Abstract
Background and Aims:
There are limited data on the potential role of preoperative non-invasive markers, specifically the aspartate-to-alanine aminotransferase ratio and the aspartate aminotransferase-to-platelet ratio index, in predicting perioperative liver-related complications after hepatectomy for colorectal cancer metastases.
Methods:
Patients undergoing liver resection for colorectal cancer metastases in a European institution during 2003–2010 were retrospectively enrolled. Relevant data, such as neoadjuvant chemotherapy, preoperative liver function tests, and perioperative complications, were collected from medical records. The nontumorous liver parenchyma in the surgical specimens of 31 patients was re-evaluated.
Results:
Overall, 215 patients were included. In total, 40% underwent neoadjuvant chemotherapy and 47% major resection, while 47% had perioperative complications (6% liver-related). In multivariate regression analysis, the aspartate aminotransferase-to-platelet ratio index was independently associated with liver-related complications (odds ratio: 1.149, p = 0.003) and perioperative liver failure (odds ratio: 1.155, p = 0.012). The latter was also true in the subcohort of patients with neoadjuvant chemotherapy (odds ratio: 1.157, p = 0.004) but not in those without such therapy (p = 0.062). The aspartate-to-alanine aminotransferase ratio was not related to liver-related complications (p = 0.929). The area under the receiver operating characteristics curve for the aspartate aminotransferase-to-platelet ratio index as a predictor of liver-related complications was 0.857 (p = 0.008) in patients with neoadjuvant chemotherapy. Increasing aspartate aminotransferase-to-platelet ratio index was observed with an increase in degrees of sinusoidal obstruction syndrome (p = 0.01) but not for fibrosis (p = 0.175) or steatosis (p = 0.173) in the nontumorous liver in surgical specimens.
Conclusion:
The preoperative aspartate aminotransferase-to-platelet ratio index, but not the aspartate-to-alanine aminotransferase ratio, predicts perioperative liver-related complications following hepatectomy due to colorectal cancer metastases, in particular after neoadjuvant chemotherapy. The aspartate aminotransferase-to-platelet ratio index is related to sinusoidal obstruction syndrome in the nontumorous liver.
Keywords
Introduction
Liver resection is the only potentially curative treatment for patients with colorectal cancer (CRC) liver metastases (1). Although mortality rates after liver resection have improved in recent years, the perioperative morbidity rate remains high (2). Liver-related complications, in particular liver failure, are of concern following hepatectomy (3). Neoadjuvant chemotherapy, frequently administered in patients with CRC liver metastases before liver resection, may contribute to the risk of complications by exerting a toxic effect on the nontumorous liver parenchyma. Chemotherapy-induced histopathologic liver lesions, such as steatohepatitis and sinusoidal obstruction syndrome (SOS), are considered regimen-specific (4). In particular, SOS occurs mainly in patients receiving oxaliplatin-based chemotherapy regimens and it has been associated with a poor functional liver reserve and a higher complication rate after hepatectomy, including liver dysfunction (5–7).
The preoperative aspartate aminotransferase (AST)-to-platelet ratio index (APRI) was initially developed as a non-invasive marker of significant fibrosis in chronic hepatitis C (8). Recently, it has been proposed as a non-invasive marker of high-grade SOS lesions in the nontumorous liver parenchyma of patients undergoing hepatectomy due to CRC metastases after neoadjuvant chemotherapy (7). However, there are very few data on the potential relation of the preoperative APRI with the occurrence of perioperative complications in these patients. Furthermore, the aspartate-to-alanine aminotransferase (AST/ALT) ratio, a simple non-invasive marker widely used in chronic liver disease to assess liver disease severity (9–11), has not been evaluated in the setting of hepatectomy for CRC liver metastases. Finally, although the presence of SOS has been reported to have an impact on long-term outcome in patients undergoing hepatectomy due to CRC metastases (12), it is unclear whether non-invasive biomarkers of liver pathology, such as the APRI and the AST/ALT ratio, may also be predictors of long-term outcome.
The main aim of this study was to investigate the potential role of preoperative non-invasive markers, specifically the APRI and the AST/ALT ratio, as predictors of perioperative complications after liver resection for CRC metastases. A secondary aim was to assess the relation of these non-invasive markers with long-term outcome in these patients.
