Abstract
Purpose:
Resectability assessment of patients with colorectal liver metastases is based on computed tomography and liver magnetic resonance imaging. Addition of fluorine-18-fluorodeoxyglucose positron emission tomography/computed tomography has been recommended, but the impact of the added information remains unclear. The primary aim of this study was to determine how preoperative positron emission tomography/computed tomography changed management in patients with potentially resectable colorectal liver metastases. The secondary aim was to investigate whether findings on positron emission tomography/computed tomography correlated to metastatic disease in cases with extended surgery and influenced oncological outcomes.
Methods:
A retrospective observational study of the impact of adding positron emission tomography/computed tomography to conventional imaging in the surgical decision-making of colorectal liver metastases. All patients with colorectal liver metastases diagnosed by conventional imaging were included and assessed by a multidisciplinary team conference at Umeå University Hospital between June 2013 and December 2017. Eligibility criteria were all patients with potentially resectable colorectal liver metastases. Patients who underwent preoperative positron emission tomography/computed tomography in addition to conventional radiology were compared with those who underwent conventional imaging only.
Results:
151/220 patients underwent preoperative positron emission tomography/computed tomography. Findings on positron emission tomography/computed tomography changed the management in 10.6% of the patients. Eight patients were excluded from surgery after detection by positron emission tomography/computed tomography of extrahepatic disease. Eight patients underwent more extended surgery than initially planned due to positron emission tomography/computed tomography. Five of these positron emission tomography-positive resected sites were verified by pathology as metastatic disease. No difference in overall survival was seen following surgical resection in patients with and without a preoperative positron emission tomography/computed tomography.
Conclusions:
Preoperative positron emission tomography/computed tomography resulted in a changed surgical management in 10.6% of cases in a selected cohort.
Introduction
Colorectal cancer (CRC) is one of the most common cancers globally and the incidence was estimated at 1.8 million new cases in 2018. 1 Metastases to the liver are diagnosed in one-third of patients. 2 Patients with resectable colorectal liver metastases (CLM) can achieve a long-term cure with liver surgery, often in combination with oncological treatment. The reported 5-year overall survival (OS) for these patients exceeds 50%.3 –5 The extent and location of intrahepatic disease and the presence of extrahepatic spread examined by preoperative imaging significantly affect the chances of improving patient survival by radical surgical resection. Patients with CLM are assessed by conventional imaging using abdominal and thoracic contrast–enhanced computed tomography (CT) and by liver magnetic resonance imaging (MRI) with liver-specific contrast. The combination of CT and MRI is used to detect extrahepatic disease (EHD) and to map liver metastases, respectively. 6 At surgery, it is reported that up to 5% of patients have technically irresectable CLM, or widespread EHD, despite an extensive radiological evaluation. 7 To select the patients that might benefit from liver resection, it is essential that the preoperative evaluation accurately assesses the tumor stage and metastatic involvement.
Fluorine-18 fluorodeoxyglucose (18F-FDG) positron emission tomography (PET)/CT may reveal additional EHD compared with CT, thus excluding patients from surgery. 8 However, the use of PET/CT in the routine preoperative evaluation of CLM is under debate. Observational studies suggest a change in patient management of between 8% and 43%.9 –20 A recently published meta-analysis based on two randomized trials and 11 retrospective studies concluded that PET/CT changed the management of patients by between 8% and 20%. 21 In spite of this, the authors did not find any OS benefit from performing PET/CT, and there was no significant reduction in open–close surgery.
The European Society for Medical Oncology (ESMO) guidelines from 2016 advocate the stepwise utilization of imaging resources based on the low-level evidence of incorporating PET/CT in routine staging. 6 During this study period, national guidelines recommended PET/CT for all patients diagnosed with CLM 22 or for patients with CLM who were being considered for curative resection. 23
The primary aim of this study was to evaluate the impact of adding preoperative PET/CT to conventional radiological examinations on the management of patients with potentially resectable CLM. The secondary aim was to evaluate whether PET/CT positive findings correlated with metastatic disease in cases with extended surgery and whether PET/CT affected oncological outcomes after curatively intended surgery.
Material and methods
Study design
Umeå University Hospital is a tertiary hepatobiliary center in Sweden. Patients with CLM are discussed at a multidisciplinary team (MDT) conference with colorectal surgeons, liver surgeons, radiologists, oncologists, and pathologists. Patients with potentially resectable CLM were studied retrospectively.
The inclusion criterion for this study was patients evaluated as potentially resectable CLM (including ablation) based on conventional imaging (abdominal and thoracic CT and/or liver MRI and/or contrast-enhanced ultrasound of the liver) at the initial MDT between 1 May 2013 and 31 December 2017. A preoperative liver MRI was clinical routine, but when contraindicated (e.g. medical implants not compatible with MRI, body constitution, or claustrophobia), an abdominal CT or contrast-enhanced ultrasound of the liver was performed to assess resectability. The national guidelines recommended PET/CT in newly diagnosed CLM 22 or in all patients potentially or up-front resectable 23 during the study period. The exclusion criterion was patients deemed as not being resectable based on conventional imaging, low performance status, or comorbidities. Patients receiving preoperative oncological therapy were discussed at additional MDTs with follow-up radiological examinations to assess new information and evaluate treatment effect and resectability. The last follow-up MDT is referred to as the final treatment decision. We divided the included patients into two groups based on whether or not they underwent a preoperative PET/CT.
