Abstract
Acute pancreatitis is a common general surgical emergency presentation. Up to 20% of cases are severe and can involve necrosis with high associated morbidity and mortality. It is most commonly due to gallstones and excess alcohol consumption. All patients with acute pancreatitis need to be scored for severity and patients with severe acute pancreatitis should be managed on the high dependency unit. The mainstay of early treatment is supportive, with care to ensure strict fluid balance and optimisation of end organ perfusion. There is no role for early antibiotic use in acute necrotising pancreatitis and antibiotics should only be used in the presence of positive cultures. Nutritional support is vitally important in improving outcomes in necrotising pancreatitis. This should ideally be provided enterally using an naso-jejunal tube if the patient cannot tolerate oral intake. Patients with significant early necrosis, persisting organ dysfunction, infected walled off necrosis requiring intervention or haemorrhagic pancreatitis should be referred to a regional hepato-pancreatico-biliary unit for advice or transfer. Percutaneous and endoscopic necrosectomy has replaced open surgery due to improved outcomes. Acute necrotising pancreatitis remains a complex surgical emergency with high morbidity and mortality that requires a multidisciplinary approach to attain optimum outcomes. The mainstay of treatment is supportive care and nutritional support. Patients with significant pancreatic necrosis or infected collections requiring drainage require input from a tertiary HPB unit to guide management.
Introduction
Acute pancreatitis is one of the commonest general surgical emergency presentations to hospital in the UK. The incidence of acute pancreatitis is rising with the condition affecting 13–45 per 100,000 people. 