Abstract
Background
Necrotizing pancreatitis (NP) may result de novo or following procedures such as ERCP or partial pancreatectomy (post-procedural), and may require surgical debridement. Video-assisted retroperitoneal debridement (VARD) is a standard approach for NP that employs a 5 cm incision with varying degrees of blind and open debridement. We describe our technique and outcomes of a modified VARD called laparoscopic-assisted pancreatic necrosectomy (LAPN) performed through a single 12 mm incision that uses direct laparoscopic visualization during debridement.
Methods
At one medical center, all LAPN patients (2012-2020) were assessed for demographics, disease factors, and outcomes. Bivariate logistic regression analyses were performed to identify factors independently associated with recovery after LAPN for patients with de novo vs post-procedural necrosum.
Results
Over 9 years, 60 patients underwent LAPN for NP. Median age was 57 years (IQR: 47-66) and 43 (69%) were men. Pancreas necrosum was de novo in 39 (63%) patients and post-procedural in 23 (37%). NP resolved with a median of 1 LAPN procedure and median hospitalization was 33 days. The LAPN major morbidity rate and in-hospital mortality rate were 47% and 5%. No significant differences were seen between NP etiology cohorts, although post-procedure NP patients trended towards a faster clinical recovery to baseline compared to de novo patients (193 vs 394 days; p-value = .07).
Conclusions
LAPN offers a smaller incision with excellent visualization and non-inferior outcomes, regardless of etiology, with likely faster recovery for patients with post-procedural vs de novo necrotizing pancreatitis.
Keywords
Introduction
Acute pancreatitis accounts for over 200,000 hospital admissions in the United States every year, and approximately 20% of these patients develop necrotizing pancreatitis (NP), associated with a high risk for complications such as organ failure, sepsis, and death.1-3 NP can develop within a few days of the onset of pancreatitis and is characterized by non-enhancing areas of pancreatic parenchyma and/or extra-pancreatic tissue on contrast-enhanced computed tomography (CT).3-5 The necrosum can become infected in up to 30% of patients within a month after the onset of NP resulting in sepsis and rapid clinical deterioration. 4 NP infection increases mortality rates from 15% in non-infected patients to 39%. 6
Until the late 1990s, open necrosectomy was widely utilized early for infected NP, with high complication rates and mortality ranging from 11 to 39%. 6 The “step-up” approach, consisting of initial percutaneous drainage and clinical stabilization followed by delayed minimally invasive necrosectomy, has become standard of care. This approach obviates the need for surgery in 30% of patients and leads to lower rates of new-onset diabetes, organ failure, and incisional hernias when compared with open necrosectomy.6,7
Video-assisted retroperitoneal debridement (VARD) is an integral part of the step-up approach which typically involves a 4-5 cm incision, followed by open debridement of loose necrosum, and final inspection with a zero-degree laparoscope. While this technique has comparable postoperative outcomes to endoscopic methods and superior outcomes to traditional open surgical approaches, it requires a 4-5 cm incision with each successive procedure and provides for varying degrees of blind debridement, resulting in some risk of disrupting adherent vasculature and organs. 6
Laparoscopic pancreatic necrosectomy is described in several small retrospective studies with significant variability in approach, including hand-assisted technique.8-11 In this report, we describe our systematic approach to laparoscopic-assisted pancreatic necrosectomy (LAPN) through a single 12 mm incision, which minimizes blind debridement through directed simultaneous laparoscopy. We report our institution’s initial outcomes and assess the results of LAPN according to etiology of NP formation, either due to acute pancreatitis (de novo) or secondary to post-procedure etiology, with the goal of further defining the benefits of this approach.
Materials and Methods
Timing of Surgical Intervention and Preoperative Planning
The appropriate timing for an operative intervention depends on both the patient’s clinical stability and the length of time required for the NP to develop a fibrotic rind around the necrosum. In the interim, percutaneous drainage is frequently an important early tool to address a variety of symptoms including sepsis, pain, and mass effect causing biliary or gastrointestinal obstruction, either as definitive therapy or as a means of guidance for directed surgical therapy.4,12,13
The surgeon should be involved in discussions with interventional radiology (IR) as the placement of percutaneous drains ideally will define the course of future operative tractotomies. The optimal course should minimize the distance from the skin to the target and maximize distance from all major vascular structures and neighboring organs. We find that a 20F or larger drain size allows for adequate efflux of material and best facilitates tractotomy in the cases where operative intervention is necessary. Even with larger diameter and multiple catheters, drainage via percutaneous catheter fails in approximately 50% of cases. 12 In some situations, the optimal tract takes a trans-diaphragmatic course. When this is the case, a plan for pleural drainage to prevent empyema should be made.
Once the necrosum is matured, typically around 4 weeks post symptom onset, the percutaneous drains are in place, and the patient is physiologically optimized, a surgical intervention can be planned. Given the potential for significant blood loss as well as developing a marked systemic inflammatory response following surgery, complications are extensively discussed with the patient, patient’s family, and the team. The most common complications include bleeding requiring conversion to a laparotomy or angioembolization, enterocutaneous fistula, prolonged intubation, and the need for further operation and debridement.