Clinical data were retrieved from medical charts. The dates of death were collected from the Swedish population register. The Fong Clinical Risk Score was calculated (node-positive primary tumor, disease-free interval from primary to metastases < 12 months, number of CLM > 1, largest CLM > 5 cm, carcinoembryonic antigen (CEA) > 200 µg/L) and the patients were divided into high3 –5 and low (0–2) scores, according to Fong et al. 24 The two groups were compared for potential differences in prognostic factors and OS was calculated for patients undergoing curatively intended surgery. The TNM stage of the primary tumor was determined from the pathological report in the case of chemonaive CRC and from the radiologist’s staging of MRI on preoperatively radiotherapy-treated rectal cancer or CT on chemotherapy-treated colon cancer. At the time of the final treatment decision, the EHD status was documented according to all available information, including radiological information. Abdominal and thoracic CT, liver MRI, and PET/CT data were collected from the Picture Archiving and Communication System (PACS) and radiological information system (RIS). All preoperative radiological examinations were reviewed by a minimum of two trained radiologists and nuclear medicine specialists at Umeå University Hospital, according to clinical routines. All resected specimens were examined by a pathologist specializing in gastrointestinal pathology.
Conventional imaging
A routine clinical assessment of patients with CRC includes a CT in the supine position with the arms raised above the head, from the orbitomeatal plane to the proximal thighs. The image acquisition is made with a 64-slice CT scanner (GE Healthcare, Milwaukee, WI, USA) with intravenous contrast and an image reconstruction of 2.5 mm slice thickness. A pelvic MRI was performed with a 1.5T MRI scanner (Philips Healthcare) with a standardized rectal cancer protocol in line with the European Society of Gastrointestinal Radiology consensus guidelines. The standard pulse sequences consisted of multiplanar two-dimensional (2D) T2-weighted fast spin-echo (transaxial, sagittal, and coronal) with 3–4 mm slice thickness, a high-resolution oblique sequence perpendicular to the rectal tumor with a slice thickness of 3 mm and a transaxial diffusion-weighted sequence.
If CLM was suspected, patients underwent a liver MRI with a 1.5T MRI scanner, according to standard protocols using liver-specific contrast (PrimovistTM). The patients fasted for 4 h before the examination and were not allowed to drink any fluids 1 h before the examination. The examination was performed with a routine MRI liver protocol, including one 0.1 ml/kg Gadolinium (Gd) contrast scan, with contrast administered intravenously at a rate of 1 mL/s. The three sequences that were used in this study are called Gd-e THRIVE BH SENSE, performed in axial, sagittal, and coronal views, and they were acquired in the late hepatocellular contrast phase performed one after another with an interval of 1 min. A contrast-enhanced ultrasound of the liver was performed on patients that were unable to undergo MRI for medical reasons if the CT was inconclusive.
18F-FDG PET/CT protocol and reconstruction
18F-FDG PET/CT was performed with a Discovery 690 PET/CT with a 64 slice CT (General Electric, WI, USA). The patients had been fasting for 6 h prior to the examination. Weight, height, and blood glucose levels were obtained. Static PET/CT scans were acquired from the proximal femur to the base of the skull with the arms above the head using a clinical routine protocol. The CT was acquired with 120 kV, 0.5 s tube rotation time, 2.5 mm section thickness, 2.5 mm section interval and 30–160 mA automatic tube-current modulation (AutomA). PET acquisition started 60 min after the injection of 18F-FDG. Attenuation-corrected images were created using the Vue Point FX reconstruction supplied with the scanner, employing a 6.4 mm filter cut-off, two iterations, 24 subsets.
Impact on management
The added value of PET/CT was defined as imaging results that changed the management because of new findings not revealed in conventional preoperative radiological examinations. Two hepatobiliary surgeons (O.H. and H.N.) independently and retrospectively assessed information from imaging reports and medical charts from MDTs to determine whether PET/CT changed the management. Changed management was classified as canceled surgery, a change in surgical approach with more extensive hepatic surgery, or a widened surgical operating field with the removal of limited suspected EHD.
Statistical analysis
Statistical analyses were performed using IBM SPSS Statistics version 25.0 (Mac OS X, Armonk, New York, USA: IBM Corp.) and R 4.0.2. 25 The chi-square test of association or Fisher’s exact test were used to detect differences in binary variables. Quantitative variables were analyzed for normal distribution. The Mann–Whitney U test was used to detect differences in quantitative variables. Survival rates were estimated using the Kaplan–Meier method. The Kaplan–Meier estimate was calculated from the date of surgery to death when comparing OS for patients undergoing curatively intended liver surgery. The two-sample multiple-direction logrank test in the R package mdir.logrank was used to determine statistically significant differences in survival rates. 26 Patients who were alive were censored at the date of the last follow-up, 19 September 2019. Median follow-up was calculated from the date of surgery to the date of the last follow-up. Statistical significance was defined as a p-value less than 0.05.
Research ethics
This study was conducted in agreement with the Declaration of Helsinki and was approved by the Swedish Ethical Review Authority (DNR 2009-175, DNR 2019-05690).