1 Most district general hospitals (DGHs) admit around 100 cases of pancreatitis per year. 2 The resulting inflammatory process in the pancreas can precipitate the systemic inflammatory response syndrome (SIRS) and result in multi-organ dysfunction syndrome and death. The majority (80%) of cases of acute pancreatitis are mild and resolve spontaneously with supportive care. By contrast, 20% of cases are severe and can involve organ necrosis, may require critical care support and have a reported mortality of up to 30%. 2
The aetiology of acute pancreatitis is varied. In the UK, the commonest causes are gallstones (approximately 40%) and alcohol (25–35% of cases). 3 Rarer causes include ERCP-induced pancreatitis, trauma, drugs (azathioprine, corticosteroids, 5-aminosalicylic acids), hyperlipidaemia, hypercalcaemia, viral pancreatitis, hereditary pancreatitis, malignancy and autoimmune pancreatitis. Current UK guidelines aim to confirm the cause of pancreatitis is 80% of cases, thereby leaving only 20% of cases labelled as idiopathic. 4 This review will focus on severe necrotising pancreatitis and recommendations for both local management and indications for transfer to a tertiary HPB unit.
Predicting the severity of pancreatitis
Traditionally, the severity of pancreatitis was predicted using the Glasgow–Imrie criteria for acute pancreatitis. 5 However, these criteria had the disadvantage of only being applicable 48 h after admission. At presentation, in addition to thorough clinical examination, the APACHE II score should be calculated. An APACHE II > 8 is predictive of severe pancreatitis. Both the Glasgow–Imrie score and the APACHE II have an accuracy of 84%. 6 Other parameters that are useful in predicting severity include CRP > 150 mg/l after 24 h and persisting organ failure 48 h after admission. 4
The revised Atlanta classification of acute pancreatitis
7
should now be used to predict severity and guide future management in all patients with severe acute pancreatitis. This classification is based upon both:
local factors (pancreatic necrosis) systemic factors (organ failure)
