Abstract
Backgrounds:
The optimal surgical strategy for the treatment of chronic pain after inguinal hernia repair is controversial and based on relatively weak evidence. The purpose of this study was to analyze pain-related functional impairment using a simplified clinical treatment algorithm for a standardized surgical treatment. The algorithm was predefined, and the indication to operate was based on strict criteria.
Methods:
This was a prospective, non-controlled, explorative study. The pain operation was either open triple neurectomy with total mesh removal or laparoscopic retroperitoneal triple neurectomy. A clinically relevant postoperative change was defined as ⩾25% change from the baseline level. Primary outcome was pain-related impairment of physical function using the Activity Assessment Scale. Secondary outcomes included Individual Patient-Reported Outcome Measures, Hospital Anxiety and Depression Scale, and PainDETECT Questionnaire.
Results:
A total of 240 patients were referred (2016–2019). Sixty-six patients were included for the analysis. A total of 25% of referred patients were offered a pain operation. Follow-up was a median 3 months (range: 3–13). Activity Assessment Scale scores were clinically relevant improved in 43 patients (68%), not clinically relevant different in 19 (30%), and clinically relevant worsened in one (2%). Secondary outcome scores were all significantly improved (P < 0.05) except for the risk of postoperative depression (P = 0.092). Fifty-one patients (77%) reported that chronic groin pain was reduced after the operation.
Conclusions:
Pain-related functional impairment was improved with clinical relevance in roughly 70% of patients through a simplified clinical algorithm for surgical treatment of severe chronic pain after an inguinal hernia repair.
Introduction
Groin hernia repair is one of the most common general surgery procedures. Approximately 12,000 and 800,000 were operated per year in Denmark and the United States, respectively,1,2. In Denmark, elective primary inguinal hernia repairs are performed exclusively by a Lichtenstein procedure or laparoscopic transabdominal preperitoneal patch (TAPP)1,3. The introduction of mesh reinforcement in groin hernia repair has significantly reduced recurrence rate 2 , and the main clinical challenge has changed to chronic pain 4 . Well-defined severe chronic pain is reported by 3%–5% of patients after groin hernia repair and probably less after TAPP or total retroperitoneal repair (TEP)4 –7.
Pain following groin hernia repair can be nociceptive or neuropathic in nature. Nociceptive pain arises from actual or threatened damage to non-neural tissue, such as from musculoskeletal inflammation or the mesh (mechanical), whereas neuropathic pain is defined as a lesion or damage to the somatosensory nervous system 8 . A recent questionnaire survey (among 47 international expert hernia surgeons (response rate 60%)) 9 recommended a treatment algorithm using routine preoperative imaging (ultrasound/magnetic resonance (MR) scan) before offering a pain operation and preoperative evaluation including quantitative sensory testing (QST) performed by a specialized pain team. QST is a meticulous and time-consuming technique to diagnose peripheral nervous system disorder in patients with postoperative chronic pain 9 . The algorithm also implied local analgesic (LA) trigger point injection9,10 and dermatosensory mapping9,11.
Preliminary results from clinical pain outcome studies have suggested an overall analgesic effect of a pain operation10 –15. Unfortunately, only a few studies were consistent in using a well-defined treatment algorithm or a clear description of the indication to offer a pain operation 10 . Overall, the suggested algorithm is based on sparse evidence.
The aim of this study was to compare pre- versus postoperative clinical pain–related outcomes in patients with severe chronic pain after inguinal hernia repair. Patients were enrolled using a simplified, standardized clinical algorithm without preoperative routine imaging or QST evaluation by a specialized pain team. Before study enrollment, specific preoperative criteria to offer a pain operation were defined.
Methods
This was a prospective, non-controlled, explorative, quality assessment study. Patients were enrolled from a university hospital serving as a national referral center for surgical treatment of chronic postherniotomy pain. Thus, preoperative information on groin pain before the index hernia repair was unknown. The treatment algorithm, questionnaire battery, and surgical logistics were developed before study initiation (January 2015–April 2016). The surgical learning curve for neurectomy and groin re-exploration was overcome after 13 open neurectomy procedures with mesh removal and 14 laparoscopic retroperitoneal triple neurectomy procedures (guided by two internationally recognized experts). At the beginning of study start, the duration of surgery had plateaued at 40–60 min with no apparent difference (although not systematically analyzed).
One pain operation was defined as one patient. Thus, a patient undergoing first an open and subsequently a laparoscopic pain operation (second operation due to failure of the first, see below) counted as two patients (one patient in the open group and one in the laparoscopic group).
The Simplified Treatment Algorithm
In this study, a simplified clinical standardized treatment algorithm was developed from a previous international consensus report 9 . At the development phase of the present simplified algorithm, there was no or only limited evidence of the predictive value of the mentioned elements from the report. Therefore, it was chosen not to include the potentially predictive elements 9 in the present simplified algorithm.
The indication to qualify for a pain operation was based solely on clinical criteria, anamnesis, and pain characteristics (Fig. 1). A diagnosis of neuropathic inguinodynia was supported by dermatosensory mapping to identify involved nerves in the groin.
