Abstract
Chronic pancreatitis is a long-term illness leading to hospital admissions and readmission. This disease is often caused by heavy alcohol consumption and smoking. Patients with chronic pancreatitis suffer from acute or chronic pain episodes, recurrent pancreatitis, and complications, such as pseudocysts, biliary duct strictures, and pancreatic duct fistulas. Pancreatic duct strictures and stones may increase intraductal pressure and cause pain. Endoscopic therapy is aiming at decompressing the pressure and relieving the pain, most commonly with pancreatic duct stents and pancreatic duct stone retrieval. Early surgery is another option to treat the pain. In addition, endotherapy has been successful in treating complications related to chronic pancreatitis. The therapy should be individually chosen in a multidisciplinary meeting. Endoscopic therapy and surgery as treatment options for chronic pancreatitis are discussed in this review.
Keywords
Pain in Chronic Pancreatitis
Patients with chronic pancreatitis (CP) can be classified with TIGAR-O system into one of the six etiological categories: toxic (T), idiopathic (I), genetic (G), autoimmune (A), recurrent acute and severe pancreatitis (R), and obstructive cause (O) (1). In Western countries, CP is mostly related to excess alcohol use (in 53%–66%) and smoking (in 53%–75%), and patients can have multiple risk factors (2–5).
Pain is present in majority of the patients with CP. Patients may suffer from continuous or intermittent pain or episodes of recurrent pancreatitis. Pain in CP is multifactorial and poorly understood. In plumbing problems, pancreatic fibrosis and pancreatic duct (PD) hypertension are responsible for pain and decompressing therapy is usually effective. In wiring problems, peripheral nerve damage results in both stimulus-dependent and spontaneous, neuropathic pain. In this situation, endoscopic therapy or surgery is usually ineffective. Celiac plexus block or neurolysis may be attempted, but these are out of the scope of this review. CP complications may also cause pain (6). In step-up approach, endoscopic therapy should be attempted if analgesics are not sufficient to treat pain, especially if there is a need of opioids. The idea of endoscopic therapy is to decrease the increased PD pressure by relieving PD stricture with pneumatic dilatation and PD stent, removing the PD stones with extracorporeal shockwave lithotripsy (ESWL) and endoscopic retrograde cholangiopancreatography (ERCP). When treating pain in CP, endoscopic therapy or surgery should be performed early in the disease course to avoid pain centralization and narcotic dependence. Treatment modality should be chosen individually.
Endoscopic therapy is not indicated if there is excess pain and no PD dilatation or stricture or no intraductal stone. Accordingly, if patient with pancreatic calcification and PD dilatation has no pain, there is no indication for endoscopic interventions.
CP patients with alcoholic etiology have more complications and hospitalizations compared to CP cases with other etiologies (7). The rate of hospital readmissions is decreased in those patients undergoing ERCP or pancreatic surgery (8). Patients with CP undergoing invasive treatments should be encouraged to stop alcohol abuse and smoking.
Endoscopic Therapy
PD Strictures
Moderate to severe PD lesions including strictures were found in 53% among 1071 CP patients (5). Painful uncomplicated CP with obstructed main pancreatic duct (MPD) is a treatment indication. According to European Society of Gastrointestinal Endoscopy (ESGE) guideline, the definition of clinical success of endoscopic therapy is no pain during the year following stent removal (9). Dominant PD strictures may be treated with PD dilatation and stenting. Often, 10 Fr stents are recommended and stenting time should be at least 1 year (10–12). Malignancy should be excluded with brush cytology and abdominal imaging. Especially, in patients older than 40 years, new CP diagnosis should be confirmed, as up to 5% of pancreatic cancer patients are first misdiagnosed as CP and their treatment is delayed (13). Prior to plastic stent placement, dilatation with wire-guided balloons of 4–6 mm in diameter is usually necessary. If tight, scarred stricture still exists, Soehendra stent retriever (7, 8.5, or 10 Fr) can “drill” route through the stricture over the guidewire (14). Diameter of the plastic stent should be as wide as possible to prevent clogging and to sufficiently dilatate the stricture. Pancreatic stents with S-form resembling PD or “tilde” mark have large diameter, flaps, and side holes. They were developed to prevent migration and occlusion and work better than biliary stents in PD (15).
Different stenting policies have been suggested. In three studies, PD stents were exchanged only on demand, not regularly. In 3.8–14.4 years of follow-up, 66%–73% were free of pain. Only a few complications were related to stent clogging (10, 11, 16). Obstructed pancreatic stent can act as a wick, which conveys pancreatic juice between duct and outer stent wall from dilated PD to duodenum. However, repeated stent exchanges to avoid stent clogging even every 3 months have been suggested (17, 18). In painful PD stricture, endoscopic therapy with one 10 Fr stent for uninterrupted year with stent exchange combined with control imaging every 6 months is recommended by ESGE guidelines. This is based on non-randomized observational studies, where recommendation is weak and with low-quality evidence. After 6–8 weeks, clinical response to stenting should be checked (9). If patient reports no pain relief, stent should be removed, but if patient reports symptom relief, then stent treatment should last at least a year.