Results
Characterization of the cohort
Characteristics of all patients with potentially resectable CLM
A total of 293 patients with CLM were assessed at a regional MDT conference. Patients deemed unresectable based on conventional abdominal CT and/or liver MRI were excluded (n = 73). Patients with CLM deemed potentially resectable on conventional imaging (n = 220) at the initial MDT were included, based on the intention to treat, see the flow chart of inclusion in Fig. 1. One hundred and fifty-one (69%) patients had a preoperative PET/CT performed in addition to the conventional radiological examination. Fifty-seven of 90 (63%) patients in first half of the study period (2013–2015), and 94 of 130 (72%) patients in the second half of the study period (2015–2017) underwent preoperative PET/CT (p = 0.158). Seven patients did not undergo liver MRI in the PET/CT group, and four patients in the conventional imaging group. The reasons for not evaluating the patient with MRI included a high body mass index (BMI; n = 4), pacemaker (n = 4), claustrophobia (n = 1), ear bone prosthesis (n = 1), and spine prosthesis (n = 1). Among patients assessed as potentially resectable at the initial MDT, 22 patients in the PET/CT group and 5 in the conventional imaging group did not undergo surgery (Fig. 1).
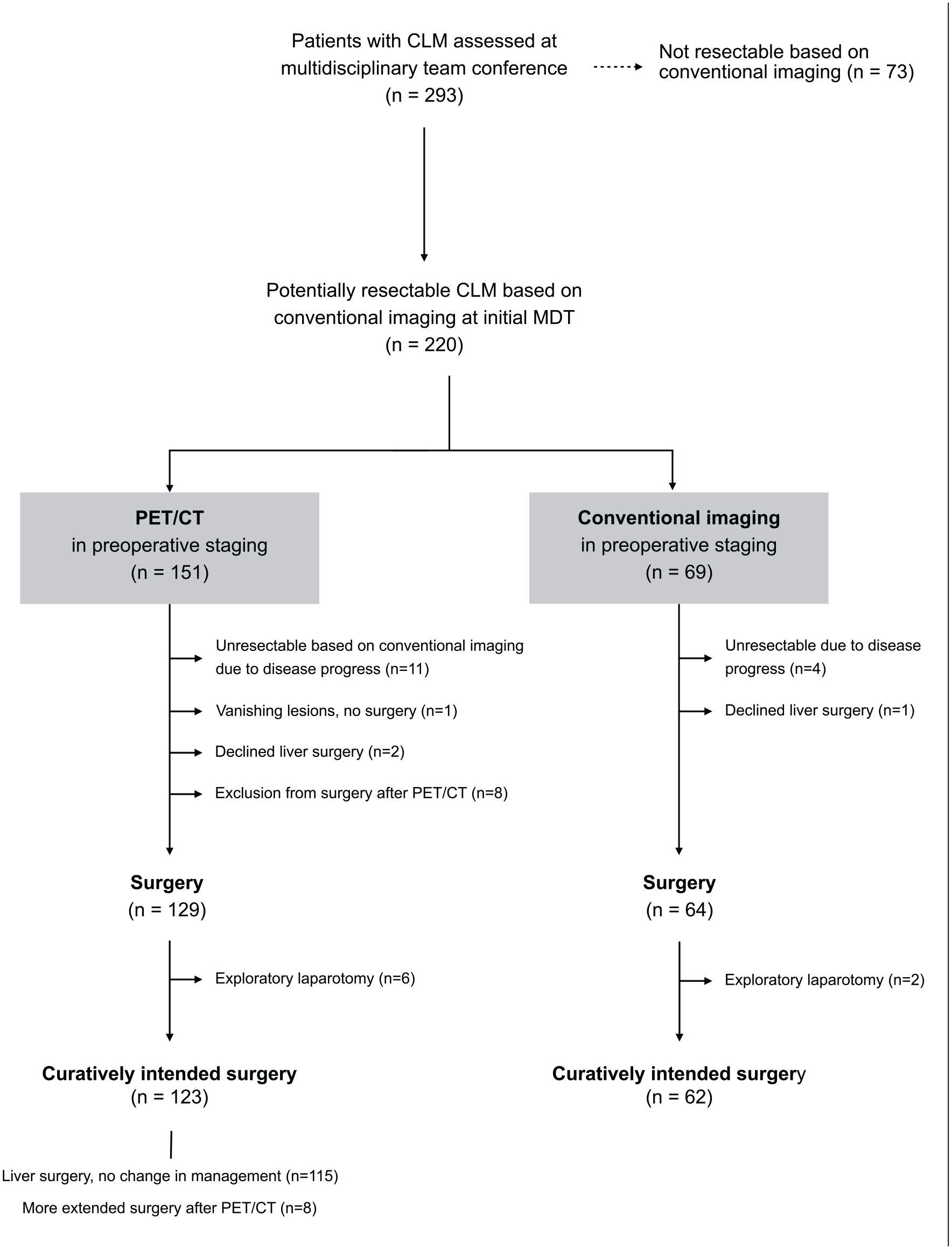
Flow chart of inclusion and exclusion and the management of patients with potentially resectable colorectal liver metastases.
The clinical characteristics of all patients with potentially resectable CLM are presented in Table 1 and Supplementary Table 1. Patients evaluated with PET/CT were younger (p = 0.035), had a higher number of CLM (p = 0.009), more frequent bilobar distribution (p = 0.010), and suspected EHD at initial MDT (p = 0.020).
