Abstract
Background and objective:
There are still gaps in knowledge concerning the adherence to different multimodal pathways in pancreatic surgery. The aim of this trial was to explore and evaluate an Enhanced Recovery After Surgery (ERAS®) and prehabilitation protocol in patients undergoing open pancreatic surgery.
Methods:
Three groups of patients were included: two prospective series of 75 patients undergoing open pancreatic surgery following an ERAS® protocol with or without prehabilitation, and one group of 55 historical controls. Variables regarding adherence to, and effects of the protocols, were collected from the local database and the patients’ hospital records. Patients’ adherence to advice given pre-operatively was followed up using a study-specific questionnaire.
Results:
The patients reported high adherence to remembered advice given. The health care professionals’ adherence to the various parts of the concepts varied. ERAS® implementation resulted in more frequent gut motility stimulation (p < 0.001) and shorter duration of epidural anesthesia, site drains, and urinary catheter (p = 0.001). With prehabilitation, more patients were screened concerning nutritional status and prescribed preoperative training (p < 001). There was a significant change in weight before surgery, a shorter time to first flatus and a shorter length of stay after implementation of the concepts (p < 0.05). Complications were rare in all three groups and there were no significant differences between the groups.
Conclusion:
The implementation of an ERAS® and a prehabilitation protocol increased adherence to the protocols by both patients and healthcare professionals. An implementation of an ERAS® protocol with and without prehabilitation decreases length of stay and may decrease preoperative weight loss and time to bowel movement.
Context and relevance
There are still gaps in knowledge concerning the adherence to different multimodal pathways after different kind of surgery as in pancreatic surgery. The results of our study indicate that imple-mentation of an ERAS® and a prehabilitation protocol can increase adherence to the protocols by both patients and healthcare professionals and improve postoperative outcome. The evidence increases that implementation of ERAS® and prehabilitation protocols have an important impact in the development of care of patients undergoing pancreatic surgery.
Introduction
Surgery induces a major physiological stress response to the human body. 1 To reduce the risk of complications and to improve care, various care pathways have been developed such as “Enhanced recovery After Surgery” (ERAS®). 2 There are now several guidelines based on such pathways for a variety of procedures, for example colorectal, cardiac, and gynecological surgery.3–5 In these pathways the team of surgeons, anesthetists, physiotherapists, dietitians, and nurses cooperate with the patient, following standardized protocols covering the whole surgical process.
Despite progress in pancreatic surgery regarding mortality rates, the procedures are still associated with a morbidity of 40 to 60%.6,7 Hence, it is of importance to further develop the care and ERAS® or “fast-track” concepts that have been established.8,9 These are based on best available evidence and have been found to have an impact on variables such as length of stay, morbidity, mortality, and hospital readmission rates, and there is no evidence of harm.9–14 However, positive results are not found in all trials, which may be related to the health care professionals’ and the patients’ adherence to the protocols. This has been addressed previously and it was found that adherence to variables included in the protocols significantly increased with the implementation and that adherence was higher for the pre- and intraoperative items than for the postoperative ones.15,16 However, its effect on postoperative complication rates and hospital length of stay are contradictory, and more studies are needed to further explore the subject.
In addition, over the last few years, there has been an increasing focus on how to optimize and prepare the patients pre-operatively to reduce complications and facilitate recovery.17,18 Studies where patients’ adherence to such specific prehabilitation protocols before pancreatic surgery is evaluated are currently lacking.
The main aim of this trial was, therefore, to explore patients’ and health care professionals’ adherence to an ERAS® and prehabilitation protocol. An additional aim was to evaluate clinical outcome after implementation of these protocols for patients undergoing pancreatic surgery.
Methods
Development of the modules
In 2014, a multidisciplinary group consisting of surgeons, anesthesiologists, nurses, physiotherapists, and dietitians at Sahlgrenska University Hospital, Sweden, started the development of an ERAS® concept for patients undergoing pancreatic surgery. The concept was based on the Swedish concept for colorectal surgery but adjusted for patients undergoing upper abdominal surgery, specifically on the pancreas, focusing on their specific needs of care. The final concept is presented in Table 1, and it was implemented in January 2015.
Content in the pre-admission, preoperative, intraoperative, and postoperative phases in the care before ERAS® was implemented, with the ERAS® concept and after the additional preoperative module was implemented.
