Abstract
Adrenal glands can host metastases from various malignancies. While surgical resection offers potential benefits in solitary- or oligometastatic cases, many patients have systemic disease unsuitable for surgery when adrenal metastases are diagnosed. Most adrenal metastases originate from malignancies in the lungs, kidneys, or liver. This review explores diagnostic and treatment strategies for adrenal metastases including data from a single center. In particular, it is shown that surgical resection of metastases from colorectal cancer to the adrenals may offer a potential survival benefit. Personalized treatment plans and further research are critical for improving outcomes for patients with adrenal metastases.
Introduction
Adrenal glands are paired, hormonally active, Y-shaped organs, located retroperitoneally on the cranial margin of each kidney. They may harbor metastases from various malignant tumors. Although surgical resection of adrenal metastases is considered a technically feasible treatment option for patients with solitary metastases or oligometastatic disease, many patients are not candidates for this approach, as adrenal metastases often indicate widespread, systemic disease. Current studies indicate that the most common primary tumors associated with adrenal metastases are lungs, kidneys and liver, constituting more than half of the cases suitable for surgical resection.
This narrative review summarizes current knowledge on therapeutic strategies for detection, diagnosis, and treatment of patients with adrenal metastases, and present updated results from a tertiary center cohort, with special emphasis colorectal cancer adrenal metastases (CRAMs).
Mechanisms for adrenal metastases
The hormonal activity of the adrenal glands and their multiple functions most likely play a role in the development of local metastatic disease. They contain a cortex in which steroid hormones are being produced and a medulla, from which catecholamines are being released into the systemic circulation. Their active role in homeostasis of numerous hormonal feedback loops, may be at least in part be attributable to their hypervascularity, making them particularly susceptible for harboring and growth of metastatic malignant cells. Since the adrenal glands do not represent a biological “filter” like the liver and lungs, the exact mechanism by which malignant cells end up in these organs is not known. 1
There are numerous reports of malignant tumors that may metastasize to the adrenal glands, the most frequent being small-cell lung carcinoma (SCLC), renal cell carcinoma (RCC), hepatocellular carcinoma (HCC), and pancreatic adenocarcinoma2,3. In a previous study from the authors unit, it was demonstrated that 31 out of 41 patients (75.6%) with a history of malignant disease who later underwent surgical removal of adrenal incidentaloma, were found to have metastases in the adrenal gland based on the final pathology report. 4
Diagnostic workup and preoperative staging
Several imaging modalities including computed tomography (CT), magnetic resonance imaging (MRI), and positron emission tomography (PET) scans all play a central role in the differentiation of adrenal lesions. Distinguishing metastatic lesions from benign adrenal nodules by radiology alone, can, however, be challenging, necessitating a comprehensive approach in which imaging characteristics, biochemical testing, and sometimes tissue biopsy for histopathological confirmation are required.
Biopsy
In most cases, the differentiation between a benign and a metastatic adrenal lesion can be made on radiology alone. CT- or ultrasound-guided biopsy or fine-needle aspiration (FNA) may, however, be justified. 5 In case of inconclusive radiological imaging, no evidence of hormonal activity, and, in particular, if the histological report may impact the clinical management of the patient, a biopsy is warranted.
Adrenal metastases from colorectal cancer
Colorectal cancer (CRC) is one of most common cancers and is the second leading cause of cancer-related death worldwide. The precise incidence of CRAMs is not known and most likely depends on the stage of the disease upon diagnosis. Such data can be obtained either by radiological imaging of patient cohorts during diagnostic assessment, as part of clinical follow-up images, or by post-mortem autopsies. It has been reported that as many as 14.4% of patients with CRC have malignant cells in their adrenals if examined by microscopy upon autopsy. 6 This may indicate that some patients dying from CRC have systemic disease at the time of death. However, the extent to which clinically significant metastatic disease, defined as disease influencing therapeutic strategies or treatment outcomes, is present remains unclear.