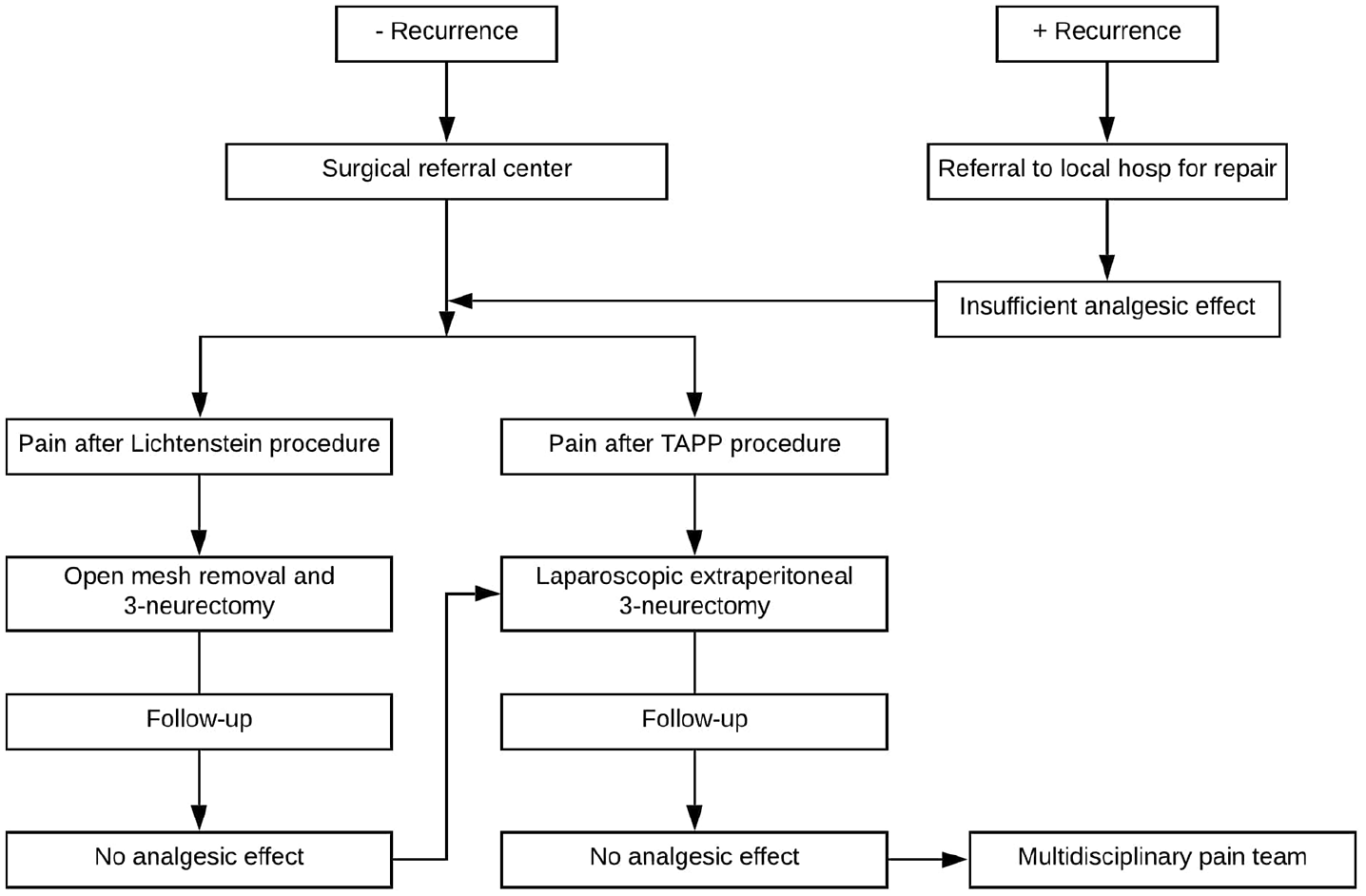
The modified treatment algorithm. Patients with a recurrent hernia are referred to a local hospital for repair. Patients without a recurrent hernia and patients without analgesic effect after repair of recurrence are referred to a surgical center. Patients with pain after Lichtenstein’s procedure are offered open triple neurectomy with mesh removal. Patients without sufficient analgesic effect after the open neurectomy and patients with pain after transabdominal preperitoneal patch (TAPP) procedures are offered laparoscopic retroperitoneal triple neurectomy. Patients are referred to a multidisciplinary pain team if there is an insufficient analgesic effect.
Indication for Pain Operation
Indication to offer an operation was pain localized in the groin classified as moderate or severe. Pain was preoperatively registered by the patients themselves in a structured questionnaire (see pain Numerical Rating Scale (NRS) below). The characteristics and intensity were confirmed during the subsequent clinical assessment and interview. Patients were not offered a pain operation if the index hernia repair and pain anamnesis were less than 6 months before referral, if multi-modal analgesic treatment had not been attempted for at least 3 months (analgesic administration within a specialized multidisciplinary pain team was not compulsory), a preoperative chronic pain syndrome unrelated to the inguinal intervention was present, the patient was deemed non-compliant, alcohol or recreational drug abuse was present according to the criteria of the Danish National Board of Health (<3 months up to evaluation), or <18 years of age.
The index repair, either Lichtenstein’s procedure or TAPP, was defined as the pain-triggering operation (as mentioned above, TEP is not performed in Denmark). Patients with chronic pain after the open Lichtenstein procedure were offered a planned open triple neurectomy and total mesh removal9,12. A laparoscopic retroperitoneal triple neurectomy was planned after TAPP9,11,14. If the dermatosensory mapping was negative in the TAPP index patients and there was no neuropathic clinical pain (see below), a total mesh removal without neurectomy was planned.