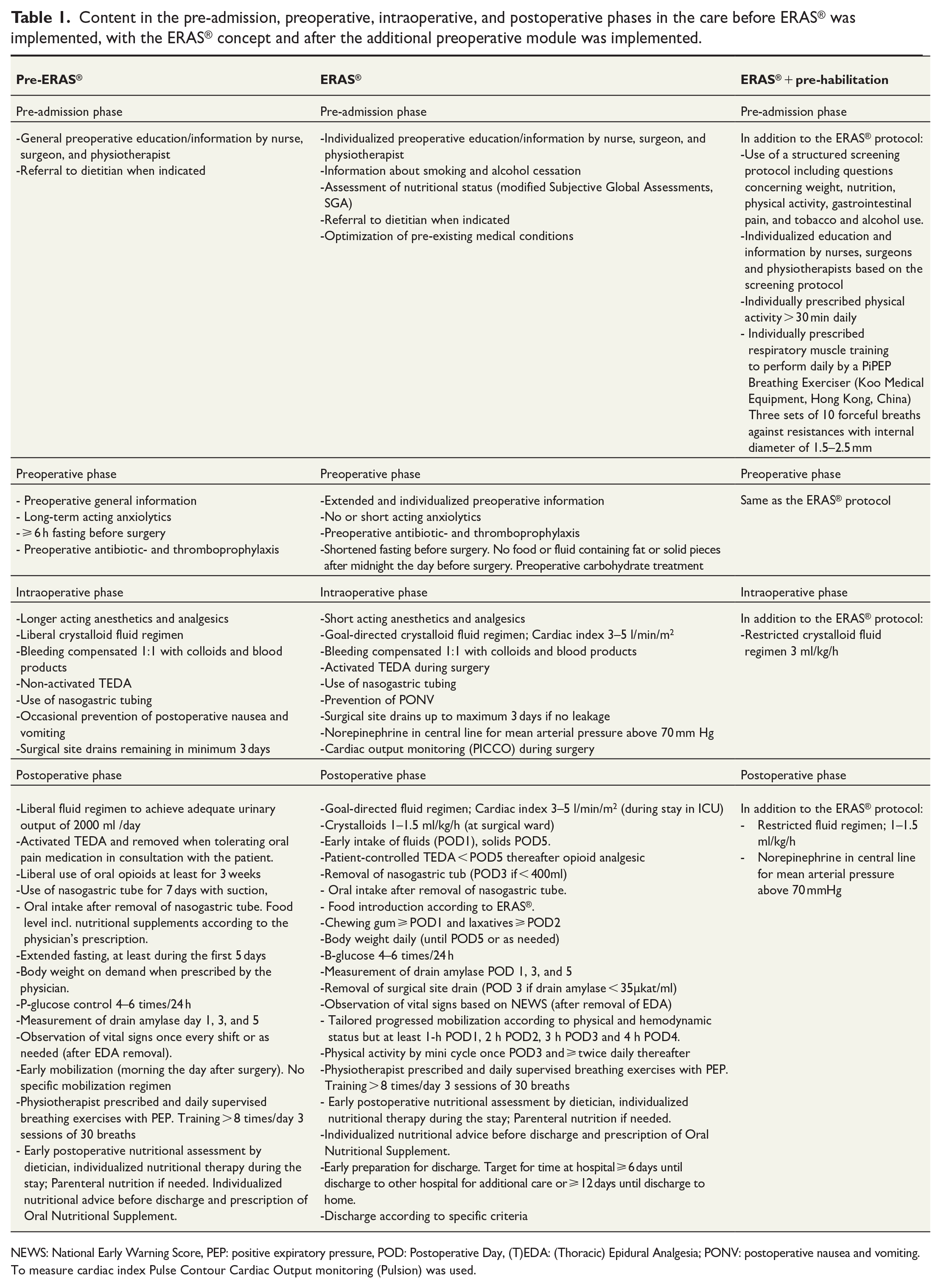
NEWS: National Early Warning Score, PEP: positive expiratory pressure, POD: Postoperative Day, (T)EDA: (Thoracic) Epidural Analgesia; PONV: postoperative nausea and vomiting.
To measure cardiac index Pulse Contour Cardiac Output monitoring (Pulsion) was used.
To further improve the healthcare, the multidisciplinary team developed a pre-admission screening protocol during 2017 to gain more detailed and structured information about the patients’ preoperative status. The screening included questions about nutrition, bowel function, alcohol intake, smoking habits, and level of physical activity. The protocol was completed by or with the patient before or during the visit at the outpatient clinic when the planning of surgery was initiated. The results of the protocol were to be used in the preparation of the patient before surgery. By October 2018, all members of the team also used this prehabilitation protocol. The risk of malnutrition was assessed by the contact nurse and patients at risk were referred to a dietician. All patients met a physiotherapist who emphasized the importance of increased physical activity before surgery. The patients also received a respiratory muscle training program based on their individual status and needs, to be performed daily.
A description of the different phases of care before ERAS® was implemented, together with the ERAS® concept and the prehabilitation protocol, is presented in detail in Table 1.
Materials
In this study, three groups were compared:
Pre-ERAS®: A group of 55 historical controls who underwent surgery 1 to 2 years before the ERAS® implementation.
ERAS®: A prospective consecutive series of 75 patients who underwent surgery from first of August 2017 until the 30 June 2018 followed the ERAS® protocol.
ERAS® + prehabilitation: A prospective consecutive series of 75 patients who underwent surgery from the first of October 2018 until the 31 August 2019 followed both the ERAS® protocol and the prehabilitation module.
Patients submitted to open pancreatic surgery, with either pancreatoduodenectomy ad modum Whipple or total pancreatectomy, were included. Both procedures include resection of the distal part of the stomach and reconstruction with a gastrojejunostomy and hepaticojejunostomy and in the case of the Whipple procedure, a pancreatogastrostomy.
Evaluation of adherence and clinical outcome
To explore patient adherence to the prehabilitation module, patients in the ERAS® + prehabilitation group who underwent surgery from 1 January to 30 May 2019 (n = 32) were contacted by letter and asked to participate in a follow-up. All patients in this sub-group accepted and participated in a telephone interview in June 2019. During this interview, the patients were asked the same questions as in the preoperative screening protocol, Table 1. Thereafter, related to their answers, structured follow-up questions were asked about the content of the information and of their adherence to the advice given. The applicability of each form of advice was assessed on a five-level scale from “not applicable” (1) to “fully applicable” (5), adherence to the advice was assessed on a three-level scale from “no” (1), “partly” (2), or “yes, completely” (3) and whether the advice had improved their situation before surgery was assessed on a five-level scale from no change (1) to much improved (5). In addition, the patients were asked to what extent they thought that written information had prepared them, using a five-graded scale from not at all (1) to totally (5), and to give feedback about the quality of the information and interventions given. The replies were registered in a study-specific response sheet.