Radiological staging of patients with newly diagnosed CRC reveals adrenal masses in approximately 2%–4% of cases. 7 The presence of an adrenal mass in a patient with resectable CRC does, however, not necessarily indicate metastatic disease, as the reported prevalence of adrenal metastases varies widely. Adrenal metastases from CRC typically present radiologically as an adrenal mass with non-adenoma enhancement (absolute washout <60% or relative washout <40%) and without lipid on chemical shift MRI or on non-contrast CT. Absence of signs of hemorrhage, calcification, and lack of pheochromocytoma hyperenhancement aids in excluding primary adrenal malignancy. 8
A recent study of 6474 patients with potentially resectable CRC, identified on CT scans, found adrenal nodules larger than 10 mm in 363 patients. Among these, three nodules were malignant, 11 were indeterminate and 349 were benign, emphasizing that further investigation beyond CT scans is warranted for adrenal lesions in patients with CRC. 9 This contrasts with the earlier findings from the authors center, where 38% of patients with a history of CRC and an adrenal mass had confirmed CRC metastases.
A recent article from a European database (Eurocrine®) on adrenalectomy for secondary malignancies reported that less than 10% of adrenal metastases originated from CRC. Lung cancer accounted for the majority (46.7%), followed by RCC (22.1%). 10
To update our understanding, all adrenalectomies performed for secondary malignancies at Oslo University Hospital during the past decade (January 2014–June 2024) were reviewed. Among 46 patients with confirmed adrenal metastases, 13 cases (28.2%) were CRC metastases. Other primary cancers included SCLC (11 cases, 23.9%), RCC (8 cases, 17.4%), and HCC (6 cases, 13.0%). The median time from initial treatment of the primary tumor to adrenalectomy was 28.8 months for CRC, 13.5 months for lung carcinoma, 47.7 months for RCC, and 7.4 months for HCC. For patients with colorectal adrenal metastases (CRAM), overall survival was 34.5 months, with five patients alive at data analysis. There was, however, no cumulative 5-year overall survival in patients who underwent adrenalectomy for CRAM (Fig. 1B). Median survival for metastases from pulmonary, renal, and HCC was not reached. The cumulative overall survival for all patients and grouped according to the respective entities are depicted in Fig. 1A, C, respectively. Detailed findings are summarized in Table 1.
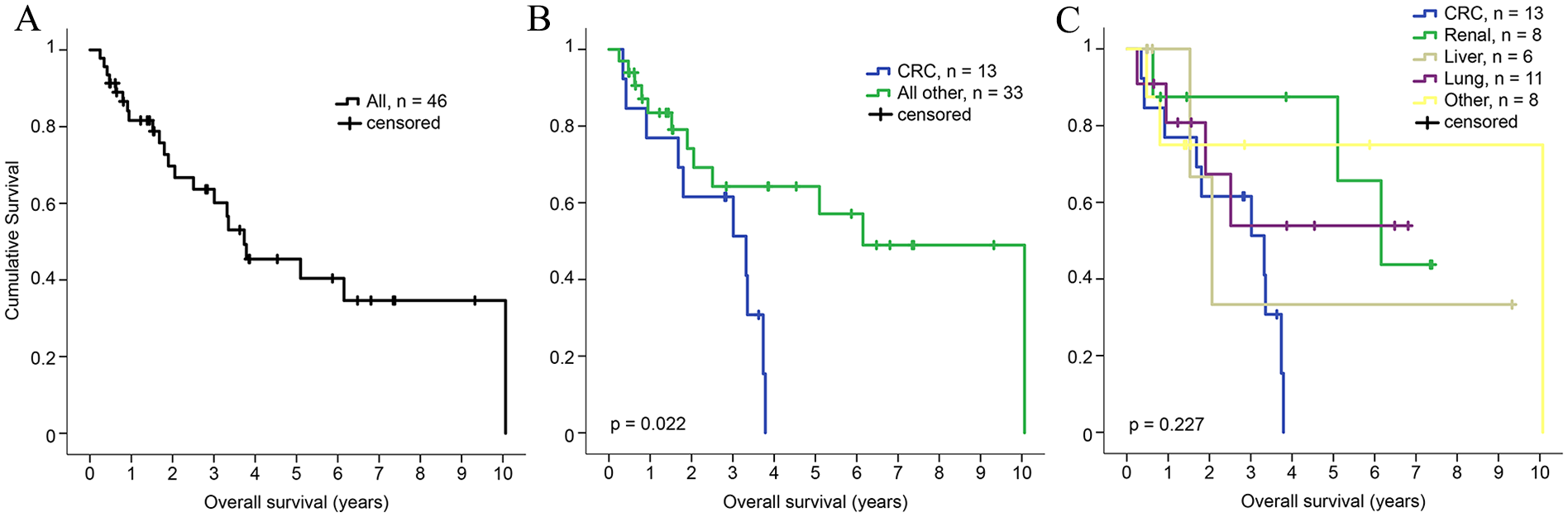
Kaplan–Meier cumulative overall survival analysis of patients who underwent adrenalectomy for metastatic disease at Oslo University Hospital. (A) All patients irrespective of origin of the primary tumor. (B) Comparison of adrenal metastases from colorectal cancer (CRC) adrenal metastasis versus those from other primary tumor origins. (C) Survival analysis of adrenal metastases from CRC, renal cell carcinoma, hepatocellular carcinoma, lung cancer, and other primary tumors.