Patients and Methods
Patients
All consecutive patients who underwent hepatectomy due to CRC liver metastases at the Skåne University Hospital in Lund, Sweden, between 2003 and 2010 were retrospectively included after a computerized search of the discharge register of the hospital. Repeat resections were excluded. The same cohort has been used in a study on the impact of obesity and diabetes mellitus in survival following hepatectomy for CRC metastases (13). In short, medical records were scrutinized and relevant data were extracted, such as demographics, site of the primary tumor, preoperative chemotherapy and its intent, date and extent of hepatectomy, American Society of Anesthesiologists (ASA) score, and preoperative locoregional treatment (such as radiofrequency ablation). Preoperative liver function tests and platelet counts, routinely measured usually within the last week prior to liver resection, were registered for calculation of the AST/ALT ratio and APRI score (8). As a measure of comorbidity, the Charlson comorbidity index (14) was calculated after exclusion of malignant tumors as all patients had this in common. In the surgical specimen, a resection margin of <1 mm was considered positive, while a margin of ⩾1 mm was considered a tumor-free margin. Resections of ⩾3 liver segments were considered major resections.
Perioperative Complications and Survival Following Hepatectomy
Complications were retrospectively identified. They were graded according to the Dindo–Clavien classification (15), and those with a score of ⩾3 were considered severe. Complications were further classified as non-liver related (cardiovascular, respiratory, infection, bleeding, acute kidney injury, and other) and liver related. Bleeding was defined as need for intra- or post-operative blood transfusions. Acute kidney injury was defined as an abrupt increase in serum creatinine of ⩾26.4 µmol/L, a percentage increase in serum creatinine of ⩾1.5-fold from baseline, or a reduction in urine output (in cases of documented oliguria) (16). Liver-related complications included post-operative hepatic failure, defined as peak post-operative total bilirubin >119.7 µmol/L (3) and/or ascites, and bile leak requiring drainage. Dead versus alive status after hepatectomy was ascertained through medical records and the regional healthcare administration system. Alive patients were censored at last follow-up and overall survival (from hepatectomy until death from any cause) was calculated. Disease-free survival (from hepatectomy until tumor recurrence or death due to any cause) was also calculated.
Histopathologic Examination
The nontumorous liver parenchyma in the surgical specimens of a subset of 31 patients was re-evaluated for the presence of SOS, fibrosis, and steatosis by a liver pathologist (P.R.) blinded to the clinical data. Sinusoidal dilation was graded semiquantitatively: 0, absent; 1, mild (centrilobular involvement limited to one-third of the lobular surface); 2, moderate (centrilobular involvement in up to two-third of the lobular surface); 3, severe (complete lobular involvement) (17). Steatosis and fibrosis were assessed as the non-alcoholic steatohepatitis activity score (NAS), in which the following features are graded: steatosis (0–3), lobular inflammation (0–2), ballooning (0–4), and fibrosis (0–4) (18).
Statistics
Data are presented as mean and standard deviation (SD) or n (%) as appropriate. Fisher’s exact test was used for categorical variables, and the Mann–Whitney test was used for continuous variables for group comparisons. Logistic regression analysis was used to adjust for confounders in the relation between complications and the APRI. Confounding factors taken into consideration were as follows: age, gender, the ASA score, the Charlson comorbidity index, type of resection, and diabetes, as well as any parameter in the cohort found to be related to post-operative complications at p < 0.1 in univariate analysis. The discriminant validity of the preoperative APRI score in predicting the occurrence of complications was assessed by means of the area under the receiver operating characteristics curve (AUROC). The relationships of the APRI and the AST/ALT ratio with overall and disease-free survival were assessed with Cox regression analysis. Multivariate Cox proportional-hazards models were used to adjust for confounders, that is, node positive primary, >3 liver metastases, size of the largest ⩾5 cm, carcinoembryonic antigen >60 mg/mL, positive resection margin, and extrahepatic disease (19), as well as any parameter found to be univariately related to overall or disease-free survival at p < 0.1 in this cohort. Reported p-values are two-tailed and the significance level was set at p < 0.05.
Results
A total of 215 patients underwent first-time hepatectomy for CRC metastases during the study period (Table 1). Preoperative AST and ALT were available in all patients, but there were no available preoperative platelet counts in 17 (7.9%) patients. Thus, all patients (n = 215) were included in the study cohort (Table 1), but analyses regarding the APRI were performed in 198 patients. In all, 40% received neoadjuvant chemotherapy (30% oxaliplatin-based) and 47% underwent a major resection (Table 1). Overall, 3/215 (1.4%) of patients were known to have chronic liver disease (n = 2, hemochromatosis; n = 1, hepatitis C) but none was known to have liver cirrhosis.