Health care professionals’ adherence to the protocols was extracted from the ERAS® Interactive Audit System® (EIAS). The clinical outcome was also extracted from this database as well as from the medical records. Data were divided into factors associated with adherence to the ERAS® and ERAS® + prehabilitation protocols and results of the protocols. Variables are presented in Table 2. Definition of descriptive and outcome variables included in the protocols used in EIAS are presented in a supplementary file.
Demographic data of the patients (early). Mean (SD), n [%].
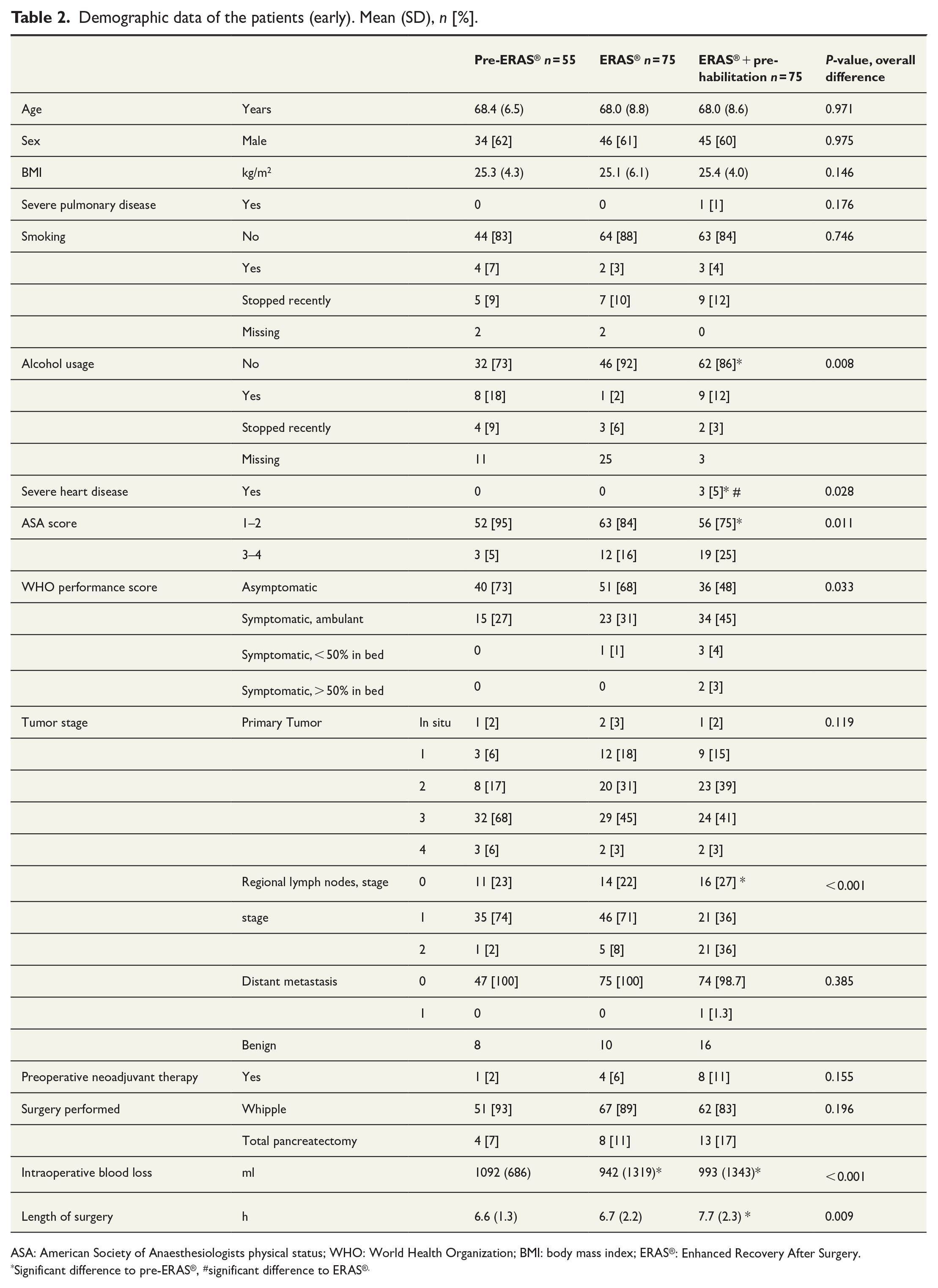
ASA: American Society of Anaesthesiologists physical status; WHO: World Health Organization; BMI: body mass index; ERAS®: Enhanced Recovery After Surgery.
Significant difference to pre-ERAS®, #significant difference to ERAS®.
Clinical data were extracted using EIAS®, as well as from the medical records. The same person extracted the data for all groups. EIAS® contains clinical variables collected for each patient during pre-admission and the preoperative, perioperative, and postoperative phases.
Ethics and statistical analyses
This study protocol was approved by the Regional Ethics Committee for the region of Västra Götaland, Sweden. (Registration number: 437-17). The patients were included based on their consent to participate in EIAS®. This consent was given after oral and written information was provided which covered that participation in the database might also entail contributing to research.