Resected nerves were confirmed by histology. Triple neurectomy and not selective neurectomy was performed due to anatomical nerve variations and cross-innervation in the inguinal canal and the retroperitoneum 11 . The surgical open and laparoscopic techniques are described in detail elsewhere11,12,16. If the analgesic response in the open group was clinically unsatisfactory or worsened at the follow-up, a laparoscopic retroperitoneal triple neurectomy was offered. Patients with treatment failure after laparoscopic triple neurectomy were referred to a multidisciplinary pain team.
Pain Characteristics
Pain characteristics were clinically classified into either inflammatory (deep, throbbing, constant), neuropathic (shooting, burning, stabbing, electric shock, and/or radiating), testicular (nociceptive/neuropathic), and/or mechanical pain (movement aggravated, pain improvement when stretching from a sitting position, foreign body feeling). Nociceptive and/or neuropathic pain characteristics were not quantified, but characterization of the pain was routinely included in patients’ pain history.
Outcomes
Outcomes were pre- versus postoperative clinical changes in questionnaire variables (see below). The intended postoperative follow-up was arbitrarily chosen as 3 months.
A pre- and postoperative structured questionnaire was filled out by the patients. Postoperative registrations were blinded toward patients’ preoperative registrations. Based on previous studies17,18, a cut-off value of ⩾25% was chosen as a clinically relevant change for continuous variables (i.e. a reduction from 100% to 75% (0.75 × 100 is a reduction of 25%) or from 50% to 37.5% (0.75 × 50 is a reduction of ⩾25%)).
The primary outcome was pain-related functional impairment assessed by the Activity Assessment Scale (AAS) 19 . The following four secondary outcomes were used: Individual Patient-Reported Outcome Measures (I-PROM), three questions rated on an NRS (pain-NRS), The Hospital Anxiety Depression Scale (HADS), and The PainDETECT Questionnaire (PDQ).
Questionnaires
The AAS is a validated questionnaire measuring physical impairment due to groin pain. The AAS provides a score from 0 to 65 which is converted to 0%–100% (continuous variable; 0% = no pain-related impairment of physical function, 100% = maximal impairment)19,20.
I-PROM is a non-published, non-validated situational patient–defined pain assessment instrument developed for this study. Patients themselves chose three specific daily-life activities/situations where the groin pain was most bothersome. Pain was rated on an NRS (0 = no pain, 1 = little pain, 2 = moderate pain, and 3 = severe pain. The total score was 3 × 3 = 9; scores were converted to a 0%–100% scale; continuous variable). Before the operation, patients were carefully instructed to fill out the I-PROM questionnaire to prevent potential misunderstandings.
The pain-NRS consisted of three questions rated on an NRS: pain during the last week in the operated groin, pain after ejaculation, and pain-related social activity disruption 21 . The pain-NRS was dichotomized to no/mild pain and moderate/severe pain 21 .
The HADS is a validated questionnaire assessing the risk of anxiety and depression (a score of 11–21 indicates high risk of either anxiety or depression) 22 . The PDQ is a validated questionnaire used to screen for the likelihood of neuropathic pain (a score of 19–38 indicates neuropathic pain) 23 .
Due to the statistic categorical nature of the secondary outcomes (pain-NRS, HADS, and PDQ), these were not analyzed according to a 25% pre- versus post-change. Instead, the variables were analyzed according to the number of patients changing group from moderate/severe to no/mild complaints (see section “STATISTICS”). Finally, at the postoperative follow-up, patients registered whether the operation had improved their chronic groin pain (yes/no).
Pre-study, it was decided to exclude patients with intraoperative complications (except from minor bleeding) from the study analysis and patients with treatment-demanding complications within the first postoperative week were excluded.
Statistics
This study was explorative in nature, and a sample size calculation was not possible. A per-protocol analysis was performed (see above). Preoperatively, it was arbitrarily decided to include a minimum of 60 patients for the study. Patients were analyzed as one group (all patients) and separately as two groups (open and laparoscopic). Patients served as their own controls (pre- vs postoperative changes). Females were excluded from the analysis of post-ejaculatory pain. No intergroup analysis was performed (open vs laparoscopic). Due to the low number of included patients, non-parametric statistics were used. The Wilcoxon signed-rank test (continuous data) and the McNemar test (2 × 2 tables) were used. The latter applied to group changes (preoperative vs postoperative moderate/severe or no/mild pain-NRS, HADS, and PDQ; see section “METHODS”). Data are presented as numbers (percentages) and medians (range). Multiple imputation by fully conditional specification was used for missing questionnaire variables. Patients who refused to fill out the preoperative or postoperative questionnaires or failed to meet for the 3-month postoperative follow-up were excluded from the analysis. As mentioned above, pain-NRS, the HADS, and PDQ questionnaires were dichotomized. P < 0.05 was considered statistically significant. Data were entered in a REDCap database hosted by the Capital Region of Denmark 24 . Data analysis was performed using SPSS (IBM SPSS Statistics for Windows, Version 25, released in 2017, Armonk, NY, USA; IBM Corporation).
