Abstract
Background:
Esophagectomy is associated with pulmonary complications. This study evaluated if thoracoscopic minimally invasive esophagectomy (MIE) mitigates these risks by comparing pulmonary findings on postoperative computed tomography (CT) between open esophagectomy and MIE.
Method:
Postoperative (day 5) thoracic CT from 40 patients (20 open and 20 MIE) who had undergone esophagectomy with epidural analgesia and right-sided thoracic drainage were reviewed. On an axial view, the areas of atelectasis and pleural effusion were measured at 1 and 5 cm above the right diaphragmatic dome and at the level of the carina. In addition, the total distribution of atelectasis and pleural effusion was estimated on an ordinal scale (0–5), with ≥3 considered clinically important.
Results:
The groups were well-matched in terms of age, sex, and smoking status. There were no differences in the areas of atelectasis or pleural effusion for open surgery compared with MIE. The groups did not differ in the proportion of patients with clinically important atelectasis (right: 30% vs. 25%, left: 65% vs. 65%) or pleural effusion (right: 15% vs. 15%, left: 65% vs. 45%). More pleural effusion and atelectasis at the 1-cm level was present on the left side at day 5 in both open and MIE patients.
Conclusion:
Despite major differences in surgical trauma and ventilation strategies between open and MIE, CT evaluation at day 5 was surprisingly similar. Less right-sided pleural effusion demonstrates the effect of surgical drains. We believe that the defined levels of measurement used in this study, performed at clear anatomical landmarks, can be of value in future studies.
Keywords
Context and Relevance
No difference was seen in atelectasis and pleural effusion on postoperative (day 5) thoracic computed tomography between open and minimally invasive esophagectomy in this material. Previously described differences in postoperative pulmonary complications must be explained by other factors. The measurement at clear anatomical landmarks used in this study may be of value in future studies.
Introduction
Esophageal and gastroesophageal junction (GEJ) cancer is the seventh most common cancer and, because of its grim prognosis with less than one fifth of the patients surviving the first 5 years from diagnosis, it is the sixth leading cause of cancer death. 1 Despite recent advances in surgical care, 2 esophagectomy results in postoperative complications in more than half of the patients, with pulmonary pathology being most common. 3 Hybrid minimally invasive approach has reduced pulmonary complications in trial settings.4,5 These findings have, however, not always been demonstrated outside of the controlled environment of clinical trials,6,7 and concerns of too hasty introduction of minimally invasive approach in routine care have also been raised. 8
After esophageal resection, a gastric conduit is anastomosed to the remaining part of the esophagus, either intrathoracically (Ivor Lewis esophagectomy) 9 or on the neck (McKeowns three-field esophagectomy). 10 During the thoracotomy part of open procedures, the right lung is purposely collapsed to give room for the surgeon, influencing lung pathophysiology in several ways despite efforts to use protective ventilation techniques. 11 Single-lung ventilation also exposes the isolated lung to additional inflammation, besides direct surgical trauma, thus increasing the risk for developing lung injury and postoperative pulmonary complications.12,13 Based on the open techniques, minimally invasive esophagectomy (MIE) has been developed with a thoracoscopic approach, using four or five 12 mm trocars. With thoracoscopy in the prone position, first described by A. Cuscheiri et al, 14 single-lung ventilation can be avoided as the lung does not interfere with the operative field during capnothorax due to gravity. This is theoretically beneficial for the patient.
In modern healthcare, the diagnosis and assessment of pulmonary complications are commonly performed with computed tomography (CT) scan of the chest. We hypothesized that there would be radiological differences on postoperative CT regarding the amount of atelectasis and pleural effusion when comparing open surgery to MIE. We therefore aimed to perform detailed descriptions of said pathologies and compare the findings after open and MIE. As descriptions of postoperative radiologic appearance after esophagectomy are scarce, we also present a suggestion for standardized CT evaluation, based on clear anatomical landmarks, in this group of patients.
