Abstract
Background and Aims:
The impact of biliary invasion on recurrence and survival, after resection of colorectal cancer liver metastases, is not well known as publications are limited to small patient series. The aim was to investigate if biliary invasion in liver resected patients associated with liver relapses and recurrence-free survival. Secondary endpoints included association with other prognostic factors, disease-free survival and overall survival.
Materials and Methods:
All patients with histologically verified biliary invasion (n = 31, 9%) were identified among 344 patients with liver resection between January 2009 and March 2015. Controls (n = 78) were selected from the same time period and matched for, among others, size and number of colorectal cancer liver metastasis.
Results:
Median liver recurrence-free survival was significantly shorter in patients with biliary invasion than in controls (15.3 months versus not reached; p = 0.031) and more relapses were noted in the liver (61.3% versus 33.3%; p = 0.010), respectively. In univariate analyses for liver recurrence-free survival, biliary invasion was the only significant prognostic factor; p = 0.034. There were no statistical differences in disease-free and overall survival between the groups.
Conclusion:
Biliary invasion was associated with higher liver recurrence rates and shorter liver recurrence-free survival in patients with resected colorectal cancer liver metastasis.
Introduction
Colorectal cancer (CRC) is the third most common cancer worldwide and liver is the most common site of metastases (1). About 20% have synchronous colorectal liver metastases (CRLM) at presentation and approximately 30% of patients will develop metachronous CRLM (1, 2). Median overall survival (OS) with modern oncologic therapy has reached 30 months, but long-term survival is rare (3, 4). Liver resection has become a golden standard, as a potentially curative treatment (1,5, 6). Approximately 10%–20% of patients with CRLM were initially candidates for hepatic resection (1, 7), and another 10%–20% were downstaged with neoadjuvant chemotherapy and biologics, increasing the rate of resectability to over 30% (2).
The 5-year survival rates after liver resection and chemotherapy were 45%–60% (7–12), but two-thirds develop recurrence, often in the liver (2, 13). Pooled analysis of two randomized studies showed marginal favor of adjuvant chemotherapy for both progression-free and OS (16). Neoadjuvant and adjuvant chemotherapy have improved disease-free survival (DFS) and consensus guidelines suggest perioperative chemotherapy which is thus increasingly used (1, 2, 8, 14–16).
Several studies have proposed prognostic scoring systems to predict liver recurrences after resection of CRLM (11, 12, 17), but no consensus of clinically meaningful risk factors have been reached. Number and size of hepatic metastases, extrahepatic disease, high carcinoembryonic antigen (CEA), synchronous CRLM, and lymph node positive primary and positive (R1) resection margins have consistently been suggested as risk factors for liver recurrence (8, 11, 12). RAS and BRAF mutations are emerging predictive and prognostic factors in CRLM, with impaired survival after liver resection (18–22), effect on relapse profile, with more lung relapses in RAS mutated (22) and negative predictive effect of epidermal growth factor receptor (EGFR) therapy (23).
To date only a few patient series have explored the prognostic value of histological factors, such as vascular, perineural, and biliary invasions (24–26).
The incidence of biliary invasion has varied between 8.8% and 36% (10, 24, 27) and prognostic significance has been contradictory in these small patient series, and thus it is still a matter of debate (10–12, 24–26, 28).
The primary aim of the study was to investigate whether biliary invasion in resected CRLM was associated with liver relapses and recurrence-free survival (LRFS). Secondary endpoints were impact of vascular, perineural, RAS, and BRAF mutations; neo- and adjuvant treatment on LRFS, DFS, and OS.
Methods
This is a retrospective case–control study of patients, who had histologically verified biliary invasion in CRLM and were resected with curative intent at the Transplantation and Liver Surgery Unit at Helsinki University Central Hospital (HUCH). Between January 2009 and March 2015, 344 patients were resected for CRLM and 31 patients with biliary invasion (9%) were identified from pathology department’s electronic records of all histological specimens and they composed the biliary invasion group.
For each patient with biliary invasion, we systematically searched two and whenever possible three controls. The control group was matched regarding tumor characteristics, such as size of the largest metastasis, similar number of CRLM and synchronous versus metachronous presentation of metastases, and time of operation. Matched control patients were operated as near as possible compared with biliary case operation. Only resectable extrahepatic lung metastases were allowed and matched for, if present.
The following parameters were collected from patient charts: (1) patient characteristics; (2) primary tumor characteristics (TNM staging, location); (3) characteristics of CRLM (distribution of metastases, synchronous or metachronous presentation, and extent of liver resection (minor/major); (4) histopathological characteristics (resection margin, size and number of metastases, vitality (%), vascular, perineural, and biliary invasion); (5) preoperative CEA and cancer antigen 19-9 (CA19-9) values; (6) neo- and adjuvant chemotherapy; and (7) RAS and BRAF mutation status from the primary tumor, when available.