Characteristics of patients undergoing liver resection due to CRC metastases.
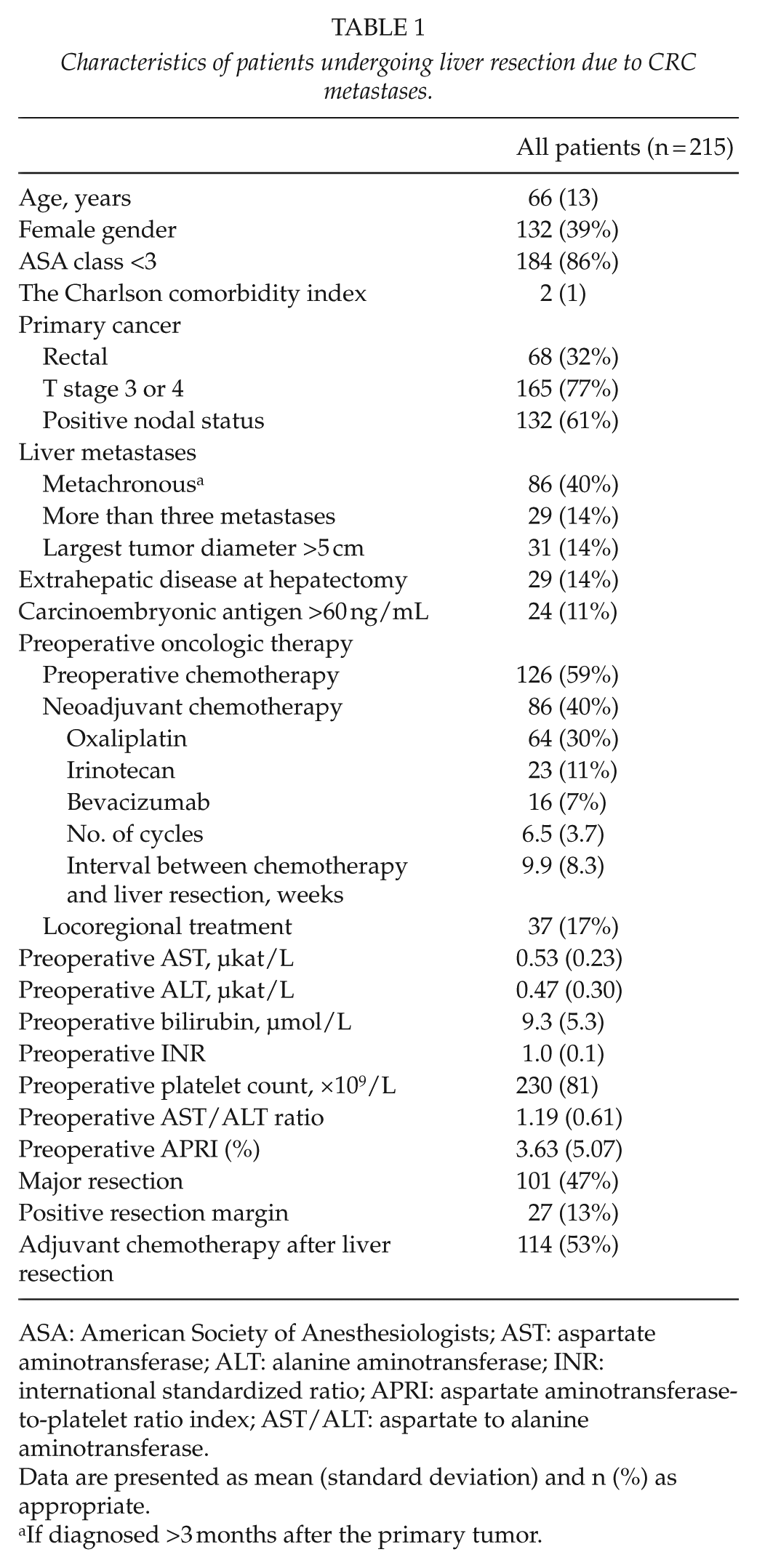
ASA: American Society of Anesthesiologists; AST: aspartate aminotransferase; ALT: alanine aminotransferase; INR: international standardized ratio; APRI: aspartate aminotransferase-to-platelet ratio index; AST/ALT: aspartate to alanine aminotransferase.
Data are presented as mean (standard deviation) and n (%) as appropriate.
If diagnosed >3 months after the primary tumor.