Methods
Forty patients who had undergone surgery for esophageal (ICD C15.0–15.9) or gastroesophageal junction cancer, Siewert I and II (ICD C16.0a and 16.0b), between 2008 and 2018, 20 with open technique and 20 with MIE, at a tertiary esophageal cancer center, were retrospectively identified and included in the study. Patients were excluded in cases of anastomotic leakage (10%) or symptomatic chyle leak (n = 0) 15 and if pre- or post-operative CT imaging could not be accessed. An identical posterolateral thoracotomy with single-lung ventilation was performed in the two open surgical procedures—Ivor Lewis esophagectomy with a circular stapled intrathoracic anastomosis and McKeown’s three-field esophagectomy with a handsewn left-side cervical anastomosis. The gastric conduit was prepared in a midline laparotomy. The minimally invasive approaches were performed by thoracoscopy in the prone position, with capnothorax and maintained double-lung ventilation, after preparing the gastric conduit by laparoscopy. In both procedures, routine (one or two) right-sided chest tubes were placed perioperatively. All patients received thoracic epidural analgesia, supplemented with systemic opioids as needed. During the postoperative phase, all patients were urged to early ambulation and instructed to use a handheld positive expiratory pressure device to promote mucus clearance. Nasal oxygen therapy was given when needed.
All patients had undergone a preoperative and a postoperative (day 5) CT thorax, performed with intravenous- and peroral contrast, according to clinical routine.
