Abstract
Introduction:
To assess the results and outcome of radiofrequency ablation in the treatment of recurrent colorectal liver metastases.
Patients and Methods:
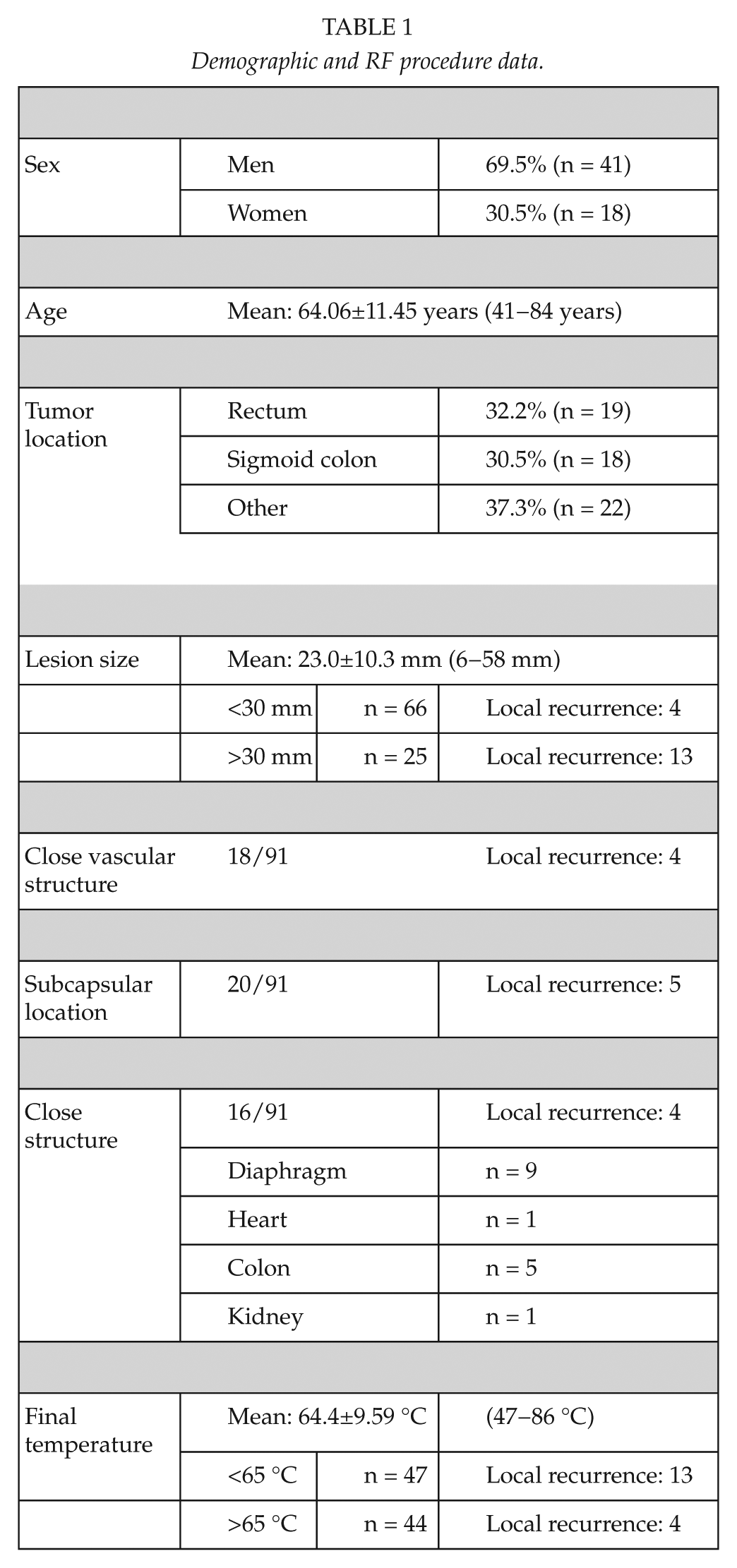
Between January 2005 and September 2012, we treated 59 patients with recurrent colorectal metastases not amenable to surgery with 77 radiofrequency ablation procedures. Radiofrequency was indicated if oncologic resection was technically not possible or the patient was not fit for major surgery. A total of 91 lesions were treated. The mean number of liver tumors per patient was 1.5, and the mean tumor diameter was 2.3 cm. In 37.5% of the cases, lesions had a subcapsular location, and 34% were close to a vascular structure.