Preoperative Tumor Imaging
Whole-body high-volume contrast-enhanced computer tomography (CT) was performed to evaluate the extent of metastatic disease. Positron emission tomography (PET) was used for extrahepatic disease evaluation, if necessary. Magnetic resonance imaging (MRI) of the liver was used to evaluate the extent and size of intrahepatic metastases in case of steatotic liver or multiple, unspecific small lesions.
Liver Surgery
All liver resections were performed by experienced liver surgeons, with extensive liver transplantation expertise. Intraoperative ultrasound was performed in all patients in collaboration with experienced radiologists. If the CRLM had radiologically disappeared during neoadjuvant chemotherapy, the area where metastasis had been located was resected. Hepatic resection was defined as major if three or more Couinaud’s segments were resected. Cavitron ultrasonic surgical aspirator (CUSA) knife was used in all liver resections.
Histopathology of the CRLM
All resected liver specimens were examined by experienced hepatobiliary pathologists and included the number of tumors, diameter of each tumor, and length of resection margin, with R0 resection defined as a negative and R1 as a positive microscopic margin. Histopathological evaluation included assessment of biliary, vascular, and nerve invasion. Biliary invasion was defined as tumor cells growing along the ductal wall replacing the normal biliary epithelium (Fig. 1A to D). Vascular invasion was defined as tumor cell growth in the vascular space and nerve invasion as tumor cell growth in the nerve sheath or perineural space. H&E, CK7, and CK20 staining were used to define cancer cell growth along the biliary ducts.
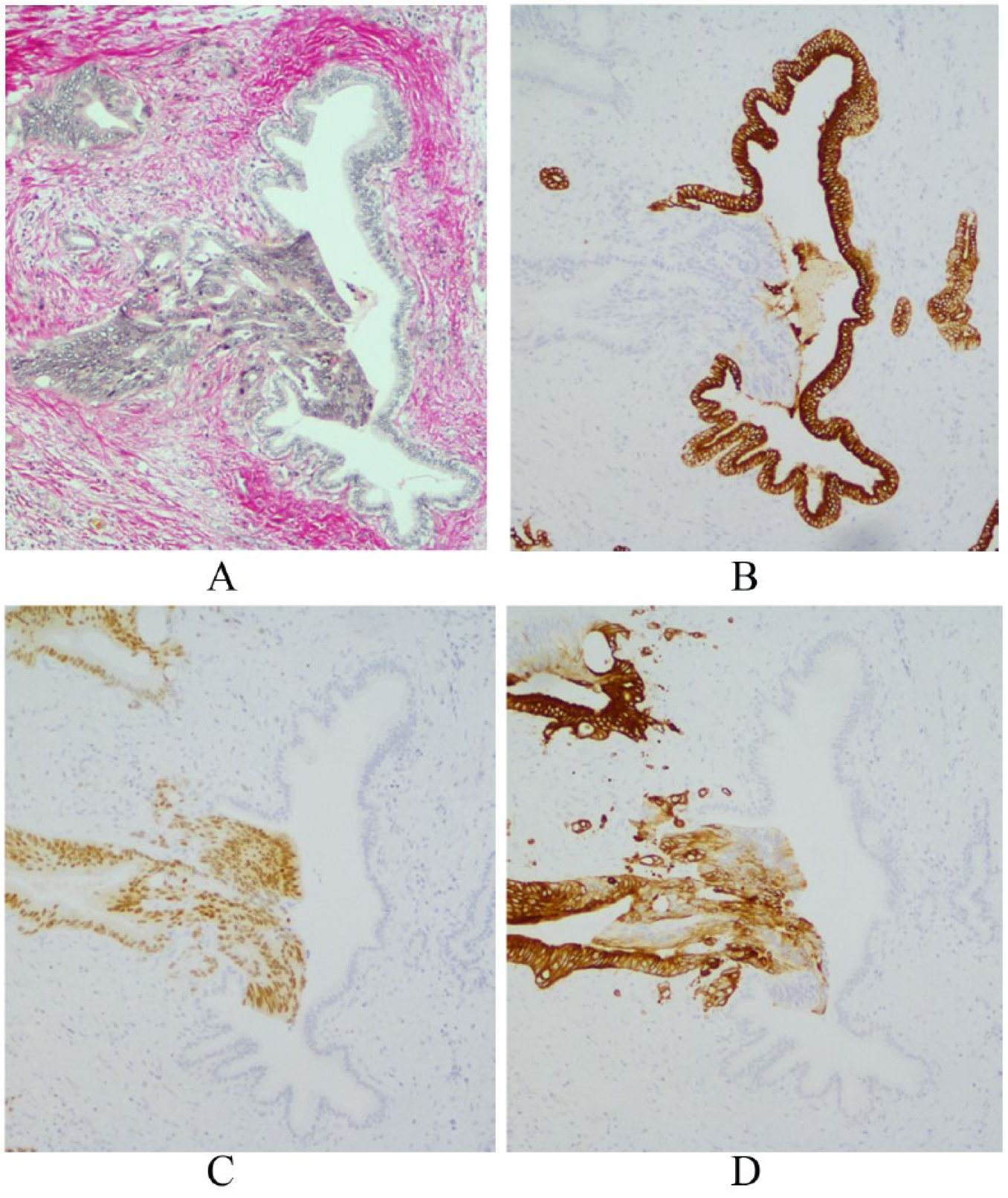
Colorectal adenocarcinoma invades left corner of the bile duct. (A) Herovici staining. (B) Normal bile duct epithelium is cytokeratin 7 positive, but tumor cells are negative. Tumor cells are (C) CDX2 and (D) cytokeratin 20 positive.
