Abstract
Background:
The benefit of surgical resection for colorectal cancer (CRC) lung metastases is unclear. The aim was to investigate the incidence, treatment strategy, and 5-year overall survival (OS) in CRC patients with isolated lung metastases.
Methods:
This registry-based retrospective cohort study included patients treated with curative resection of CRC within the county of Skåne during the period 2010–2016, who had synchrone or metachrone isolated lung metastases. Exclusion criteria were previous or concurrent metastases at other organ sites. Patients were identified in the Swedish Colorectal Cancer Registry (SCRCR) and data were retrieved from SCRCR and medical charts. Patients were divided into groups based on whether they had synchronous or metachronous lung metastases and curative or palliative treatment intent. The primary endpoint was 5-year OS. Multivariable Cox-regression and Kaplan–Meier survival analysis were performed.
Results:
Of 8457 curatively resected CRC patients, 93 (1.1%) had isolated lung metastases (53 synchronous/40 metachronous). Of these, 53 were treated with curative intent, 51 (96%) of whom were managed surgically. The remaining 40 patients were treated palliatively and either with chemotherapy or with best supportive care. Five-year OS was 42% (39/93), median 50 months (IQR: 24–60) for the entire cohort, and 68% (36/53), median 60 months (IQR 55–60) and 7.5% (3/40), median 22 months (IQR: 12–33) for curative and palliative patients, respectively. In multivariable analysis, age (hazard ratio (HR): 1.04, confidence interval (CI): 1.01–1.07), multiple lung metastases (HR: 1.64, CI: 1.08–2.47), and unilateral distribution (HR: 0.41, CI: 0.20–0.84) were predictors of OS.
Conclusions:
Isolated CRC lung metastases are rare. Curative treatment was associated with considerably better 5-year OS than palliative treatment (68% vs 8%). Age, solitary metastases, and unilateral distribution were predictors of survival.
Keywords
Introduction
Among non-communicable diseases, cancer represents a significant disease burden with colorectal cancer (CRC) consistently ranking in the top regarding incidence and mortality. 1 Stage at diagnosis is the most important prognostic factor and survival varies significantly among stages. Patients with localized disease (stages I–III) generally have advantageous outcomes with 5-year survival up to 90%, whereas the corresponding number for disseminated disease (stage IV) is only 10%–15%. 2
CRC most commonly metastasizes to the liver and/or the lungs. Improved screening and surveillance programs in combination with ever-evolving treatment options, especially oncological advances, have increased the number of potential candidates for surgical resection. Resection of colorectal liver metastases is an established part of the management of stage IV CRC due to its association with improved survival. 3 Interestingly, the pattern of metachronous metastases seems to be influenced by the treatment strategy as shown in the RAPIDO trial where short-course neoadjuvant radiotherapy followed by neoadjuvant chemotherapy reduced the occurrence of metachronous liver recurrences compared to traditional long-course chemoradiotherapy, whereas lung recurrence rates were not significantly different between the groups. 4
The benefit of pulmonary metastasectomy in CRC patients is still debated. Although some studies suggest a survival benefit for certain patients,5,6 other studies argue that a selection of favorable tumor biology resulted in increased survival, rather than the surgery itself. 7 The only randomized controlled trial (RCT) is the PulMiCC study comprising 93 patients. In this study, no survival benefit was shown with metastasectomy, and a better-than-expected survival was demonstrated for the patients receiving non-operative management. However, the study included patients with prior liver resection (27/93) and was gravely underpowered, as it was stopped due to poor recruitment before reaching even one-third of the calculated sample size. 8
The primary aim of this study was to identify the incidence, treatment strategies, 5-year overall survival (OS) and potential prognostic factors in CRC patients with isolated lung metastases. Secondary aims were to quantify the risk of recurrence after curative treatment of lung metastases as well as the risk of progression of metastatic disease with palliative treatment.