Characteristics of all patients with potentially resectable colorectal liver metastases at initial MDT.
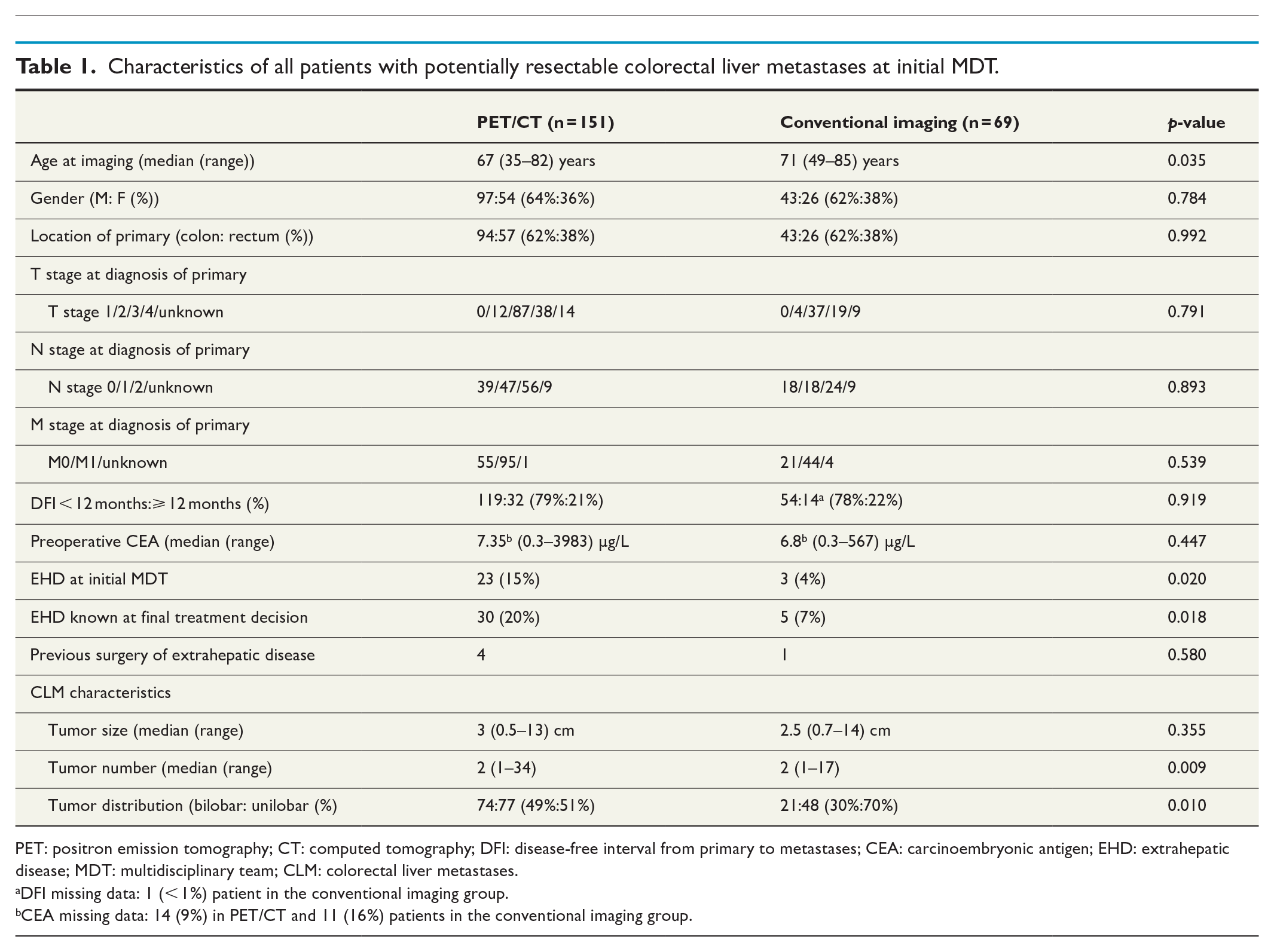
PET: positron emission tomography; CT: computed tomography; DFI: disease-free interval from primary to metastases; CEA: carcinoembryonic antigen; EHD: extrahepatic disease; MDT: multidisciplinary team; CLM: colorectal liver metastases.
DFI missing data: 1 (< 1%) patient in the conventional imaging group.
CEA missing data: 14 (9%) in PET/CT and 11 (16%) patients in the conventional imaging group.
The anatomical localization of suspected EHD at the time of the final treatment decision for patients with potentially resectable CLM is presented in Supplementary Table S2A. Six (4%) patients in the PET/CT group and two (3%) patients in the conventional imaging group only underwent an exploratory laparotomy (p = 1.000). The liver surgery was canceled due to peritoneal carcinomatosis (four in the PET/CT and one in the conventional imaging group) and the progress of liver metastases (two in the PET/CT and one in the conventional imaging group). To summarize, PET/CT showed false-negative results in six patients, and two patients had false-negative results in the conventional imaging group.