Before the study started, a power analysis was performed to estimate the sample size. A difference of 1 day in hospital between ERAS® protocols with and without prehabilitation, (from 13.5 to 12.5 days, with a standard deviation of 2 days, a power of 0.80, and alpha of 0.05) revealed an estimation of sample size of 63 participants per group. To adjust for dropouts and missing data, the sample size was increased to 75 per group in the prospective cohorts. The comparison group consists of a series of 55 patients registered retrospectively during the 2 years before implementation of the ERAS® concept.
Non-parametric analyses were used as most of the data was qualitative and the results were not normally distributed. The differences between the groups were therefore analyzed by Mann–Whitney U test and Chi 2 /Fisher’s exact test. The level of significance was set to p < 0.05.
Results
Background data
The background characteristics are given in Table 2. There were some significant differences between the groups in alcohol use, severe heart disease, American Society of Anaesthesiologists physical status (ASA) score, World Health Organization performance score, tumor stage, preoperative chemotherapy, and length of surgery. Patients in the ERAS® + prehabilitation group had more comorbidities, more advanced tumor stage, and had received preoperative chemotherapy more often compared to the pre-ERAS® group.
Patient adherence to the prehabilitation protocol
The results concerning how the patients recalled if advice had been given (primary outcome), and the usefulness, adherence and effects of the advice are given in Table 3. When the patient remembered that advice had been given, the majority found it helpful, and the adherence was high. The given advice improved the symptoms concerning preoperative difficulties with eating and gastrointestinal pain.
Patient’s recall of information given and adherence to the advice, n = 33.
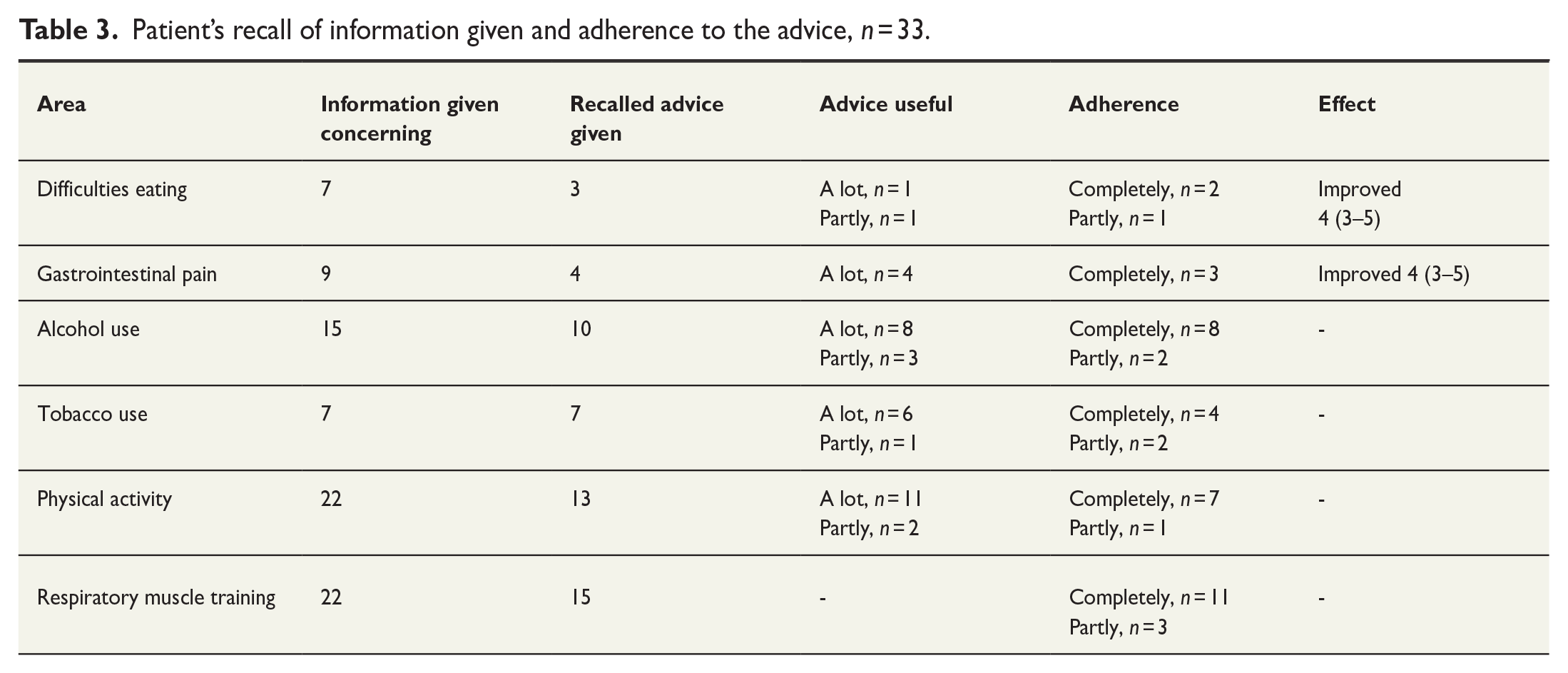
All patients were given written information before surgery, and 28 of them remembered receiving and reading it. The information was rated as fully applicable (median 5).
In addition, the patients were asked to give feedback on the preoperative information and the newly implemented preoperative interventions. Nine gave general positive feedback while one thought that too much information was given pre-operatively. Nine of the responders would have liked to receive more information about complications, prognosis and where to find answers to any questions.