Baseline characteristics and perioperative data were collected from the patients’ electronic medical records. Pre- and post-operative (day 5) CT scans as well as postoperative chest X-rays were collected and anonymized on all patients. A consultant radiologist (A.T.) reviewed all examinations. The total lung volume was acquired using the entire CT scan volume with aid from a medical software system (AW server, G.E. Healthcare version 3.2 Ext 3.3). The areas of atelectasis and pleural effusion were outlined and measured bilaterally in three anatomically defined axial levels: 1 cm above the right diaphragm dome, 5 cm above the right diaphragm dome, and at the level of the carina. In addition, a sagittal reconstruction just lateral to the main bifurcation of the left and right pulmonary arteries respectively was used for the same measurements on all patients (Fig. 1 and Fig. 2). Finally, the total amount of atelectasis and pleural effusion was estimated on a six-graded scale (0–5). Software for measurements of affected areas was provided by Sectra ris/pacs version 22.1 and AW server, G.E. Healthcare version 3.2 Ext 3.3.
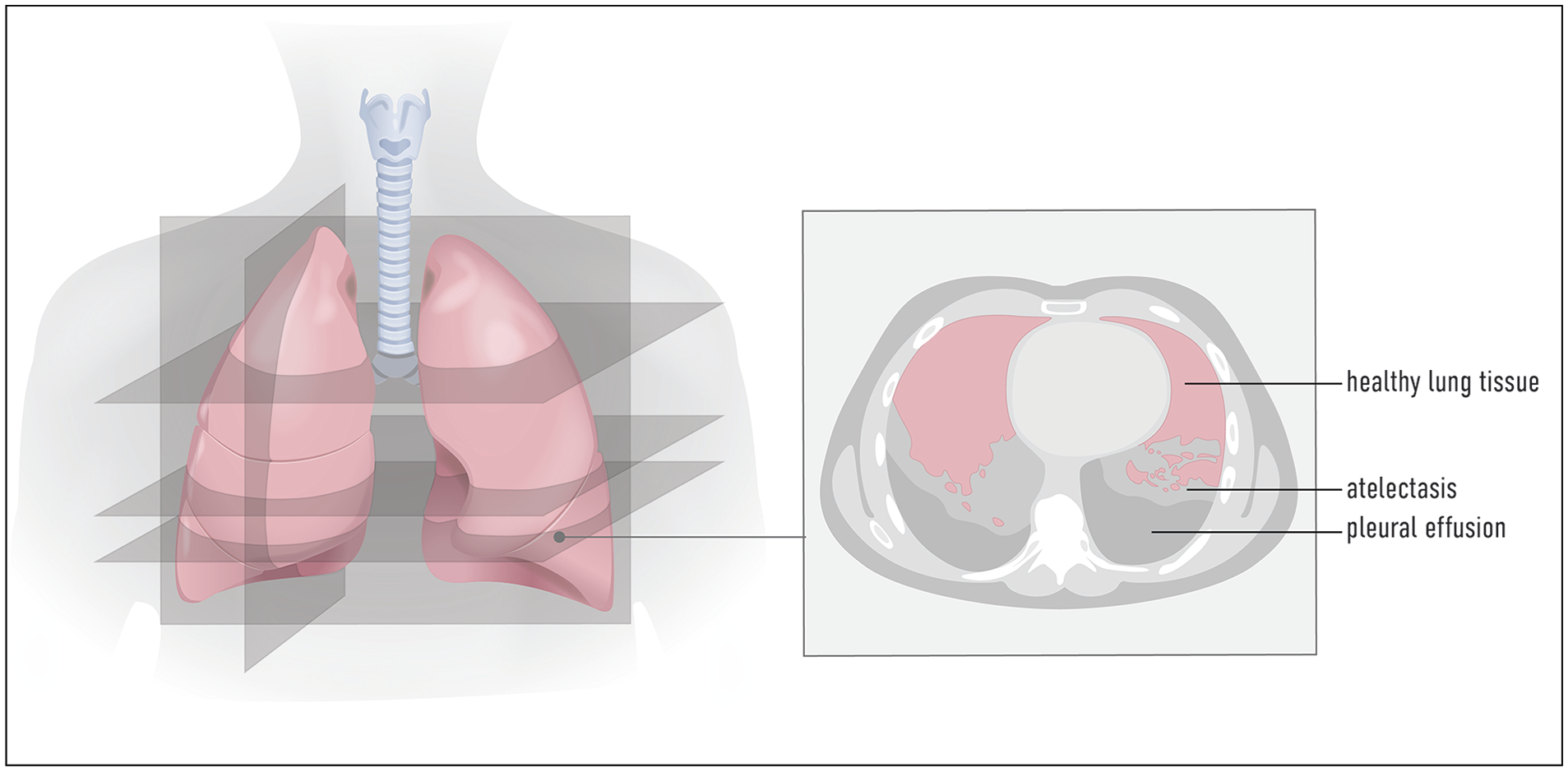
The three anatomically defined levels (at 1 and 5 cm above the right diaphragm dome and the level of the carina) used for delineation and area calculation of postoperative atelectasis (in light gray) and pleural effusion (in gray) after esophagectomy.
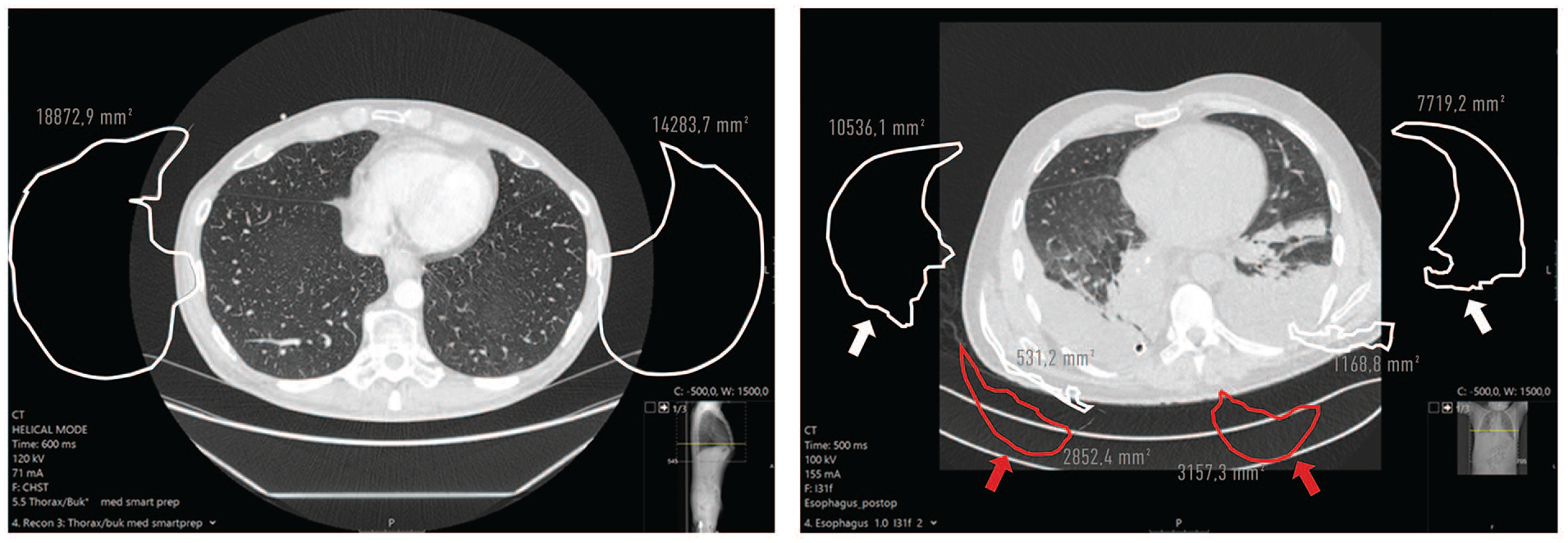
(A) Preoperative axial CT scan with normal lung parenchyma outlined. (B) Postoperative (day 5) axial CT scan 1 cm above the right diaphragm dome. Aerated lung tissue (white arrows) , dorsal pleural effusions (red arrows) and atelectasis (white outline, no arrow) are outlined.