Basic Operative Approach
Prior to beginning the operation, the required equipment is confirmed to be in the room. The authors prefer a 12 mm trocar, which allows sufficient space for a 5 mm laparoscope (0 or 30°) and a working instrument. The operative set-up can be seen in Figure 1. A 5 mm trocar is used initially if a tract runs close to concerning structures. A 15 mm trocar can be used for debridement of large cavities. Frequently used instruments include Scanlan ringed forceps (Scanlan International, Inc., Saint Paul, MN)), a suction irrigator, Robinson drains, a triple lumen sump drain (Davol, Bard, Warwick, RI), and a variety of hemostatic agents. A 5 mm Ligasure or Harmonic scalpel is occasionally used and always kept readily available. Warmed fluid for irrigation should be readily available, and the authors will occasionally use a 3:1 mixture of hydrogen peroxide or betadine with saline for irrigation. Finally, instruments required for laparotomy (major dissecting pan, Bookwalter, etc.) are prepared in case of emergent conversion. The authors occasionally use a flexible cystoscope to visualize necrosum that cannot be seen with the 30° scope. Video-assisted retroperitoneal debridement (VARD) and laparoscopic-assisted pancreatic necrosectomy (LAPN) operative techniques (a). Pancreatic necrosum with a percutaneous drain in place, the initial component of the step-up approach for necrotizing pancreatitis; (b). Depiction of the VARD operative approach, utilizing a 5 cm incision; (c). Depiction of the LAPN operative approach, utilizing a 12 mm trocar.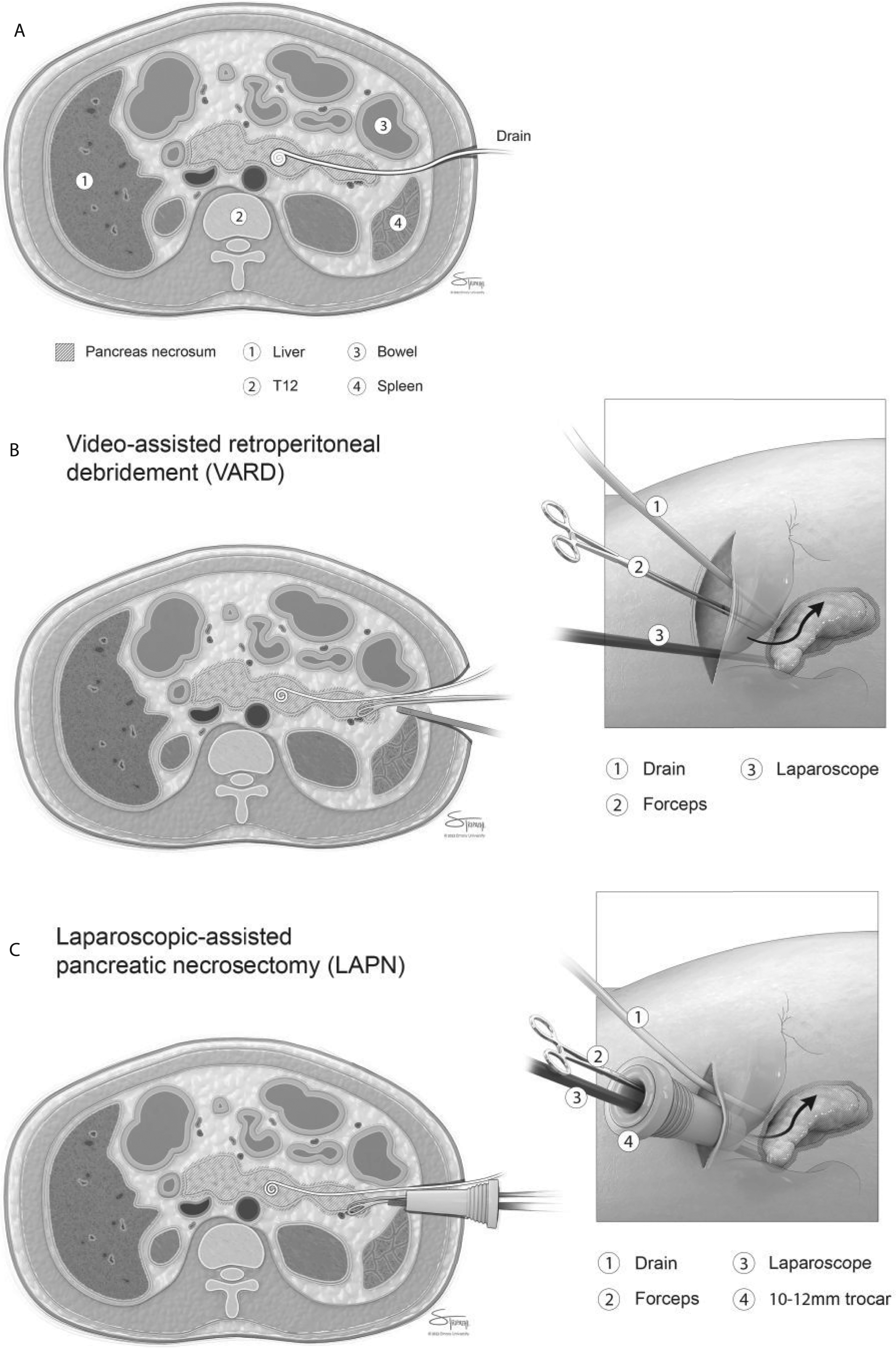
If the patient is already receiving broad-spectrum antibiotics, these are continued as scheduled through the operation. Otherwise, the authors prefer to use a carbapenem and anti-fungal within 60 min of the operative start. The patient is positioned with beanbag assistance based on necessary exposure to drain insertion sites while ensuring access to the abdominal midline. As with any operation, care is taken to ensure that all pressure points are appropriately padded.