Health care professionals’ adherence to the ERAS® + prehabilitation protocol
The adherence to the ERAS® and prehabilitation protocols is presented in Table 4. Some of the variables, such as information about cessation of smoking and drinking alcohol, use of epidural anesthesia, use of antibiotics and thrombosis prophylaxis were fulfilled already before the ERAS® concept was implemented (p > 0.05). The patients in the ERAS® and ERAS® + prehabilitation groups received postoperative nausea and vomiting prophylaxis (p = 0.016) and stimulation of gut motility (p < 0.001) significantly more often. Removal of site drains, and the urinary catheter was carried out significantly earlier with the ERAS® protocol (p < 0.01) and the thoracic epidural anesthesia was used for significantly fewer days (p = 0.001).
Adherence to the factors in the concepts.
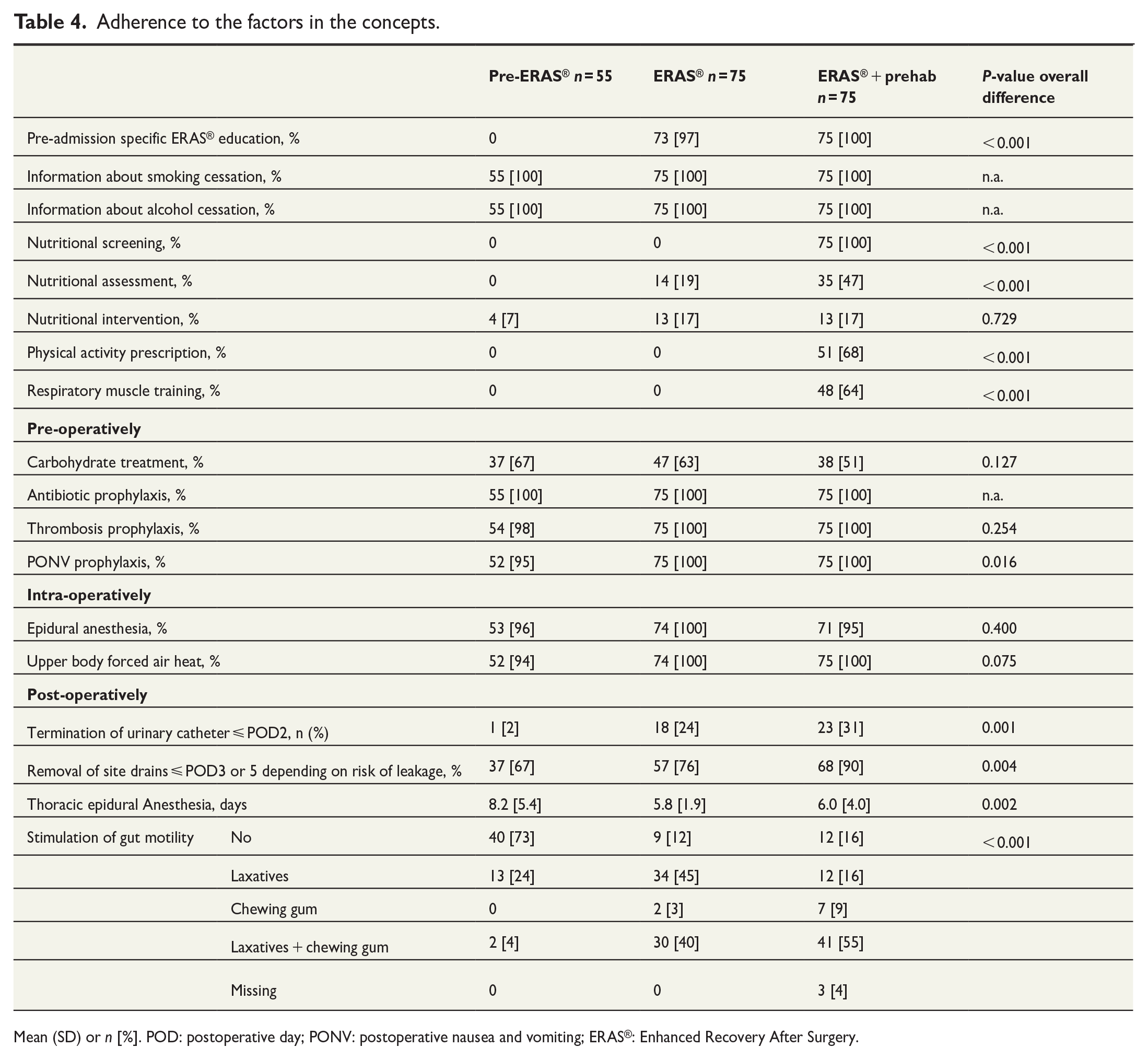
Mean (SD) or n [%]. POD: postoperative day; PONV: postoperative nausea and vomiting; ERAS®: Enhanced Recovery After Surgery.
After the addition of the preoperative protocol, significantly more patients were screened and assessed concerning nutritional status (p < 0.001), but that did not lead to significantly more nutritional interventions (p = 0.729). About two thirds of the patients in the ERAS® + prehabilitation group received the individually prescribed physical activity and respiratory muscle training before surgery.
Clinical outcomes and complications after the changes in care
The results of the perioperative and postoperative outcome variables are presented in Table 5. Length of stay (primary outcome) was significantly shorter among the patients in the ERAS® and ERAS® + prehabilitation groups compared to the patients in the pre-ERAS® group (p < 0.001).
Clinical outcomes and complications related to the ERAS® and ERAS® + prehab protocols.