Statistical analysis
Descriptive statistics were used to analyze baseline characteristics and perioperative data by using Fisher’s exact test for categorical variables and Mann–Whitney test for continuous variables. Differences in areas of atelectasis and pleural effusion in cm2 were analyzed using the Mann–Whitney test. Fisher’s exact test was used when analyzing differences in the estimated total amount of atelectasis and pleural effusion. In a preliminary analysis, between 5% and 30% of the lung area 1 cm above the diaphragm was represented by atelectatic lung tissue. A clinically relevant reduction in atelectasis area was set to 10 percentage units in MIE compared with open surgery. With a power level (beta) of 80% and a significance level (alfa) of 5%, this yielded a sample size of 32 patients (16 in each group). To account for this and a margin for logistical and technical issues, 20 patients of each operation type were reviewed. All statistical analysis was done using Stata® version 15.1 (Stata Corp, College Station, Texas, USA).
Ethics
The study was conducted in accordance with the Declaration of Helsinki (as revised in 2013). The study was approved by the regional ethics board of Uppsala (No. 2018/466 and 2019-04615), and individual consent for this retrospective analysis was waived.
Results
Both groups included 20 patients (18 men and 2 women in both groups) with 85% having adenocarcinoma and 15% squamous cell carcinoma. The groups (open vs. MIE) were well-matched in terms of age (65 years vs. 69 years), smoking status (50% vs. 60% previous/active smokers), body mass index (BMI; 27 kg/m2 vs. 25 kg/m2). The preoperative percentage of forced vital capacity (FEV1%) did not differ significantly between the patients having open or MIE (90% vs. 80%, p = 0.28). The open surgery group had a longer median duration of surgery (309 min vs. 276 min, p = 0.02) as well as length of stay (14 days vs. 10 days, p < 0.001). All patients were extubated before leaving the operating room, and none needed mechanical ventilation or intensive care postoperatively. There was no statistically significant difference in the number of days with oxygen treatment (6 days vs. 6 days, p = 0.32), nor the number of patients treated with antibiotics postoperatively (25% vs. 30%, p = 1.00). There was a statistically significant difference in preoperative oncological treatment, with neoadjuvant chemotherapy being more common in the MIE group (5% vs. 50%, p = 0.02) (Table 1).
Baseline data on patients with esophageal or GEJ cancer who had undergone esophagectomy grouped by type of surgery.