Relation Of Perioperative Complications With The Ast/Alt Ratio And The Apri
One or more perioperative complications occurred in 47% of patients, but only 9% experienced a severe event (Table 2). In univariate analysis, a higher APRI was related to the occurrence of liver-related complications, in particular liver failure, and to longer length of hospital stay (Table 2). The AST/ALT ratio was not associated with perioperative complications (Table 2).
Occurrence of perioperative complications in all patients undergoing liver resection due to CRC metastases.
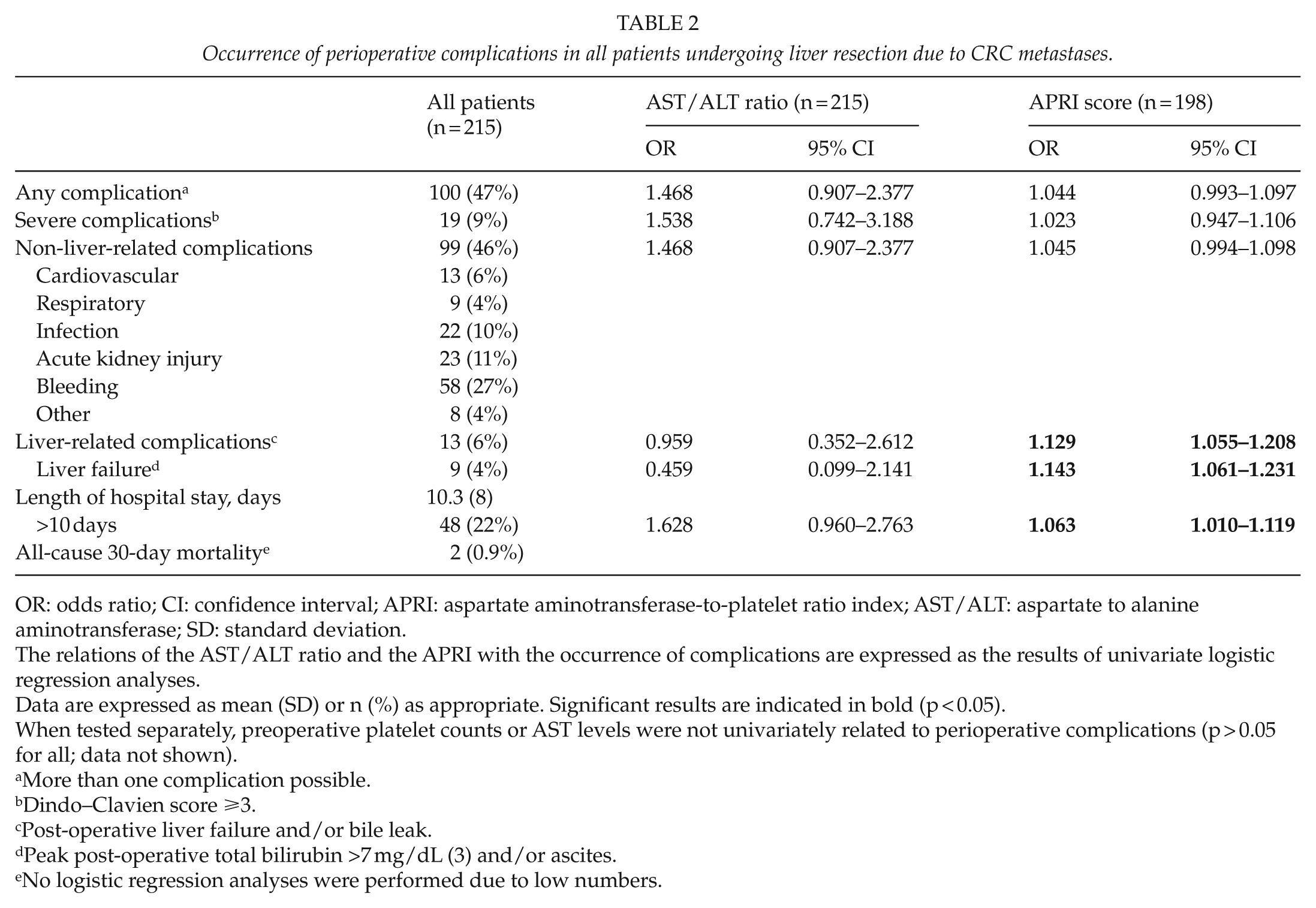
OR: odds ratio; CI: confidence interval; APRI: aspartate aminotransferase-to-platelet ratio index; AST/ALT: aspartate to alanine aminotransferase; SD: standard deviation.