Characteristics of patients undergoing curatively intended surgery
A total of 185 (84%) patients underwent curatively intended liver surgery, 123 (82%) in the PET/CT group and 62 (90%) in the conventional imaging group (p = 0.114). The characteristics of patients undergoing curatively intended surgery are presented in Table 2 and Supplementary Table 3. The patients in the PET/CT group were significantly younger (p = 0.041), had a higher number of CLM (p = 0.005), a higher proportion of bilobar distribution (p = 0.007), and suspected EHD at initial MDT (p = 0.047) compared with the conventional imaging group. Ablation therapy was used more frequently in the conventional imaging group (p < 0.001), see Supplementary Table 4.
Characteristics of patients undergoing curatively intended liver surgery including minor resection of suspected extrahepatic disease.
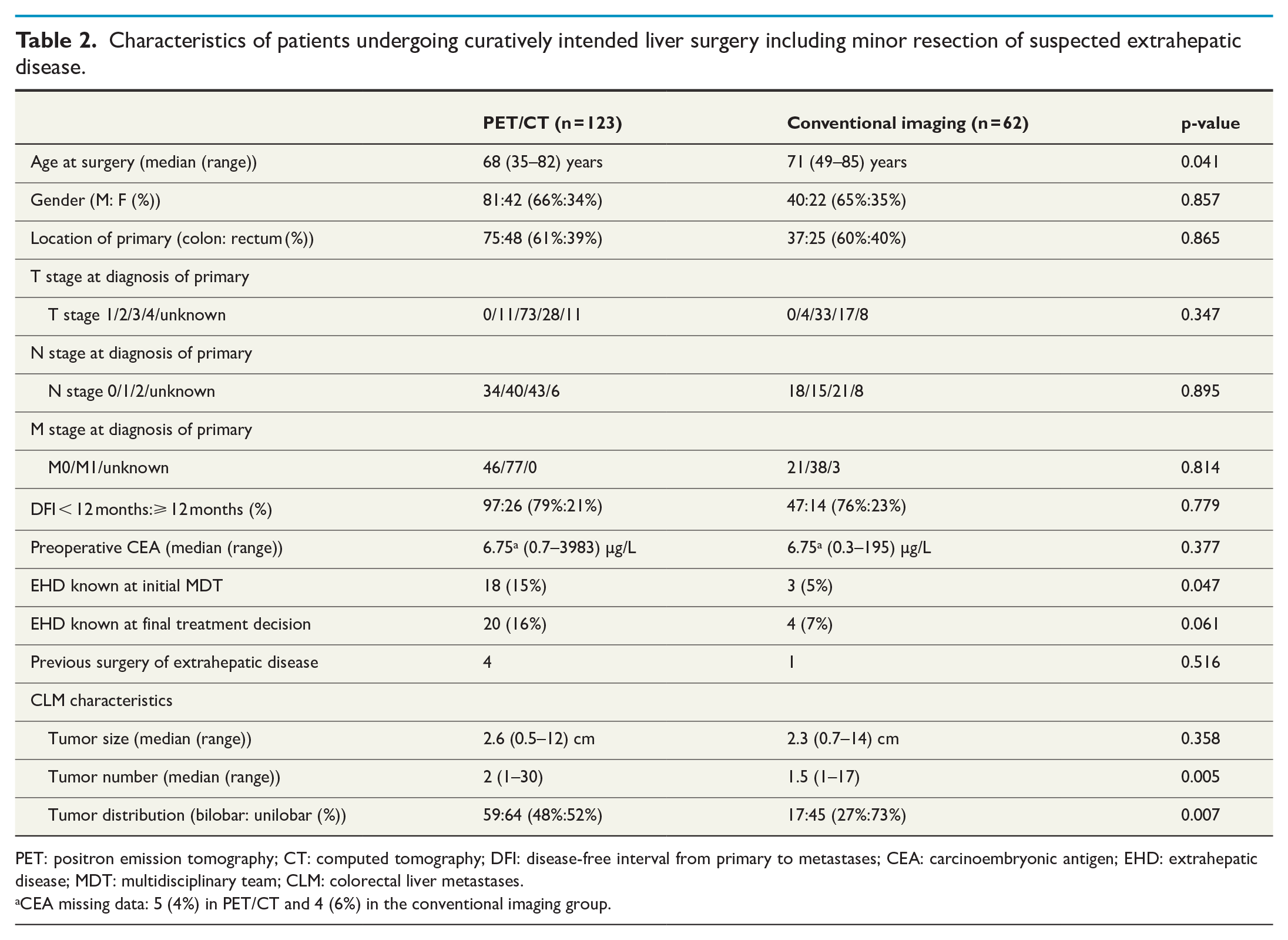
PET: positron emission tomography; CT: computed tomography; DFI: disease-free interval from primary to metastases; CEA: carcinoembryonic antigen; EHD: extrahepatic disease; MDT: multidisciplinary team; CLM: colorectal liver metastases.
CEA missing data: 5 (4%) in PET/CT and 4 (6%) in the conventional imaging group.
A total of 20 patients in the PET/CT group and four patients in the conventional imaging group had suspected EHD at the time of the final treatment decision (localization of EHD described in Supplementary Table 2B). Without discontinuation of the liver surgery, there were seven (6%) patients in the PET/CT group presenting with previously undetected EHD intraoperatively. The suspected EHD in these patients comprised minor loci of peritoneal carcinomatosis (n = 3), hepatic regional lymph node metastases (n = 2), and tumor overgrowth to the diaphragm (n = 2). All the EHD sites in these patients were resected and verified as CRC. Thus, PET/CT was false-negative regarding EHD in these seven cases. No patients in the conventional imaging group undergoing curative surgery had intraoperative findings of not previously known EHD.
