
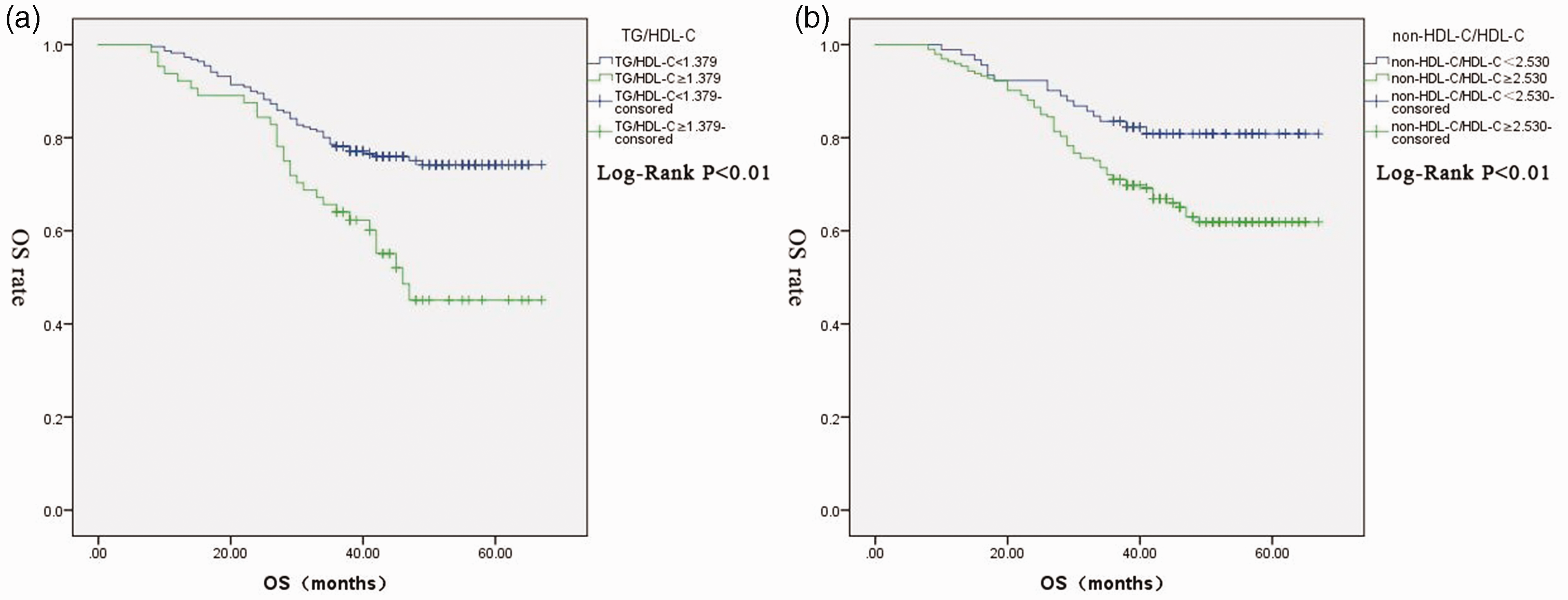
Kaplan–Meier analysis of survival. (a) Kaplan–Meier plot of OS according to TG/HDL-C and (b) Kaplan–Meier plot of OS according to non-HDL-C/HDL-C.
Univariate and multivariate analyses
The results of univariate and multivariate analyses using Cox proportional risk models are shown in Table 3. In the univariate analysis of OS, low TG/HDL-C, low non-HDL-C/HDL-C, female sex, age <60 years, pathological type of adenocarcinoma, stage I, no smoking history, BMI< 24, ECOG PS = 0 or 1, low TG and no postoperative treatment were significant factors for a longer survival time (P < 0.05). Variables with statistical significance in the univariate analysis were included in the multivariate analysis. In addition, considering its prognostic impact on patients with LC in clinical settings, CCI was included. The multivariate analysis showed that low TG/HDL-C, low non-HDL-C/HDL-C, age <60 years, no smoking history, BMI < 24, low TG and no postoperative treatment may be independent prognostic factors for long OS (P < 0.05).