Results
Baseline demographics are listed in Table 1. A total of 240 patients were assessed for eligibility (1 March 2016 to 31 June 2019) (Fig. 2). Due to the exclusion criteria, results from 66 patients were analyzed (i.e. 66 operations—33 open and 33 laparoscopic). Thus, the patient inclusion and operation rate was 25% and 28%, respectively. Fifty-seven patients underwent a single operation, and 13 patients underwent an open neurectomy with mesh removal followed by a laparoscopic retroperitoneal triple neurectomy. Two patients insisted on a laparoscopic pain operation despite the index operation being a Lichtenstein procedure (a total of 15 patients underwent a laparoscopic pain operation after Lichtenstein and 18 after TAPP, Table 1). There were no patients with bilateral chronic pain after bilateral inguinal hernia repair. None of the patients were offered selective mesh removal without neurectomy. During the pain operation, a medial hernia was found in five patients in the open group and was repaired with annuloplasty (no clinical recurrences at follow-up). All patients in the open group had a mesh removal, but no data was recorded on mesh types or intraoperative findings regarding the mesh (meshoma, mesh folding, etc.).
Data presented as medians (range) and numbers (%).
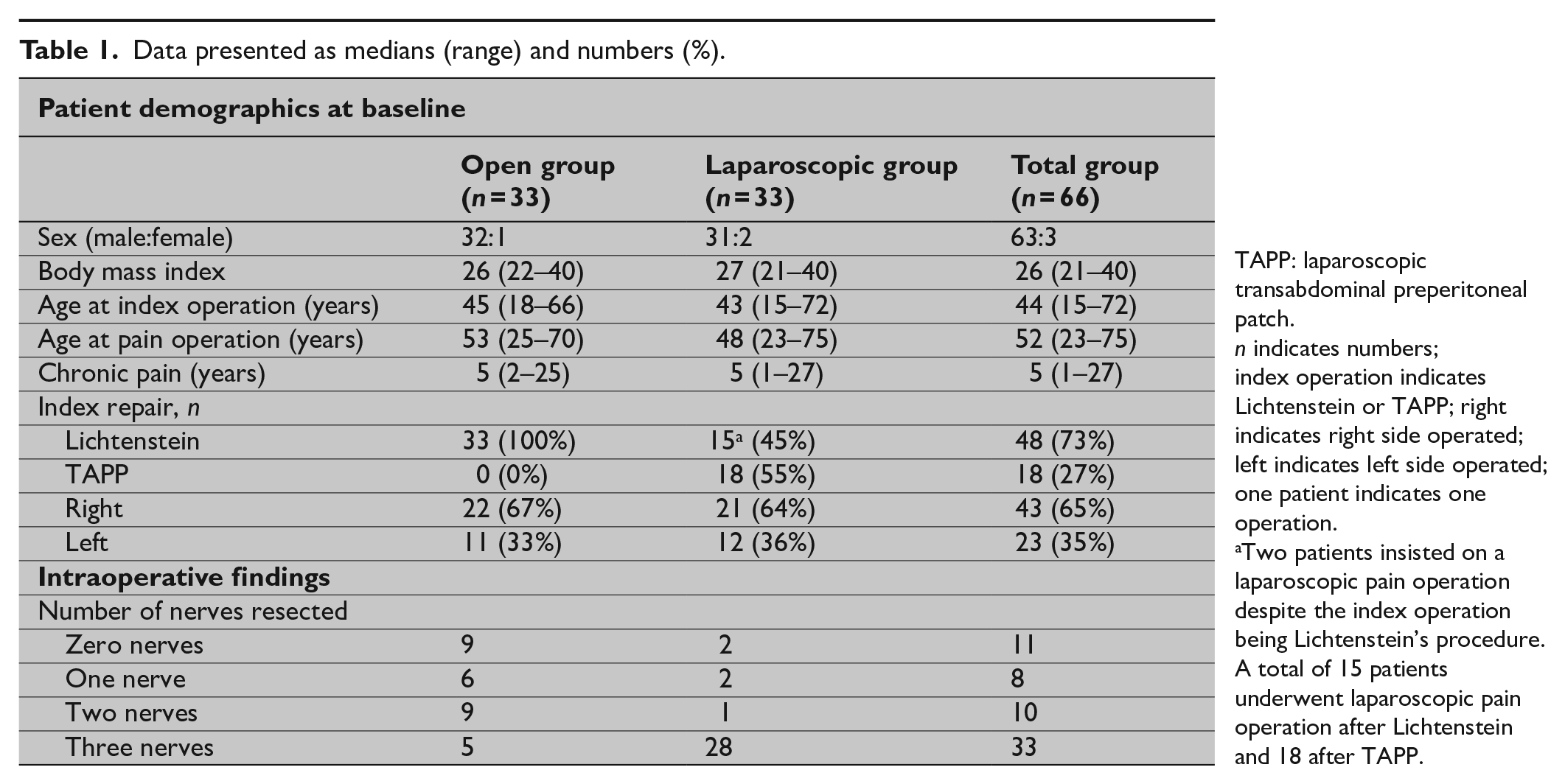
TAPP: laparoscopic transabdominal preperitoneal patch.
n indicates numbers; index operation indicates Lichtenstein or TAPP; right indicates right side operated; left indicates left side operated; one patient indicates one operation.
Two patients insisted on a laparoscopic pain operation despite the index operation being Lichtenstein’s procedure. A total of 15 patients underwent laparoscopic pain operation after Lichtenstein and 18 after TAPP.