Multicenter study from Germany including 1018 patients with follow-up data of 2–12 years showed no pain in 51.8% of the patients after endoscopic therapy (19). In Japanese study of 59 patients, stent placement succeeded in 41 (70%) patients and median stenting time was 276 days with mean of 3.5 stent exchanges. Stricture recurrence was observed in 17 (41.5%) patients. Risk factors for restenosis were remaining stones in PD after stent removal or stricture location in the body of pancreas (12). Refractory CP pain after the removal of pancreatic stent is common. After 2–4 years, 27%–38% of the patients suffer from pain relapse (10, 11). Multidisciplinary meeting should discuss therapeutic options, either repeated endoscopic therapy or surgery.
In cases of refractory PD strictures, multiple PD stenting has been effective. A total of 48 patients had refractory pancreatic strictures, and multiple plastic PD stents were placed for 6–12 months. After a mean follow-up of 9.5 years, 74.4% of the patients were asymptomatic (20). Also, covered self-expandable metallic stents (cSEMS) have been successfully used as a rescue method after symptomatic stricture recurrence by small series of 32 (21), 15 (22), and 17 patients (23). In our own data, significant differences in stricture resolution (14.3% vs 84.6%, p = .004) and pain improvement (28.6% vs 84.6%, p = 0.22) were evident in patients with stent migration compared to patients without stent migration (23). Also, de novo stricture may be a problem (21).
Biodegradable self-expandable stents were placed in PD to cover PD stricture in 19 patients with CP and preceding plastic stenting for 6 months. Stricture resolution rate was controlled at 6 months. Technical success rate was 58% and clinical success rate was 52% (24). It is possible that these stents degrade too soon to relieve the stricture. In a meta-analysis of 16 studies and 1498 patients with painful CP undergoing endotherapy with a total of 3960 ERCPs, pain was measured with visual analog scale (VAS) or Izbicki and Melzack scales. Immediate relief of pain after endotherapy was achieved in 88% and long-term relief of pain (mean follow-up of 9.7–66 months) in 67% of the patients. Complication rate was 7.9%. In these studies, the stenting time varied between 0.5 and 23 months and stent sizes were between 5 and 11 Fr (25).
In our unit, after PD stricture is crossed with the guidewire, stricture is dilated with 4–6 mm dilatator. We prefer 10 Fr S-shaped PD stent for one interrupted year at a time, depending on the PD diameter. The response to stent therapy is controlled after 8 weeks. If successful, stent exchange is planned after 1 year. Stent removal is planned after 1–2 years. In cases of refractory PD strictures, repeated PD stenting with multiple plastic stents or cSEMS is started, or a surgery is suggested.
PD Stones and Pain
Endoscopic therapy is effective especially, if patients have a short history of symptoms, if the stones are located in the pancreatic head, and if there is no stricture and PD is cleared from the stones. Extracorporeal shock wave lithotripsy (ESWL) is possible for radiopaque stones more than 5 mm in diameter, because smaller stones are difficult to find in fluoroscopy. Stones less than 5 mm are usually removable with ERCP alone (9). In randomized controlled trials comparing ESWL alone (n = 26) to ESWL + ERCP (n = 29), both were equally effective in pain relief, but cost per patient was three times higher in ESWL + ERCP group. The pain relapsed after 2 years in 38% and 45% of the patients, respectively. In this study, 10 Fr PD stents were used for a period of 12–24 months. Stents were exchanged every 6 months, which may explain part of the expenses (26). However, in the presence of downstream stricture, ESWL alone may not be sufficient for pain relief in CP. Pain can be relieved, if stent is placed beyond the stone and stricture. In our unit, we always combine ESWL with ERCP.
In a Chinese study, patients with PD stones and pancreatic pseudocysts (n = 59) were compared to a control group of 790 patients with PD stones but without pseudocyst. After ESWL + ERCP and a median follow-up of 21.9 months (range: 12–45), pseudocyst disappeared in 76% of the patients in a year. Complete or partial pain relief was achieved in 90% of the patients in pseudocyst group with no difference in complication profile. Adverse events in patients with or without pseudocyst were similar, 11.9% versus 12.4% indicating that ESWL is safe even when pseudocysts are present (27). In an Indian follow-up study of short-term effect of ESWL + ERCP in more than 5000 patients, stone clearance was achieved in 3722 (72.6%) cases. In majority, 98% patients had pancreatic sphincterotomy and 3536 (69%) patients had a pancreatic stent. Altogether 69% of the patients with 6 months follow-up were free of pain. Only in 5.3% of the cases, pain intensity remained the same (28). In 5 years follow-up after ESWL and ERCP, 60% of the patients with idiopathic CP (n = 272) had no pain (29). In our retrospective analysis of 89 patients with calcific CP of mostly alcoholic origin and stones with a median diameter of 10 mm (5–25 mm), ESWL + ERCP with PD stenting was performed. Clinical success was primarily 89%. In the median follow-up of 53 months in 63 patients, phone survey was undertaken and 93% of the interviewed patients were free of pain (4).