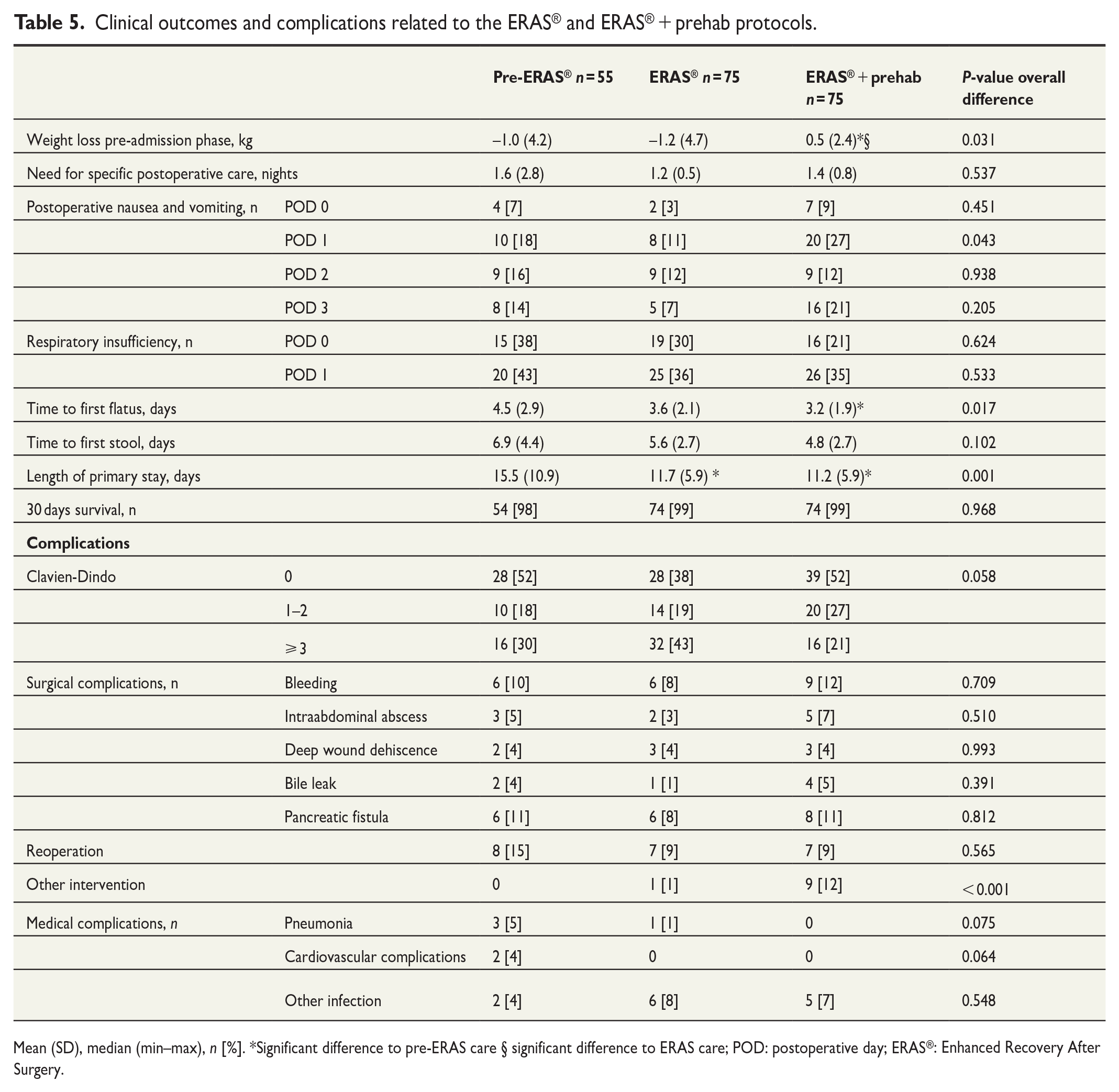
Mean (SD), median (min–max), n [%]. *Significant difference to pre-ERAS care § significant difference to ERAS care; POD: postoperative day; ERAS®: Enhanced Recovery After Surgery.
There was also a significant difference regarding weight change while waiting for surgery between the groups. The patients in the ERAS® + prehabilitation group had a slight weight gain compared to the patients in the other groups, who instead had a weight reduction (p = 0.031). The patients in the ERAS and ERAS® +prehabilitation groups were significantly more active post-operatively (both p < 0.001), Fig. 1. The patients in the ERAS® + prehabilitation group also had a significantly shorter time to first flatus (p < 0.01) compared to the patients in the pre-ERAS® group.