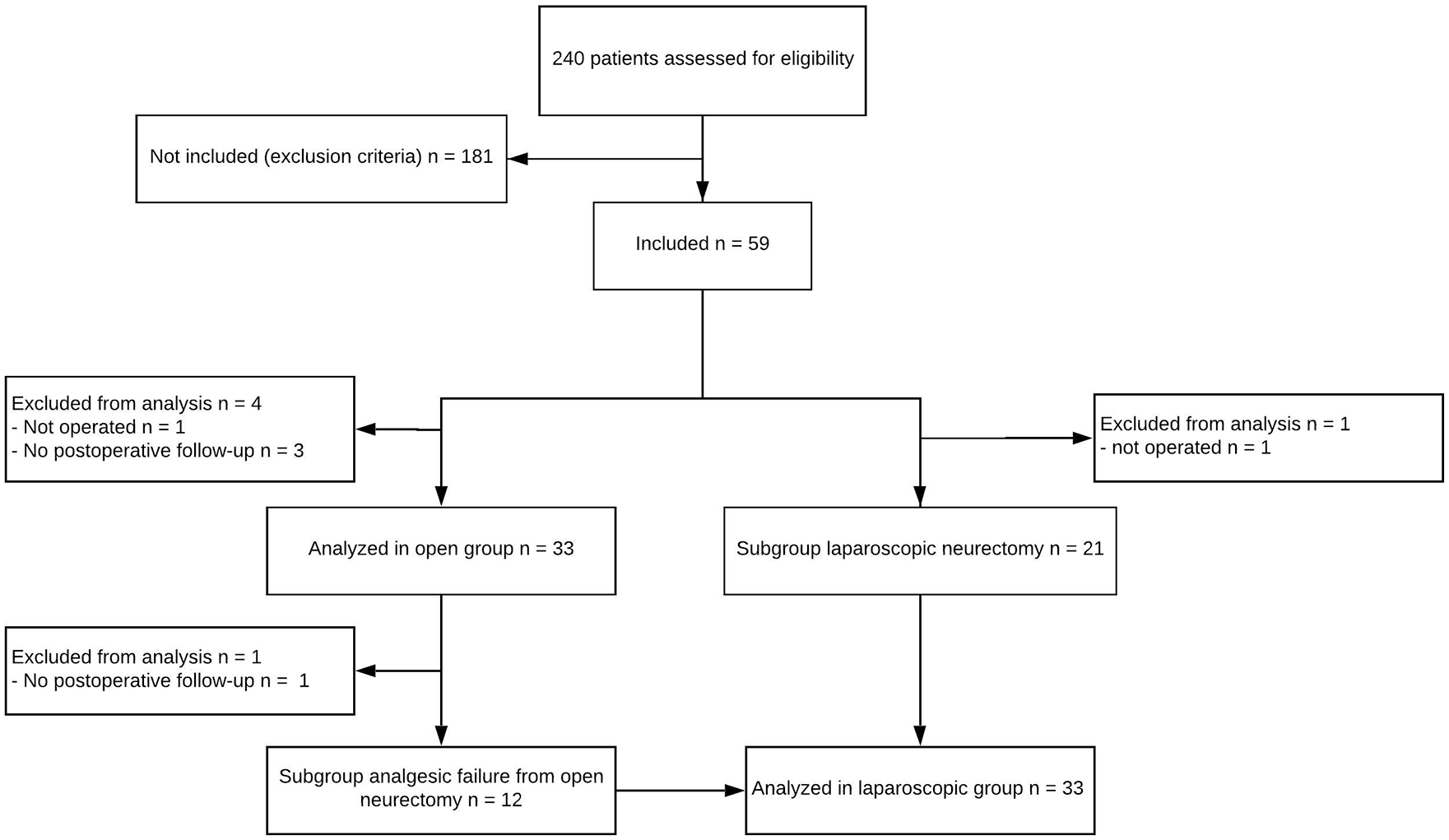
Study flowchart. A total of 240 patients were assessed for eligibility. Of those, 181 patients were excluded. Thirty-seven patients were included in the open neurectomy group. Four patients were excluded from analysis, leaving 33 patients for analysis. Thirty-five patients were included in the laparoscopic group, where 13 of the patients had already undergone an open neurectomy with insufficient analgesic effect. Two patients were excluded from analysis, leaving 33 patients for analysis.
A median of two nerves was resected in the open group (range: 0–3) and three in the laparoscopic group (range: 0–3) (Table 1). Not all patients in the open group underwent a triple neurectomy which was not intentional from the outset but resulted from severe chronic inflammation and poorly defined anatomy within the scarred operation field (i.e. the nerves could not be clearly identified during operation and subsequent histology confirmed that not all three nerves were resected). In the laparoscopic group, the reasons that not all patients underwent a successful triple neurectomy were: two patients in the laparoscopic group had no nerves removed due to mechanical pain (mesh removal only) and three patients were morbidly obese, making the anatomy difficult (two had a single nerve resected and one had two nerves resected). The median postoperative follow-up was 3 months (range: 3–12 months).
Primary Outcome (All Patients)
Postoperative AAS scores were clinically relevant improved (⩾25% change) in 43 patients (68%), not clinically relevant different in 19 (30%), and clinically relevant worsened in one (2%). In the open and laparoscopic groups, AAS scores were clinically relevant improved in 24 (77%) and 19 patients (60%), respectively. The total AAS score changed significantly in both the open group from 50 (range: 19–79) to 27 (range: 0–81) (Fig. 3A) and in the laparoscopic group from 59 (range: 27–89) to 25 (0–75) (Fig. 3A and B, respectively) (P < 0.001).