Results:
The morbidity rate was 18.7%, and there were no post-procedural deaths. Distant extrahepatic recurrence appeared in 50% of the patients. Local recurrence at the site of ablation appeared in 18% of the lesions. Local recurrence rate was 6% in lesions less than 3 cm and 52% in lesions larger than 3 cm. The size of the lesions (more than 3 cm) was an independent risk factor for local recurrence (p < 0.05). Survival rates at 1, 3, and 5 years were 94.5%, 65.3%, and 21.7%, respectively.
Discussion:
Radiofrequency ablation is a safe procedure and allows local tumor control in lesions less than 30 mm (local recurrence of 6%) and provides survival benefits in patients with recurrent colorectal liver metastases.
Keywords
Introduction
Colorectal cancer (CRC) is the third most common cause of cancer death in western countries and the fourth most common malignancy worldwide (1, 2).Up to 50% of patients with CRC will present liver metastases at some stage of their disease either at the time of presentation or during the course of their disease (3, 4). Liver resection is the first-line treatment in patients with limited colorectal liver metastases (CLM) with 5-year survival rates between 25% and 58% (5–9). Unfortunately, only 30% of patients with CLM are candidates for liver resection, and liver recurrence is detected in 50%–70% of patients following curative resection (8). Percutaneous ablative therapies such as radiofrequency ablation (RFA) are effective treatments for certain metastatic liver cancers (9–13) and are associated with lower cost and mortality. Several studies have reported the results of RFA in the treatment of liver metastases (9–11), but few have focused on RFA in recurrent CLM after initial surgery. The exact place of RFA in the treatment of recurrent liver metastases is still debated because there is no solid evidence on the impact of RFA on overall recurrence and long-term survival. Repeated surgery, when amenable, is still considered the first treatment option in that setting (14–18), but the risk of complications is higher than after primary liver resection. Therefore, percutaneous RFA could be an attractive alternative treatment in selected patients with recurrent CLM due to its lower mortality and morbidity rates.
The aim of this study was to prospectively evaluate the outcomes of percutaneous RFA in non-surgical candidates with recurrent CLM. The study end-points were complication rate, local and distant recurrence of the tumor, and patient survival.
Patients and Methods
Patients
All patients with recurrent CLM after previous surgical resection between January 2005 and September 2012 were considered for RFA. Inclusion criteria for RFA were non-resectable or non-operable patients (for medical reasons) with three tumors or less, a maximum tumor diameter of 5 cm, and no extrahepatic disease except resectable pulmonary metastases. The indication of RFA was taken in a multidisciplinary board with hepatobiliary surgeons, radiologists, and oncologists. Eligible patients were prospectively included in our database and retrospectively analyzed.
This study was conducted with the approval of the institutional ethics committee. Written informed consent was obtained from each patient prior to the procedure.
Contraindications for RFA were non-correctable coagulation disorder, platelet count less than 50,000/mL, or previous bilioenteric anastomosis. Ongoing chemotherapy was not a contraindication for RFA. Systemic chemotherapy was continued after RFA, if there was no contraindication.
Pre-RF Work-Up
All patients were studied with liver function tests and carcinoembryonic antigen (CEA) levels. Preoperative staging included thoracic and abdominal multidetector computed tomography (MDCT) and total colonoscopy. In seven patients with liver steatosis or contrast allergy, magnetic resonance imaging (MRI) was also performed.
RFA Procedure
RFA was done with 150–200 W generators (RITA Medical Systems Model 1500 and Model 1500X; RITA/AngioDynamics, Fremont, CA, USA) with a 15-G multi-tined expandable device (RITA Medical System/AngioDynamics StarBurst Talon, or XLi) that deploy from the trocar tip. The needle was advanced into the metastatic nodule under continuous US guidance, and its correct placement in the center of the lesions was checked.