Simultaneous access to cross-sectional imaging and fluoroscopy views will help guide dissection. We use an optical viewing trocar with a zero-degree, 5 mm or 10 mm scope. The trocar is advanced into the retroperitoneum by following the course of the drain with the drain in place. The appearance of pancreatic necrosum and the loss of resistance to advancing the trocar signals the entry into the necrosum cavity. The authors recommend leaving the CT guided drain in place to serve as a guide until the largest trocar is in place and the tract is well established.
Once in the cavity, the transparent obturator is removed. Liquid and small particles are aspirated using a laparoscopic suction irrigator. Samples are collected for culture. Insufflation of the cavity is rarely necessary during debridement but is used intermittently for inspection of the cavity. The ringed forceps are inserted through the trocar alongside the scope and used to gently debride the solid NP. Scanlan ringed forceps are often extremely helpful during this stage of the operation. Direct observation during the debridement should be employed whenever possible, pausing after tissue has been debrided to assess the entire cavity.
When performing debridement, it is often convenient to remove the reducer cap from the 10 mm trocar to facilitate retrieval of larger particles. Irrigation with saline or a dilute hydrogen peroxide mixture is also often helpful and is typically administered through a red rubber catheter that is placed through the port. Debridement should continue until the granulated walls of the cavity are seen. Necrotic tissue adjacent to bowel or major vascular structures should be debrided with caution and may be left in place at the surgeon’s discretion to prevent injury to bowel or vasculature.
Following satisfactory debridement, a triple lumen sump drain is placed in the cavity (Figure 2). This drain is then anchored to the skin with non-absorbable suture to prevent dislodgement. Irrigation should be initiated as soon as possible to prevent drain occlusion by clot or debris. In situations where the image guided drain takes a trans-diaphragmatic course, and the pleural space is traversed with tractotomy, consideration should be given to placing a separate chest tube under sterile conditions in the operating room. Axial radiographic series depicting interval reduction in pancreatic necrosum in a single patient (a). Pancreatic necrosum at initial evaluation; (b). Interval placement of a CT-guided percutaneous drain; (c). Transition from percutaneous drain to a Davol drain following laparoscopic-assisted pancreatic necrosectomy (LAPN); d. Transition from a Davol drain to a red rubber catheter following satisfactory LAPN.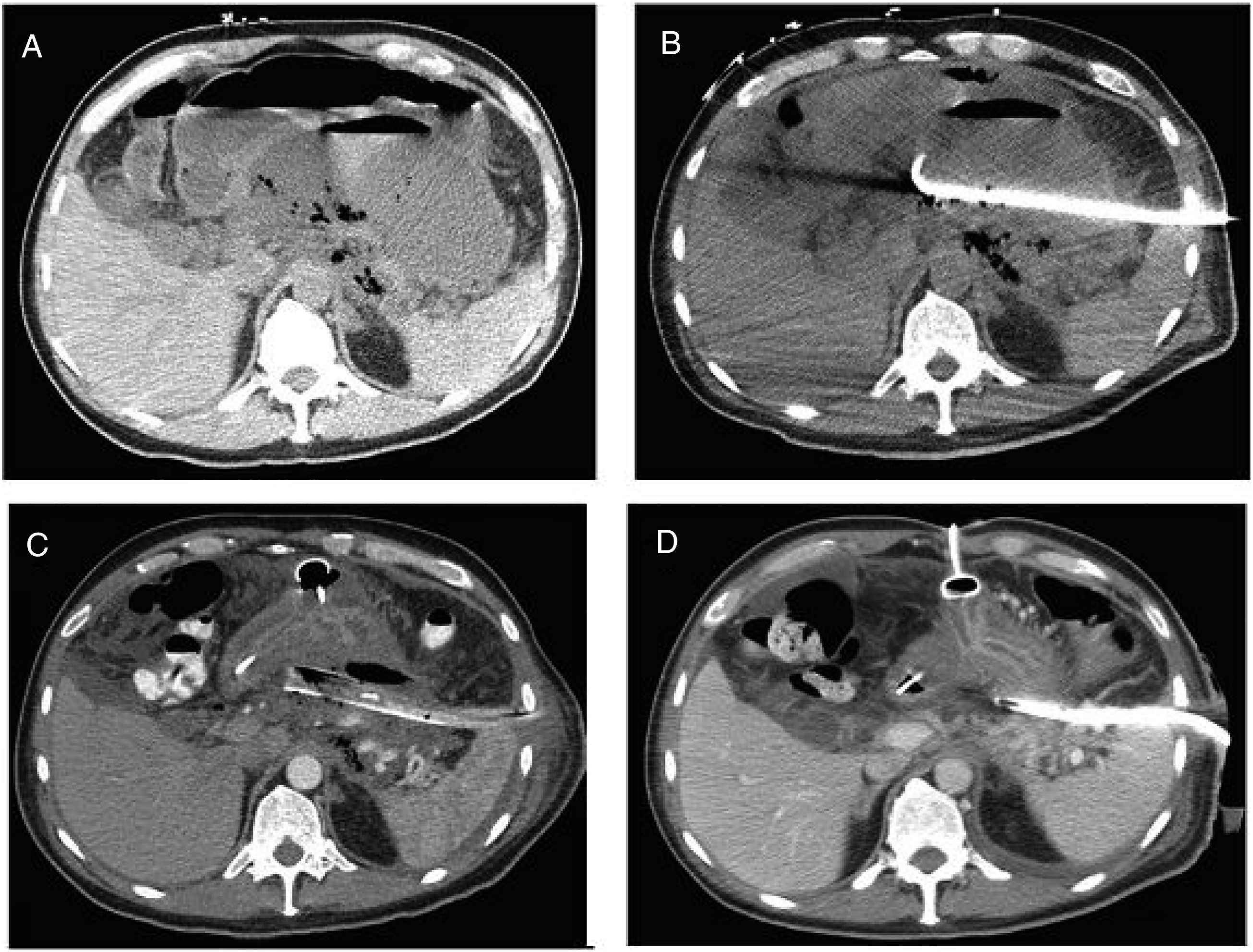
Unique Operative Considerations