No difference in OS was found between patients undergoing curatively intended surgery in the PET/CT compared with the conventional imaging group (p = 0.509), see Fig. 2. The median follow-up was 29 months (range 2–71 months) in the PET/CT group and 29 months (range 4–72 months) in the conventional imaging group. At the end of the study, 75 of 123 (61%) patients in the PET/CT group were alive compared with 34 of 62 (55%) in the conventional imaging group (p = 0.423).
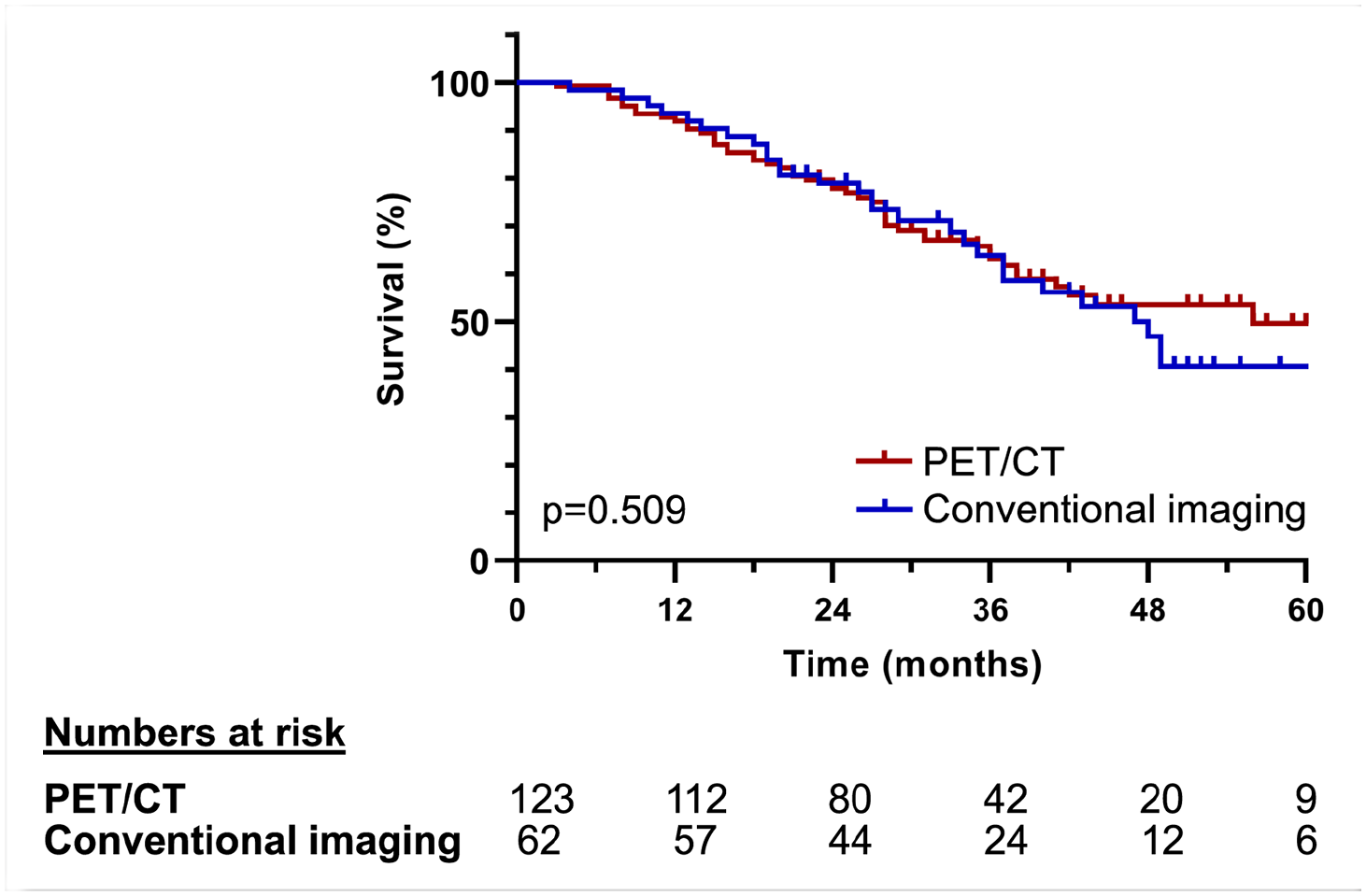
Overall survival for patients with PET/CT and conventional imaging only in the preoperative investigation undergoing curatively intended liver surgery including the resection of minor extrahepatic disease.
Impact on clinical management and changes in surgical approach after PET/CT
Additional findings on PET/CT changed the clinical management in 16 (10.6%) of 151 patients with potentially resectable CLM examined with PET/CT. Eight (5.3%) patients were omitted from curative liver surgery following the detection of unresectable EHD. The findings of EHD in this group consisted of multiple lymph node metastases (n = 3), pleural metastasis (n = 1), metastatic spread to the abdominal wall and spleen (n = 1), skeletal metastasis (n = 1), progress of liver metastases to the extent of irresectability (n = 1), and peritoneal carcinomatosis (n = 1). The MDT decision to exclude patients from liver surgery due to PET/CT positive findings was supported by contributing risk factors including disease progress on chemotherapy or high tumor burden (n = 3), evidence of correlating findings on reviewed conventional imaging (n = 3), and/or further investigation with biopsy (n = 1) or additional radiology (n = 1).