Univariable and multivariable analyses.

BMI, body mass index; ECOG PS, Eastern Cooperative Oncology Group Performance status; CCI, Charlson comorbidity index; TG, triglyceride; HDL-C, high-density lipoprotein cholesterol; TC, total cholesterol; LDL-C, low-density lipoprotein cholesterol; VLDL-C, very-low-density lipoprotein cholesterol; Apo-A1, apolipoprotein A1; Apo-B, apolipoprotein B; HR, hazard ratio; CI, confidence interval; LL, lower limit; UP, upper limit.
Subgroup comparison
Pathological type and stage showed statistical significance in the univariate analysis but not in the multivariate analysis. To further estimate the prognostic impact, we performed subgroup analyses. The survival curves are shown in Figures 3 to 5. For patients with adenocarcinoma (Figure 3a and 3b), patients at stage I (Figure 4a and 4b) and patients following lobectomy (Figure 5a and 5b), those with low TG/HDL-C and low non-HDL-C/HDL-C had higher survival rates (P < 0.05). However, TG/HDL-C and non-HDL-C/HDL-C showed no statistical significance in the survival analyses among patients with squamous cell carcinoma (Figure 3c and 3d), at stage II to IIIA (Figure 4c and 4d) and following sublobar resection (Figure 5c and 5d).

Kaplan–Meier plot of OS according to TG/HDL-C and non-HDL-C/HDL-C in stage I (3a, 3b) and stage II to IIIA (3c, 3d).

Kaplan–Meier plot of OS according to TG/HDL-C and non-HDL-C/HDL-C for different pathological subtypes: adenocarcinoma (4a, 4b) and squamous cell carcinoma (4c, 4d).

Kaplan–Meier plot of OS according to TG/HDL-C and non-HDL-C/HDL-C for different surgery types: lobectomy (5a, 5b) and sublobar resection (5c, 5d).
Discussion
With advances in medical technology, the survival rates of most malignancies have been significantly improved. 12 However, the current assessment of LC prognosis is still based on imaging data or pathological staging, which cannot always accurately predict the patient’s treatment benefit.13–14 Therefore, it is necessary to identify suitable biomarkers that easily and effectively assess the therapeutic effect.
Abnormal lipid metabolism has been reported to be related to the prognosis of various malignancies. Serum lipid markers, such as TC, TG and HDL-C, have been demonstrated to be prognostic factors for breast cancer, gastric cancer, nasopharyngeal carcinoma and NSCLC.15–26 In addition, it has been shown that statins may prevent the development of LC, 27 which further indicates the correlation between serum lipid markers and LC prognosis. This study enrolled patients with NSCLC following VATS to explore the prognostic impact of serum lipid biomarkers. Our results are generally consistent with previous studies. Ma et al. reported a correlation between elevated preoperative TG and HDL-C levels and poor prognosis in patients with NSCLC. 6 However, our results showed no independent prognostic impact of HDL-C on patients with NSCLC undergoing VATS, which may be due to differences in the basic characteristics of the cases, such as mean age and pathological type distribution.