When there is close proximity between the necrosum and named vascular structures, the authors perform this procedure in the vascular surgery operating suite and collaborate with the vascular team in order to further reduce risks for hemorrhage. This involves placement of a femoral sheath by vascular surgeons in order to offer intraoperative real-time fluoroscopic capabilities as well as act as a back-up therapeutic option for management of hemorrhagic shock. Given the high rates of strong adherence of vessels to the necrosum, especially in severe infected NP, this adjunct allows for real-time visualization of major blood vessels during the debridement as well as the presence of a secondary expert opinion during the case.
Postoperative Care and Drain Management
Patients are routinely admitted to the ICU and are monitored closely for hemorrhage or sepsis. The triple lumen drain is set up: low continuous suction is applied to the largest lumen, normal saline is delivered to the irrigation port using an IV pump at 100 ml/hour, and the sump port is left open to the atmosphere. As the patient begins to improve clinically and the character of the irrigation clears, the rate of the irrigation is weaned over days to a week. A repeat CT scan with arterial and delayed contrast phases should be obtained on postoperative day 5-7 to determine completeness of debridement and screen for pseudoaneurysm.
Repeat debridement is performed in a similar fashion when necessary. Most often, the authors are able to use the existing drain tracts to complete the debridement. Laparotomy for PN is seldom performed at the authors’ institution. Depending on the clinical status of the patient, small amounts of necrosum in the vicinity of vascular structures or bowel are left in-situ and no further debridement is performed.
Once the surgical team deems debridement complete, the triple lumen drain is replaced with a 28 French Robinson catheter that is trimmed to match the internal length of the triple lumen drain. Additional side holes are typically cut in the Robinson catheter and a safety pin is placed through the drain at skin level to mark the catheter depth. A stoma appliance or wound drainage collector is applied at skin level to protect the skin and record output. By postop day 5-7, sufficient time has elapsed that the drain tract is mature and stable, allowing for daily removal, rinsing, and replacement of catheters. This is done to prevent clogging and allow any remaining particulate necrosum to necessitate. As output decreases and the patient improves clinically, the drains are downsized and shortened over weeks to months until the tract has closed. Patients are considered ready for discharge once all drains are converted to Robinson catheters, there is an established plan for nutrition outside of the hospital, pain is controlled with non-intravenous medication, and there is no care being provided that cannot be delivered outside of the hospital setting. This approach of slowly “weaning” the drains allows for healing of the pancreatic fistula while preventing re-accumulation of the effluent. This can be completed in the outpatient setting.
Results
Demographic and Clinicopathologic Patient Data
Clinicopathologic Characteristics of Patients Who Underwent Laparoscopic-Assisted Pancreatic Necrosectomy (LAPN) With Stratification by Underlying Etiology.