The surgical approach was changed to an extended intervention in eight (5.3%) patients due to PET/CT. This was based on metabolically active lymph nodes (n = 5), new resectable liver lesions (n = 2), or suspected recurrent disease in the abdominal wall (n = 1). The removal of suspicious limited regional lymph nodes was performed in five patients simultaneously with the liver resection. The resection was performed on lymph nodes located by the hepatoduodenal ligament (n = 2), at the paraaortic region (lymph node at the left iliac artery after rectal cancer (n = 1) and near the superior mesenteric artery (n = 2) One metabolically positive focus at the anterior abdominal wall was resected, one extended hepatic resection, and one additional ablation were performed after PET/CT.
Correlation of positive PET findings with histology after extended surgery
Due to findings on PET/CT, a total of eight patients underwent extended surgery. The histology of resected regional lymph nodes showed: CRC (n = 3), follicular lymphoma (n = 1), and benign tissue (n = 1). An implantation metastasis of CRC was confirmed in the metabolically positive focus at the anterior abdominal wall (n = 1). Similarly, the additional resected CLM was confirmed as CRC (n = 1). No surgical specimen was available for analysis for the patient undergoing additional ablation. The preoperative PET/CT finding could thus be correlated to metastatic CRC in five of seven patients with tissue specimens available for histology.
Discussion
This retrospective study found that the addition of preoperative PET/CT changed the management in 10.6% of patients, leading to more extended surgery in 5.3% and excluding 5.3% from surgery. Based on our findings, a quarter of the initial cohort was deemed unresectable on conventional CT and/or liver MRI, comparable with numbers presented in a study by Yip et al. 27 Depending on study design, previous reports in the literature have suggested that preoperative PET/CT changes treatment strategy by up to 43% of patients with CLM deemed resectable on conventional imaging.9 –20 The majority have small or heterogeneous cohorts mixing both patients with known and suspected recurrent CRC located in the liver and lungs. Some studies have not specified if an MDT discussed the patients preoperatively.9,11,13,15,18 The impact of PET/CT on the outcome is likely affected by the criteria for PET/CT and timing to conventional imaging and MDT. PET/CT probably influence management more frequently in a setting where patients are first assessed at an MDT and then referred to PET/CT only in selected cases.
In our study, PET/CT upstaged 5.3% of the cohort from having resectable to unresectable disease. This result is in the lower range of the previously reported (2.7%–33.8%).10 –20 Our results comply with a randomized trial performed in 2014 by Moulton et al. 17 in which the addition of PET/CT changed management in 8% of the cohort. In their study, only 2.7% were excluded from surgery preventing exploratory laparotomy compared with 5.3% in our study. Our study had a selection of patients with higher tumor burden to PET/CT, which could explain discrepancies in the exclusion frequency between ours and the previous retrospective studies. PET/CT changed the surgical strategy in one-tenth of the patients in the PET/CT group. We hypothesize that this rate would likely be lower if all 220 patients underwent PET/CT, including patients with low suspicion of EHD.
Our results showed that PET/CT had potentially false-negative findings in a total of 13 (8.6%) patients. The intraoperative findings in these cases resulted in six exploratory laparotomies and seven extended surgeries. A limitation regarding the false-negative results in this study is to differ disease progression over time that could have been detected with radiology to actual false negativity if PET/CT had been performed very close to surgery. Since we did not have the data to adjust for this, the results should be interpreted cautiously.
One potential benefit of PET/CT is a reduction in open–close surgery, concurrently avoiding unnecessary surgery for the patient and reducing costs. Serrano et al. 28 did not find a cost reduction when including PET/CT in the preoperative investigation compared with only conventional imaging. In line with the study by Moulton et al., 17 we did not identify any difference in the rate of open–close laparotomies between our groups.
One-third of the 220 patients with potentially resectable CLM at conventional imaging did not undergo PET/CT. During the study period, national guidelines recommended PET/CT in all patients with confirmed CLM, or at least in all patients with CLM where curative intent was the target.22,23 One important limitation of this restrospective study is that we did not have access to comprehensive information for the decision to perform PET-CT or not. Our results showed that not all patients underwent preoperative PET/CT and that patients without PET/CT had more favorable prognostic factors in comparison. Despite an expected lower OS in patients with more advanced disease in the PET/CT group, we did not detect a difference in OS between the two groups with and without PET/CT. One important reason for this might be that we did not have data to calculate disease-free survival. There might be a higher proportion of patients with more advanced disease-developing recurrence later. Also, we cannot attribute the OS similarity to PET/CT since the groups are dissimilar in the type of operative intervention performed, with a higher rate of ablative procedures in the conventional imaging group.