Methods
Study design
This retrospective, observational cohort study in the Swedish county of Skåne, combined data from the national Swedish Colorectal Cancer Registry (SCRCR) with data extracted from medical charts. The region comprises approximately 1.3 million citizens, roughly 10% of Sweden’s population. The SCRCR collects data of all patients diagnosed with adenocarcinoma in the colon or rectum. All patients are informed about their data being used and have the possibility to opt out. In 2017, the coverage was 97% both for colon and for rectal cancer. This study included CRC patients operated with curative colorectal resections during the years 2010–2016 diagnosed with synchronous or metachronous isolated pulmonary metastasis within the 5-year follow-up. Eligible patients operated in Skåne (Skåne University hospital and the regional hospitals of Helsingborg, Kristianstad, Ystad and Trelleborg) were identified from the SCRCR. Exclusion criteria were extrapulmonary metastases diagnosed before or at the same time as the lung metastases, lack or withdrawal of SCRCR consent, as well as incomplete medical records caused by part of the treatment being given outside the county of Skåne. Ethical approval was obtained from the Swedish Ethical Review Authority (DNR 2022-06592-01).
Lung metastases were defined as synchronous if diagnosed concomitantly with or within 6 months of the diagnosis of the primary CRC. Metastases diagnosed between 6 months after resection of the primary tumor and the end of the 5-year follow-up were considered as metachronous. For indeterminate lung nodules, the date of diagnosis was set to when the lung lesion was deemed as a metastasis.
In Sweden, the standard follow-up after curative CRC surgery includes CEA monitoring and thoracoabdominal CT scans at 1 and 3 years after surgery, as well as a colonoscopy at 3-years post-surgery. For stage IV patients treated with curative intent, follow-up is more intense with CT scans every 6 months for the first 2 years. After this, at least one more scan at 3 years is recommended for both liver and lung metastases. There is no age limit for follow-up; however, in cases where patients are considered too frail to tolerate additional surgical or medical treatment, follow-up may be omitted.
Disease-free interval (DFI) was defined as the number of months between curative resection for the primary tumor and the date of diagnosis of the lung metastasis.
Disease-free survival (DFS) was analyzed in patients with curative treatment intent and was defined as the number of months between curative treatment of the lung metastasis to recurrence, or death, or end of follow-up.
Progression-free survival (PFS) was analyzed in patients with palliative treatment intent and was defined as the number of months between the diagnosis of the lung metastasis and the progression of pulmonary metastasis burden or diagnosis of extrapulmonary metastases, or death.
Five-year OS was calculated from the date of lung metastasis diagnosis to death.
Clinical data
Clinical data such as gender, age, American Society of Anesthesiologists (ASA) physical status score, body mass index (BMI), primary tumor localization, date of surgery for the primary tumor, stage according to the TNM classification, and date of death were obtained from the SCRCR. The tumor T and N stages are based on the postoperative pathological staging after primary tumor removal. The M stage could be derived from pre- or postoperative scans as well as peri-operative findings. Complementary data such as carcinoembryonic antigen (CEA) levels, number and distribution of lung metastases, details regarding surgical and oncological treatment and treatment intent were obtained from the patients’ medical records.
Statistics
Data are presented as median with interquartile range (IQR) for continuous variables and nominal variables are presented as numbers and percentage. Continuous variables were analyzed using the Mann–Whitney U test and nominal data were compared using the Chi-square test.
Survival was estimated using the Kaplan–Meier method. Log-rank test was used to assess differences between groups. Patients were censored at the end of follow-up.
Multivariable analysis was performed using Cox-proportional hazard models to examine factors that influenced survival such as age, primary tumor location (colon or rectum), primary tumor stage (I–IV), DFI (months), number of lung metastases, and distribution of lung metastases (unilateral or bilateral).
The significance level was set to p < 0.05. Statistics were processed using SPSS version 27.0 and Stata Basic Edition version 18.0.
Results
Baseline characteristics
A total of 8457 patients treated for CRC in the county of Skåne during the period 2010–2016 were identified in the SCRCR, 6270 of which were operated with curative intent. Of these, 524 (8.6%) patients had registered lung metastases, 124 (2.0%) of which isolated lung metastases. After review of the medical records, 93 (1.5%) patients were included in the final cohort, 53 with synchronous and 40 with metachronous lung metastases. Patients were further subdivided into the curative or palliative group depending on the treatment intent of the lung metastasis/es (see Fig. 1).