Chemotherapy
All patients were considered for neoadjuvant and adjuvant chemotherapy at a multidisciplinary team meeting (MDT). In the neoadjuvant or conversion setting, a combination of fluoropyrimidines with irinotecan or oxaliplatin was used to downsize CRLM and to improve resectability and survival (1, 15). Bevacizumab, panitumumab, and cetuximab were used in the neoadjuvant setting as appropriate (28, 29). None progressed during neoadjuvant or conversion therapy. Patients were assessed for resectability in the MDT every 2–3 months.
In the adjuvant setting, a fluoropyrimidine, as single agent or in combination with oxaliplatin or irinotecan (if contraindications present), was used. Treatment was initiated 4–8 weeks after resection, unless postoperative complications were present. If neoadjuvant or conversion therapy had been used, the same combination was continued to the total treatment time of at least 3 months post resection. Chemotherapy duration was at least 6 months. After publication of negative adjuvant studies with bevacizumab and cetuximab, biologics were omitted in the adjuvant setting (15, 16).
Follow-Up
Patients were followed up regularly after hepatic resection. The first check at 3 months was performed at the liver surgery unit. Follow-up consisted of physical examination, whole-body CT scan, and laboratory tests (full blood count, biochemistry, coagulation panel, CEA, and CA 19-9). Follow-up was continued at the local hospitals every 3 months for the first 2 years and thereafter every 6 months or more often to 5 years.
Statistical Analysis
Categorical data were presented as frequencies and proportions and compared using Pearson’s chi square or Fisher’s exact test, as appropriate. The continuous data were presented as median and range and compared with the Mann–Whitney U or Kruskal–Wallis test.
LRFS was defined as time from liver resection to recurrence in liver or death from whatever the reason, but recurrences appearing within 3 months were excluded, as they most probably already were present at surgery. DFS was defined as the time from liver resection to recurrent disease or death for whatever the reason. OS was defined as time from the liver resection to the latest follow-up or death for whatever the reason.
Survival probabilities were estimated using the Kaplan–Meier method and compared with a log-rank test. Univariate analyses were performed by Cox proportional hazard models to identify any significant differences in clinicopathological features that influenced LRFS, DFS, and OS. Multivariate analyses were performed for factors with a p value of less than 0.05 in the univariate analysis. A p value of <0.05 was considered statistically significant. No correction for multiple testing was performed. Statistical analyses were performed with IBM SPSS 22 software (USA).
Results
Patient Characteristics
Between March 2009 and March 2015, all 31 (9%) patients with biliary invasion were identified among 344 resected CRLM. Two to three controls per case, 78 in total, were matched from the resected group. Median follow-up time was 32.3 months (range: 7.0–83.6 months) in the biliary group and 25.4 months (range: 6.1–77.1 months) for the controls (p = ns).
Patient demographics are presented in Table 1. There were no statistically significant differences in demographics between the groups. There were 84% males in the biliary and 65% in the control group (p = 0.065). The median age was 65 (range: 45–80) years in the biliary and 64 (range: 39–80) years in the control group.
Demographics in patients resected for colorectal liver metastases with biliary invasion (n = 31) and matched controls without (n = 78).
