Abstract
Background and Aims:
Skeletonization has been proposed as a technique to minimize the risk of sternal devascularization during bilateral internal thoracic artery harvest for coronary artery bypass grafting. The impact of this strategy on late radiologic pleuropulmonary changes has not been addressed.
Material and Methods:
Post-operative chest radiographs from patients (n = 253 per group) undergoing bilateral internal thoracic artery harvest using skeletonized and non-skeletonized techniques were reviewed by blinded radiologists. The primary outcome was the incidence of atelectasis and pleural effusion. Multivariable linear regression models were derived to assess the relationship of radiologic pleuropulmonary outcomes to patients and operative variables.
Results and Conclusion:
Patients in the skeletonized group were older (p < 0.0001), had a lower preoperative hematocrit (p = 0.014), had higher prevalence of peripheral vascular disease (p = 0.001), were of female gender (p = 0.015), underwent off-pump surgery (p < 0.001), had urgent/emergent status (p = 0.024), and had chronic obstructive pulmonary disease (p = 0.019). There was no difference in the incidence of post-operative complications, ventilation time, or intensive care unit stay. There was no difference in the severity of post-operative atelectasis in both groups. More patients in the non-skeletonized group had a grade 2/3 left pleural effusion on the late post-operative chest X-ray (p = 0.007). The independent effect of skeletonization on the development of a late left pleural effusion was significant (odds ratio = 0.558, 95% confidence interval = 0.359–0.866, p = 0.009). Skeletonization results in a decreased incidence of late post-operative left pleural effusion with no difference in early or late atelectasis. Further studies are warranted to assess the mechanism of these pleuropulmonary changes and the impact of other factors such as pleural violation during surgery.
Introduction
Use of bilateral internal thoracic artery (BITA) harvest for coronary artery bypass grafting (CABG) has become routine due to the findings that BITA grafting improves long-term survival over single internal thoracic artery use (1–4). The enthusiasm for this strategy has been tempered by concerns that BITA grafting may result in increased sternal wound infections as a result of sternal devascularization particularly in diabetic patients (5, 6). In an effort to mitigate this complication, many surgeons have advocated skeletonization of the artery, which involves careful dissection of the conduit from adjacent tissues, preserving the accompanying vein, fascia, nerves, and muscles in situ. Although this technique is more time-consuming, studies have shown improved sternal blood supply post-operatively compared to the non-skeletonized (NSK) technique (7). Skeletonization has been associated with a decreased risk of wound complications (8–10) and decreased post-operative chest wall pain and dysesthesia (7). It has not been conclusively demonstrated whether skeletonization is also associated with improvement in pulmonary function related to the limited dissection and the subsequent decrease in chest wall pain.
The aim of this study was to determine whether skeletonization of the internal thoracic artery is associated with differences in pulmonary and pleural changes as opposed to NSK harvest techniques. We hypothesize that skeletonized (SK) BITA harvest will be associated with decreased radiologically detected atelectasis and effusion in patients undergoing coronary surgery.
Materials And Methods
Ethics
Our center has approval from its institutional research ethics board to anonymously publish data that are prospectively collected before and after CABG. As such, individual patient consent was waived.
We obtained approval from the Research Ethics Board (Protocol # 2011188-01H) to retrospectively analyze prospectively collected data from the Peri-operative Database Unit to identify patients undergoing CABG since the initiation of the use of SK internal thoracic harvest. Between the periods of 2 May 2006 and 23 October 2012, 9166 patients have undergone CABG of which 2393 cases were done with BITA. Patients were excluded if they had undergone other procedures such as valve replacement or arrhythmia surgery. Patients were also excluded if follow-up radiographs were not available. A final cohort of 253 SK cases was selected, and these were matched 1:1 with a cohort of NSK cases based on the date of surgery. The database captures detailed information on preoperative, peri-procedural, and post-operative variables for all patients undergoing cardiac surgery, and it is maintained by a team of full-time data abstractors who are responsible for data collection and an ongoing audit process. Follow-up until death or discharge was 100% in both groups.
Peripheral vascular disease was defined as the presence of claudication with confirmation of lower limb vascular stenosis on Doppler, a history of peripheral vascular intervention, abdominal aortic aneurysm, or documented renal or subclavian stenosis.