The relations of the AST/ALT ratio and the APRI with the occurrence of complications are expressed as the results of univariate logistic regression analyses.
Data are expressed as mean (SD) or n (%) as appropriate. Significant results are indicated in bold (p < 0.05).
When tested separately, preoperative platelet counts or AST levels were not univariately related to perioperative complications (p > 0.05 for all; data not shown).
More than one complication possible.
Dindo–Clavien score ⩾3.
Post-operative liver failure and/or bile leak.
Peak post-operative total bilirubin >7 mg/dL (3) and/or ascites.
No logistic regression analyses were performed due to low numbers.
Patients who experienced liver-related complications were somewhat older, they had lower preoperative platelet counts, and they underwent extensive resection more frequently compared to those without liver-related complications (Table 3). However, the two groups did not differ significantly in liver metastases characteristics nor in their preoperative liver function tests or in preoperative oncologic treatment (Table 3). After adjustment for confounders by multivariate logistic regression analysis, the APRI score was still related to the occurrence of liver-related complications (adjusted odds ratio (OR): 1.149, 95% confidence interval (CI): 1.05–1.258, p = 0.003) and, in particular, liver failure (adjusted OR: 1.197, 95% CI: 1.062–1.350, p = 0.003). The occurrence of liver-related complications did not differ significantly among patients operated early (2003–2007) versus late (2008–2010) in the study period (p > 0.05; data not shown).
Characteristics of patients with versus without perioperative liver-related complications following liver resection due to colorectal cancer metastases.
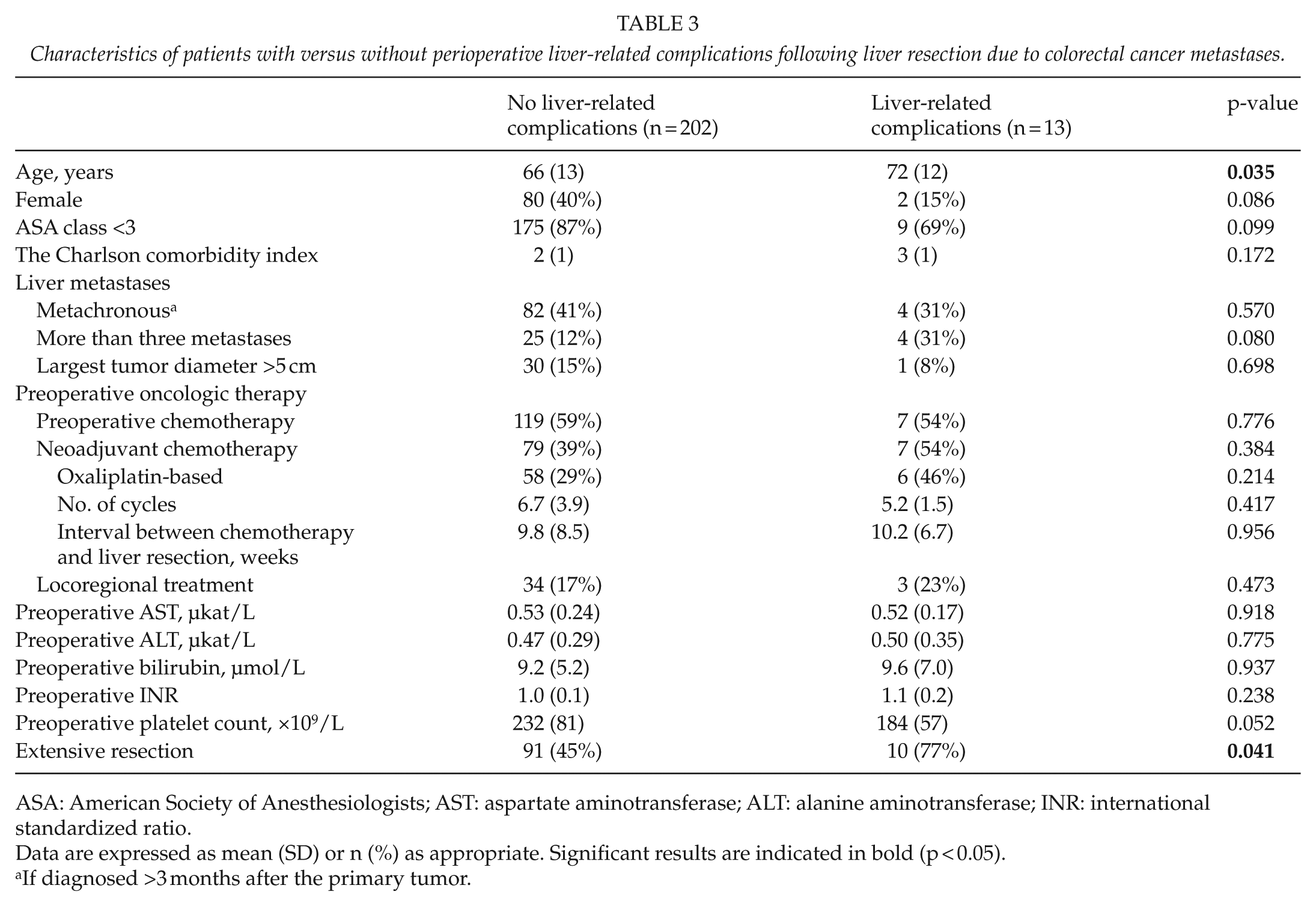
ASA: American Society of Anesthesiologists; AST: aspartate aminotransferase; ALT: alanine aminotransferase; INR: international standardized ratio.