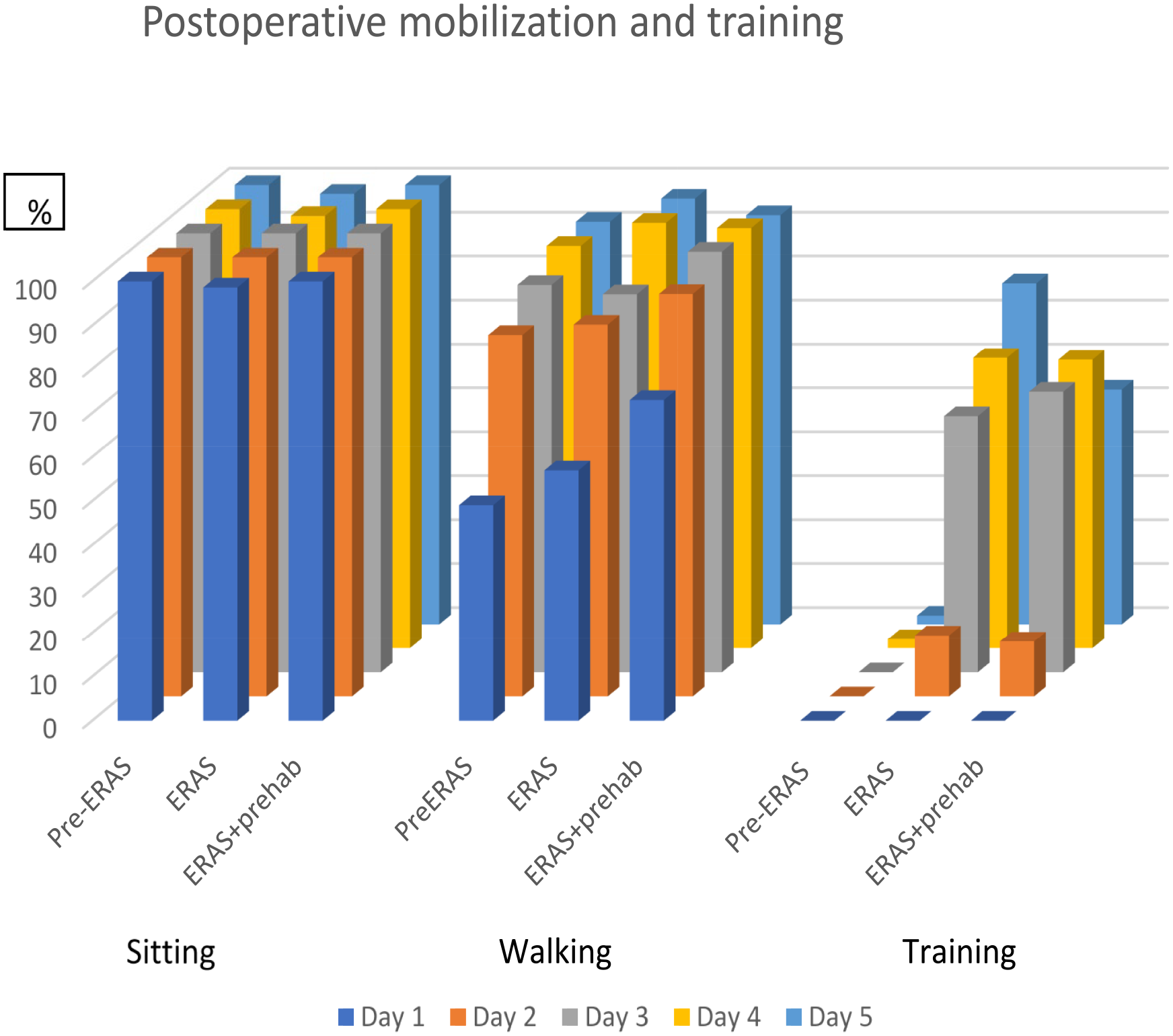
Ratio of patients who were mobilized to sitting, walking, and training during the first 5 postoperative days.
Complications were rare in all three groups and there were no significant differences between the groups regarding any complications. However, significantly more patients (n = 9) in the ERAS® + prehabilitation groups needed interventions because of complications (drainage of abscess n = 5, percutaneous transhepatic cholangiography (PTC) n = 2, radiology during anesthesia n = 1, gastroscopy 1) compared to the other groups (0 in PRE ERAS and 1 in the ERAS group (PTC), p = 0.004).
Discussion
There are multiple known barriers for a successful implementation of an ERAS® protocol such as resistance to change, insufficient education, limited resources for implementation and external factors related to patient complexity.19–21 When ERAS has been implemented in pancreatic surgery the adherence was found to be higher pre- and intra-operatively compared to post-operatively.15,16 The current study provides indications that there is a lack of adherence to some parts of the ERAS® program that need to be further addressed. Furthermore, the results indicate a need for a more individualized model of care as a complement to standardized practice. The concepts include more and individualized information to increase the patients’ understanding of the interventions, thereby improving the outcomes. On the other hand, patients’ possibilities to adhere to the pathways are confounded by postoperative symptoms. In the current trial, patients’ adherence to a preoperative intervention was assessed. Of those who recalled that information was given, the adherence was high. It is possible that patients undergoing pancreatic surgery chose to optimize their situation pre-operatively to be better prepared, decrease the risk of complications and increase the pace of recovery.
The implementation of the ERAS® concept led to a decreased length of stay by 3.8 days (p < 0.05) and the prehabilitation module an additional 0.5 days (n.s). The structured protocol and the high adherence to it, from both the health care professionals’ and patients’ perspective can therefore be interpreted to have an important impact as also shown in previous trials (2,3,8). There were though a larger proportion of patients in the ERAS® + prehabilitation groups who needed other intervention after surgery. An explanation may be that surgery was performed in patients with additional co-morbidity and with more advanced stages of the disease, Table 1.
Concerning one of the cornerstones in the preoperative protocol, nutrition, the assessment of method has been changed from a more extensive (Modified Subjective Global Assessments, SGA) to a simpler screening, but it seems to catch those at risk for malnutrition. According to our results, those at risk of malnutrition are referred to a dietician for assessment and individual nutritional counseling/treatment. This management is in accordance with applicable guidelines from the European Society of for Clinical Nutrition and Metabolism.22,23 As seen in the results of the changes in care, patients do not lose weight pre-operatively to the same extent as previously, reflecting a possible benefit for the patients taking part in the changed routine.