Chest Radiography
Radiographs were interpreted by one of three radiologists with 7–20 years of experience (S.A., A.O., C.J.D.) who were blinded to the surgical technique. Pre- and post-operative chest radiographs were obtained in all patients and assessed for the presence of basal atelectasis and pleural fluid. The first post-operative chest radiograph was a supine anteroposterior projection obtained immediately after surgery. This was defined as the early chest X-ray, and the position of the pleural tubes, if present, was assessed at that time. The second post-operative radiograph was an anteroposterior upright chest X-ray obtained after removal of the mediastinal and pleural tubes. This was defined as the mid chest X-ray, and the third chest X-ray was a posteroanterior and lateral view obtained 30 days to 6 weeks post-operatively at the time of the first out-patient follow-up appointment. Atelectasis was graded using a modified rating scale based on that described by Wilcox et al. (11).
Pleural effusions were defined as grade 1 if there was minimal blunting of the costophrenic sulcus, grade 2 if the effusion occupied between 15% and 30% of the hemithorax, and grade 3 if it occupied over 30% of the hemithorax.
Correct pleural tube position was defined as a tube curving either obliquely upward around the mid-lung or horizontally into the costophrenic sulcus (Fig. 1A and B). An incorrectly positioned tube was defined as a tube pointing vertically upward toward the apex of the lung (Fig. 1C).
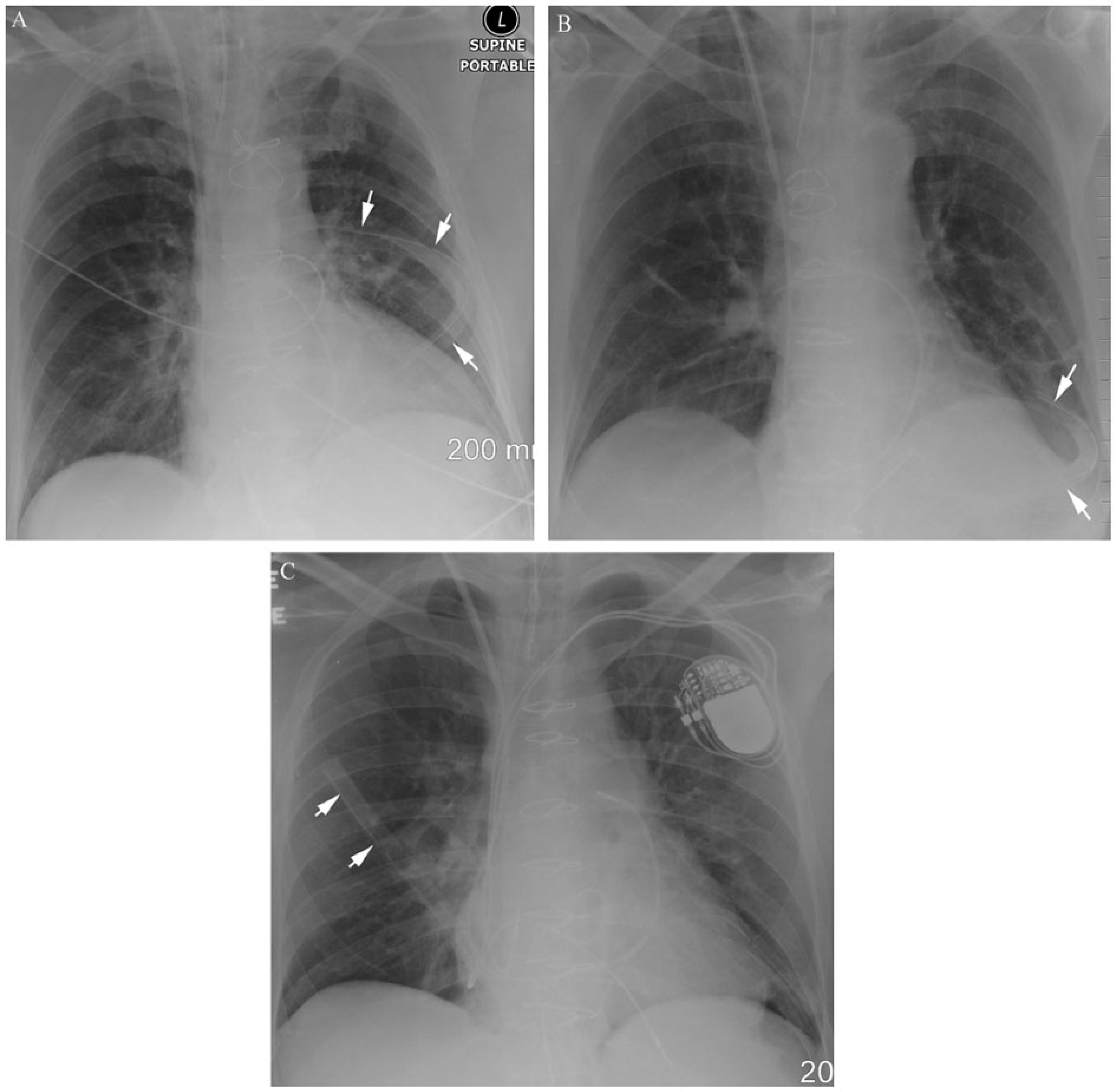