Data are expressed as mean (SD) or n (%) as appropriate. Significant results are indicated in bold (p < 0.05).
If diagnosed >3 months after the primary tumor.
The APRI was an independent predictor of liver failure (adjusted OR: 1.341, 95% CI: 1.038–1.732, p = 0.025) when analyses were performed only in patients who had received neoadjuvant chemotherapy prior to hepatectomy, but this did not reach statistical significance when analyses were restricted in patients without prior neoadjuvant therapy (adjusted OR: 1.219, 95% CI: 0.990–1.5, p = 0.062).
Receiver Operating Characteristics Analysis
In the whole cohort (n = 215), the AUROC of the APRI score for the prediction of liver-related complications was 0.698 (95% CI: 0.513–0.883, p = 0.035) and for the prediction of liver failure 0.761 (95% CI: 0.571–0.960, p = 0.019). Among patients who had received neoadjuvant chemotherapy, the discriminant validity of the APRI score was very good both for the prediction of liver-related complications (AUROC: 0.857, 95% CI: 0.722–0.991, p = 0.008) and for the prediction of liver failure (AUROC: 0.827, 95% CI: 0.667–0.987, p = 0.028).
Relation of the Apri with Histopathology Features of the Nontumorous Liver Parenchyma
The surgical specimens of a subset of 31 patients were re-evaluated by a liver histopathologist blinded to the clinical data (mean age: 64 years (SD: 2); 16% female; 45% with neoadjuvant chemotherapy, 32% oxaliplatin-based; 87% ASA class <3; 55% with extensive resection). The APRI score increased significantly with an increase in SOS grades but did not differ significantly among patients with different scores of steatosis, NAS, or fibrosis (Table 4). Two out of three liver-related complications occurring in this group of 31 patients were experienced by patients with SOS grade 2 and one-third by a patient with SOS grade 0 (p = 0.06).
Relation of the APRI score with histopathology features of the nontumorous liver parenchyma in 31 randomly selected patients undergoing liver resection due to CRC metastases whose surgical specimens were re-evaluated.
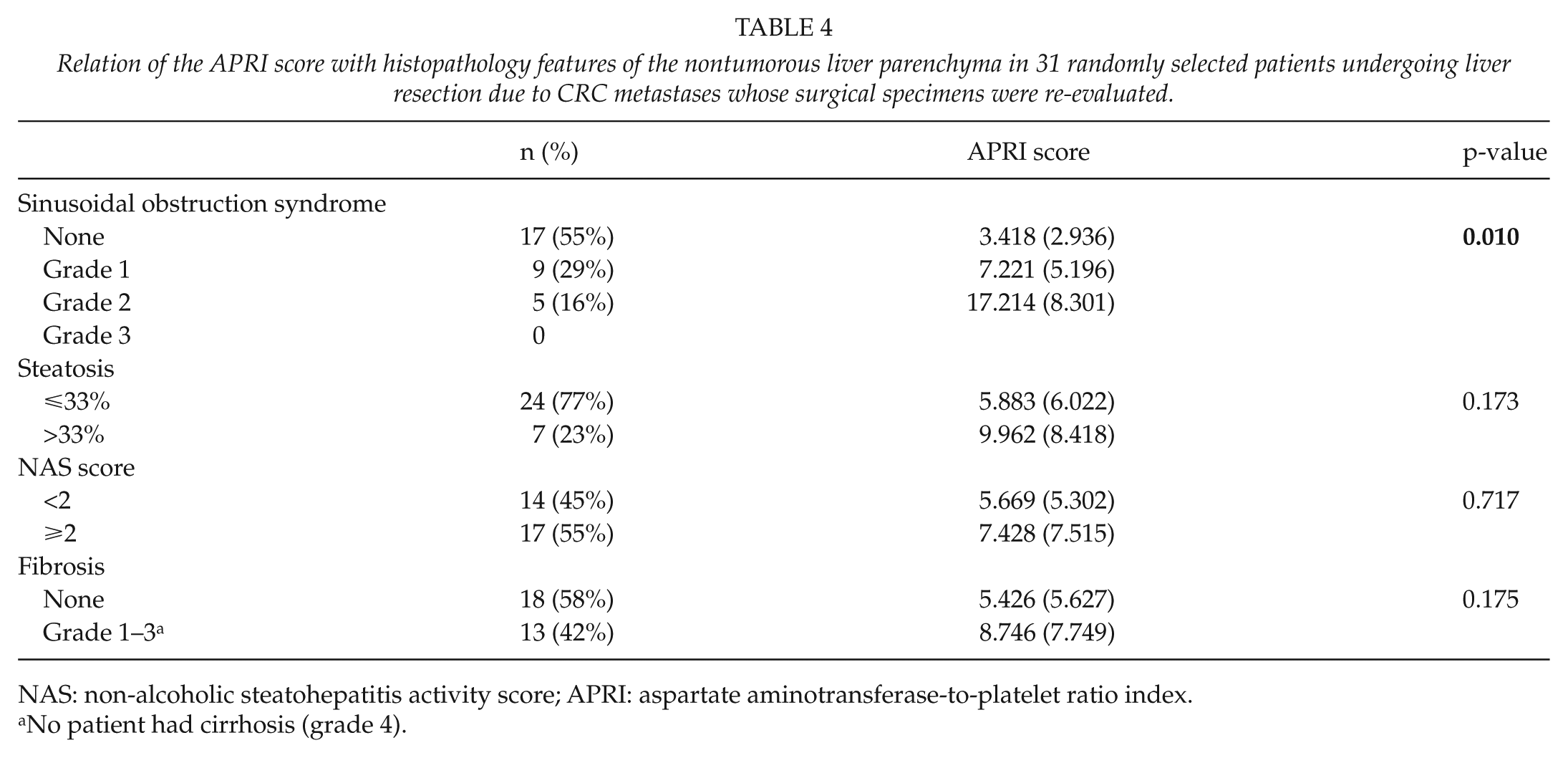
NAS: non-alcoholic steatohepatitis activity score; APRI: aspartate aminotransferase-to-platelet ratio index.