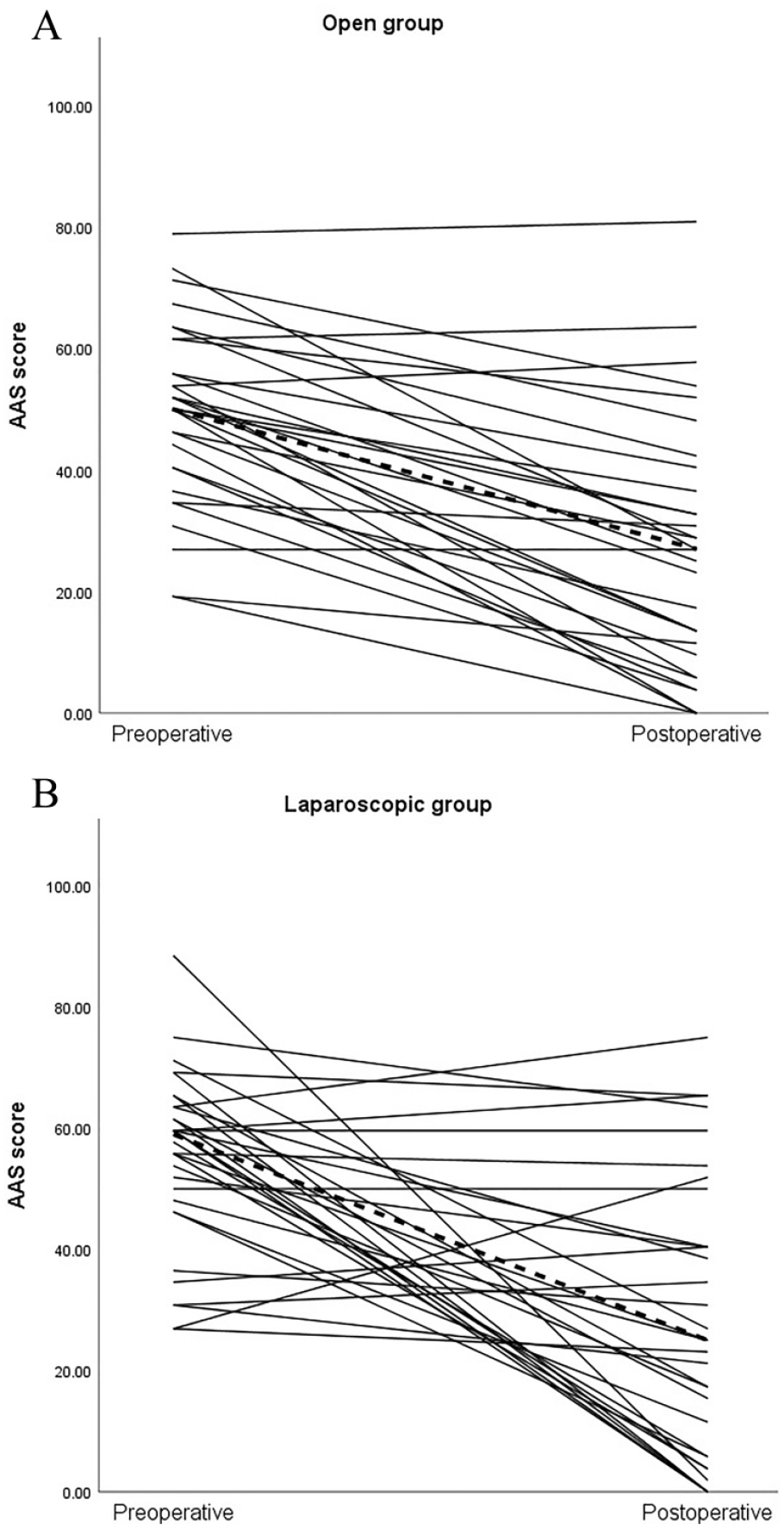
Individual AAS scores for the (A) open and the (B) laparoscopic groups displaying each patient’s pre- and postoperative AAS score.
Primary Outcome (Sub-Groups)
In the first sub-group (n = 21) undergoing laparoscopic neurectomy (no prior open pain operation) (Fig. 2), postoperative AAS scores were clinically relevant improved in 16 patients (76%) and not clinically relevant different in five (24%). The total AAS score changed significantly from 60 (range: 31–71) to 17 (range: 0–75) (P < 0.001).
In the second sub-group (n = 12) undergoing first an open neurectomy and subsequently a laparoscopic neurectomy (Fig. 2), postoperative AAS scores were clinically relevant improved in four patients (33%) and not clinically relevant different in eight (66%). The total AAS score changed from 53 (range: 27–89) to 39 (range: 0–65) (P = 0.15).
Secondary Outcomes
Results from the open, laparoscopic, and total groups are listed in Table 2. The five most bothersome activities/situations in the I-PROM were sitting down for a longer period (n = 22 (33%)), lifting heavy objects (n = 21 (32%)), walking longer distances (n = 17 (26%)), climbing stairs (n = 16 (24%)), and moving during sexual intercourse (n = 16 (24%)). I-PROM scores (total group, n = 66) were clinically relevant improved in 49 patients (74%), not clinically relevant different in 16 (24%), and worsened in one patient (2%). In the total group, the raw I-PROM scores significantly improved (P < 0.001). For the pain-NRS results, moderate/severe pain during the last week, pain-related social disability, and post-ejaculatory pain were reduced from 66 to 24 patients (64% reduction), 44 to 16 (64% reduction), and 39 to 15 (62% reduction), respectively, (P < 0.001) (Table 2). The risk of depression and anxiety was reduced in 13 to 6 patients (54% reduction) (P = 0.092) and in 19 to 6 patients (68% reduction), respectively, (P < 0.05). According to the PDQ, the number of patients with preoperative neuropathic pain was halved postoperatively (P < 0.05). Fifty-one patients (77%) reported that chronic groin pain was reduced after the operation (25 patients in the open group and 26 in the laparoscopic group).