The grade of severity is based upon both local and systemic factors. In addition, based on CT imaging (ideally a CT pancreas protocol to best quantify pancreatic necrosis), the revised Atlanta classification divides acute pancreatitis and associated fluid collections into:
interstitial oedematous pancreatitis
○ within four weeks – acute peripancreatic fluid collections ○ after four weeks – pseudocyst necrotising pancreatitis
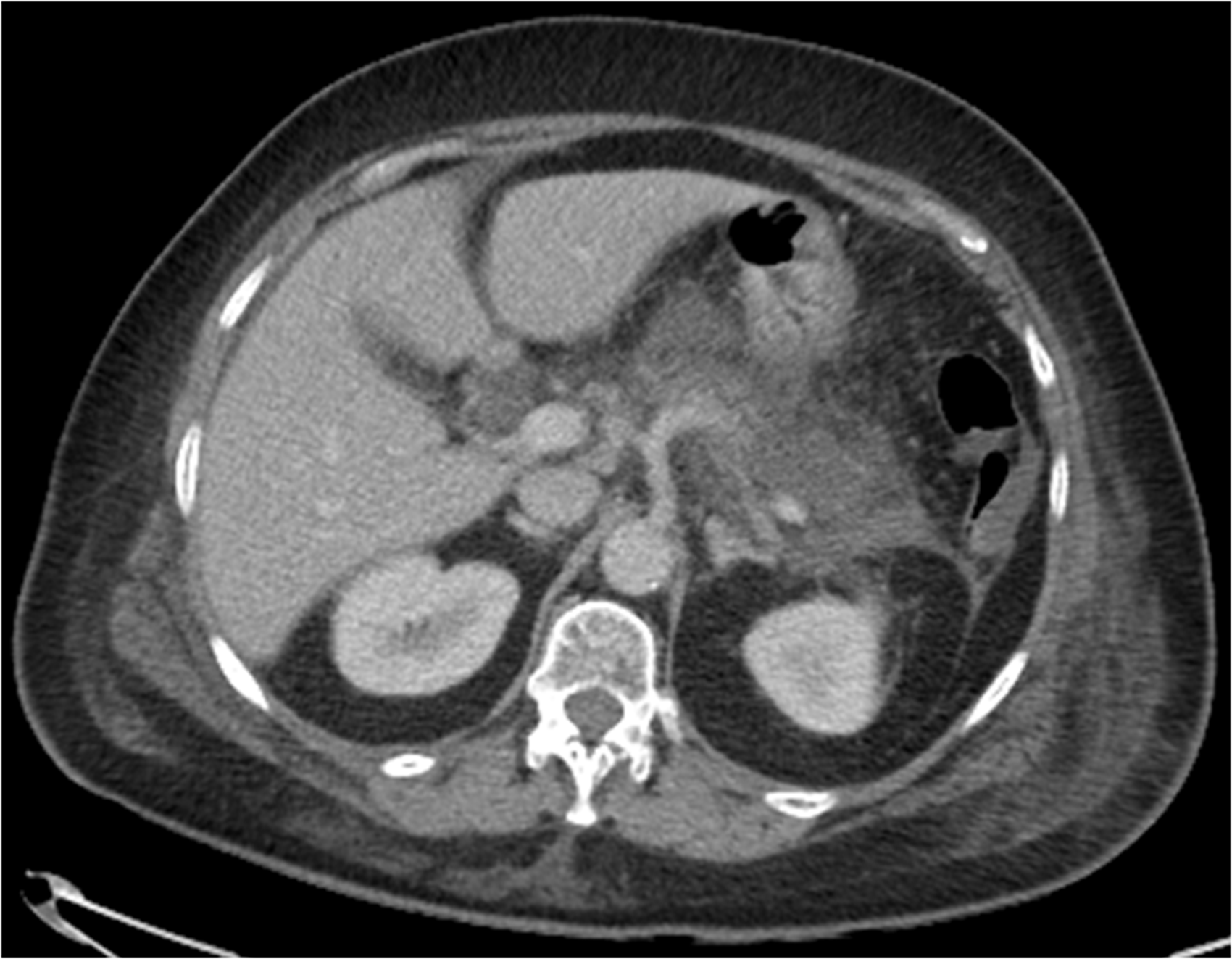
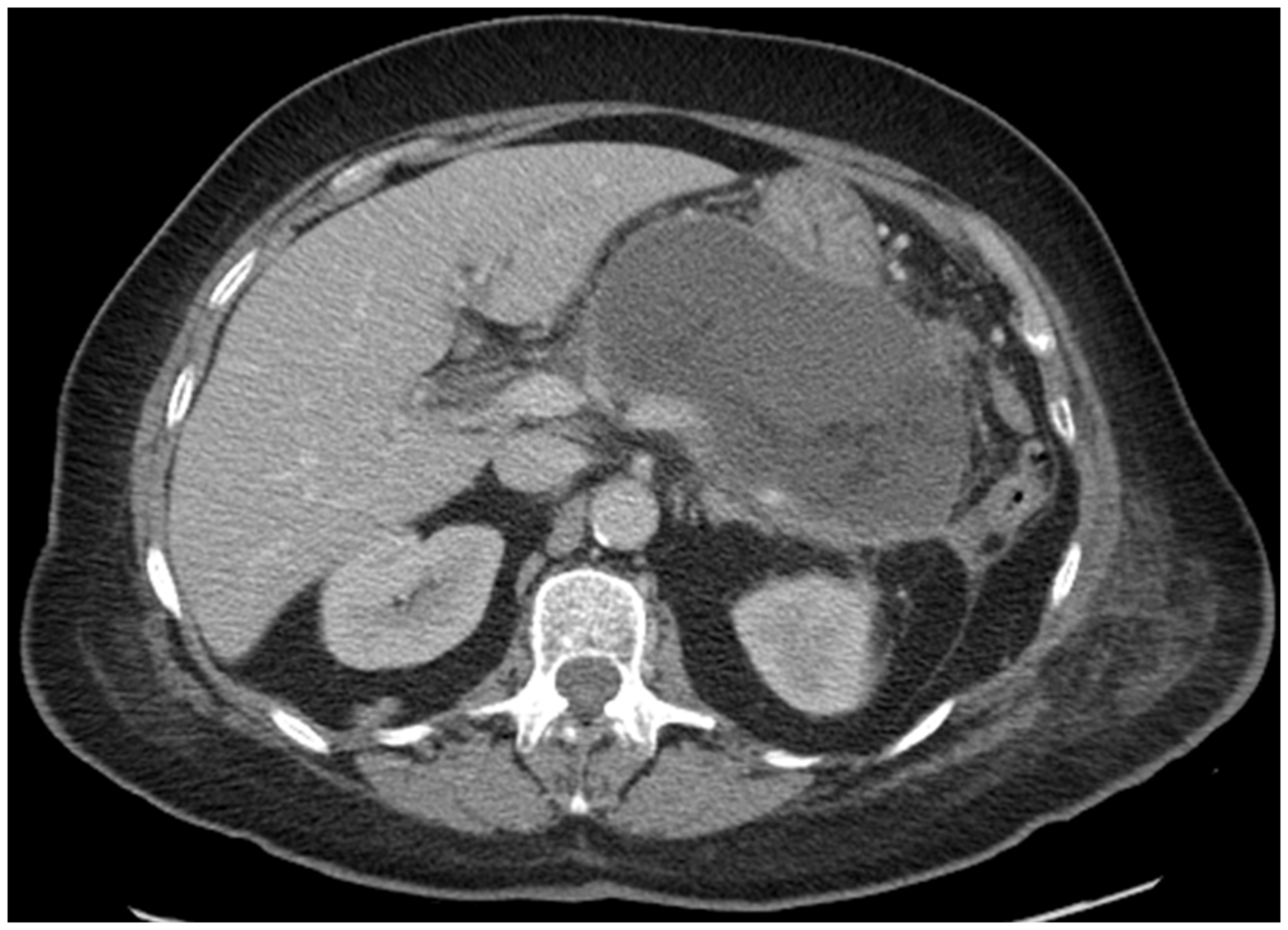
Extensive early necrosis of the body and tail of pancreas 72 h after admission with severe gallstone pancreatitis. Large collection of WON four weeks after admission with necrotising pancreatitis secondary to gallstones.
UK guidelines state that all patients with severe pancreatitis should be managed by the same team within each hospital 4 and managed on the high dependency unit (HDU). 4 This can be difficult in practice but all patients with severe pancreatitis not on HDU should be referred to the critical care outreach team for regular review and escalation of care if deterioration occurs.
Early management of necrotising pancreatitis at the DGH
The majority of patients with early necrotising pancreatitis can be managed at their local hospital and the mainstay of treatment in the early stage of the disease is supportive care. A prospective cohort study showed that 62% of patients with severe pancreatitis had SIRS on day 1 after admission. 8 SIRS predisposes patients to third space fluid losses and the acute respiratory distress syndrome. Fluid resuscitation and respiratory support via high flow oxygen to aid oxygen delivery is essential. All patients with necrotising pancreatitis require urinary catheterisation and a strict input/output chart as they are at high risk of fluid overload, which precipitates respiratory complications. The optimal fluid for resuscitation has yet to be elucidated, with a wide variety used in surgical practice. 9 Invasive monitoring and inotropic support may be required to avoid excessive fluid replacement.