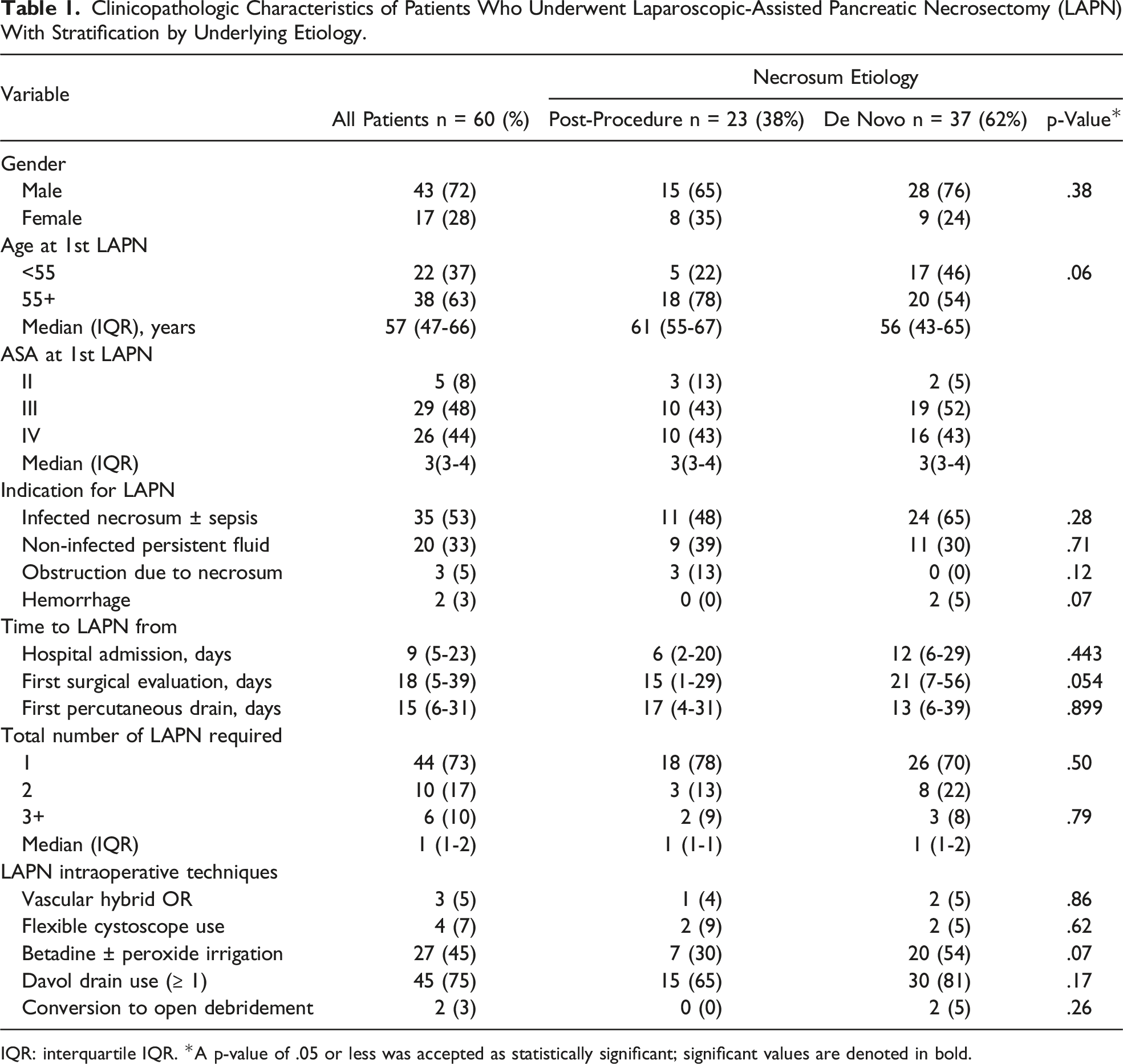
IQR: interquartile IQR. *A p-value of .05 or less was accepted as statistically significant; significant values are denoted in bold.
Operative Data
Patient Outcomes Following Laparoscopic-Assisted Pancreatic Necrosectomy (LAPN) With Stratification by Underlying Etiology.