Data are presented as numbers (%).
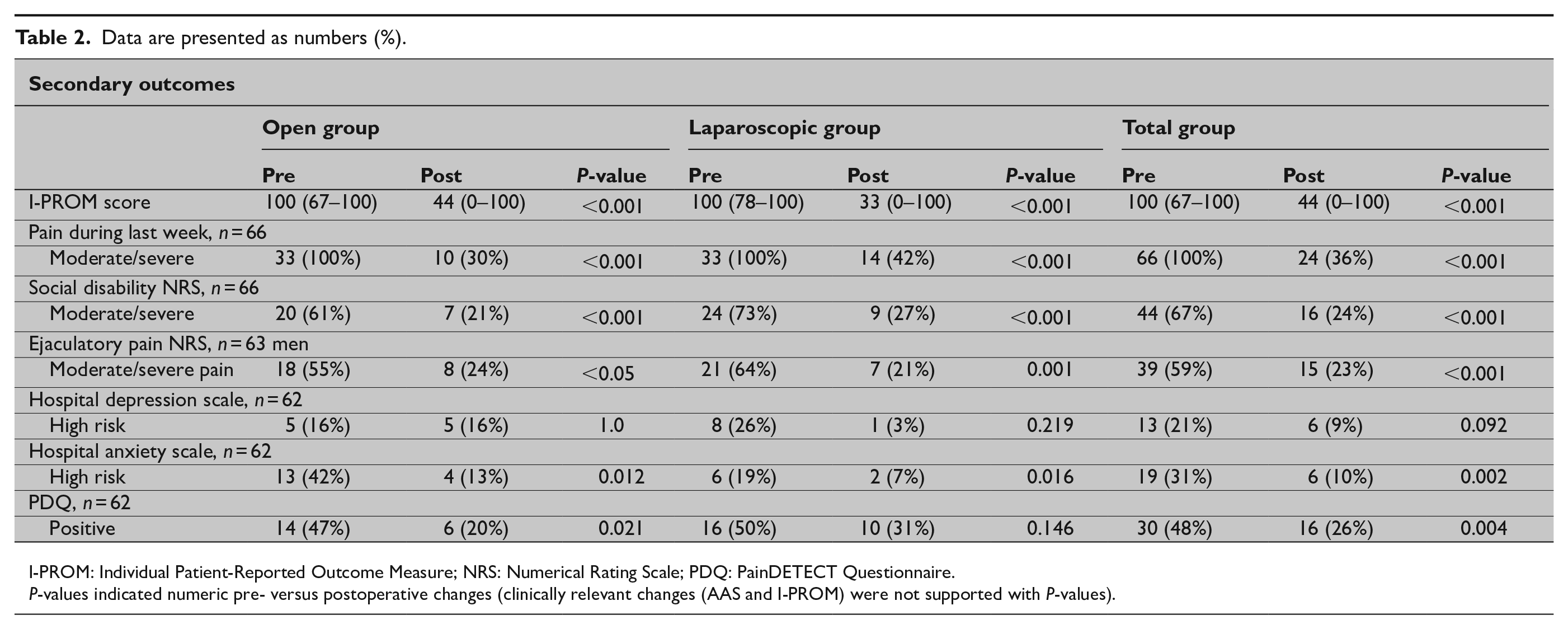
I-PROM: Individual Patient-Reported Outcome Measure; NRS: Numerical Rating Scale; PDQ: PainDETECT Questionnaire.
P-values indicated numeric pre- versus postoperative changes (clinically relevant changes (AAS and I-PROM) were not supported with P-values).
Complications
One patient in the open group needed orchiectomy at postoperative day 3 due to ischemic orchitis. One patient in the laparoscopic group with chronic pain in the right groin and a history of five previous incidents of right-sided pneumothorax (drainage and bleb surgery) experienced an intraoperative diaphragm lesion due to severe adhesions in the quadratus–diaphragm area. The diaphragm lesion was laparoscopically sutured, and neurectomy was not performed. Postoperatively, the patient was followed by specialized anesthesiologic pain team. Two patients in the open group presented with a hematoma/seroma within the first 14 postoperative days. According to the exclusion criteria, the former two were excluded whereas the two latter were included in the analysis.
Discussion
This study results suggest clinically relevant improvement in pain-related functional impairment in roughly 70% of patients with severe chronic pain after inguinal hernia repair. Patients were screened using a simplified clinical treatment algorithm (without routine use of preoperative pain team, imaging, QST, or LA injection), including strict indications for a pain operation. Based on the present treatment algorithm, only 25% of referred patients were offered a pain operation. The remaining patients were referred to local hospitals/pain teams. The number of patients undergoing a second pain operation was too limited to draw any definitive conclusion on a beneficial effect of a subsequent operation.