No patient had cirrhosis (grade 4).
Relation of the Apri and Ast/Alt Ratio with Long-Term Outcome after Liver Resection
Patients included in this study were followed up for a mean of 37 months (SD: 28). Overall, 72 patients died during follow-up. Recurrence status or recurrence dates could not be ascertained in 12 patients who were excluded from disease-free survival analyses (all referred back to referring institutions) (13). In univariate analysis, the APRI was related to overall survival only in patients who had received neoadjuvant chemotherapy (hazard ratio (HR): 1.084, 95 CI: 1.016–1.157, p = 0.015) but not the whole cohort (HR: 1.007, 95% CI: 0.970–1.046, p = 0.71) or in patients without neoadjuvant treatment (HR: 0.97, 95% CI: 0.915–1.028, p = 0.309). The APRI was not found to be a predictor of disease-free survival in the whole cohort nor in the subcohorts with or without neoadjuvant therapy (p > 0.05 for all; data not shown). The AST/ALT ratio was not related to overall or disease-free survival (p > 0.05 for all; data not shown).
After adjustment for confounders by means of multivariate Cox regression analysis, there was only a trend for an independent relationship between APRI and overall survival (HR: 1.093, 95% CI: 0.992–1.205, p = 0.072). Surgery early (2003–2007) versus late (2008–2010) in this study period was not significantly related to overall survival (p > 0.05; data not shown).
Discussion
The main finding of this study is that the preoperative APRI, a non-invasive biomarker easily calculated from routine blood tests, is an independent predictor of liver-related complications, and in particular liver failure, in patients undergoing hepatectomy due to CRC metastases in a non high-volume center. This is in line with a recent retrospective case–control study showing that patients with perioperative liver failure following resection for CRC metastases have significantly higher APRI compared to controls without liver failure (20). Our findings further suggest that the APRI may be useful in discerning patients with a higher risk of developing liver-related complications post-hepatectomy, especially among unselected patients who had undergone neoadjuvant chemotherapy.