Antibiotic use in early necrotising pancreatitis
Prophylactic antibiotics have no role in the early management of necrotising pancreatitis. Trials performed in this area show great heterogeneity with a variety of antibiotics used for different durations. 4 One randomised control trial showed no difference in the incidence of infected necrosis or mortality between patients given ciprofloxacin and metronidazole versus placebo. 10 Antibiotic therapy should only be instituted in the presence of positive bacterial culture (either from pancreatic necrosis aspirate or peripheral blood culture) and the appropriate agent used based on microbiological sensitivities. 11
Nutritional support in necrotising pancreatitis
Approximately 80% of patients with necrotising pancreatitis are in a prolonged catabolic state 12 and the negative nitrogen balance can reach 40 g/day. 13 Therefore, nutritional support is essential to prevent or minimise malnutrition. Oral intake is generally poorly tolerated by patients with necrotising pancreatitis due to nausea associated with SIRS and ANCs within the lesser sac leading to gastric outflow obstruction. We advocate early NJ tube insertion to allow enteral feeding with supplemental parenteral nutrition if required. This is in keeping with ESPEN guidelines. 12 Endoscopic NJ insertion should be performed at the local hospital and a slowly increasing continuous feeding regime implemented under dietitian guidance. NJ feeding is generally well tolerated in most patients with necrotising pancreatitis. 14 Total parenteral nutrition can be used if NJ feeding is not tolerated.
Who should be referred to a tertiary HPB centre?
All patients with necrotising pancreatitis with the following features should be referred to a regional HPB centre for discussion:
greater than 30% necrosis of the pancreas or ANCs on early CT (within seven days of admission)
4
persisting organ dysfunction for greater than 48 h requiring organ support (e.g. ventilation, renal replacement therapy) infected WON requiring drainage or necrosectomy gastric or duodenal outflow obstruction secondary to acute peri pancreatic collections, pseudocyst or WON evidence of haemorrhage on CT
Some HPB units have developed regional pancreatitis networks with formal online referrals, clinical nurse specialists (CNSs) and dedicated pancreatitis multidisciplinary team meetings. Given that the backbone of management for necrotising pancreatitis is supportive care, this provides referring DGHs with regular input from a tertiary specialist unit whilst local units retain autonomy of care. In addition, if patients do require transfer for specialist intervention, this can be co-ordinated by the CNS to ensure that planned procedures are performed expeditiously after arrival.
Drainage of infected WON. What route to take?
All patients with infected WON require drainage. 4 The optimum route of drainage remains controversial. Historically, patients underwent open necrosectomy. This involved a laparotomy and transperitoneal approach to the lesser sac. The necrotic pancreas was debrided gently taking great care to avoid damage to the major retropancreatic vascular structures. The retroperitoneum was widely drained with up to four wide bore drains used for flushing and drainage. However, the open approach is associated with high morbidity and mortality.
The landmark PANTER trial changed the approach to drainage of infected necrotic pancreatic collections. This randomised controlled trial compared open necrosectomy to a ‘step-up’ approach involving percutaneous drainage and minimally invasive necrosectomy. 15 Although no difference in mortality was observed, major complications (multi-organ failure, onset of diabetes mellitus) were significantly lower in the ‘step-up’ approach group. 15 Most HPB units now use variations on the step-up approach.
The aim of drainage is to remove infected necrotic material from the retroperitoneum without breaching the intraperitoneal cavity. Radiological, percutaneous drainage is a safe approach in experienced hands and some case series have demonstrated that this is the only intervention required in 50% of cases. 16 This approach is favoured for infected flank collections. In most cases, these patients require fluoroscopic upsizing of these drains to ensure that the drains are not blocked by necrotic material. This involves balloon dilatation of drain tracts up to 30Fr to allow passage of a rigid nephroscope. 17 Under vision, the necrotic cavity can be irrigated and evacuated via suction. In addition, graspers can be passed down the operating channel to remove necrotic tissue. 17 Complications include immediate or delayed haemorrhage, septic shock and enteric fistula.