Severe chronic pain with functional impairment, compromised ability to perform daily activities and work, sexual dysfunction, or disrupted quality of life is reported by 3%–5% of patients after Lichtenstein’s procedure but probably less after minimally invasive TAPP or TEP4 –7. Prospective studies to reduce chronic pain following inguinal hernia repair have, overall, suggested a beneficial analgesic effect in the majority of patients with varying success rates11,13,14,18,20,25. Unfortunately, most studies were small and/or often with unclear defined clinical pain outcome measures. Larger (n ⩾ 100) well-conducted studies have suggested a significant analgesic effect in 69%–95% of patients12,15 in accordance with this study’s results, although a clinically relevant analgesic effect of the surgical treatment was not defined.
Chronic pain can be life-threatening as some experience suicidal ideation, but the risk to reward ratio (suicidal ideation or suicide) is not known. In this study, depression scores (HADS) were unaffected by the operation for chronic pain.
The clinical diagnosis of chronic post-herniotomy pain and the indication to offer an operation are dependent upon a meticulous pain anamnesis focusing on pain characteristics (nociceptive/neuropathic/mechanical)9 –11,18. A recent systematic review 26 suggested that preoperative QST findings showed no consistent association with pain intensity after pain surgery for several types of operations (orthopedic, cesarean procedure, cholecystectomy, etc.), and only a few studies on chronic pain after inguinal hernia repair correlated QST with postoperative clinical pain–related outcomes20,25. A new study used QST to distinguish between inflammatory and neuropathic pain and offered a pain operation (mesh removal) to patients with significant inflammatory pain; however, QST was not able to predict the outcome 27 . The only study, so far, analyzing preoperative predictors suggested that female gender and opioid use were associated with poorer clinical outcome in 136 patients with chronic pain after Lichtenstein’s hernia repair 28 .
The present explorative study is the first to analyze the clinical outcome using solely clinical criteria (anamnesis, exclusion of differential diagnoses, and pain characteristics/intensity) and a pre-determined surgical strategy with open neurectomy/mesh removal for chronic pain after Lichtenstein’s procedure and laparoscopic retroperitoneal triple neurectomy for chronic pain after TAPP. The non-validated I-PROM questionnaire used in this study focuses on patients’ selection of individual complaints. I-PROM was based on individual complaints defined by patients themselves and not by surgeons. Surgeon-defined Patient-Reported Outcome Measures (PROMs) are exclusively used in PROM studies after hernia repair 17 . The I-PROM scores demonstrated clinically relevant improvement. The most bothersome complaints were aggravated pain when sitting down for a longer period, lifting heavy objects, walking longer distances, climbing stairs, and physical activity during sexual intercourse. This specific information has not been addressed in earlier studies.
There are several limitations to the study. First, the relatively few included patients precluded multivariate statistics for the hypothetical predictive power of preoperative variables for the pain-related outcomes. Second, given the lack of a control group, blinding for intervention, and randomization, there is risk of selection-, interviewer-, and response bias. However, the primary and secondary outcomes were registered by patients themselves, where postoperative registrations were blinded toward their preoperative registrations. Third, the low sample size increases the risk of type II errors. Fourth, while the AAS is validated in hernia patients, it is not validated for patients undergoing surgical treatment of chronic pain after inguinal hernia repair. Fifth, the 25% cut-off value for clinically relevant changes was arbitrarily selected but based on previous studies. Other studies have used 15%–30% cut-off values 17 , and 25% has previously been used for chronic pain after inguinal hernia repair 18 . However, the optimal cut-off value does not exist. A higher cut-off value lowers the sensitivity but increases the specificity, and a lower cut-off value increases the sensitivity but lowers the specificity. Sixth, the follow-up time was only 3 months. A 6 or 12 months or even longer follow-up may have been preferable, although there is no consensus in the literature except for a minimum of 3-month follow-up. Seventh, the concept of central sensitization as a neuropathophysiological mechanism for chronic pain was not addressed in the present treatment algorithm since the phenomenon is mainly demonstrated in experimental pain trials and regarded controversial in a clinical setting 29 . Finally, a lack of surgical experience (learning curve) to perform the pain operations cannot be ruled out. In this study, the definition of learning curve was subjective and not based on strict criteria. However, before initiating this study, several operations were performed under international surgical expert guidance prior to patient enrollment. At the time of the study start, the duration of surgery was reduced and more uniform.
As described in the IDEAL framework for surgical innovation 30 , observational studies, like this study, should be large and prospective (to secure external validity) using standardized treatment interventions with well-defined procedure-specific pain outcomes and generic quality of life scores. These chronic pain data can be subjected to future randomized controlled clinical trials for individual surgical pain strategies (for instance, mesh removal vs no mesh removal and open vs laparoscopic neurectomy after Lichtenstein’s index operation). The effect of preoperative LA trigger point injection to predict postoperative outcome should be investigated in a large randomized placebo–controlled trial.
Conclusion
Roughly 70% of the patients achieved clinically relevant improvement using a simplified surgical treatment algorithm for patients with severe chronic pain after inguinal hernia repair.
Footnotes
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: D.C.C. reports personal fees from Medtronic and BD for lectures not related to this study. All other authors declare no conflicts of interest.
Ethical approval
Danish Data Protection Agency, #2019-522-0122. The study was performed as a quality assessment of a novel standard surgical strategy, and approval from the Ethics Committee and the Danish Patient Safety Authority was not required. According to the Danish regulations, the study was not preregistered due to being a quality assessment study.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