There are several studies showing that neoadjuvant chemotherapy (in particular oxaliplatin-based regimens) is associated with SOS (21–23). Sinusoidal injury may persist several months after chemotherapy, while associated fibrosis may progress (17). We could confirm a previous report showing that the APRI is an indicator of the occurrence and severity of SOS (7). This, in turn, is in accordance with published data on the relation of post-chemotherapy sinusoidal injury with a poorer functional liver reserve and perioperative morbidity (6). Should our results be confirmed in larger cohorts of patients undergoing hepatectomy for CRC metastases in which reliable cut-off values for the detection of patients at high risk for perioperative liver failure could be identified, it is conceivable that preoperative APRI may be used for discerning these patients prior to hepatectomy. Such patients could, potentially, benefit from reducing the number of chemotherapy cycles (24), prolonging the interval between cessation of chemotherapy and liver resection (25), and/or adding bevacizumab in the chemotherapy regimen (26, 27).
A single previous report has shown that SOS in the nontumorous liver parenchyma of patients undergoing liver resection for CRC metastases may be related to poor overall and disease-free survival (12). This could be associated with the higher risk of perioperative complications these patients are exposed to, which, in turn, could have an impact on long-term outcome (28) or with endothelial factors implicated in the tumor cell adhesion and invasion into the liver parenchyma (12). In this cohort, although the APRI was significantly related to overall survival in univariate analysis, after adjustment for confounders there remained only a statistical trend (p = 0.072). Furthermore, no association between the APRI and disease-free survival was found. Further studies are warranted to delineate the potential relation between chemotherapy-induced SOS and long-term outcome following liver resection for CRC metastases and to evaluate the potential role of preoperative APRI, as a surrogate of the former in predicting the latter. However, our findings suggest that APRI is unlikely to be a major predictor of long-term outcome in these patients.
Although the preoperative AST/ALT ratio has been used as a measure of liver disease severity in patients with various types of chronic liver disease (9–11), it was not a predictor of perioperative liver failure in this cohort. This could be explained by the fact that neither AST nor ALT differed significantly between patients with versus those without liver-related complications. However, the former group had significantly lower preoperative platelet counts, which, in turn, may explain why preoperative APRI was related to perioperative liver-related complications and liver failure. This is in line with recent studies showing that spleen volume increase during oxaliplatin-based neoadjuvant chemotherapy is associated with SOS, thrombocytopenia, and complications after liver resection for CRC metastases (29, 30). As these patients frequently undergo computed tomography scans, it is possible that measuring spleen volume during neoadjuvant chemotherapy may be complementary to preoperative APRI as non-invasive markers predicting perioperative liver-related complications. This is a hypothesis that would need to be addressed in future studies.
Limitations of this study include its retrospective nature, and thus subject to bias, including but not limited to information bias, and the fact that it was conducted in a single center. Also, the study spans 8 years. Despite that neither the occurrence of liver-related complications nor overall survival differed significantly among patients operated early versus late in the study period, it is conceivable that changes in surgical and oncological practices over the years may have had an impact on our findings. Finally, patients with liver-related complications were older and underwent major resection more frequently compared to those without such complications. Although patient’s age and extent of resection were adjusted for in multivariate analyses for complications, it is conceivable that these differences may have confounded our results. Thus, although our findings are in line with those of previous studies (6, 7, 20), it is clear that they need to be confirmed in larger multicenter cohorts, ideally comprising prospectively collected data. Also, the predictive value of preoperative APRI should be validated in an external larger patient sample, in which relevant cut-off values for the prediction of post-operative complications could be identified.
Conclusion
Preoperative APRI, but not AST/ALT ratio, is an independent predictor of perioperative liver-related complications following liver resection due to CRC metastases, in particular after neoadjuvant chemotherapy. It is also related to the degree of SOS lesions in the nontumorous liver parenchyma, but it does not appear to be a major predictor of long-term outcome in these patients. Should our findings be confirmed in larger patient cohorts, the preoperative APRI, which is easily calculated from routine blood tests, may turn out to be useful non-invasive biomarkers in patients with CRC liver metastases receiving neoadjuvant chemotherapy prior to hepatectomy.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
The study protocol was approved by the regional ethics committee in Lund, Sweden (2012/132). No written informed consent was required for this retrospective study.
Funding
This work was supported by a grant from the Inger Persson fund for cancer research.