(A) Supine AP chest radiograph of a 70-year-old man with a correctly placed pleural tube curving around to the back of the lung in the left mid zone (arrows). (B) Supine AP chest radiograph of a 58-year-old man showing a correctly placed pleural tube curving around the base of the left lung just above the hemidiaphragm (arrows). (C) Supine AP chest radiograph of a 52-year-old man depicts poor positioning of the pleural tube. The tube has been placed over the surface of the right lung rather than curving around it to drain fluid in the dependent portion of the pleural space (arrows).
Statistical Analysis
Continuous variables were reported as mean ± standard deviation or median ± interquartile range for non-normally distributed variables. Categorical variables were reported as counts and percentages. Student’s t-tests or Wilcoxon rank-sum tests were used to compare continuous variables between the two BITA groups. For categorical variables, chi-square or Fisher’s exact tests were used to compare the differences between the two groups. The impact of treatment type (SK vs NSK) on the development of late pleural effusion was assessed for both univariable and multivariable associations using logistic regression models. Univariable predictors included age, urgent/emergent status, chronic obstructive pulmonary disease (COPD), current smoking, female gender, incorrect pleural tube placement, off-pump surgery, and skeletonization. For the multivariable analysis, the independent effect of the treatment type on the development of late pleural effusion was assessed by controlling for baseline characteristics related to pleural effusion in the univariable analysis (p < 0.1). A p < 0.05 was considered significant. All statistical analyses and plots were performed with STATA® version 13.1 (College Station, Texas).