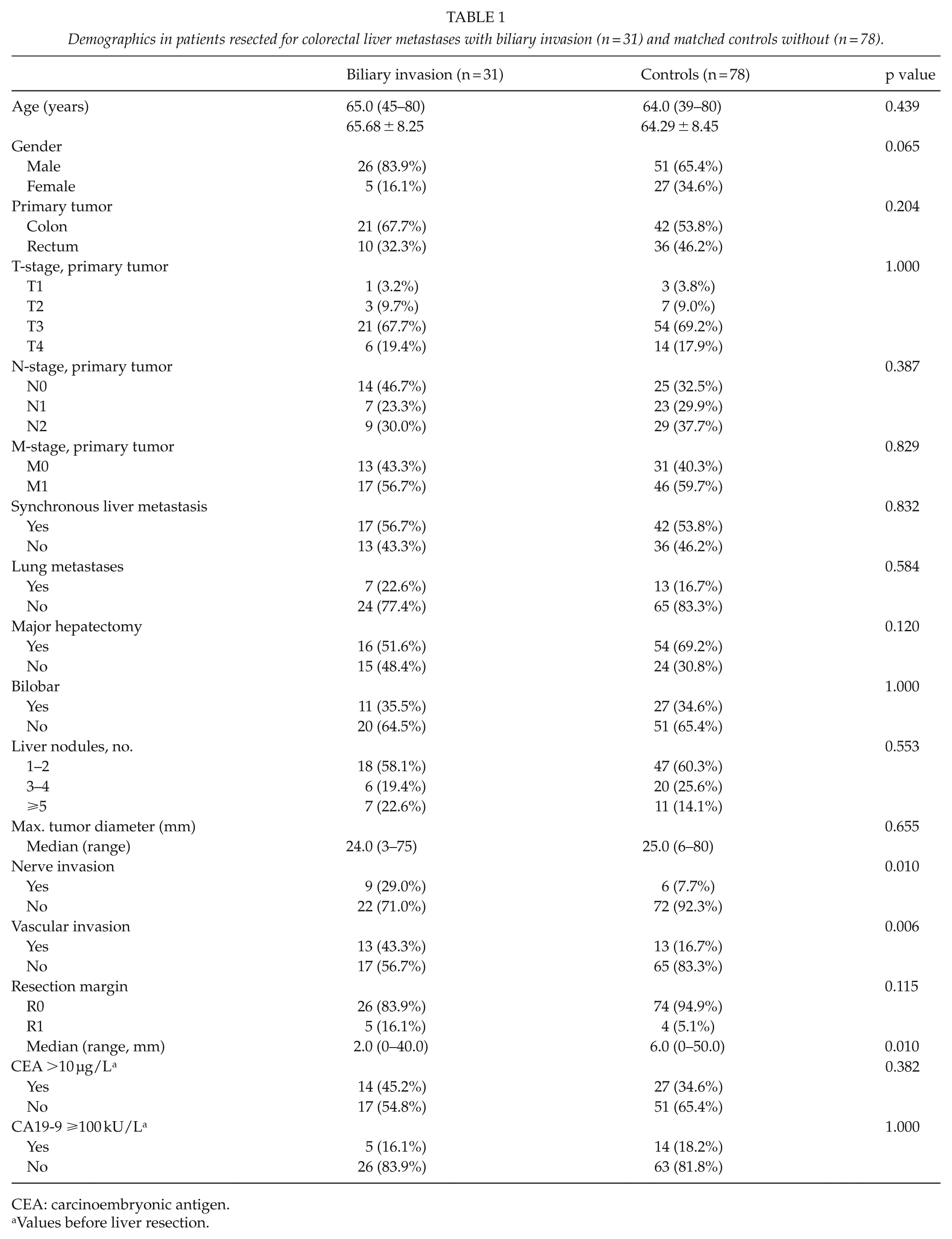
CEA: carcinoembryonic antigen.
Values before liver resection.
Most patients had synchronous metastases and T3-4 primaries with lymph node positivity. These were equally distributed between the groups. So was the presence of resectable lung metastases and preoperative CEA and CA19-9 levels (Table 1). There were no differences regarding R0 resection rates (biliary invasion 83.9% versus controls 94.9%; Table 1; p = 0.115).
Patients with biliary invasion had a shorter histological resection margin (2.0 versus 6.0 mm; p = 0.010). Perineural (29.0% versus 7.7%; p = 0.010) and vascular (43.3% versus 16.7%; p = 0.006) invasions were more common in the biliary invasion group.
Incidence of KRAS mutation was 26.9% in the biliary invasion group and 37.7% in controls. All RAS (KRAS and NRAS) mutations were equally distributed (Table 2). One BRAF mutation (in the biliary group) was present.
Chemotherapy—neoadjuvant and adjuvant therapy and mutational status of primary tumor, divided by biliary invasion and controls.
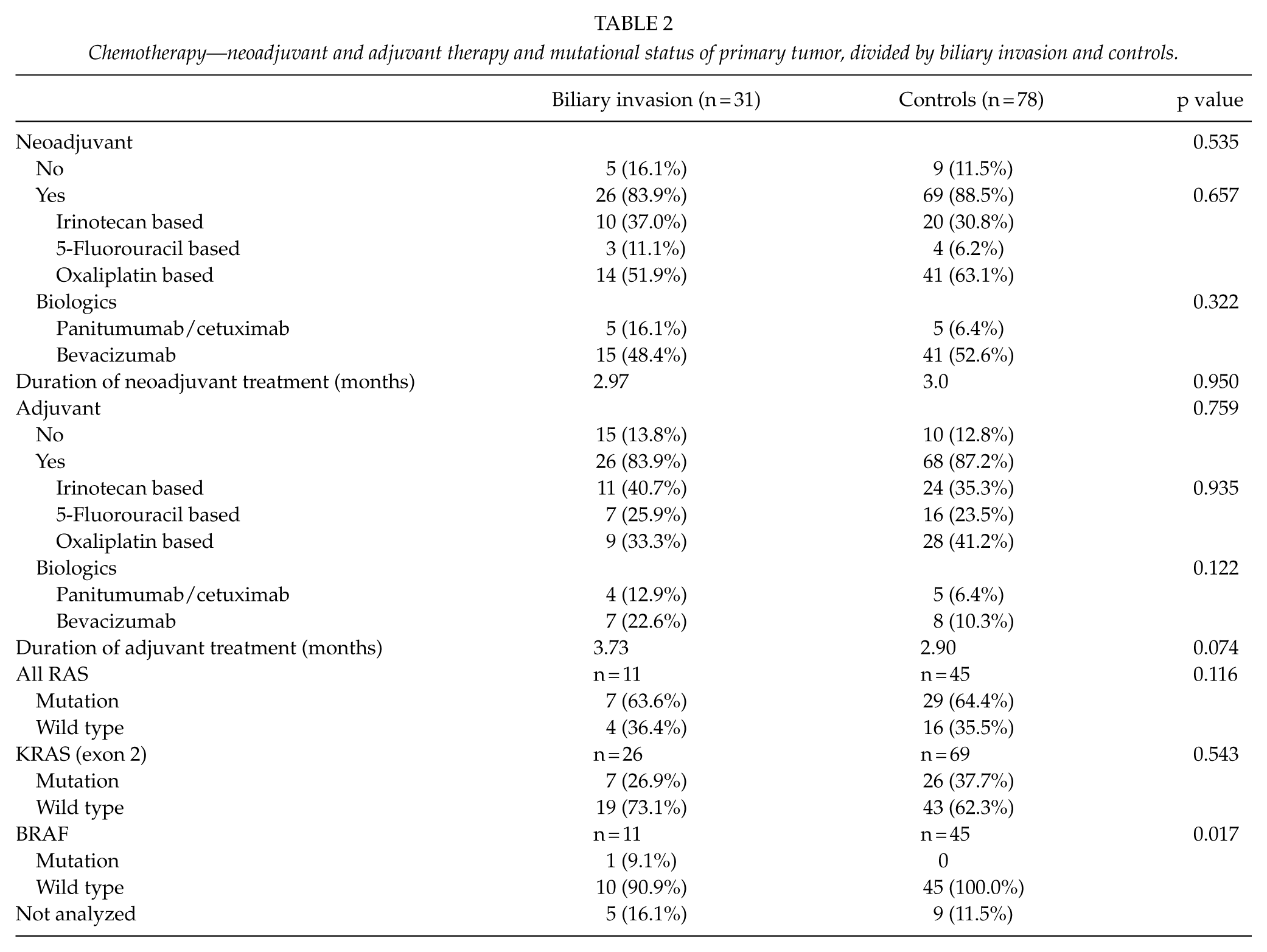