Clinical presentation and oncological outcome for patients resected for adrenal metastases.
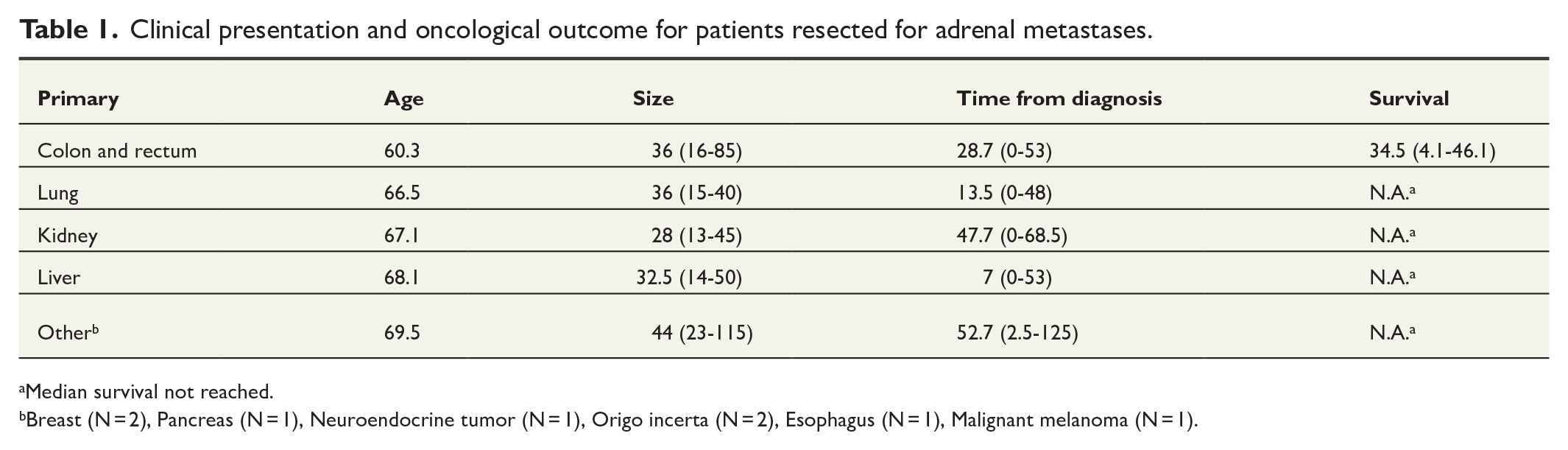
Median survival not reached.
Breast (N = 2), Pancreas (N = 1), Neuroendocrine tumor (N = 1), Origo incerta (N = 2), Esophagus (N = 1), Malignant melanoma (N = 1).
Surgery for adrenal metastases
Most reports on surgical treatment for CRAM are case presentations or case series. Due to the scarcity of patients with oligometastatic CRAM, conducting a randomized controlled trial (RCT) is unlikely, necessitating an individualized evaluation for each patient. Managing adrenal metastases from CRC requires a multidisciplinary approach tailored to the patient`s clinical profile and tumor biology. Surgical resection remains the cornerstone of treatment for isolated adrenal metastases, offering the potential for durable disease control and even cure in select cases. Not all patients are suitable candidates for surgery, particularly those with advanced disease or significant comorbidities. In such cases, systemic therapies, including chemotherapy, targeted treatments, or immunotherapy, may be employed alone or in combination with surgery to maximize disease control and prolong survival.