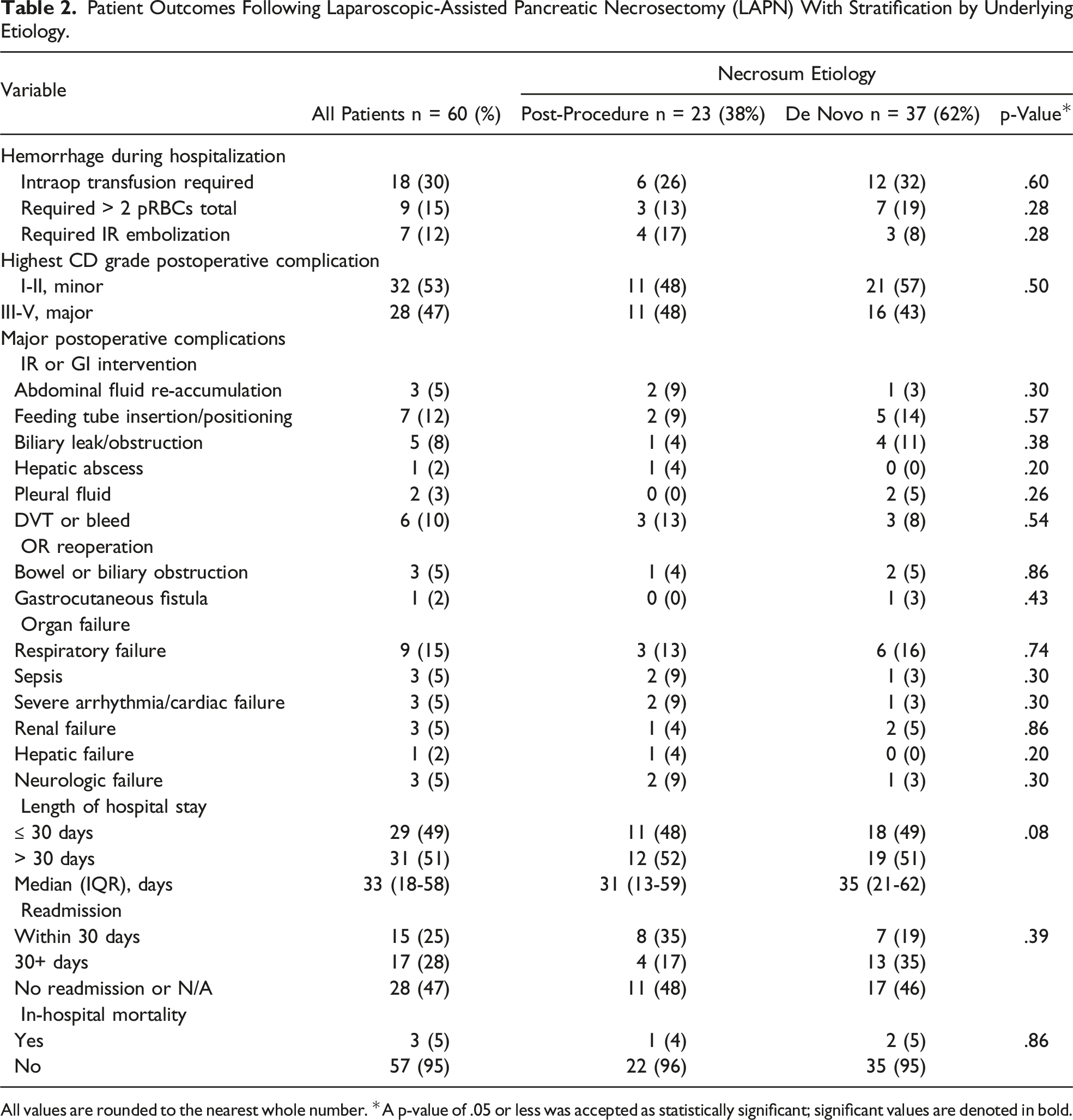
All values are rounded to the nearest whole number. *A p-value of .05 or less was accepted as statistically significant; significant values are denoted in bold.
Postoperative Patient Outcomes
Fifty-three percent (n = 32) of patients experienced a minor postoperative complication (CD grades I-II), including electrolyte derangements, dehydration, asymptomatic anemia, and malnutrition requiring total parenteral nutrition (TPN). Forty-seven percent (n = 27) of patients experienced a major postoperative complication (CD grades III-V), the most common of which were respiratory failure requiring escalation of care (15%, n = 9), malnutrition requiring the insertion or re-positioning of a feeding tube (12%, n = 7), and internal hemorrhage or thrombosis requiring an IR intervention (10%, n = 6). The median length of hospital stay from was 33 days (IQR: 18-58). Of the 60 patients, 53% (n = 32) of patients were readmitted at least once, 47% (n = 15) of whom were readmitted within 30 days of discharge. The most common reasons for readmission were failure to thrive and/or nutrition concerns (31%, n = 10) and recurrence of abdominal pain (34%, n = 11). The total in-hospital mortality rate was 5% (n = 3). All postoperative outcomes are delineated in Table 2.
Twenty percent (n = 12) of patients experienced complete radiologic resolution of necrosum at the time of discharge. The median number of days to radiologic resolution was 120 days (IQR: 55-230). Patients required at least one drain for a median of 104 days (IQR: 69-155) from the first LAPN. Based on available clinic notes within our institution’s EMR, 53% (n = 32) of patients returned to clinical baseline. The median number of days to clinical baseline from first LAPN was 277 days (IQR: 123-483).
LAPN Outcomes Stratified by Necrosum Etiology
Timing of Resolution With Stratification by Underlying Etiology.