Neoadjuvant treatment was given to 83.9% of patients with biliary invasion and in 88.5% of controls (p = 0.535) and adjuvant in 83.9% versus 87.2%, respectively (p = 0.759; Table 2). Biologics were given in 66% in the neoadjuvant setting and in 22% in the adjuvant setting.
Liver Recurrence-Free Survival
Patients with biliary invasion developed significantly more liver recurrences than the controls (61.3 versus 33.3%; p = 0.010). LRFS was significantly shorter in patients with biliary invasion (p = 0.031; Fig. 2A). Median LRFS was 15.3 months (CI95% 3.3–27.3) in biliary invasion and has not yet been reached in controls. For the biliary invasion and controls, LRFS rates at 1 year were 58.3% versus 73.5% and at 2 years 39.7% versus 62.5%, respectively.
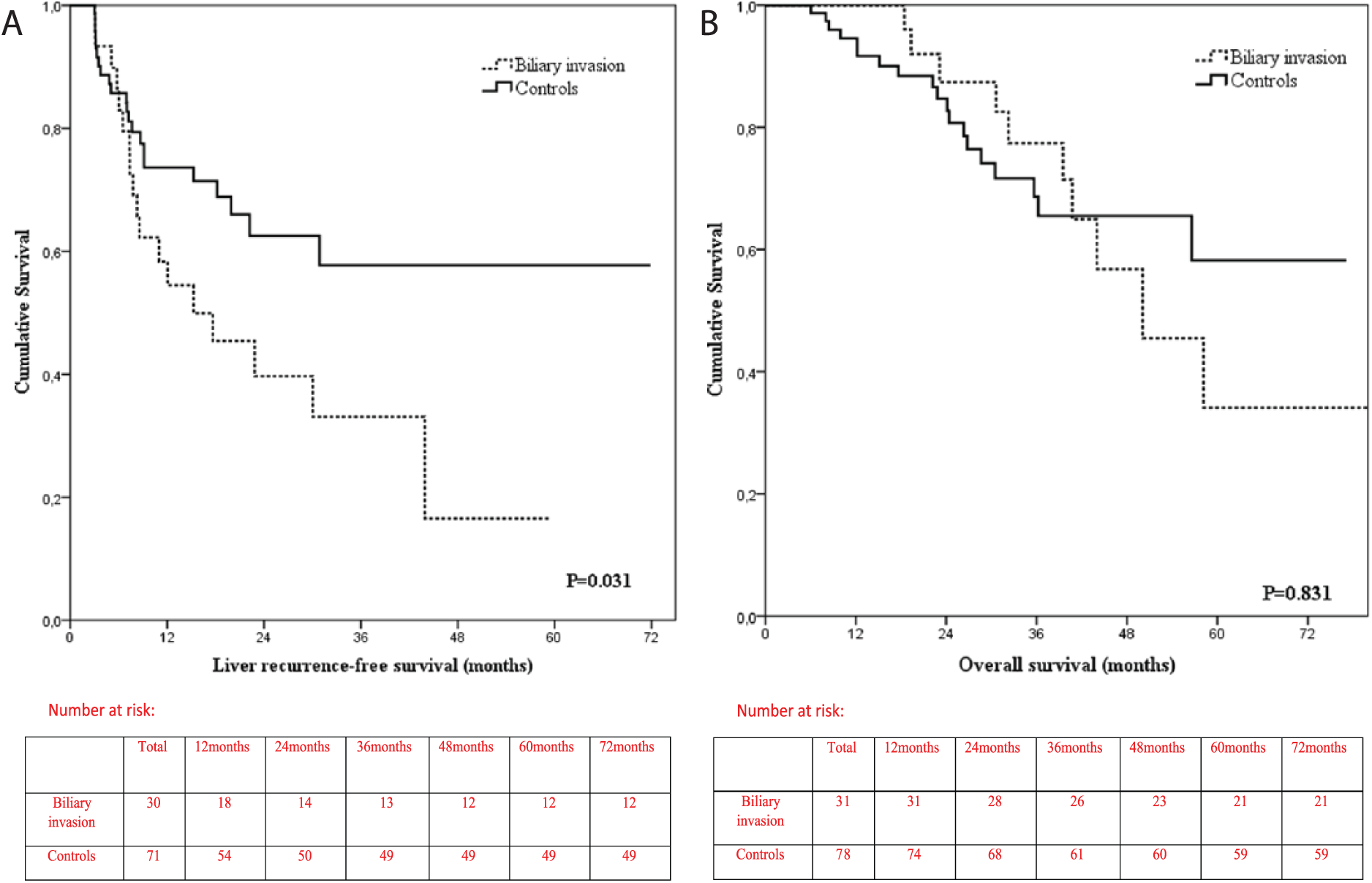
Kaplan–Meier survival curve associated with (A) LRFS and (B) OS.
In univariate analysis, biliary invasion was the only significant prognostic factor affecting the LRFS (p = 0.034).
DFS
Median DFS was 13.0 (CI95% 6.2–19.8) months in the biliary group and 15.3 (CI95% 6.2–24.4) months in the controls (p = 0.560). DFS rates at 6 months, 1 year, and 2 years were 83.0% versus 74.0%, 55.1% versus 56.0%, and 27.7% versus 35.4% for the biliary versus controls, respectively. Univariate analysis showed