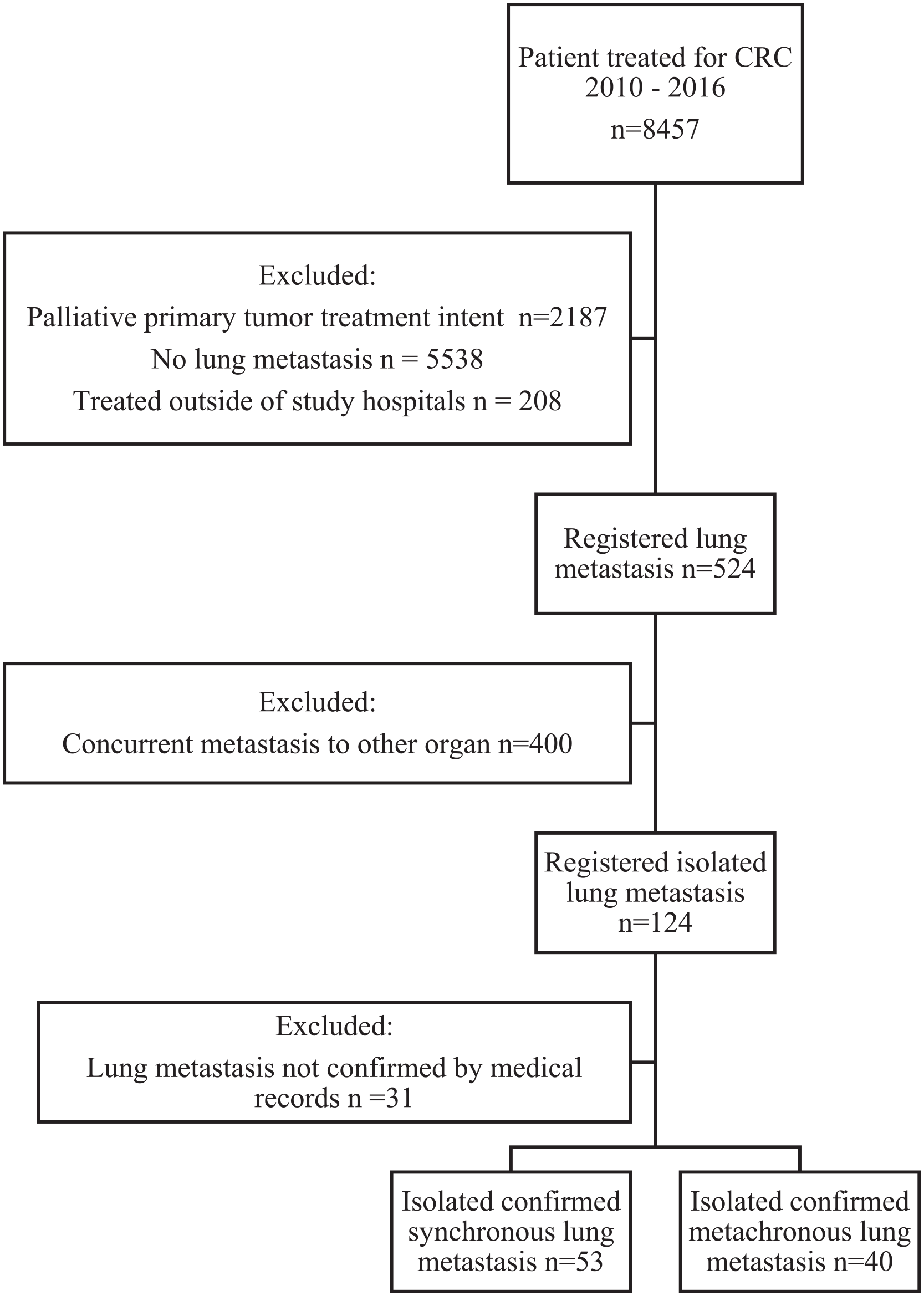
Flow chart of patient inclusion.
Patient and primary tumor characteristics are shown in Table 1. Patients with curative intent were younger, had lower ASA scores, lower preoperative CEA levels and had more often received neoadjuvant radiotherapy and adjuvant chemotherapy for the primary tumor. Regarding the lung metastases, patients with curative treatment intent were more likely to have unilateral distribution, a lower number of metastases, and a lower CEA at metastasis diagnosis.
Patient and tumor characteristics.