After the removal of PD stone, patients chose if their PD stricture was either stented (n = 20) or not (n = 22). In a follow-up of 3 and more years, 15% of the stented patients suffered from recurrence of pain compared to 50% of those without PD stent (30). In a meta-analysis of 27 studies and 3189 patients, ductal clearance was obtained in 70.7% and pain was absent in 52.7% (95% confidence interval (CI): 50.85–54.56) of the cases. Quality of life (QoL) improved in more than 88% of the patients after ESWL + ERCP (31).
Peroral pancreatoscopy (POP)-guided intraductal treatment with electrohydraulic lithotripsy (EHL) or laser is an option when ESWL fails or as an alternative to ESWL. In a retrospective analysis, 39 out of 46 patients underwent POP-guided PD stone extraction with EHL (n = 31) or laser (n = 15), with a technical success of 87% versus 100%. Clinical success was 74% and complication rate was 10% (32). In a review of 17 studies and a total of 225 patients, clinical success varied between 74% and 100% and adverse event rate was 0%–30% (33). In a retrospective analysis of 109 patients from 17 centers, patients with PD stones underwent POP-guided lithotripsy with either EHL (n = 59) or laser (n = 50). Clearance was achieved in 89% of patients. This was possible in single session in 74% of the patients. ERCP prior to lithotripsy had been performed to 88% of the cases. PD stents were placed in 98% of the cases. Adverse event rate was 10%, including five patients with pancreatitis (34). It may be difficult to get access to PD with pancreatoscope (position of the scope, strictures in PD, form of PD, and diameter less than 5 mm) especially in native papilla scenario. Also, post-ERCP pancreatitis risk is increased with prolonged manipulation of papilla. Therefore, POP should be second-line treatment option, if ESWL fails or is not available.
Pancreatic Pseudocysts
In 20 years of follow-up in 245 CP patients, 56 (23%) developed pseudocysts (35). By Chinese multicenter study during 1994–2004 and with 2008 patients, 26% developed pseudocysts (36). In a study cohort of 1071 CP patients, the prevalence of pseudocysts was 42% (5). Pseudocysts associated with CP has lower spontaneous resolution tendency than those related to acute pancreatitis. Altogether 25 (26.3%) of 95 pseudocysts resolved spontaneously (37). Therapeutic measures are indicated only in pseudocysts associated with pain, outlet obstruction, weight loss, early satiety, jaundice, or complications like infection and bleeding, rupture, or fistulization to nearby organs (9).
Secretin magnetic resonance cholangiopancreatography (MRCP) can give information of PD rupture or disconnected duct (9). Conscious sedation or intubation especially in treating large pseudocysts is recommended. Pseudocyst drainage can be performed with transgastric route and preferably two pigtails are left in place to resolve pseudocyst. If performed under endoscopic control, pseudocyst is entered with Zimmon needle, and guidewire-controlled dilatation (8 mm) of the punction site is followed by double-pigtail placement. Transpapillary route with PD stent is another option. In a prospective study of 50 patients and pseudocysts with mean size of 8 cm, 58% did not bulge to digestive wall. Endoscopic ultrasound (EUS) guidance was necessary in 56% of the cases. Combining EUS, conventional transmural, and transpapillary methods, final success rate was 96% (38).
In a randomized trial of pseudocysts drained with pigtails, recurrence rate was significantly lower in cases when pigtails were left in place (0/15) than in cases with stent retrieval (5/13) after pseudocyst resolution (39). Especially, in CP, removal of pigtail may increase the recurrence rate in the presence of PD stricture or stone or if tail of the pancreas is isolated as in disconnected duct. Antibiotic prophylaxis is indicated when pseudocysts are drained (40). Samples for amylase level and bacterial culture can be taken.
In a prospective randomized trial of 20 patients in each treatment arm, endoscopic and surgical cystogastrostomies were compared. After 24 months of follow-up, recurrence and complication rates were similar. With endoscopic therapy, mental and physical health was better after endoscopic treatment, hospital stay was shorter, and costs were lower (41).
In a meta-analysis of 12 studies and 342 patients with pancreatic pseudocysts, method equivalence of surgery and endotherapy (pseudocystogastrostomy in 92% and transpapillary PD stenting in 8%) was similar, p = 0.07, with 81%–100% and 53%–95% treatment successes, respectively. Also, the rate of adverse effects was similar, that is, 0%–18.6% in surgery and 0%–23.5% in endotherapy groups, respectively, p = 0.48. With endotherapy, time of hospitalization was shorter and treatment costs were lower (42).
In a meta-analysis of 17 studies and 881 patients, lumen-apposing metal stents (LAMS) were compared with plastic stents, when transmural drainage of pseudocysts or walled-off necrosis was performed. Treatment success was 85% (95% CI: 81%–89%) versus 83% (95% CI: 74%–89%), respectively, when pseudocysts were treated and lower, 70% (95% CI: 62%–76%) versus 78% (95% CI: 50%–93%), in cases of walled of necrosis. Adverse event rate and recurrence were similar (43). Double pigtails are cheaper and recommended by ESGE guidelines (9).