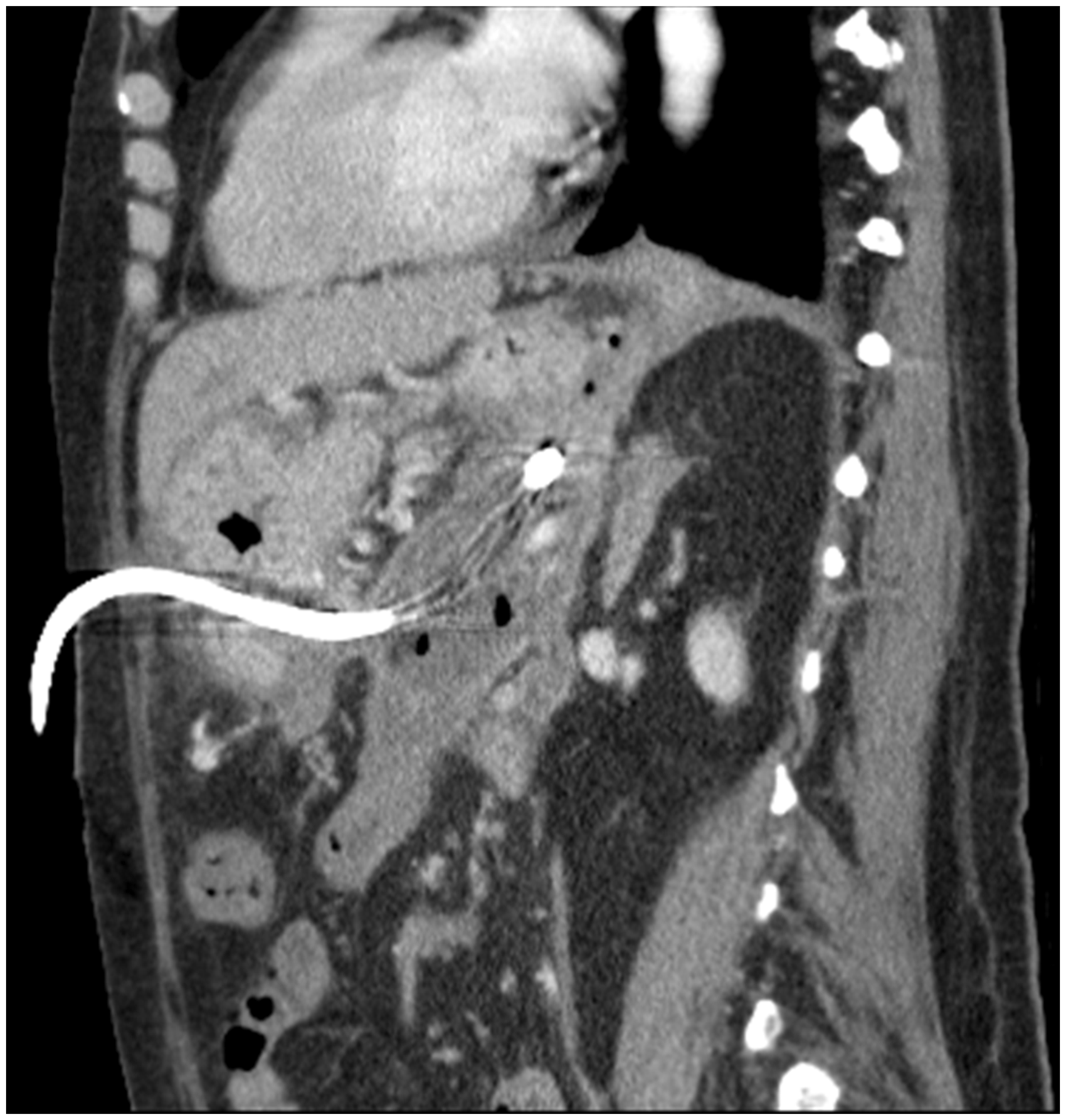
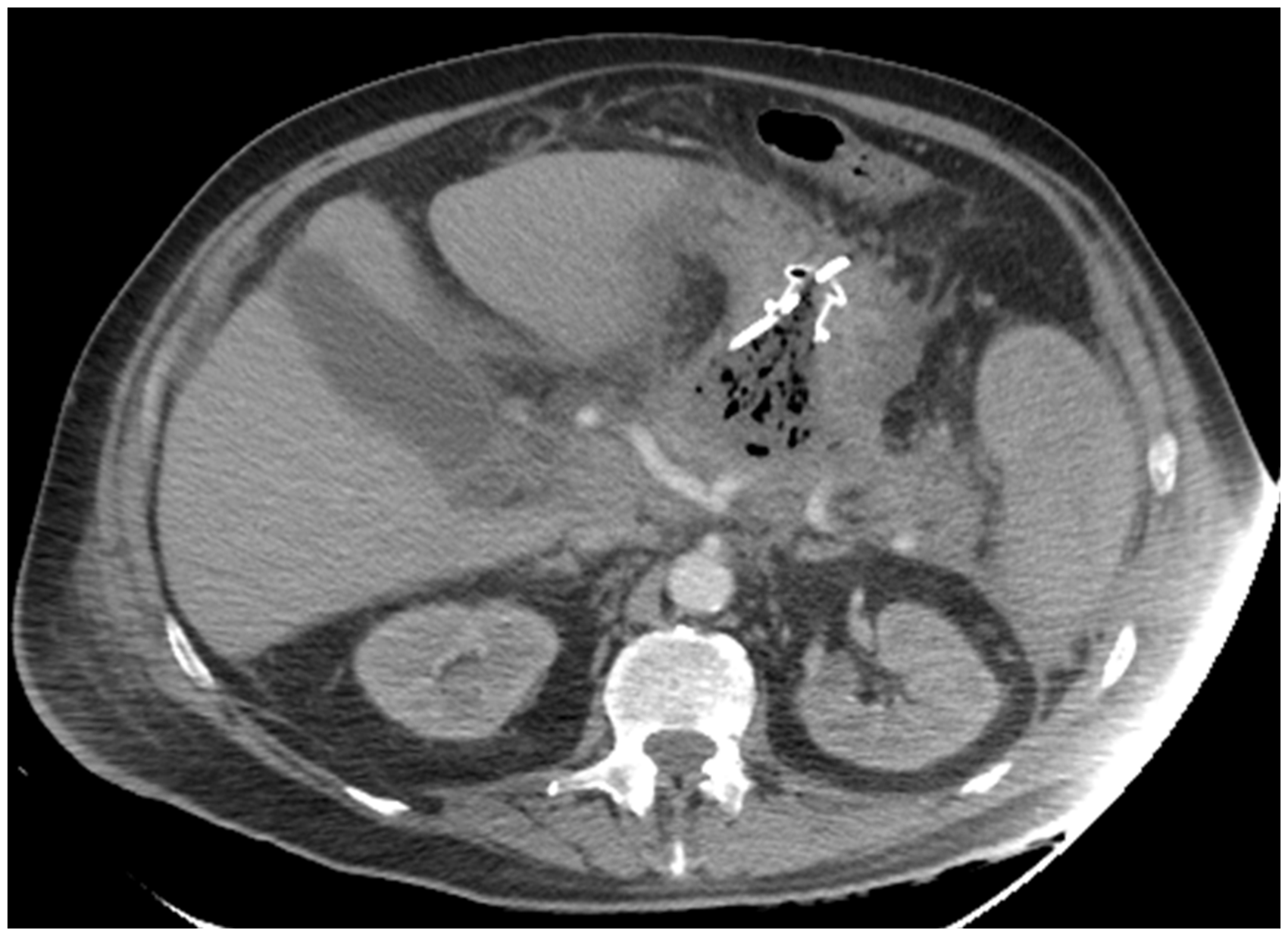
Infected collections located centrally within the lesser sac are less amenable to percutaneous, retroperitoneal drainage. Transgastric drainage can either be performed percutaneously or endoscopically. The percutaneous approach was first described in 1988
18
and involves CT-guided puncture of the anterior and posterior walls of the stomach to enter into the necrotic collection (Figures 3 and 4). As with other percutaneous approaches, the initial drain (usually 12Fr) can be flushed regularly and upsized or internalised if required. Six weeks after insertion, the drain can generally be removed as a cyst-gastrostomy tract will have formed and the anterior gastrocutaneous fistula will close up over time. Endoscopic ultrasound-guided drainage has now become a favoured route in many HPB centres. The necrotic collection can be identified accurately and the distance between the stomach and collection wall measured to ensure stent deployment will be successful. In addition, Doppler ultrasound can be used to avoid vascular structures overlying the collection.