Another important part of the protocol is the preoperative level of physical activity. Previous studies have reported improvement in muscle mass or markers of muscle function following prehabilitation. 24 In addition, the adherence to exercise programs has been found to be higher with supervision of the programs. Only 65% of the patients in our ERAS® + prehabilitation group received individual advice to increase physical activity or perform breathing exercises pre-operatively. This may partly be explained by the time between the pre-admission visit and surgery being too short (< 2 week). A period of > 2 weeks of respiratory muscle training before surgery has been suggested to be efficient by Hulzebos et al. 25 The aim of the respiratory muscle training was to prepare the patients for the deteriorations seen in respiratory function after major open abdominal surgery and avoid postoperative pulmonary complications.26,27 For several decades Swedish’ clinical practice has included preoperative information about postoperative pulmonary complications and how to prevent and treat them, in combination with postoperative follow-up and guidance of treatment given. 27 The number of patients who developed pneumonia has therefore been low during the whole study period. Neither were there any differences in PaO2/FiO2 nor number of patients with respiratory insufficiency between the groups. It seems therefore that the supplement with additional respiratory muscle training cannot be justified, at least not at a group level. In accordance with nutritional screening and dietary advice there might be a place for a respiratory risk scoring tool to select patients for respiratory training.
During the development of the ERAS® protocol, the level of mobilization and physical activity was set arbitrarily, based on earlier published protocols and clinical practice. The new protocol led to significantly increased mobilization and training (Fig. 1). Several other changes in the care pathway may have contributed such as the more active approach concerning a low fluid regimen to maintain arterial blood pressure with an infusion of norepinephrine during surgery but also a strict low fluid regimen after surgery. However, whether these activities had an impact on the significantly earlier time to bowel movement in combination with the changed regimens for gut stimulation is not known.
The trial has some strengths. The three series of patients underwent surgery at the same study site. Already before the implementation of the ERAS® protocol, patients from a larger region were having surgery at the hospital. This ensures a large enough group of patients to keep up a high quality of surgery and care. Another advantage is the cooperation between the different professions in the team. The development of the ERAS® and ERAS® + prehabilitation protocols started from a clinical perspective and is tailored to the needs of the specific group of patients in the actual context.
There are also some limitations. The two prospective groups were compared to historical controls where data was retraced retrospectively from medical records. To avoid heterogeneity in the care given during the period, only patients who underwent surgery during the 2 years before implementation were included. The pre-ERAS® group therefore included a series of 55 patients instead of the 75 anticipated by the power analysis. This may have had an impact on the results of the trial. In addition, the pre-, peri and postoperative pancreatic care has developed over the years and patients with more co-morbidity and with more advanced stages of the disease, who were not able to undergo surgery during the pre-ERAS® period, were included in the later series of patients. This may have had an impact on the results. An analysis of the additional costs could also have added important knowledge as previous research has found reduced costs with enhanced recovery clinical pathways for patients undergoing pancreatic surgery. 28 Furthermore, even though the ERAS® concepts and EIAS® have been developed there are still discrepancies to consider between various categories of patients. For instance, the level of mobilization ought to be adjusted after various kinds of surgery. The results from the register were therefore not included in our study. Finally, the evaluation of the patients’ adherence to the advice given in the last cohort of patient was only performed on a subgroup of the patients. The number of patients who remembered that information was given and gave feedback was low. There is a risk that only the patients who considered the information relevant responded and the results have therefore to be interpreted with that in mind.
During recent years, a growing number of patients have been operated for premalignant lesions in the pancreas, which often comprise anastomoses with a higher risk of leakage. There is also a trend toward a more frequent use of neoadjuvant chemotherapy. For these groups there is potentially a longer window for preoperative interventions compared to patients operated upfront for manifest cancer. A challenge with a detailed protocol is to fulfill the needs of a heterogeneous patient group. Such interventions generate costs. In our concept, nutritional interventions were prescribed after screening, in contrary to physical activity and breathing exercises. The importance and effects of these separate interventions are not known but it is possible that patients with a different preoperative physical activity level would have gained from a more patient-centered intervention. Future trials should focus on the effects of such tailored protocols.
Conclusion
The aim of this trial was to explore the adherence to and effects of an ERAS® concept with and without prehabilitation, on patients undergoing open pancreatic surgery. Most of the included variables of adherence were rated highly, by the patients who remembered advice given, and we therefore conclude that adherence was satisfactory. However, there are important aspects to consider. The patients sometimes did not remember that advice was given. Furthermore, some of the variables are included in traditional care of the patients undergoing pancreatic surgery and were already fulfilled before ERAS® was implemented. Other variables had lower adherence. However, even with a high standard of surgery and care there is always room for improvement.
Supplemental Material
sj-docx-1-sjs-10.1177_14574969231186274 – Supplemental material for Development of and adherence to an ERAS® and prehabilitation protocol for patients undergoing pancreatic surgery: An observational study
Supplemental material, sj-docx-1-sjs-10.1177_14574969231186274 for Development of and adherence to an ERAS® and prehabilitation protocol for patients undergoing pancreatic surgery: An observational study by Monika Fagevik Olsén, Thomas Andersson, Micheline Al Nouh, Erik Johnson, Linda Block, My Vakk and Johanna Wennerblom in Scandinavian Journal of Surgery
Footnotes
Author contributions
All authors planned the study. T.A., M.A.N., M.F.O., and J.W.H. retrieved the material from the database and patient files. M.A.N. interviewed the patients. All authors interpreted the results, wrote, and approved the final manuscript.
Clinical trial registration
Clinical Trials: NCT03466593, R&D in Sweden/ FoU i VGR: 238701.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Work was supported by grants from the Swedish state under the agreement between the Swedish government and the county councils, the ALF-agreement (ALFGBG-718811).
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