According to our data, in cases of pancreatic pseudocyst and pancreatic pseudoaneurysm, after bleeding control with angioembolization, endoscopic therapy with transmural or transpapillary route to drain the pseudocyst should be the treatment of choice for the majority of CP patients, and the surgery is preserved for failures of embolization (44). Among 32 patients with pseudocyst associated with bleeding pseudoaneurysm, endoscopic therapy with transmural (n = 8, stents left in place) or transpancreatic route (n = 24, median stenting time 15 (4–41) months) was effective in 30 (93.8%) patients. Of note, 27% of the pseudocysts disappeared after embolization spontaneously and no endoscopic treatment was needed (45).
Biliary Strictures in CP
Acute inflammation episodes with edema, fibrosis, and even pseudocyst may cause bile duct obstruction in CP. Biliary strictures are present in 13%–23% of the CP patients (5, 36, 46). Malignancy should be ruled out with imaging and brush cytology. Resolution of edema can resolve bile duct obstruction. Therefore, persistent asymptomatic elevation of alkaline phosphatase, bilirubin, or both for at least a month is an indication for therapy. Challenge with endoscopic therapy is tight scarred stricture and suboptimal patient compliance. Multiple plastic stents side by side with 3 months exchange for 14 months were superior to single stenting with 3 months exchange for 21 months, when rate of cholangitis, diameter of bile duct, and liver function test were concerned (47). On control ERCPs, increasing the number of plastic stents after pneumatic dilatation in 3-month intervals gradually modifies stricture area and relieves it. Multiple side-by-side plastic stents have been the golden standard for benign biliary stricture (48).
The cSEMS have become an alternative to multiple plastic stents. In a Finnish multicenter randomized controlled trial (RCT) multiple plastic stents (n = 30, adding stents in 3 month intervals and a total of 6 stents) and cSEMS (n = 30) for 6 months were compared. Biliary stricture resolution in 2 years follow-up was similar (90% vs 92%). Procedure time with cSEMS was shorter (49). In a multicenter study (112 benign biliary stricture patients including 35 cases with CP), a comparison between 55 patients (multiple plastic stents) and 58 patients (cSEMS) was performed. Stricture resolution rate was 85.4% for plastic stents and 92.6% for cSEMS. Mean number of ERCP to achieve stricture resolution was significantly lower with cSEMS (3.24 with plastic stents and 2.14 with cSEMS) (50). In addition, placement of cSEMS is easier, dilatation prior to stent placement is not necessary, and the number of ERCP is limited to placement and removal, instead of repeated procedures after 3 month intervals. Also, procedure time is shorter and with a wider stent diameter, stent obstruction rate is lower. However, stent migration risk and high price are disadvantages with cSEMS. In a multicenter study, after 10–12 months of stenting, cSEMS were removed. After a median of 58 months, the probability of remaining stent free was 61.6% (95% CI: 52.5%–70.7%). If at the stent removal stricture was resolved, the probability to remain stent free was 77.4% (95% CI: 68.4%–86.4%). The authors suggest cSEMS as the first-line treatment of biliary stricture in CP (51).
Pancreatic Fistula, Pancreatic Ascites, and Pleural Effusion
Pancreatic ascites and pleural effusion are rare incidents and can be related to pancreatic surgery, pancreatic trauma, and acute or CP. Pancreatic ascites may be present in 2% of the patients with CP (36). Pleural effusion is of pancreatic origin in 1% (52) and mostly on the left side. In ascites or pleural effusion, amylase level is over the upper limit of normal for serum value, usually hundreds or thousands. In a multicenter study of 1071 CP patients from Scandinavia and Baltic countries, ascites, pleural effusion, and pancreatic fistula were classified as inflammatory complications with the prevalence of 1%–6% (5). In our unit, ERCP was effective in 82% of pancreatic fistulas (53). In a retrospective study from India, 17 out of 29 patients had CP, and 15 patients had ascites, 6 patients had pleural effusion, and 7 patients had both. PD stenting was effective in 96.4% of the cases, when stents were removed after 3–6 weeks. There was no recurrence after follow-up of 17 months (54). Endoscopic treatment is the first-line treatment in pancreatic fistula, and the surgery is preserved for cases where endotherapy is not successful. Treatment algorithm of endotherapy and surgery in CP is presented in Fig. 1.