that biliary invasion had no significant impact on DFS (p = 0.560). Five or more CRLM, T3-4 primary, R1 resection, CEA >10 µg/L, and CA19.9 ⩾100 kU/L were identified as independent prognostic factors for DFS in the univariate analysis. In multivariate analysis, T3-4 tumor stage, R1 resection, and CEA >10 µg/L remained prognostic.
OS
Median OS in the biliary group was 50.1 (CI95% 32.8–67.4) months and has not been reached in the control group (p = 0.831, Fig. 2B). For the biliary versus control group 1-, 3-, and 5-year OS was 100.0% versus 94.5%, 77.4% versus 68.6%, and 34.1% versus 58.2%, respectively (Fig. 2B). We also performed survival analyses of unmatched patients (n = 246) resected in the same time period. The 3-year OS for unmatched group was 79.2% and 5-year OS for unmatched group was 60.2%.
In univariate analyses, biliary invasion had no impact on OS (p = 0.831), but R1 resection, perineural invasion, CEA >10 µg/L, and CA19-9 ⩾100 kU/L were associated with decreased OS (Table 3). R1 resection, perineural invasion, and CA19-9 ⩾100 kU/L remained independent prognostic factors in multivariate analyses (Table 4).
Univariate analysis of prognostic factors on OS and LRFS.