A systematic review analyzing 55 studies of surgical resection of CRAM identified 145 cases. 11 Of these, 41 patients had a colonic primary tumor, 36 had a rectal primary, and the location of the primary tumor was not specified in 68 cases. Solitary adrenal metastases were reported in less than half (44.8%), while synchronous metastases in other organs were reported in 37.9%.
Both laparoscopic and open surgery have been utilized for adrenalectomy in patients with CRAM. The choice between minimally invasive surgery (MIS) and open surgery depends on tumor size, extent of infiltration into surrounding tissues, and surgeon expertise. For solitary adrenal metastasis without adjacent organ involvement, MIS is generally sufficient, offering advantages such as reduced blood loss and shorter hospital stays compared to open surgery. Similar outcomes have been observed in liver metastases from CRC, 12 but no large-scale studies have confirmed this for adrenalectomy in CRAM. In the series from the authors unit, all patients underwent minimally invasive transabdominal laparoscopic surgery, with one case converted to open surgery due to a concomitant liver resection.
Simultaneous resection of CRC and CRAM
Simultaneous resection of adrenal metastases and the primary tumor in CRC has rarely been reported. While technically feasible, it is often inadvisable due to the likelihood of advanced disease, where surgery may only add surgical risk without clear clinical benefit. In the authors’ series, one patient underwent simultaneous adrenalectomy during a “liver first” procedure after neoadjuvant chemoradiation for advanced rectal cancer. Unfortunately, the patient died due to disease progression within 5 months before removal of the primary tumor.
Given the limited data on treatment outcomes for simultaneous CRAM, no evidence-based recommendations can currently guide management strategies in such cases.
Focal therapies
Stereotactic body radiation therapy (SBRT) has been reported to be an option for treating adrenal metastases from renal and pulmonary cancers, 13 but evidence for its use in CRAM is limited. Similarly, thermal ablation techniques, such as radiofrequency and cryoablation, have been reported for adrenal metastases, 14 although none of the patients in the authors series underwent these treatments.
Perioperative chemotherapy
CRAM resections are often performed after a period of observation, during which systemic therapy may be administered to evaluate tumor biology. Chemotherapy regimens for CRAM should follow standard guidelines for metastatic CRC. Among the 13 patients from the authors unit, the median time from diagnosis to CRAM resection was 29 months, during which patients often received interval chemotherapy in conjunction with treatment of other metastases.
For microsatellite instability (MSI) tumors, checkpoint inhibitors (immunotherapy) may be an option. However, adrenal glands have been suggested as a potential “sanctuary” site where metastatic growth persists despite effective systemic treatment elsewhere. This resistance may be linked to impaired antigen presentation in the adrenal gland microenvironment, influenced by glucocorticoids. 15
Oncological outcome
Survival following resection of CRAM varies widely, depending on the disease stage, response to treatment, and the presence of synchronous or metachronous metastases at other sites. While there is robust evidence supporting survival benefits from resection of CRC liver and lung metastases, data on CRAM resection remain sparse.
The previous series from the authors unit reported a median survival of 24 months for patients with CRAM tumors smaller than 6 cm, compared to 13 months for larger tumors. 4 In the updated dataset, median survival improved to 34.5 months (range: 5–46.1 months), with tumor size showing no impact on survival. This improvement may reflect better patient selection due to neoadjuvant chemotherapy, now standard for CRAM management. Comparable outcomes were reported in a European multicentre series 16 and a systematic review by Nguyen et al. 11 where median survival ranged from 23 to 34.7 months. However, there was no cumulative 5-year overall survival, and there is no evidence in the authors’ data, to date, that adrenalectomy for CRAM provides cure (Fig. 1B).
Follow-up
Patients with adrenal metastases should be followed as other metastatic CRC patients, but there is no specific guidance for follow-up strategies. In the systematic review by Nguyen et al. 11 only 20.7% of patients received adjuvant chemotherapy, with no documented clinical benefit.
Conclusion
Adrenal metastases from CRC represent a significant manifestation of advanced disease, posing diagnostic and therapeutic challenges. A multidisciplinary approach integrating surgery, systemic therapy, and supportive care is crucial for addressing these challenges and improving outcomes. Surgery for CRAM in selected patients with oligometastatic disease can achieve survival exceeding 3 years. Advances in systemic therapies, including targeted agents and immunotherapy, hold promise for improving patient outcomes, underscoring the need for ongoing research to refine treatment strategies.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