The electrode array deployment and the duration of the effective time of the ablation (time at the target temperature of 100 °C–105 °C) depended on the desired volume of ablation. In lesions smaller than 30 mm, a 4-cm device (Rita StarBurst Talon) was used with an effective radiofrequency time of 10 min. Lesions measuring more than 30 mm were treated with a 4- to 7-cm electrode (Rita Xli) with deployments at 5, 6, or 7 cm with an effective radiofrequency time of 16 min. At the end of the procedure, the tines were retracted and the generator was reactivated to ablate the track from the tumor to the subcutaneous tissue to prevent bleeding or tumor-cell dissemination. In lesions greater than 20 mm, the electrode was withdrawn 10−15 mm at the end of the first ablation, and a second, overlapping ablation was performed (“pull-back procedure”).
The procedure was done with intravenous sedation performed by an anesthesiologist under standard cardiac, pressure, and oxygen monitoring with continuous oxygen administration. The patients were admitted the same day of the procedure and were usually discharged 24 h later.
Follow-Up
All patients were followed at 1 and 3 months after treatment and every 6 months therein with liver function tests, MDCT, and CEA levels. CT scans were interpreted by experienced abdominal radiologists to define the treatment response.
Complications
Complications were defined as major (life-threatening complications requiring active treatment) or minor complications (requiring only symptomatic treatment).
Treatment Efficacy
Treatment efficacy was classified as follows:
Complete local response: The ablation zone (hypovascular area) surrounds completely the treated metastasis.
Local tumor persistence: Tumor persistence was defined as evidence of solid hypo/isodense lesion in the periphery of the ablation zone on the CT performed 1 month after the procedure. Repeated RFA treatment was completed as soon as possible.
Local tumor recurrence: Local recurrence was defined as any hypodense lesion in the boundaries of the ablation zone (less than 10 mm of the site of RFA) appearing during the follow-up, after a previous CT showing adequate ablation (Fig. 1).
Distant hepatic recurrence: Distant hepatic recurrence was defined as evidence of new hepatic tumor growth at a different location (more than 10 mm away) of the site of the RFA (Fig. 1).
Extrahepatic recurrences: It was defined as evidence of any extrahepatic tumor growth. The overall local or distant progression rates were calculated. The overall, hepatic disease–free, and disease-free survivals were estimated with Kaplan−Meier analysis.
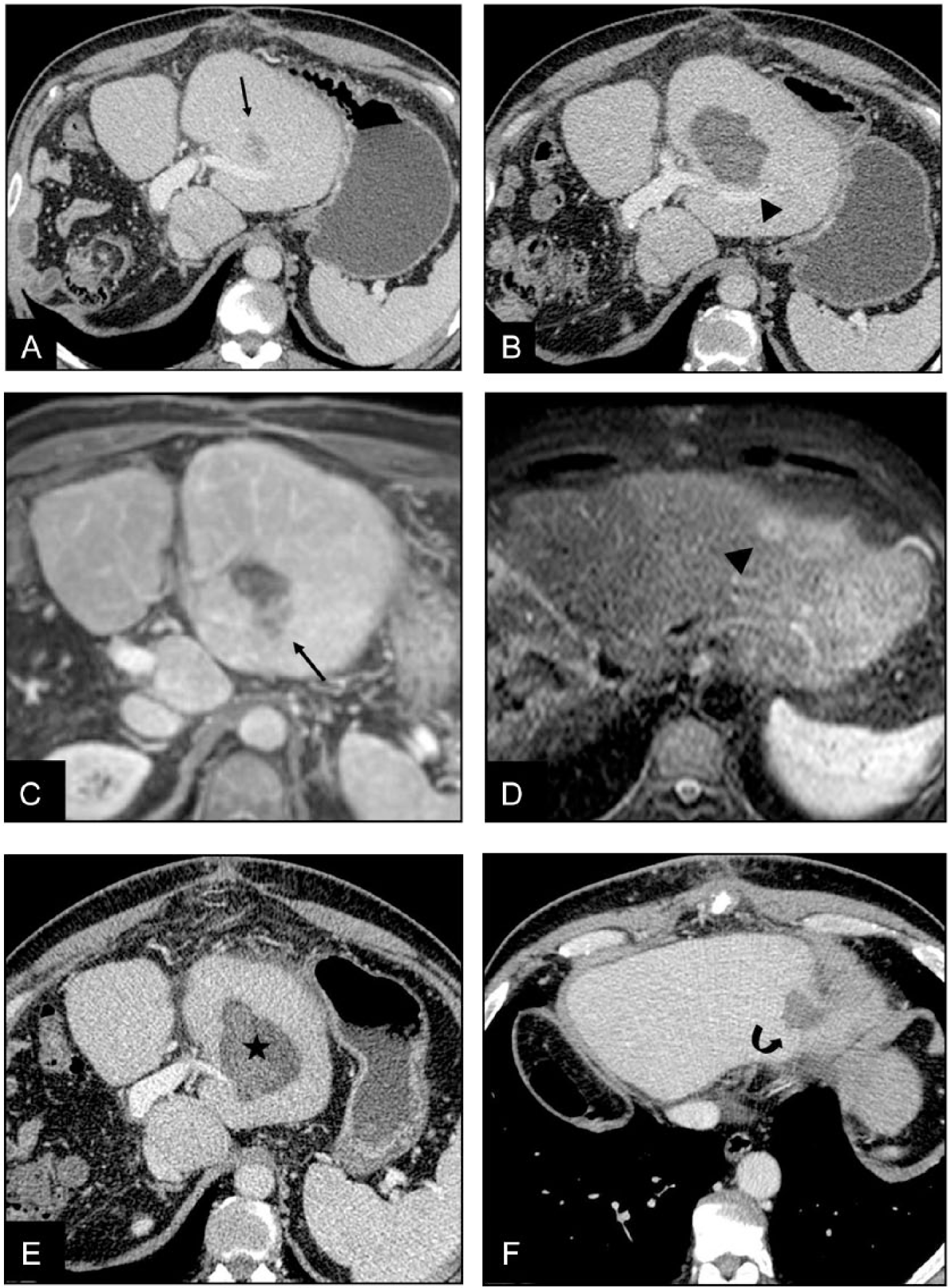