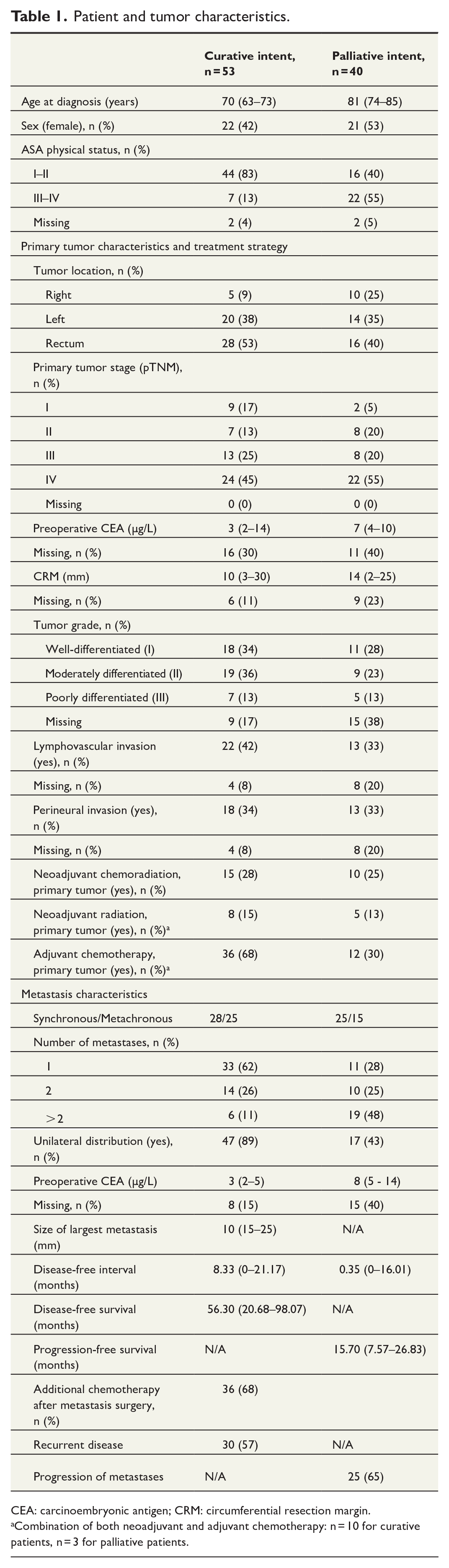
CEA: carcinoembryonic antigen; CRM: circumferential resection margin.
Combination of both neoadjuvant and adjuvant chemotherapy: n = 10 for curative patients, n = 3 for palliative patients.
Treatment strategies
Patients were categorized based on the treatment intent of the lung metastasis. Patients with curative treatment intent upon metastasis diagnosis were predominantly managed surgically (51/53, 96%). Of these, 36/53 (68%) received additional chemotherapy following pulmonary metastasectomy. The time from lung metastasis diagnosis to metastasectomy was 91 days (IQR: 68–140). Two patients were treated with curative stereotactic body radiotherapy (SBRT).
Palliative treatment was assigned to patients with either unresectable lung metastasis burden (n = 26), resectable metastases deemed inoperable due to comorbidities (n = 13), or patient’s desire (n = 1). Palliative patients received chemotherapy (n = 14), radiation (n = 1), or best supportive care (n = 25). No palliative surgical interventions for lung metastases were performed.
Survival analysis and factors associated with survival
Five-year OS was 42% (39/93), 50 months (IQR: 24–60) for the entire cohort. On a group level, 5-year OS was 68% (36/53), 60 months (IQR: 55–60) and 7.5% (3/40), 22 months (IQR: 12–33) for curative and palliative patients, respectively.
Survival over time is shown in Supplementary Figure 1. Patients treated for lung metastases with curative intent had longer survival than those in the palliative group (p < 0.001).
In multivariable analysis, age (hazard ratio (HR): 1.04, confidence interval (CI): 1.01–1.07), multiple metastases (HR: 1.64, CI: 1.08–2.47), and unilateral distribution (HR: 0.41, CI: 0.20–0.84) were associated with overall survival, whereas primary tumor location (HR: 0.64, CI: 0.38–1.09) and stage (HR: 1.15, CI: 0.78–1.72) as well as the DFI (HR: 1.0, CI: 0.99–1.00) were not.
Disease progression
During follow-up, 30/53 (57%) curatively treated patients had a recurrence of metastasis. Locations were as follows: liver (n = 5), lung (n = 8), abdominal cavity (n = 2), or multiple (n = 15).