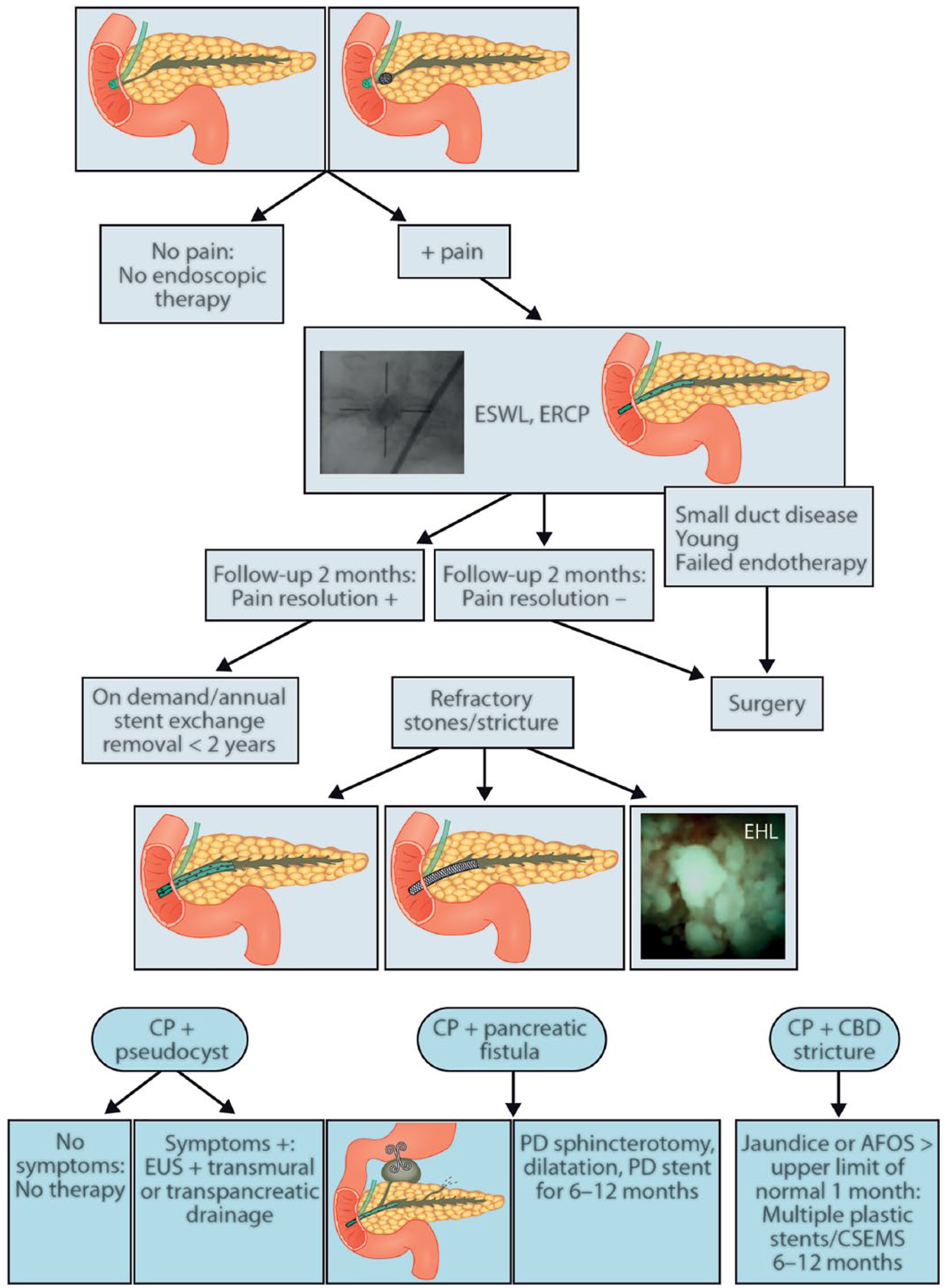
Treatment algorithm of endotherapy and surgery in CP pain and complications.
Complications Related to Endoscopic Treatment
Stent migration distally can lead to stent impaction to duodenal wall and even perforation. S-shaped stent may have lower migration tendency (15) than straight stents originally designed for biliary tract. cSEMS have also migration tendency despite antimigration flaps (23) and risk of de novo strictures (21). In case of proximal stent migration dilatators, rat forceps, Soehendra stent retriever, or even POP-guided procedures are needed. Infections following pseudocyst drainage are possible, and antibiotic therapy or stent exchange may be needed. Post-ERCP pancreatitis, sepsis, cholangitis, and post-sphincterotomy bleeding have risk of 6%, 2.6%, 2.3%, and 1.5%, respectively. Long-term stent therapy may cause ductal injury (9).
Endoscopic Therapy Versus Surgery
In painful CP, surgery is considered superior to endoscopic therapy in pain relief. Two randomized trials compared endoscopic intervention with surgical therapy (17, 55) in painful obstructive CP. Surgical group had a higher proportion of patients with pain relief in 2–5 years and ⩾5 years of follow-up as compared to endoscopic group. The surgery resulted in improved QoL and improved preservation of exocrine pancreatic function at 3–5 years of follow-up, but not thereafter.