Local tumor recurrence and distant hepatic recurrence in a 57-year-old man with previous right hepatectomy. A) Portal phase CT shows a hypovascular metastasis (arrow) in Segment III 1 year after surgery. This lesion was treated with RFA. B) Portal phase CT scan 1 month after RF shows an ablation zone in Segment III with no evidence of tumoral viability (arrowhead). C) Gadolinium-enhanced MRI (3.3–4.5/1.4–1.9; flip angle, 12°) in the axial plane 6 months later shows local tumor recurrence in contact with the posterior margin of the ablation area (arrow). D) Axial T2-weighted MRI (TR-TE 1600/70) shows a new metastasis in Segment II (arrowhead). These lesions were retreated with RFA. E) Contrast-enhanced CT scan performed 1 month after treatment shows complete ablation with no signs of tumoral persistence in Segment III (star). F) Contrast-enhanced CT scan performed 1 month after treatment shows complete ablation with no signs of tumoral persistence in Segment II (curved arrow).
Statistics
Demographic information, details of post-RF course, and follow-up data were entered prospectively into a computer database. All statistical analyses were performed using the statistical software SPSS 17.0. For the comparison of patient’s characteristics, cross tables including the chi-square or Wilcoxon test were used. The overall, hepatic disease–free, and disease-free survivals were estimated with the Kaplan−Meier analysis in an intention-to-treat analysis according to the Kaplan−Meier method, and differences were estimated by the log-rank test. Terminal event for estimation was any death, any hepatic recurrence, and any tumoral recurrence, respectively. Multivariate analyses based on prognostic factors significant in the univariate analysis were calculated using Cox regression analysis. All reported p values are two-sided and denoted as statistically significant if they did not exceed 0.05.
Results
From January 2005 to September 2012, 59 consecutive patients with 91 CLM who had previously undergone hepatectomy were treated with percutaneous RFA. In 17 patients, contrast-enhanced ultrasound (US), sulfur hexafluoride (Sonovue Bracco, Milano, Italy) was used during the procedure to enhance the visualization of 13 tumors that were not adequately visible with unenhanced US. After injection of contrast material, the tumor was seen in the portal phase, and the electrode was rapidly inserted into the lesion. The remaining 42 lesions in those patients were adequately visualized with non-enhanced US.
Demographic and RF procedures are summarized in Table 1. The majority of patients were men, with a mean age of 64.05 years. A total of 76 RFA procedures were performed in 59 patients. In 45 patients, only one RFA was performed. A total of 12 patients underwent two RFA procedures and two patients had three RFA procedures. The mean number of liver tumors was 1.5 per patient. All patients had undergone previous hepatic resection with a mean interval of 22.4 months (range 1−60 months) prior to the initial RFA procedure.