19
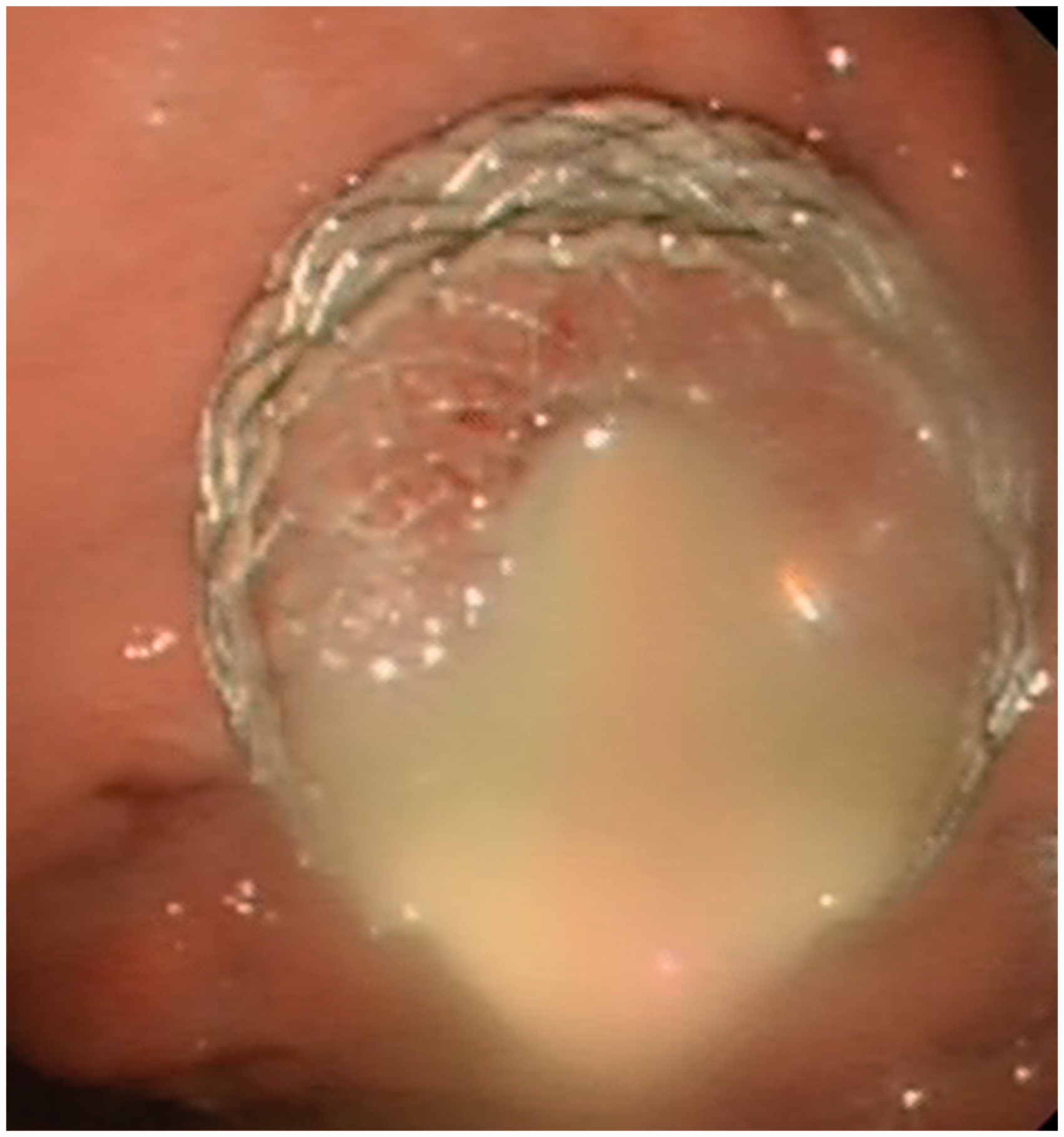
Following diathermy puncture of the stomach and collection, a fully covered self-expanding metal stent is inserted to allow drainage of infected necrosis into the alimentary tract (Figures 5 and 6). After stent deployment, a naso-cystic irrigation catheter can be inserted to allow irrigation of the necrotic cavity
20
to facilitate drainage. Repeat procedures with a gastroscope to irrigate and debride the cavity are occasionally required if there are concerns regarding stent blockage. In order to minimise stent-related complications, it is advised to remove the stent four weeks following insertion. No randomised studies have yet compared endoscopic and percutaneous approaches to identify the superior approach.