In Czech Republic study of Dite et al., 52% of patients agreed to be randomized: 36 to surgery and 36 to endoscopic therapy. Those who refused for randomization chose between surgery (n = 40) and endotherapy (n = 28). Altogether 68 patients had endoscopic therapy: pancreatic sphincterotomy (100%), PD stenting (52%), and stone extraction (23.4%). Stents were exchanged every 2–4 months with a median stenting time of 16 months (range of 12–27). In a surgery group, 61 patients underwent resections (80%) and 15 cases underwent draining procedure (20%). Initial pain relieving effect was similar in both groups (90%). After 5 years of follow-up, complete absence of pain was achieved in 15% versus 33.8% patients with endotherapy and surgery, respectively. ESWL was not available and in endotherapy group, PD stents were used in only half of the patients (17). In Netherlands, Cahen et al. randomized CP patients to either endoscopic treatment including ESWL (n = 19) or surgery (n = 20). Nine patients had PD stents (Amsterdam type 10 Fr biliary stent without side holes) for a median of 27 (6–67) weeks. Stents were exchanged at 3-month intervals, until stricture resolution. At 2 years follow-up, partial or complete pain relief was achieved significantly more often after surgery, in 75% patients, than after endoscopic treatment, in 32% patients (55). Sphincterotomy alone was considered as a sufficient endotherapy and ESWL was not routine in these studies. With biliary stents instead of PD stents, there may be an increased rate of stent migration and stent obstruction. So, endoscopic therapy in these studies has been suboptimal according to the present guidelines (9) and there is limited comparative data of this issue.
Surgical Therapy in CP
Asymptomatic patients with CP should not be operated. Early surgery seems better than surgery at an advanced stage of disease in achieving long-term pain relief with lower risk of pancreatic insufficiency and lower reintervention rates (56). CP often leads to numerous inpatient readmissions. Surgical intervention only occurs in a minority of cases (57).
There are several operative techniques which may be used in CP (Table 1). In patients with dilated MPD (⩾5 mm) and normal-sized pancreatic head, a lateral pancreaticojejunostomy with a Roux-n-Y loop and Frey’s procedure may be used (58–60). Both techniques seem to be equal in terms of pain control. Lateral pancreaticojejunostomy is a relatively safe procedure. Up to 90% of patients experience pain relief initially, but the long-term pain relief can decline to 33%–53% by 5 years, and additional procedures may be needed.
Different operation techniques for the treatment of chronic pancreatitis.
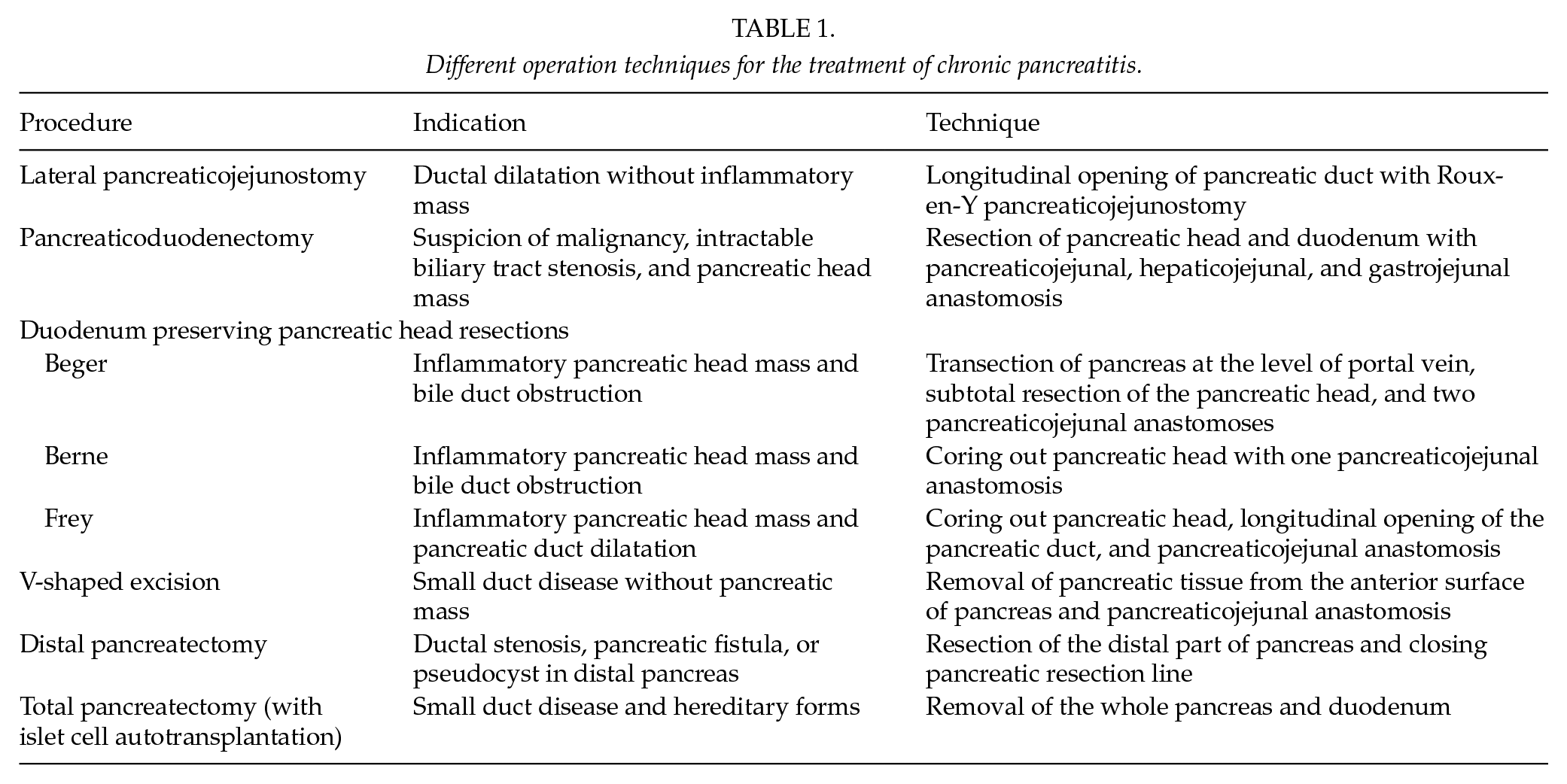
The head of the pancreas often acts as a trigger for CP. Patients with a pancreatic head mass may be treated with conventional pancreaticoduodenectomy or with duodenum preserving pancreatic head resections (58–60). Both operations carry a significant risk of morbidity (16%–53%). No significant differences have been observed between conventional pancreaticoduodenectomy and duodenum preserving pancreatic head resection in long-term pain relief, QoL, or exocrine and endocrine pancreatic insufficiency (24%–51% and 12%–48%, respectively). Occupational rehabilitation is better after duodenum preserving pancreatic head resection. There are two modifications of duodenum preserving pancreatic head resections: Beger and Berne procedures. In Beger operation, pancreas is cut at the level of portal vein and the head of the pancreas is subtotally resected. Two pancreaticojejunal anastomoses are performed to a Roux-en-Y limb. In Berne modification, pancreatic head is cored out and pancreaticojejunal anastomosis is performed to the jejunal Roux-en-Y limb.