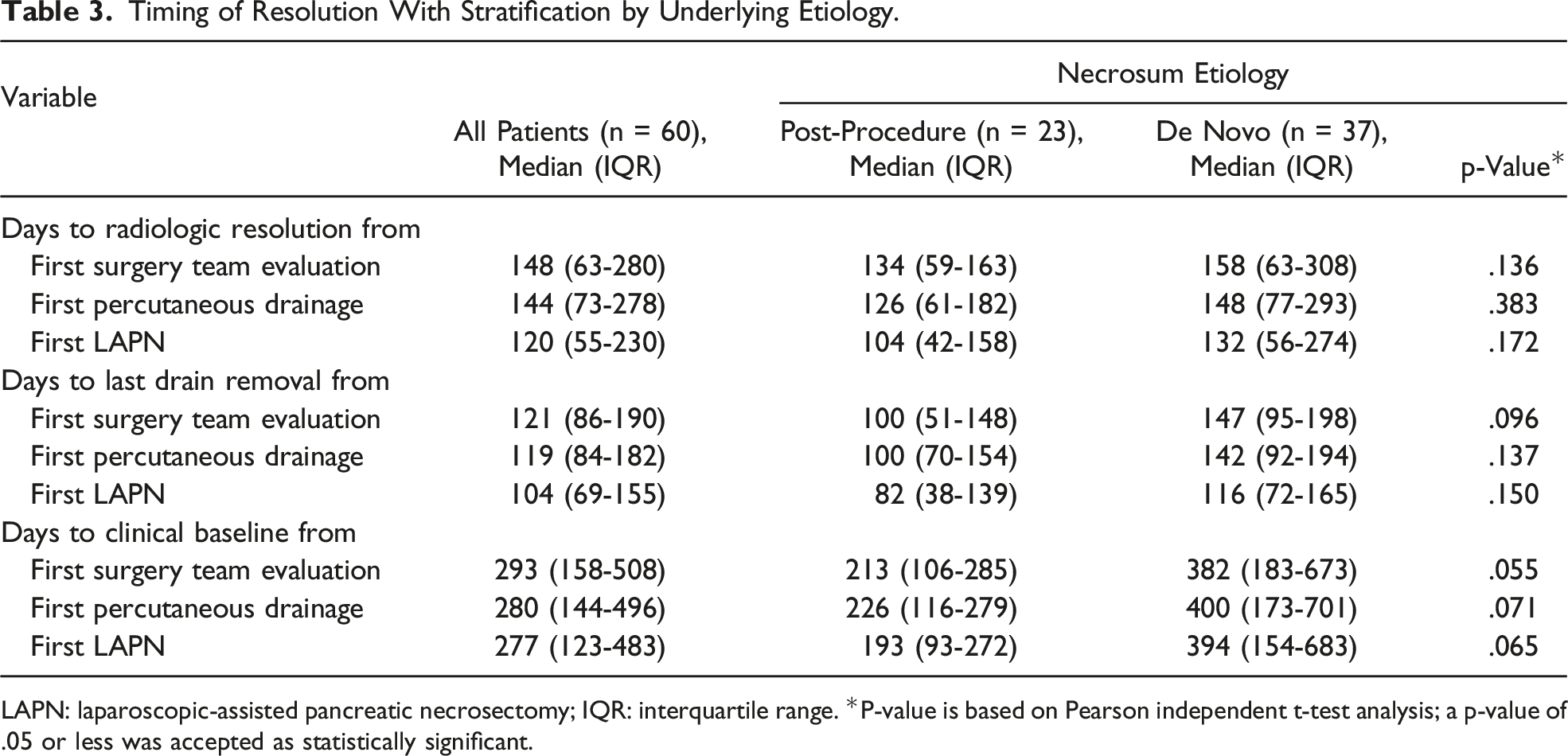
LAPN: laparoscopic-assisted pancreatic necrosectomy; IQR: interquartile range. *P-value is based on Pearson independent t-test analysis; a p-value of .05 or less was accepted as statistically significant.
Discussion
In this report, we described our approach to laparoscopic-assisted pancreatic necrosectomy and provided the results of our experience. We also assessed the impact of LAPN on patients with pancreatic necrosum who developed it as a result of de novo pancreatitis or due to post-procedure pancreatitis. We found our approach successful at mitigating the effects of pancreatic necrosum, and that patients who developed necrosum following a surgical or interventional procedure had a shorter hospital course than their counterparts who developed necrosum due to non-procedure-related acute pancreatitis.