Of these, 23 had lung involvement, either isolated (n = 8) or in combination with other distant metastases (n = 15). Most patients (18/23, 78%) underwent repeat pulmonary metastasectomy. A separate survival analysis was conducted on curative patients 5-year OS. Patients were divided into three groups: no recurrence (n = 23), isolated lung recurrence (n = 8), and extra-pulmonary recurrence (n = 22). Kaplan–Meier estimated 5-year OS is shown in Supplementary Figure 2. Details regarding the second metastasis surgery is shown in Supplementary Table 1.
In the palliative group, 25/40 (63%) developed extra-pulmonary metastases with the following locations: liver (n = 6), lung (n = 4), abdominal cavity (n = 2), central nervous system (n = 2), bone (n = 1), local recurrence (n = 1), or multiple (n = 9).
Discussion
This study demonstrates that isolated lung metastases are rare among patients treated with curative resection of CRC (1.5%). The curative treatment strategy employed in this cohort was aggressive with most patients undergoing surgery, often in combination with neoadjuvant and adjuvant oncological treatment regimes. The results show that long-term DFS can be achieved after curative treatment of isolated lung metastases. Patients treated with curative intent had a considerably better 5-year OS than patients treated with palliative intent (68% vs 8%). However, it remains unclear how much the treatment approach and how much patients selection contributed to this difference.
The incidence of isolated lung metastases (1.5%) in this study was somewhat lower than in previous studies. A population-based study from France between the years 1976 and 2005 reported an incidence of 4.3% for synchronous isolated lung metastases. 9 An Australian study comprising 2006 patients diagnosed between 2006 and 2011 reported isolated lung metastases in 7.1% of patients, with a more favorable prognosis compared to other single-site metastases. In this study, 4-year survival in surgically treated patients was 89%, compared to 49% in patients with non-surgical treatment of the lung metastases. 10 The COLOFOL study that investigated metachronous lung recurrence in 2442 patients from Sweden, Denmark, and Uruguay between 2006 and 2010, reported an incidence of 3.6% for isolated lung metastases. Among these, 60% underwent surgical treatment with curative intent, closely mirroring the 57% observed in this study. In addition, findings consistent with the COLOFOL study included 5-year survival for curatively treated patients and recurrence of metastasis after metastasis surgery 63% and 58% vs 68% and 57% in this study. 11 A larger study including 43,271 US CRC patients during 2010–2015 reported isolated synchronous lung metastases in 3.6% of patients. In this cohort, median survival in surgically treated patients was 49 months compared to 19 months in those managed non-operatively. 12 The reason for the lower-than-expected incidence of isolated lung metastases found in this study is unknown. It is possible that some lung metastases were undetected as only the records with a reported lung recurrence were reviewed. Another reason could be the lack of review of medical charts and pathology reports in other studies. In this study, this review excluded roughly 10% of reported isolated lung metastases as the diagnosis could not be confirmed in the medical records. Furthermore, a recent validation study of the SCRCR reported wrongly registered synchronous metastases in 3.6%, meaning that some isolated pulmonary metastases might have been hidden among these. 13
Several studies examining survival following surgical resection of CRC lung metastases exist. The reported 5-year OS rate varies significantly between comparably recent studies (40%–78%),14 –16 reflecting the heterogeneous population and likely the impact of patient selection and different treatment strategies. Previous studies investigating survival following curative metastasis resection have reported similar 11 or superior 10 long-term survival compared to this study. Suggested predictors of poor survival have included higher age, more advanced primary tumor stage, rectal tumor, shorter DFI, elevated preoperative CEA level, multiple lung lesions, bilateral distribution, lymph node involvement, and metastasis size.17 –20 In this study, age, number of metastases, and unilateral distribution were associated with survival, whereas DFI, primary tumor location and stage were not. It is likely that younger age generally lead to more aggressive surveillance and a higher tolerance for both oncological and surgical treatment, which might explain better outcomes. It also seems intuitive that less widespread metastatic disease, that is, solitary lesion or lesions confined to one lung would be beneficial, either due to earlier diagnosis and treatment or due to a more favorable tumor biology. CEA level, lymph node involvement, and size of the metastases could not be analyzed due to a large fraction of missing data for these variables. CRC lung metastases can be treated curatively with stereotactic radiation instead of surgery. In this study, only two patients received this treatment, and meaningful conclusions cannot be drawn. However, it remains a viable treatment option, especially for frail patients not suitable for surgery. Previous studies have shown comparable short-term survival rates 21 although other studies suggest that surgery might provide better long-term survival. 22
Most lung metastases in this study were found to originate from the left colon or rectum, a finding consistent with previous reports.23,24 This disparity is believed to stem from variations in embryological origin as well as molecular, genetic, and immunological characteristics. Some studies suggest a poorer survival for lung-metastasized CRC when the primary tumor is in the rectum compared to the colon; however, this presumed relationship is likely multifaceted15,25,26 and was not investigated in this study as the low number of patients in each group would prevent any meaningful analyses.