Results
A total of 506 patients were included in the study (SK = 253, NSK = 253). Patient characteristics are outlined in Table 1. Patients in the SK group were older (p < 0.0001), had a lower preoperative hematocrit (p = 0.014), and had a higher prevalence of peripheral vascular disease (p = 0.001) as well as COPD (p = 0.019). There were more females (p = 0.015) in the SK group. The SK cases were also more commonly done off-pump (p < 0.0001) and on an emergent/urgent basis (p = 0.024).
Patient characteristics.
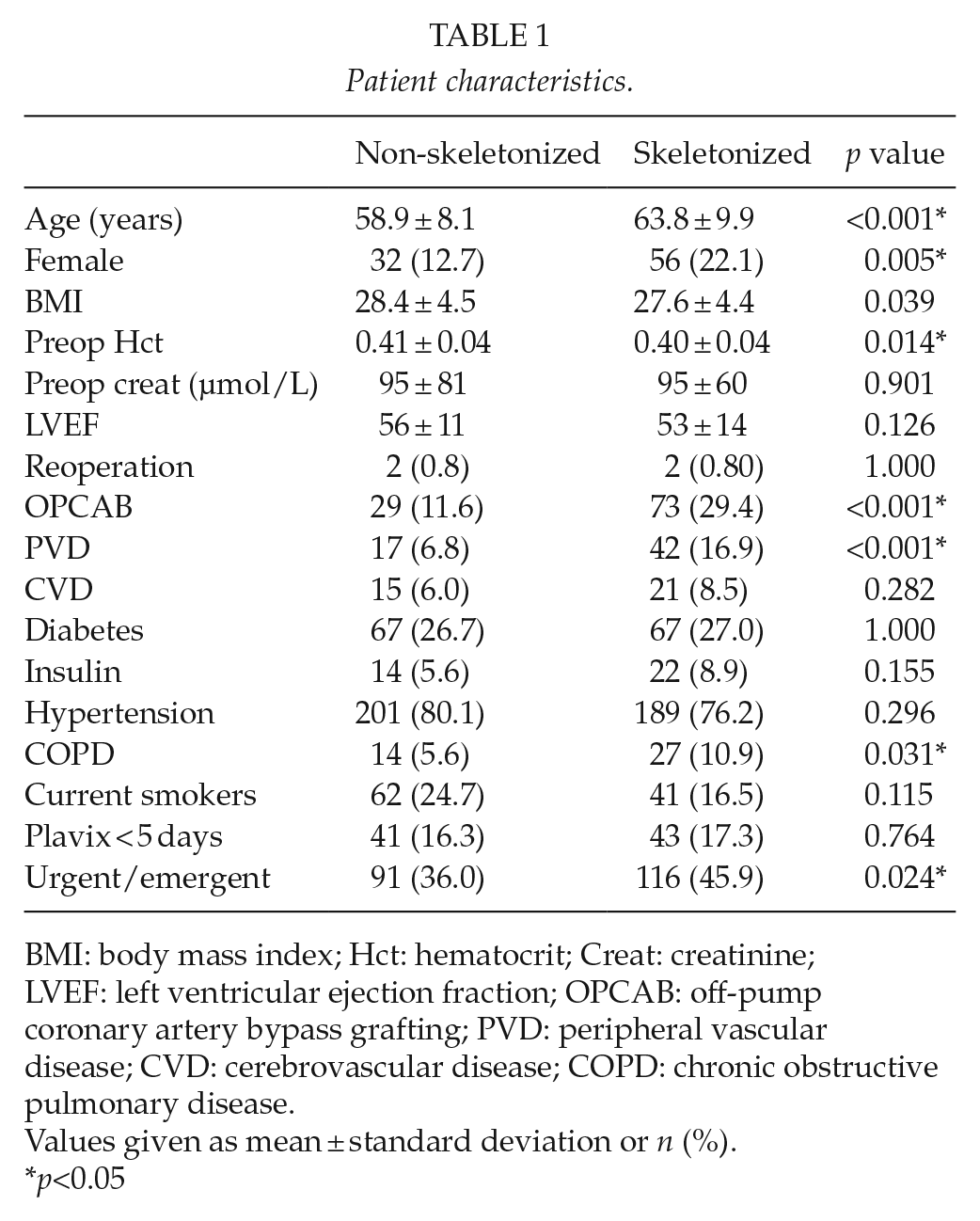
BMI: body mass index; Hct: hematocrit; Creat: creatinine; LVEF: left ventricular ejection fraction; OPCAB: off-pump coronary artery bypass grafting; PVD: peripheral vascular disease; CVD: cerebrovascular disease; COPD: chronic obstructive pulmonary disease.
Values given as mean ± standard deviation or n (%).
p<0.05
Post-operative patient characteristics and complications were similar in both groups except for a larger volume of chest tube drainage in the NSK group (p = 0.028) (Table 2). Phrenic nerve injury was uncommon, occurring in one patient in the NSK group (0.40%) and three patients in the SK group (1.19%) (p = 0.624). There was no difference in the duration of ventilation or the length of intensive care unit stay.
Post-operative clinical parameters and complications.