The now revised national guidelines advocate the more selective use of preoperative PET/CT according to recommendations given by ESMO. 6 ESMO suggests that patients considered having a high risk of EHD should have a PET/CT, although there is no consensus on the selection criteria for PET/CT. In this study, patients who underwent PET/CT had a higher rate of known EHD at the initial MDT, a higher number of metastases, and bilobar distribution. Due to the retrospective nature of our study, we cannot identify explicit patient criteria for selecting patients that should undergo PET/CT. However, we can hypothesize that patients with a low risk for EHD probably may not benefit from staging with PET/CT and that patients with a high risk for EHD may. In addition, patients with increased risk for EHD who are subject to preoperative oncological therapy could likely be staged more accurately with a baseline PET/CT. The long-term follow-up of the randomized trial by Moulton et al. did, however, not reveal any differences in disease-free survival or OS questioning the existence of PET/CT in the preoperative examination of potentially resectable CLM. 29 Half of their cohort had received chemotherapy within 12 weeks of surgery. 17 Since chemotherapy reduced the diagnostic accuracy of PET/CT, 30 it would be interesting to conduct a randomized study to evaluate whether a baseline PET/CT can improve selection in patients planned for neoadjuvant or conversion chemotherapy. To our knowledge, this has not been investigated in the setting of CLM before. The above-mentioned results highlight the need for precise indications for PET/CT in the preoperative investigation of metastatic CRC.
In five (3.3%) patients, metabolically active lymph nodes were detected on PET/CT, a finding which subsequently changed the surgical approach. EHD is considered a relative contraindication to liver resection, and debulking surgery remains controversial in CLM. Improvements in surgical technique and systemic oncological treatment may justify liver surgery in selected cases with EHD. 5 It is still uncertain whether patients with CLM and lymph node metastases achieve a higher survival rate with lymph node resection. Observational studies evaluating this topic have reported a 5-year OS of 16.2%–21%.3 –5 Final histology revealed that two of five patients with suspected lymph node metastases did not have any metastatic spread of CRC to lymph nodes, despite positive findings on PET/CT. This result disputes the specificity of PET/CT and raises the question of whether excluding a patient from potentially curable surgery based on an increase in metabolic activity in isolated lymph nodes on PET/CT is justified. Maffione et al. 8 presented false-positive PET findings in 3.1% of cases. In the randomized study by Serrano et al. 29 metabolically active portal lymph nodes were not viewed as a contradiction to liver surgery. Due to the risk of false-positive findings on PET/CT and a potential survival benefit for patients, it seems that limited PET-positive findings of patients with CLM, especially in the lymph nodes, should likely not be viewed as a general contradiction to surgery.
One limitation of this study is its retrospective nature, and it is thus subject to selection bias. Based on the results, it is apparent that patients with more advanced disease underwent PET/CT compared with those who did not. In addition, the disease distribution between the groups makes the timing to PET/CT different since some patients receive surgery upfront, and some receive preoperative chemotherapy. Considering the study is in a tertiary setting, we did not have access to all the data needed to calculate this time interval. Although the groups were not comparable considering prognostic factors, there was still no difference in OS for the patients undergoing curatively intended surgery including resection of minor suspected EHD. This article, however represents the reality of the clinical setting at a tertiary hepatobiliary center and the heterogeneous nature of patients with CLM. During a period in which nearly two-thirds of patients with potentially resectable CLM were evaluated with PET/CT, we found that PET/CT changed the management in one-tenth of cases but with less than one-tenth of patients having potentially false-negative PET/CT results.
Conclusion
In this retrospective study, we found a change in the surgical management of one-tenth in a selected cohort after the addition of PET/CT. The OS after curatively intended surgery was similar between patients with and without preoperative PET/CT. However, the groups differed in prognostic factors and type of liver-directed procedure. Based on our results, the use of PET/CT should be more selective, identifying patients with a high hepatic tumor burden and an increased risk of EHD. Further studies are needed to clarify the selection criteria and the potential survival benefit of PET/CT in patients with metastatic CRC.
Supplemental Material
sj-docx-1-sjs-10.1177_14574969221083144 – Supplemental material for Does 18F-FDG PET/CT change the surgical management of potentially resectable colorectal liver metastases?
Supplemental material, sj-docx-1-sjs-10.1177_14574969221083144 for Does 18F-FDG PET/CT change the surgical management of potentially resectable colorectal liver metastases? by Josefin Jonsson, Oskar Hemmingsson, Rebecca Strengbom, Jan Axelsson, Katrine Riklund and Hanna Nyström in Scandinavian Journal of Surgery
Footnotes
Acknowledgements
The authors acknowledge research nurse Agneta Karhu for administrative assistance and Mathias Lundin for statistical assistance.
Author contributions
O.H., K.R., J.A., and H.N. were involved in the conceptualization. O.H., K.R., and H.N. contributed toward methodology. Formal analysis and visualization was done by J.J. Investigation was done by J.J. and R.S. J.J., R.S., and H.N. were involved in writing—original draft. All the authors were involved in writing—review and editing. Supervision was done by K.R. and H.N. Project administration was done by H.N. Funding acquisition was done by H.N.
Availability of data and material
The data presented in this study can be shared in response to a reasonable request to the corresponding author. This study was not preregistered in an institutional registry.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the Swedish Research Council, Wallenberg Foundations/Knut and Alice Wallenberg Foundation, region Västerbotten, the Swedish Cancer Society, the Cancer Research Foundation in Northern Sweden, and the JC Kempe Memorial Foundation Scholarship Fund, Umeå University.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