Patients with rectal tumors are more likely to receive neoadjuvant treatment compared to colon tumors. The choice of neoadjuvant therapy has been suggested to influence not only the risk of local recurrence but also the pattern of metastasis. CRC lung metastases have been shown to exhibit chemoresistance more frequently than other sites, perhaps due to tissue-specific characteristics. 27 In one previous study, the lung was the predominate site of metastasis in rectal tumors treated with both neoadjuvant CRT and adjuvant chemotherapy, 28 while in a recent trial, neoadjuvant chemotherapy decreased the occurrence of liver metastases whereas no change was observed for lung metastases. 4
Roughly half of the patients treated with a curative intent had a pulmonary recurrence during follow-up. Repeat lung resection was performed in most cases, further emphasizing the aggressive treatment strategy in these patients. Previous studies on repeat lung resection have shown comparable 5-year OS rates of 75%, with especially favorable outcomes for those without extra-pulmonary disease. 15 In this study, all patients were referred to the same cardiothoracic department for preoperative evaluation and surgery. Hence, no differences in the aggressiveness of surgical treatment are expected between patients from different referring hospitals included in the study. A previous nation-wide study on Swedish CRC patients with both liver and lung metastases demonstrated significant differences in surgical treatment strategy between different healthcare regions. 29 It would, therefore, be of interest to expand the study population to investigate whether the same is true for isolated lung metastases.
The study included all consecutive patients of the SCRCR diagnosed with isolated lung metastases in the county of Skåne during the study period. This provides a real-world perspective on treatment strategies employed and demonstrates results comparable to other studies. However, the study has several limitations. It is retrospective and registry-based, inherently susceptible to biases associated with these designs. Furthermore, there was a significant amount of missing data, precluding analyses of important predictor variables such as CEA value. The extent of missing data surpassed the acceptable limit for statistical methods like imputation. Furthermore, the observational design of the study makes any conclusions on the causality of differences in survival impossible. Frankly, the observed associations between the therapeutic approach and survival may be the result of tumor biology and patient selection. In addition to disease burden, many other contributing factors could confound the results. For instance, in the palliative group, nearly one-third of patients had resectable metastases but were deemed inoperable due to their general condition, which naturally also affects the overall survival in this group. Due to the low number of palliative patients, no further subgroup analysis such as comparing palliative oncological treatment to best supportive care was performed.
Conclusion
Isolated lung metastases from CRC are rare. Although curative treatment was associated with better survival than palliative treatment, a majority of patients recurred which impaired survival, especially if extra-pulmonary organs were affected. In multivariate analysis, age, solitary lesions, and a unilateral distribution were predictors of survival.
Supplemental Material
sj-docx-1-sjs-10.1177_14574969251319849 – Supplemental material for Incidence, treatment, and survival of patients with isolated colorectal lung metastases: A registry-based retrospective cohort study
Supplemental material, sj-docx-1-sjs-10.1177_14574969251319849 for Incidence, treatment, and survival of patients with isolated colorectal lung metastases: A registry-based retrospective cohort study by Sofia Dahlberg, Fredrik Jörgren, Pamela Buchwald and Halla Vidarsdottir in Scandinavian Journal of Surgery
Footnotes
Data availability
Anonymous data are available and can be provided upon request.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by a grant from the Foundation of Stig and Ragna Gorthon, Helsingborg, Sweden
Ethical considerations
Ethical approval was obtained from the Swedish Ethical Review Authority (DNR 2022-06592-01).
Consent to participate
All study participants have consented to be included in the registry and that registry data are analyzed and published for research purposes.
Consent for publication
Not applicable.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