Transgastric drain within infected WON inserted under CT guidance (axial view). Sagittal view of transgastric drain within infected WON inserted under CT guidance. Infected WON drained by endoscopic ultrasound-guided stent (Hot AXIOS, Boston Scientific, USA). Endoscopic view of endoscopic stent with infected necrosis draining into the stomach.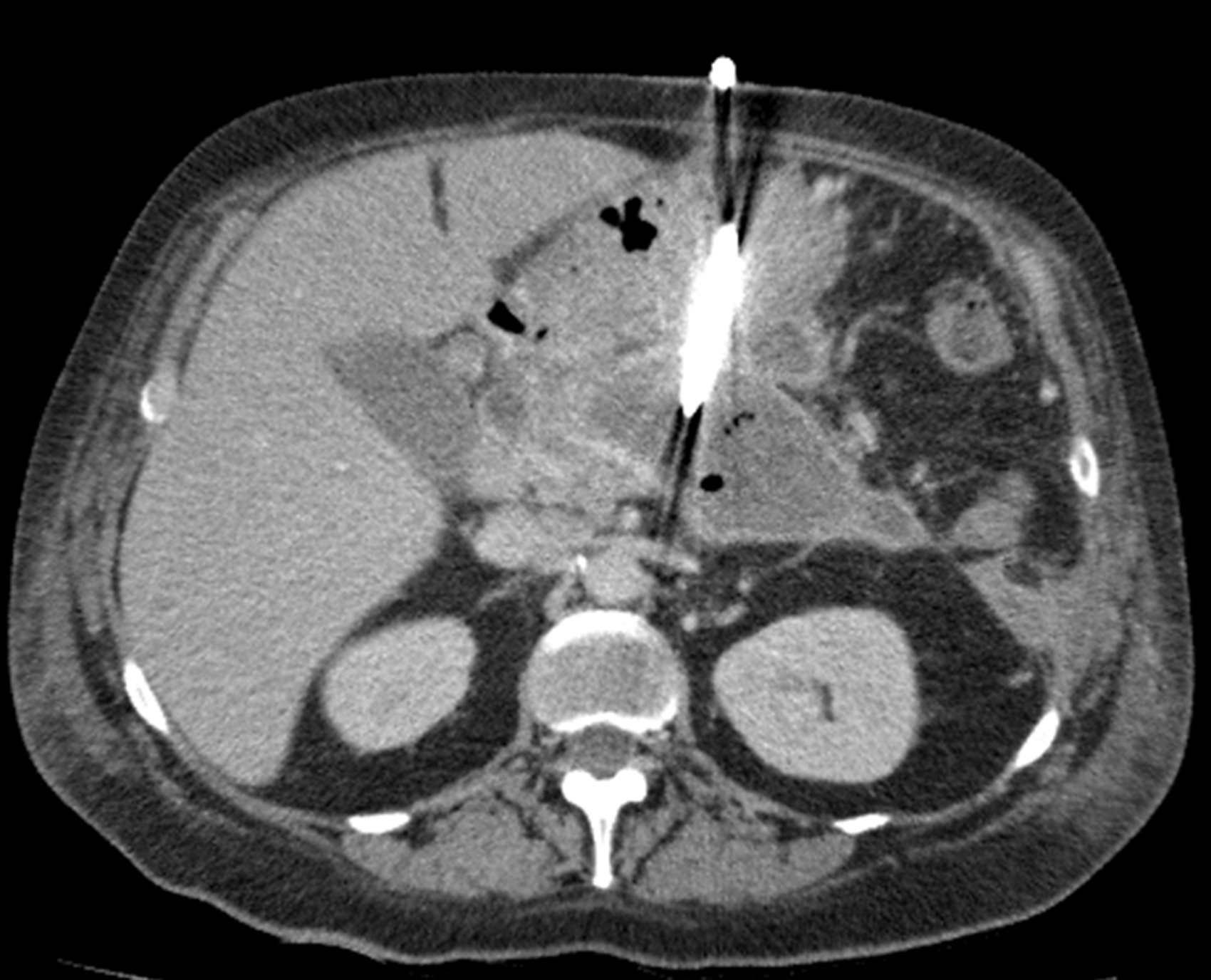
Conclusion
Severe necrotising pancreatitis is a complex surgical emergency with high morbidity and mortality that requires a multidisciplinary approach to attain optimum outcomes. Supportive care and nutritional support form the mainstay of early management. Early discussion with the regional HPB unit can facilitate transfer for intervention at an appropriate time. A step-up approach to drainage and necrosectomy is now accepted as standard of care and provide the best outcomes for patients with this disease.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