Demographic and RF procedure data.
Local Tumor Response
A total of 91 lesions were treated. Complete coagulative necrosis of the treated tumor by the ablation zone after the RFA was demonstrated on follow-up CT at 1 month in 82 of 91 tumors (91%). Nine nodules (9%) showed local tumor persistence. In four of the nine incompletely treated tumors, new RFA was not performed because of important deterioration of general condition or appearance of new distant lesions on the first follow-up CT and were considered treatment failure. The other five nodules in five patients were successfully retreated with a new RFA, and the treatment was considered completed.
Analysis of Predictive Factors for Recurrence
Cox Regression analysis was performed in the 87 nodules that were considered adequately treated after one (n = 82) or two (n = 5) RFA procedures to assess predictive factors for local recurrence. The four patients with persistent tumor after incomplete ablation not amenable to new RF treatment were excluded from the analysis of local recurrence but were included in the intention-to-treat analysis of overall survival.
Subcapsular location, size, proximity to vascular structure, final temperature after RFA, and proximity to other anatomical structures (heart, pleura, gallbladder, etc.) were analyzed. On the univariate analysis, only size >3 cm and final temperature less than 65 °C were significant prognostic factors for local recurrence (p < 0.05). Local recurrence on a per-patient basis was 25% (15/59; Table 1). Overall local tumor recurrence on a per-lesion basis was 19% (17/91). In nodules of less than 30 mm, local recurrence was observed in 6% of the cases, whereas in nodules larger than 30 mm, local recurrence was found in 52% of the cases. Lesions located away from vascular structures had local recurrence in 14% of the cases (8/59), whereas subcapsular tumors or in close vicinity of vascular structures had local recurrence in 28% (9/32). Recurrence when final temperature was below 65 °C was higher (28% vs 9%). On the multivariate analysis, only the size larger than 30 mm emerged as a predictive factor for local recurrence (p < 0.05). The odds ratio for local recurrence in the group of lesions larger than 30 mm was 14.30 (95% confidence interval (CI): 4,303−47,519).
Complications
There were no post-procedural deaths in the 59 patients of the series. Procedure-related complications were observed in 14 of 77 ablation procedures (18%). There were six major complications (8%) that required surgical or interventional treatment: pericardial abscess, ileal perforation (n = 3), and biliary stenosis (n = 2). The patient with pericardial abscess eventually required surgical pericardiectomy. Two patients with ileal perforation were treated surgically, and one was managed conservatively with parenteral nutrition and antibiotics. Two patients developed biliary stenosis 2 and 10 months after RF procedure and were treated with a biliary stent.
There were also eight minor complications (10%): parietal abscess (n = 2), segmental portal thrombosis (n = 1), minimal perihepatic hematoma (n = 1), asymptomatic pneumothorax (n = 1), bronchospasm (n = 1), and severe post-procedural pain (n = 2). Those minor complications were treated symptomatically with antibiotics, analgesia, anticoagulation, and bronchodilators. Median hospital stay was 1.1 days (range: 1−85 days). In 91% of the procedures (70/77), patients were discharged at 1 or 2 days. In six patients with major complications, hospital discharge was between 10 and 84 days.
Follow-Up and Survival
After a median follow-up of 25.3 months, 29 patients have died and 30 patients (51%) were still alive. Seven of these patients (17%) were alive and free of disease. During follow-up, 34 patients (58%) presented extrahepatic tumor with or without intrahepatic recurrence, and 30 patients (51%) had distant intrahepatic recurrence away from the treated lesions (Fig. 1). The main locations of extrahepatic recurrence were the lungs (31%), local recurrence of the primary tumor (15%), lymph nodes (5%), and bone metastasis (2%).
At the end of follow-up, 15 (25%) of 59 patients were free of disease in the liver. Median follow-up in patients without recurrence was higher (3 vs 1.5 years) because patients with recurrence have lower survival. In 13 patients, a second (n = 10), third (n = 2), or fourth (n = 1) RFA was performed to treat new liver recurrences (Fig. 1). The indications for retreatment were persistent viable tumor at 1-month follow-up (n = 5), local tumoral recurrence (n = 6), and hepatic recurrence distant from the previously treated lesions (n = 9).