Frey’s operation combines coring out the head of the pancreas and lateral pancreaticojejunostomy. Frey’s operation is indicated if the PD is dilated in addition to the pancreatic head mass. A meta-analysis of 323 patients comparing Berger, Frey, and Berne operations could not show any differences in post-operative pain, morbidity, mortality, or exocrine and endocrine insufficiency (61). According to another meta-analysis of 423 patients, there were no differences in pain, post-operative morbidity, and mortality. However, weight gain after surgery was better, and the operation time and hospital stay were shorter after duodenum preserving operations as compared to pancreaticoduodenectomy (62). In a 15-year follow-up, overall survival has been better after Frey operation than that after pancreaticoduodenectomy (63). Operating time and the length of hospital stay are shorter after Berne procedure than for that of Beger and Frey operations (64). A total pancreatectomy should be considered in patients without PD dilatation, who are resistant to medical, endoscopic, and previous surgical treatment and who have severe pain. Especially, those patients who have hereditary forms of pancreatitis are candidates for total pancreatectomy (58, 59). Total pancreatectomy provides long-term pain relief in 80%–85% of patients. The procedure is associated with a high rate of post-operative morbidity and a complete endocrine and exocrine insufficiency. Islet cell autotransplantation has been used to help to maintain glycemic control even though majority of patients do not gain insulin independence (65).
Distal pancreatectomy has been used to treat patients with a major obstruction of the MPD in the body or tail of the pancreas or those with disconnected PD (58, 59). The efficacy in long-term pain relief ranges from 55% to 81%. Distal pancreatectomy carries a significant risk of post-operative morbidity (15%–35%), especially pancreatic fistula formation. After distal pancreatectomy, 29%–47% and 38%–69% of the patients experience exocrine and endocrine insufficiency to some degree, respectively.
Initial therapy for groove (paraduodenal) pancreatitis should be medical or occasionally endoscopic. The success rate after medical and endoscopic therapy is 50% and 57%, respectively. If these are unhelpful, the patient should be referred for surgery. The surgery should be aimed for pain resolution and should also solve patient’s nutritional problem. The most suitable surgical option in experienced hand is pancreaticoduodenectomy (58–60, 66). Complete symptom relief is achieved in 79% of the patients after surgery (66). Duodenum preserving pancreatic head resections seem to be ineffective in groove pancreatitis. Flowchart of surgical techniques in different clinical scenarios is presented in Fig. 2.