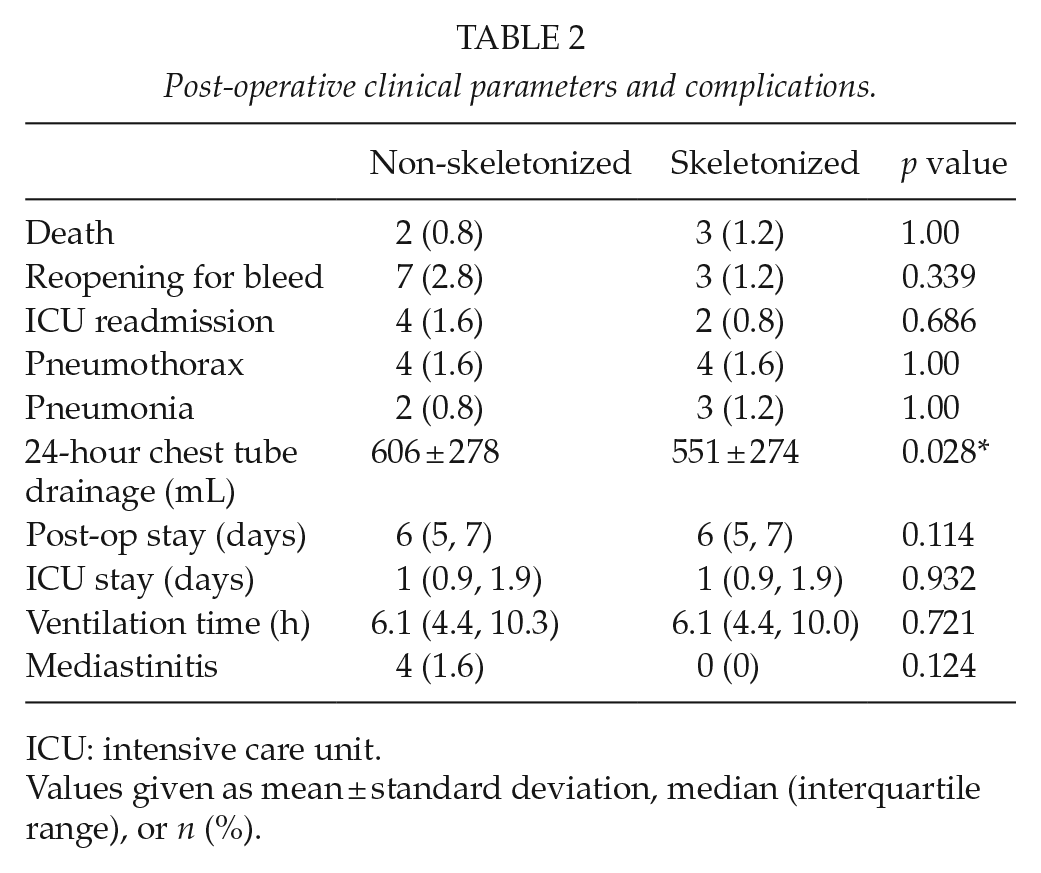
ICU: intensive care unit.
Values given as mean ± standard deviation, median (interquartile range), or n (%).
Results of the chest radiographic interpretation are outlined in Table 3. The average time elapsed between surgery and the second post-operative (mid) radiograph was 3 days in both groups (p = 0.755). The average time to the late follow-up chest X-ray was 36 days for the SK group and 30.5 days for the NSK group (p = 0.002). There was no statistically significant difference in the severity of post-operative atelectasis in the SK and NSK groups. Non-breech of the left pleura occurred in 28 patients in the SK group and 9 patients in the NSK group. Late pleural effusion occurred in 25% and 33% of cases, respectively (p = n.s.). Non-breech of the right pleura occurred in eight patients in the SK group and four patients in the NSK group. Late pleural effusion occurred in 13% and 50% of cases, respectively (p = n.s.).
Post-operative grade 2/3 atelectasis and pleural effusion on chest X-ray.
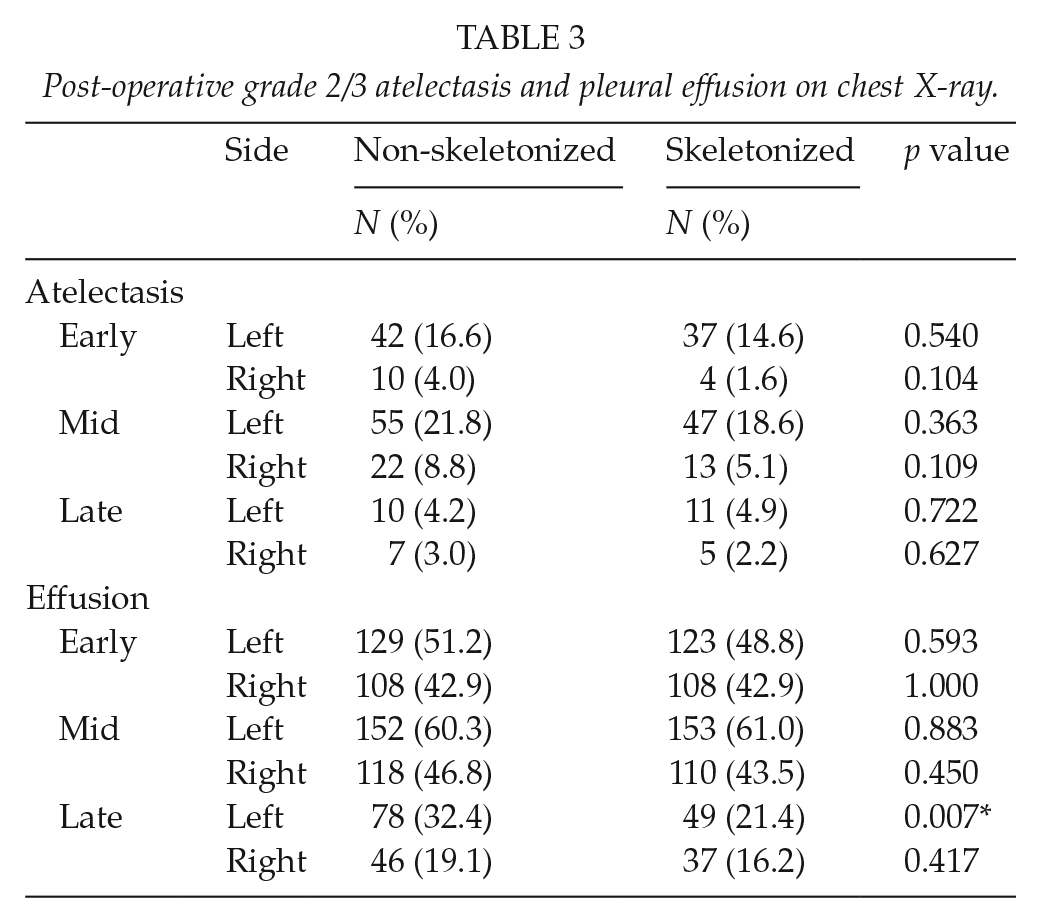