Survival rates at 1, 3, and 5 years were 95%, 65%, and 22%, respectively (Fig. 2), with a median survival of 43.5 months. Median disease-free survival was 10.9 months and actuarial disease-free survival rate was 45% at 1 year and 12% at 3 years.
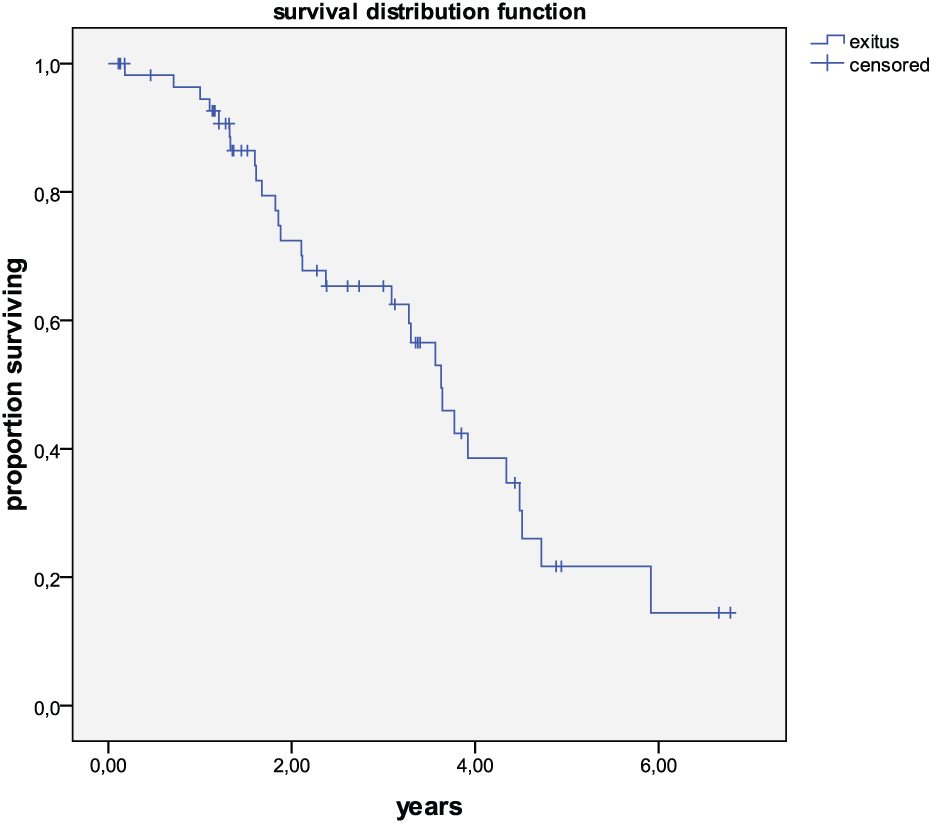
Actuarial survival of 59 patients treated with radiofrequency ablation.
Median specific hepatic disease-free survival was 17.6 months. Actuarial specific hepatic-free recurrence rate was 54% at 1 year and 24% at 2 years.
Discussion
Repeat hepatectomy is the gold standard for treating recurrent liver metastases (16–18) with reported low local recurrence. However, repeat hepatectomy is technically demanding, with a mortality rate close to 2% (17). Hence, RFA has been considered a potential option for the treatment of recurrent liver metastases (19). This study focuses primarily on the outcome and complications of patients with recurrent CLM after surgical resection, treated with percutaneous RFA.
RFA can be safely performed in non-operable patients with low morbidity rates (20, 21). However, the main concern about RFA is the risk of local tumor recurrence that has been reported to range from 4% to 40% (11, 22). This higher local recurrence rate compared to surgical resection remains a limitation for a more widespread use of the technique. Still, treatment of local recurrence can be easily repeated with percutaneous RFA, and this is a major advantage compared to surgery because liver recurrence is detected in 50%–70% of patients after curative resection (8).