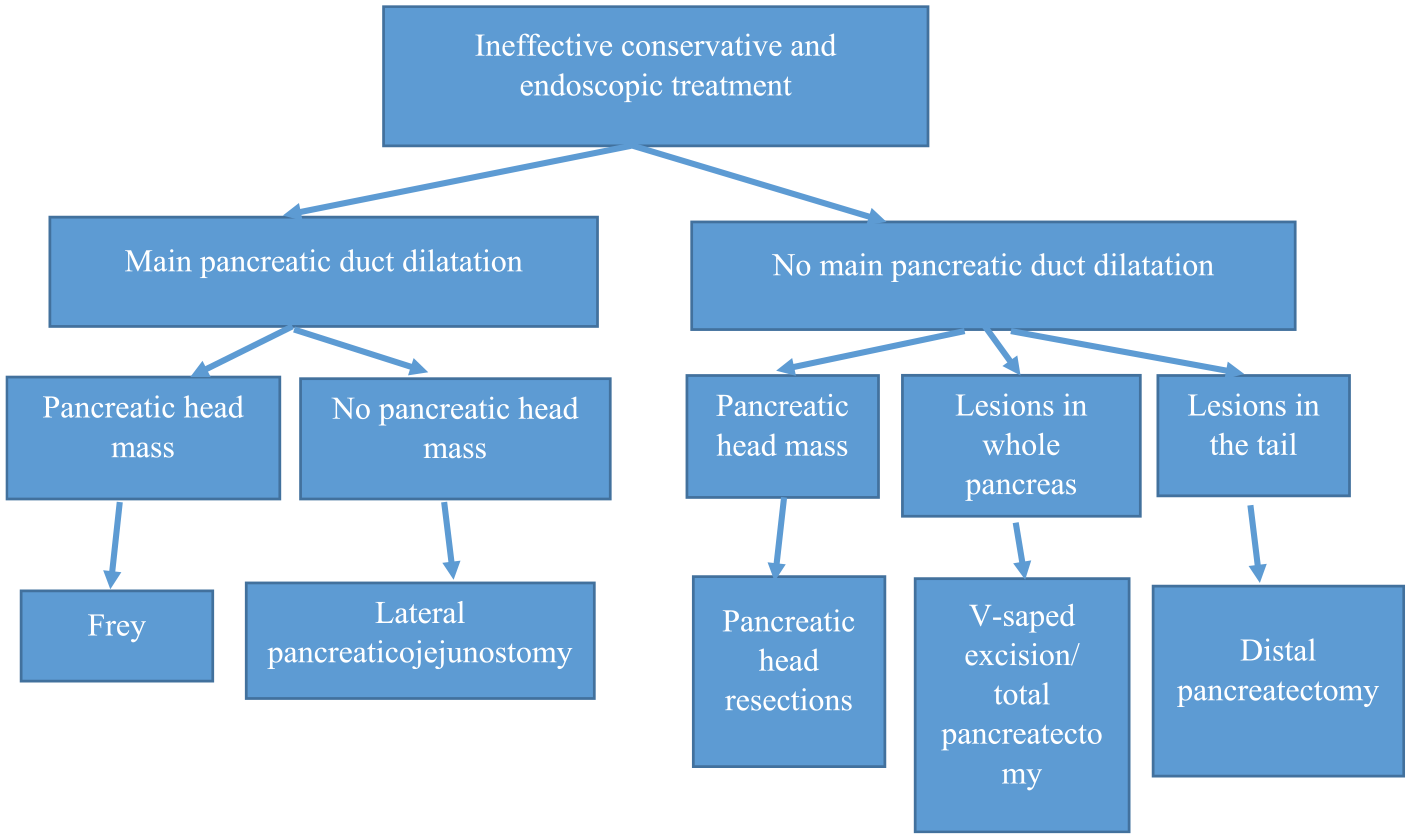
Surgical treatment of chronic pancreatitis.
The Role of Endoscopic Therapy and Surgery in the Treatment of CP
There are no placebo-controlled or sham-controlled trials of the effect of endoscopic therapy in CP. Seven randomized trials and 202 CP patients treated with placebo showed that pooled estimate of the placebo rate for pain remission was 19.9% (95% CI: 9%–36%) (67). Endoscopic therapy seems to be beneficial, but pain related to CP is intermittent. Computed tomography (CT) and magnetic resonance imaging (MRI) images, dilatation of the PD and strictures, and duct calcifications do not necessarily correlate with pain.
Ideally, therapeutic procedures are performed early in the disease course, when patient is not opioid dependent yet and without wiring problem. An online survey of opinions regarding diagnosis and treatment of CP was sent to 288 pancreatologists. Majority chose total pancreatectomy for pain and without MPD dilatation or if other methods failed. In cases of enlarged pancreatic head, initial pancreaticoduodenectomy was recommended by 58%, whereas 42% preferred initial endoscopic therapy. In cases of PD stone and dilated duct, 59% of the respondents chose ESWL + ERCP and 29% preferred surgery. The authors concluded that decision making is based on local expertise and beliefs (68). In addition to numerous national and continental guidelines and review articles of CP, high-quality prospective randomized trials and evidence-based guidelines are needed. Then, daily doses of pain killers must be carefully monitored, and the severity of pain scales should be uniform so that the studies are comparable. The role of endoscopic therapy in CP pain compared to placebo or surgery must be clarified (69). Endoscopic therapy is less invasive, and the surgery is irreversible. However, endoscopic therapy related to CP is demanding and may be operator-dependent. The complexities of CP warrant early multidisciplinary evaluation and consideration of surgical and non-surgical options. A step-up approach is currently preferred. Endoscopic therapy is the first-line effective and safe treatment for complications related to CP. Invasive treatment of CP should be centralized in high-volume centers.
The patients with CP must be encouraged to quit smoking and stop drinking alcohol. If they do not, we still have to take care of them and offer them some form of treatment.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