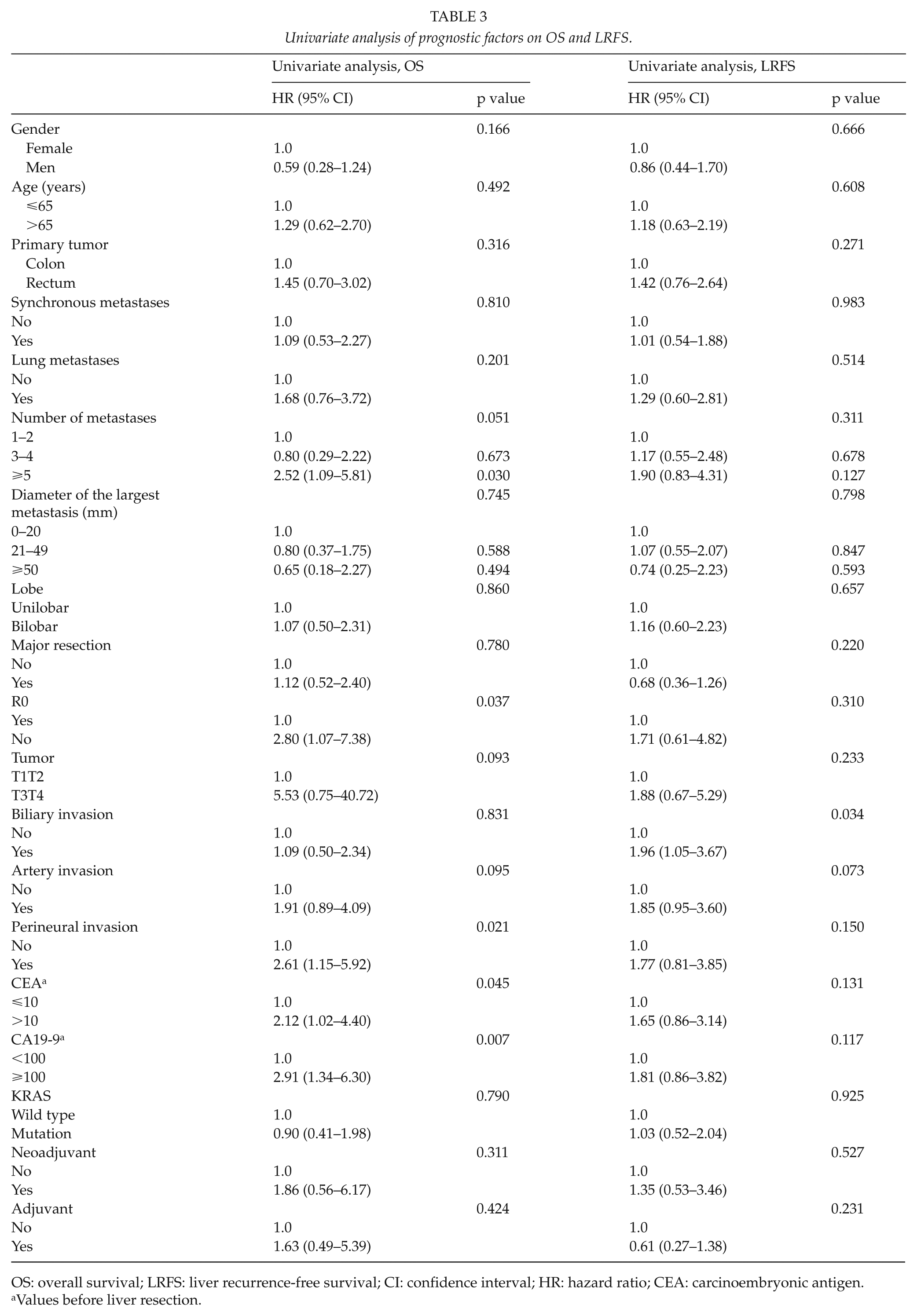
OS: overall survival; LRFS: liver recurrence-free survival; CI: confidence interval; HR: hazard ratio; CEA: carcinoembryonic antigen.
Values before liver resection.
Multivariate analysis, prognostic factors of OS.
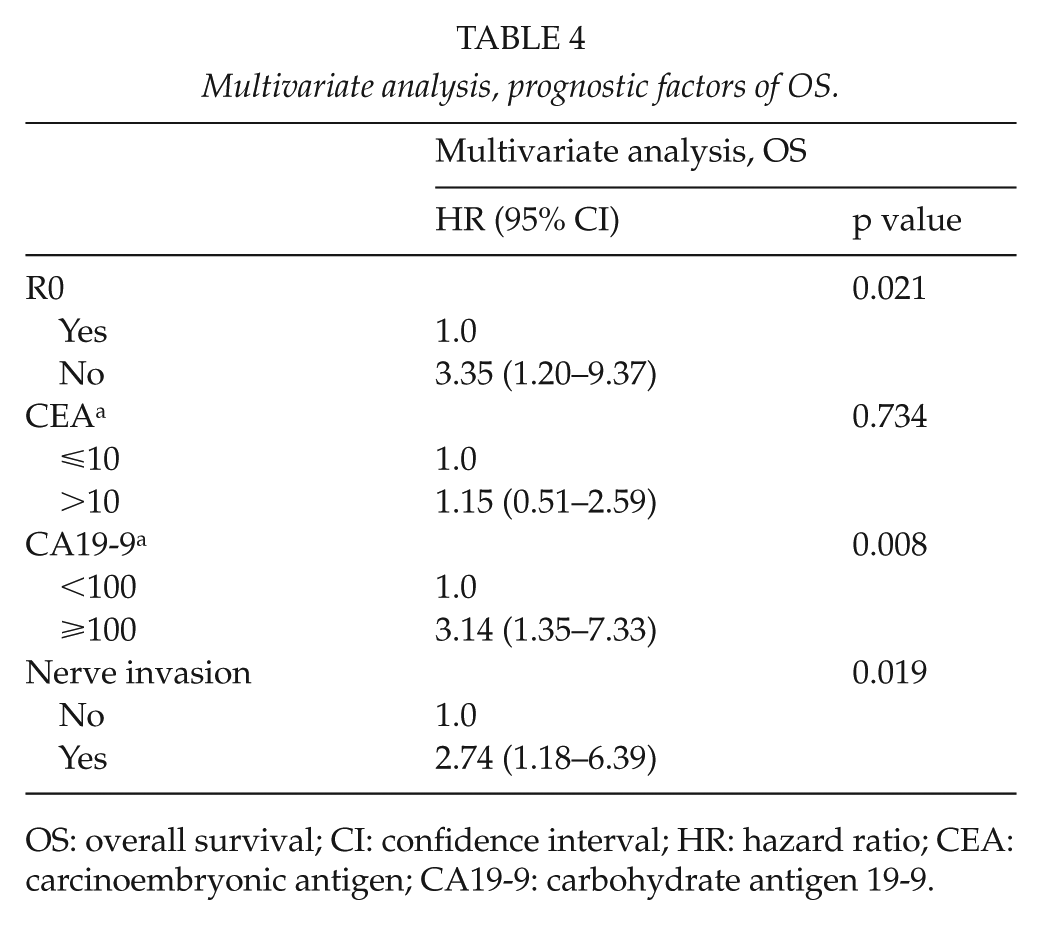
OS: overall survival; CI: confidence interval; HR: hazard ratio; CEA: carcinoembryonic antigen; CA19-9: carbohydrate antigen 19-9.