In older initial series, local recurrence after percutaneous RFA was relatively high. Solbiati et al. (22) report local recurrence in 39% of the nodules and 55% of the patients, although the majority of the patients had not been operated before RF. In the series of Livraghi et al. (23), local recurrence was seen in 40% of the patients despite retreatment. A recent larger series from Solbiati et al. (24) in 99 patients with 10-years follow-up shows better results. In that series with a median follow-up of 53 months, survival rates of 3, 5, and 10 years were 69.3%, 47.8%, and 18%, respectively. The authors report a local tumor recurrence rate of 32.3% in a per-patient basis and 11.9% on a per-lesion basis. Interestingly, 54% of local recurrence could be successfully re-ablated with RF, and these patients had a better outcome than non-reablated patients. However, in that series, only 20% of the patients had undergone previous surgical resection of liver metastases. Another series from Sofocleous et al. (25) reports a series of 54 patients who underwent salvage RFA of CRC liver metastases after surgical resection. The reported rate of local tumor progression was relatively high (55% on a per-lesion basis), but in this series, all patients had recurrence from previous resection and this is probably a subgroup with worse prognosis. Yet, after re-ablation of patients with local tumor progression only, the authors report an overall survival rate of 41% at 3 years.
In our series, which consisted only of patients with hepatic recurrence after surgery, survival rates at 3 and 5 years were 65% and 22%, respectively, with a median survival of 43.5 months. Local recurrence on a per-patient basis was 25%. However, on a per-lesion basis, local recurrence was 18%, and in the subgroup of lesions less than 30 mm, local recurrence was only 6%. In addition, in our series the size of the lesion (>30 mm) was the only predictive factor for local recurrence.
It has been suggested that open surgical RFA is preferable to a percutaneous approach, due to the added benefit of detecting unsuspected disease. However, strong evidence is lacking in the literature. In the series of Reuter et al. (26), comparing RFA and surgery in a cohort study, local recurrence was higher after RFA (17% vs 2%), but survival rates were not different (21% vs 23% actuarial survival rate at 5 years). In the series of Nielsen et al. (27) concerning intraoperative RFA, local recurrence was 14% and overall 1-, 3-, and 5-year survival rates were 93%, 60%, and 31%, respectively, which are not significantly different from the reported percutaneous series (24). In addition, a recent systematic review by Lam et al. (28) has shown that postoperative morbidity and mortality after the second hepatectomy (23% and 1.2%) are significantly higher than those after RFA. However, median overall survival after second hepatectomy was better than the results achieved with RFA.
In our series, development of new hepatic and extrahepatic metastases was a frequent event (51% and 58%, respectively). This may be attributed to a more aggressive nature of the treated tumors since all patients had recurrence after previous hepatic resection. This pattern of non-local recurrence after RFA is not different after surgical resection. Topal (14) reported the development of new liver metastases in 43% of patients and new extrahepatic disease in 60% after successful complete surgical resection with clear margins. In the series of Jones (16), recurrence rate after repeat hepatectomy was 80% at 2 years, which is not significantly different from our results with percutaneous RFA (76% at 2 years).
Our results are also supported by the results of a recently published study (29) that reports local tumor recurrence after RFA and surgical resection in two independent European Organisation for Research and Treatment of Cancer (EORTC) studies. In that study, local recurrence rate per lesion after RFA or surgery was not significantly different in lesions up to 3 cm.
Our study has several limitations. First, the design of the study was a prospective observational study. Ideally, a randomized controlled trial should be performed to compare RFA and re-hepatectomy. However, design of a phase-3 randomized trial would require a very large number of patients, making very unlikely that such a trial ever takes place. There were also some limitations concerning differences in chemotherapy treatment as a part of multidisciplinary management, but these limitations are also found in surgical patients.
In conclusion, percutaneous US-guided RFA is safe and allows local tumor control in lesions less than 30 mm (local recurrence of 6%) and provides acceptable survival in patients with recurrent CLM (survival rates at 3 and 5 years of 65% and 22%, respectively). In the absence of available randomized trials, our results may be helpful to decide treatment options in patients with recurrent CLM. RFA should be considered the first option in patients with recurrent metastases from CRC not candidates to resection. In addition, our results show that CLM of less than 3 cm are especially suitable for percutaneous ablation with curative intent, with very few procedure-related complications and limited local recurrence.
Our data do not warrant systematic use of percutaneous RFA as first-line treatment in the setting of resectable recurrent CLM. However, further studies would be needed to assess its results as first-line therapy in fragile patients with small lesions (<3 cm), especially if they are centrally located lesions requiring major hepatectomy for treatment.
Footnotes
Declaration of Conflicting Interests
All named authors hereby declare that they have no conflicts of interest to disclose.