More patients in the NSK group had a grade 2/3 left pleural effusion on the late post-operative chest X-ray (p = 0.007). One patient in the NSK group required late thoracentesis. All of these were treated successfully medically. Univariable predictors for the development of a late left pleural effusion were the use of the NSK surgical technique (odds ratio (OR) = 0.569, 95% confidence interval, CI = 0.376–0.862, p = 0.008) (Table 4). For the multivariable analysis, the independent effect of skeletonization on the development of a late left pleural effusion was assessed and remained significant (OR = 0.558, 95% CI = 0.359–0.866, p = 0.009). There was an association with poor chest tube position, but this was not significant (OR = 1.571, 95% CI = 0.905–2.727, p = 0.108).
Univariable modeling of late left pleural effusion.
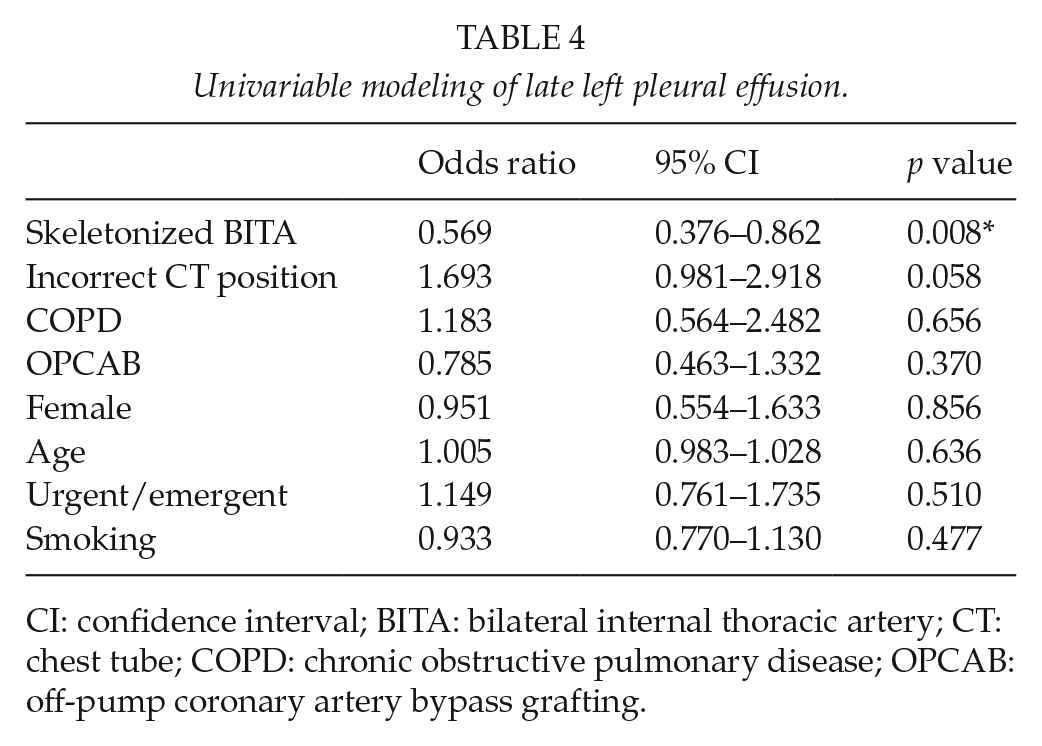
CI: confidence interval; BITA: bilateral internal thoracic artery; CT: chest tube; COPD: chronic obstructive pulmonary disease; OPCAB: off-pump coronary artery bypass grafting.