The surgical management of necrotizing pancreatitis has evolved substantially over the last three decades, shifting from an immediate, open debridement to a combination of percutaneous drainage and minimally invasive, delayed debridement, commonly referred to as the “step-up” approach.2,3,15,17 Up-front percutaneous intervention has been shown to reduce morbidity and mortality associated with immediate surgical intervention and even obviate the requirement for surgical debridement in up to 50% of patients.2,6
When surgical intervention is required, the standard operation is open or laparoscopic necrosectomy through a lateral approach, tracking along the existing percutaneous drain. The novel transition from an anterior abdomen approach to the lateral retroperitoneum was first reported by Fagniez et al (1989) and was originally performed via a 15-20 cm lateral subcostal incision. 18 This method was not widely adopted due to high morbidity and mortality rates but was instrumental in paving the way for similar retroperitoneal approaches, such as VARD.16,19 Horvath et al (2001) and Van Santvoort et al (2007) first described VARD as a hybrid between the traditional open approach and minimally invasive tractotomy, reducing the risk for damage to abdominal organs and eliminating the large operative wound.16,20 VARD consists of a 4-5 cm subcostal incision, followed by open debridement of the percutaneous tract under direct visualization and insertion of a laparoscope for final visualization. 16
True laparoscopic approaches with a variety of technique variations such as hand assistance via Gelport, traditional insufflation, and cyst gastrostomy have been reported in numerous small, retrospective studies.21-23 These approaches have been less widely adopted despite acceptable postoperative morbidity and mortality, largely due to the increased technical difficulty compared to VARD. In this manuscript, we report our institution’s method for laparoscopic-assisted pancreatic necrosectomy (LAPN) as it may offer some advantages previously described debridement methods due to the reduction in the incision length and the width of the debridement tract.
LAPN utilizes an approximately 1 cm incision adjacent to the percutaneous drain, with immediate introduction of the laparoscope through an optical viewing obturator, allowing the surgeon to follow the percutaneous tract closely. The trocar itself allows simultaneous access to a 5 mm scope and laparoscopic graspers or Scanlan forceps, allowing improved visualization during the debridement. The trocar also stabilizes the scope and offers some protection of the lens against fluids and tissue, which helps maintain optical clarity aiding technical ease and flow of the operation. We also have more recently utilized certain adjuncts, such as use of a flexible cystoscope instead of the rigid laparoscope to improve debridement visualization, as well as collaboration with vascular surgery for real-time fluoroscopy and insertion of a femoral sheath for theoretical hemorrhage emergency control. Although there were not enough patients to perform statistical analyses focusing on these adjuncts, these adjuncts are cost effective and collaborative options for minimizing the main risks involved in all pancreatic debridement, especially life-threatening hemorrhage and inadvertent damage to bowel.
Of all 60 patients who have undergone LAPN at our institution, we report a major morbidity rate of 47%, an in-hospital mortality rate of 5%, and a median length of hospital stay of 33 days (IQR: 18-58). The most common major complications were respiratory distress (n = 9, 15%) and malnutrition requiring feeding tube insertion of repositioning (n = 7, 12%). The majority of patients (73%) only required one LAPN procedure for resolution. Finally, though not statistically analyzed, we found that hepatic involvement in the necrosum and duodenal perforation was indicative of poor perioperative outcomes, whereas splenic bleeding was adequately manageable with IR embolization even when formal splenectomy was not possible. Complete radiologic resolution of the necrosum was also not required for successful discharge with recovery to clinical baseline.
At our institution, we only perform LAPN for necrotizing pancreatitis, making it difficult to directly compare with the methods used at other institutions. When comparing to the existing VARD literature primarily published by members from the Dutch pancreatitis research group between 2010 and 2013, LAPN appears to be non-inferior in terms of the major morbidity rate, which ranges from 35 to 55%. Similarly, the percentage of patients only requiring one procedure for resolution was similar, with literature reporting rates ranging from 50-83%. LAPN may offer non-inferior to reduced in-hospital mortality rates and length of hospital stay compared to that reported in VARD literature, which range from 0 to 33% and 62 to 130 days, respectively.6-7,24,26
We examined whether the etiology of the intraabdominal necrosum had an impact on perioperative outcomes and found that while there were no statistically significant differences, patients who developed procedure associated necrosum trended toward recovering back to baseline from surgery faster than those who with de novo necrosum (193 vs 394 days; p-value = .07). We suspect this difference in recovery time could be attributable to the differences in patient population, as those who develop de novo pancreatitis frequently have associated risk factors, including history of alcohol use, gallstone disease, and obesity. 27
This study was limited by its retrospective nature and small patient sample size. Given the relatively rare frequency of LAPN at our institution, a prospective or larger study would not have been feasible. The primary strength of this study lies in the inclusion of every patient who has undergone LAPN at our institution over the last decade. Fully laparoscopic pancreatic debridement has not been widely adopted due to technical challenges, and the authors hope that this manuscript is able to bolster surgeons who are looking for a true minimally invasive approach to necrosectomy.
Conclusion
Laparoscopic-assisted pancreatic necrosectomy (LAPN) is a modification of the lateral, retroperitoneal approach to pancreatic debridement and utilizes a 1 cm incision along the radiographically placed drain, an optical viewing trocar, and simultaneous use of the scope and debridement instrument. Adjuncts such as the use of a flexible cystoscope, intraoperative fluoroscopy, and operation in the vascular suite assist in minimizing the feared risks of hemorrhage and damage to adjacent structures. Compared to existing standard debridement techniques, such as video-assisted retrograde debridement (VARD), LAPN patient outcomes appear to be non-inferior and may improve in-hospital mortality rates and length of stay due to the minimized physiologic stress and improved intraoperative visualization.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