In this study, we found that the ability of non-HDL-C/HDL-C and TG/HDL-C to predict OS may be better than that of other blood lipids through time-dependent ROC curves and AUCs. A prognostic impact of TG/HDL-C and non-HDL-C/HDL-C on patients with gastric cancer, peritoneal dialysis and COVID-19 has been suggested,28,29 but the relevance of these biomarkers for LC has not been investigated. This study suggests that non-HDL-C/HDL-C and TG/HDL-C are independent biomarkers that predict the prognosis of NSCLC following VATS. HDL-C and TG are involved in energy metabolism and signal transduction and reflect the physiological and pathological states of the human body. 30 TG inhibits the degradation of ApoB-100, thereby increasing the production and secretion of VLDL. 31 Cholesteryl ester transfer protein mediates the exchange of TG-rich VLDL with other lipids. As a result, TG levels are negatively correlated with HDL-C levels. 32 HDL prevents inflammatory responses triggered by intracellular oxidative stress and oxidized low-density lipoprotein by inhibiting the nuclear factor kappa B signaling pathway and related inflammatory events, such as the expression of adhesion molecules. 33 The systemic inflammatory response is closely related to the occurrence and development of tumors and is significantly associated with the prognosis of LC.34,35 Serum lipid levels also have certain effects on the human immune system by mediating various signaling pathways and related proteins in the cell membrane. 36 HDL-C impairs cholesterol transport and metabolism, and cholesterol disturbances caused by reduced HDL-C levels may be responsible for cancer progression. 36 The above mechanisms partially explain our results. However, the specific mechanism still needs to be confirmed by further basic research.
Our findings demonstrate the prognostic importance of preoperative lipid levels in patients with NSCLC at stage I to IIIA undergoing VATS. We identified TG/HDL-C, non-HDL-C/HDL-C, TG, sex, age, pathological type, stage, smoking history, BMI, ECOG PS and postoperative treatment as prognostic factors in patients with operable NSCLC. Furthermore, non-HDL-C/HDL-C, TG/HDL-C, TG, age, smoking history, BMI and postoperative treatment may be independent prognostic factors.
Analyzing the prognostic factors of patients with NSCLC following VATS will improve the assessment of the prognostic risk for patients and guide postoperative treatment plans, thereby effectively prolonging the survival period of patients.
The study still has some limitations: 1) This study is a single-center retrospective study, and there may be selection bias in the absence of long-term blood lipid levels after surgery. 2) The number of cases was limited. Because non-HDL-C/HDL-C and TG/HDL-C may be independent prognostic factors, large-scale multicenter prospective validation studies are needed to establish more reliable and independent data. 3) Blood lipid levels can be influenced by various factors, such as lifestyle habits, diet and health perceptions, and the mechanisms should be further investigated. 4) This study only used OS as the endpoint and lacked the inclusion of patients with recurrence and metastasis.
Conclusions
In this study, we found that the predictive ability of non-HDL-C/HDL-C and TG/HDL-C may be better than that of other blood lipids through time-dependent ROC curve and AUC calculation. We used the Youden Index to determine optimal cutoff values for non-HDL-C/HDL-C, TG/HDL-C and other indicators. High non-HDL-C/HDL-C and high TG/HDL-C may be significant independent prognostic factors for worse OS. Finally, we concluded that non-HDL-C/HDL-C and TG/HDL-C ratios may be potential independent prognostic factors for patients with stage I to IIIA NSCLC undergoing surgery.
Supplemental Material
sj-pdf-1-imr-10.1177_03000605221117211 - Supplemental material for Prognostic significance of TG/HDL-C and non-HDL-C/HDL-C ratios in patients with non-small cell lung cancer: a retrospective study
Supplemental material, sj-pdf-1-imr-10.1177_03000605221117211 for Prognostic significance of TG/HDL-C and non-HDL-C/HDL-C ratios in patients with non-small cell lung cancer: a retrospective study by Lingxin Kong, Qingtao Zhao, Zhaohui Han, Wenfei Xue, Zhonghui Hu, Zhancong Niu and Guochen Duan in Journal of International Medical Research
Footnotes
Author contributions
K.L. analyzed and interpreted the patient data and was a major contributor to writing the manuscript. Z.Q. performed statistical univariate and multifactor analysis and graphing. All other authors reviewed the data statistics. All authors read and approved the final manuscript.
Declaration of conflicting interest
The authors declared that no competing interest exists.
Funding
This research received no specific grant from any funding agency in the public, commercial or not-for-profit sector.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