Discussion
Minimization of chest wall trauma by skeletonization of the internal thoracic arteries during CABG potentially may impact peri-operative respiratory function. This study has demonstrated that skeletonization is not associated with a change in the incidence of early or late moderate to severe atelectasis as detected radiologically, but it is associated with a decreased incidence of late left pleural effusion. On the other hand, the majority of these effusions did not require intervention and there was no difference in the incidence of right-sided pleural effusion between the groups. Location of chest tube placement did not affect the development of a pleural effusion in either group.
Atelectasis is a common complication of cardiac surgery with a multifactorial etiology. Peri-operative factors may include impaired clearance of secretions, gastric distension, limited mobility, as well as lung collapse due to the lack of positive pressure ventilation during cardiopulmonary bypass (11). The predominance of left-sided atelectasis after cardiac surgery with the left internal thoracic artery in previous reports has been attributed to phrenic nerve injury possibly related to topical ice administration during cardiac arrest (11) and also due to potential mechanical damage during the harvest of the internal thoracic artery (12).
The impact of NSK internal thoracic harvest on atelectasis was evaluated in an early study by Daganou et al. (13). This retrospective cohort study compared radiographs of BITA cases to cases with only left internal thoracic artery harvest. The incidence of right lower lobe atelectasis increased in the former group strongly supporting the effect of the incremental right internal thoracic NSK harvest technique.
Pleural violation has also been suggested to contribute to atelectasis possibly due to the need for a chest tube, which may increase pain and limit respiratory reserve. This compromise may translate into deleterious clinical outcomes in patients undergoing CABG. Bonacchi et al. (14) reviewed their experience with SK BITA grafting with and without pleural violation and compared these groups to NSK BITA grafting cases, all with pleural violation. Pleural violation was associated with increased ventilation time, increased pain, and an increased incidence of prolonged ventilation as opposed to cases with an intact pleura. Blood loss was increased in NSK cases, similar to that demonstrated in this study; however, there were no other consistent major respiratory benefits of skeletonization over non-skeletonization when the pleura was violated.
Early effusions occupying over 15% (Grade 2/3) of the pleural space are a common radiologic finding after CABG. These effusions persist over 1 month post-operatively in 16%–27% of patients and are more common on the left particularly in patients undergoing CABG using an internal thoracic artery graft in addition to a saphenous vein graft as compared to using saphenous vein grafts alone (15, 16). The incidence of persistent pleural effusions in this study was 25% with predominance in the NSK group.
Limitations of this study include its retrospective nature and the potential for unknown confounders that may have influenced the incidence of pulmonary changes in the two groups. Late chest radiographs in the SK group were obtained 5.5 days later than those in the NSK group; therefore it is possible that some of the left effusions in this group were resolved. However as this only represented a 15% difference in time and no therapy was initiated in either group before the radiographs were obtained to hasten resolution, it is unlikely that this was contributory to this difference.There were also potential technical differences in the surgical techniques, and surgeon-specific results between groups may have been a factor as a single operator completed the majority of the SK cases. Finally, only one pleural effusion required an intervention, and therefore, the clinical relevance is not clear. For example, it is possible that there were other objective differences between the groups such as dyspnea, which were not measured.
In conclusion, skeletonization during BITA grafting for coronary artery bypass surgery is associated with a significant decrease in the incidence of persistent left-sided pleural effusion, whereas there is no difference in early or late atelectasis with this strategy. Further studies should be directed to evaluate techniques to further minimize chest wall injury and to isolate the benefit of avoidance of pleural violation.
Footnotes
Acknowledgements
Dr Subhapradha Anand contributed to chest radiographic interpretation.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