Discussion
The prognostic significance of histopathological characteristics of CRLM has been assessed by various studies over the past decade, but the role of biliary invasion is still a matter of controversy (10–12, 24, 27). This study thus investigates the effect of biliary invasion on liver recurrences and outcome. The most interesting finding is that biliary invasion is associated with higher rate of liver recurrences and shorter liver LRFS. To our knowledge, this association has not been investigated in earlier studies. Biliary invasion was also related to shorter resection margins, although no difference in R0 resection rate was noted. Reason for the higher recurrence rate and smaller margin could be explained by cancer cells spreading discontinuously and causing smaller, undetectable positive histological margins, even though macroscopic and microscopic evaluation showed R0 resection. This finding raises an important clinical question that the operation should be done with wider margins if there is a suspicion of biliary invasion pre- or preoperatively. It is a very challenging diagnostic problem to detect biliary invasion before or during the operation even using frozen-section investigations. Until diagnostics have been developed to sufficient level, the patients with postoperatively detected biliary invasion should have more intensive follow-up surveillance in order to find resectable local hepatic recurrences early enough.
In this study, biliary invasion did not have statistically significant impact on DFS or OS. Other studies support this outcome (10–12, 24, 26, 27). However, in a recent study by John et al. (12), 5-year OS was slightly lower, 39.9% in patients with biliary invasion versus 42.5% in patients without, the same numerical trend is seen in our study with 34.1% with biliary invasion versus 58.2% without.
Perineural invasion was associated with impaired OS in the univariate and multivariate analyses in our study. Gomez et al. (27) confirm this result, but Ridder (24) did not find this association. Variation in these results may be explained by the small number (n = 11–13) of perineural invasions in all studies including ours (n = 15) (24,27).
Incidence of KRAS mutation was 27% among the biliary invasion and 38% in controls (ns), which is well in line with the published literature of 28%–30% (18, 19). In this study, KRAS as secondary endpoint had no significant impact on LRFS, DFS, and OS, which is contrary to a recent meta-analysis and review articles (18, 19), where patients with KRAS mutation had decreased OS and DFS. This may be due to a diminished drug arsenal in patients with KRAS mutations, as cetuximab and panitumumab are contraindicated, in this patient group or due to negative impact on survival as shown in a British randomized study (30). Only one patient had a BRAF mutation known to be a strong negative predictive factor (18, 22).
Chemotherapy was administered evenly between the biliary invasion and controls and there were no difference in survival outcome between patients with or without oncological treatments. Most patients had 3 months of neoadjuvant and adjuvant treatment as in the EORTC 40983 study (15), mostly with oxaliplatin in combination with a fluoropyrimidine. In the conversion setting biologics, bevacizumab in 50% and panitumumab or cetuximab in 9% were used to improve resectability as in the OLIVIA and CELIM studies (28, 29). In the adjuvant setting, bevacizumab, panitumumab, and cetuximab were used in 22% of the patients and after publication of negative adjuvant results with cetuximab and bevacizumab, biologics have not been used afterward.
The limitations of this study were the retrospective nature, the relatively small number of patients with biliary invasion, and the case–control design as propensity score matching might have been a better method but not technically feasible due to the small study group.
In conclusion, biliary invasion had prognostic significance on liver recurrences and LRFS, but not on DFS or OS. To our knowledge, this is the first study to assign increased rate of hepatic recurrences in patients with biliary invasion in the CRLM resected. If biliary invasion could be detected preoperatively or peroperatively, wider resection margins could reduce the rate of liver recurrences, but as of now, we are left with active postoperative surveillance to detect local resectable liver recurrences earlier.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Finnish government funding for health research.