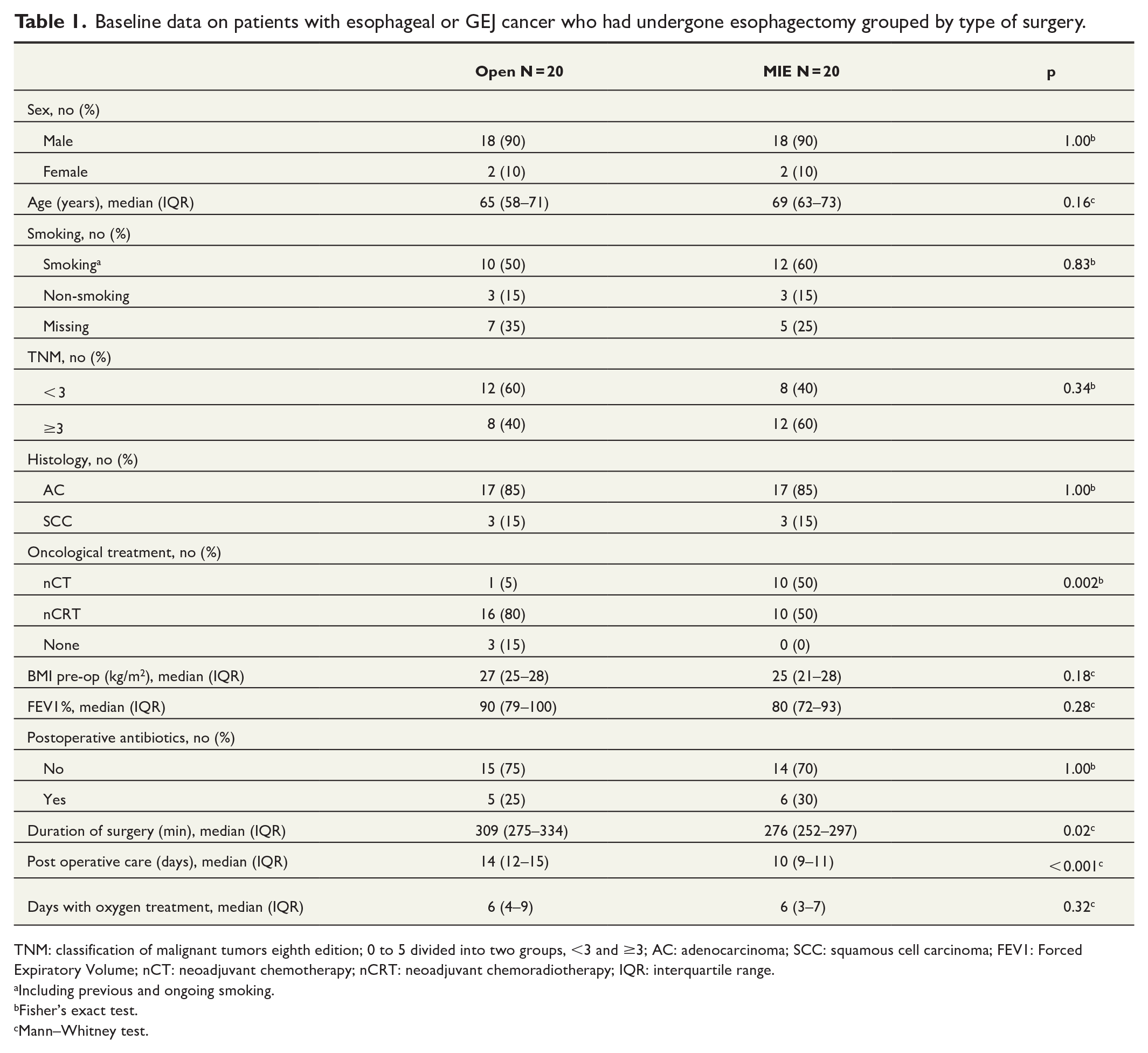
TNM: classification of malignant tumors eighth edition; 0 to 5 divided into two groups, <3 and ≥3; AC: adenocarcinoma; SCC: squamous cell carcinoma; FEV1: Forced Expiratory Volume; nCT: neoadjuvant chemotherapy; nCRT: neoadjuvant chemoradiotherapy; IQR: interquartile range.
Including previous and ongoing smoking.
Fisher’s exact test.
Mann–Whitney test.
As demonstrated in Table 2, there were no significant differences in atelectasis or pleural effusion between open and minimally invasive surgery. On postoperative day 5, the chest tubes had been extracted in two patients (one open and one MIE). No chest tubes were seen in the left hemithorax and more pleural effusion as well as atelectasis on a 1-cm level was present on the left side at day 5. In the postoperative CT scan, the occurrence of pneumomediastinum was similar between the two groups (5 vs. 4 patients). A trend toward noticing intra-abdominal gas more frequently (13 vs. 7 patients, p = 0.11) was seen after open surgery, although the difference was not statistically significant. The volume of the gastric conduit, placed anatomically in the dorsal mediastinum, was somewhat larger after open surgery compared to MIE (294 mL vs. 208 mL p = 0.01).
Post operative data on patients with esophageal or GEJ cancer who had undergone esophagectomy grouped by type of surgery.
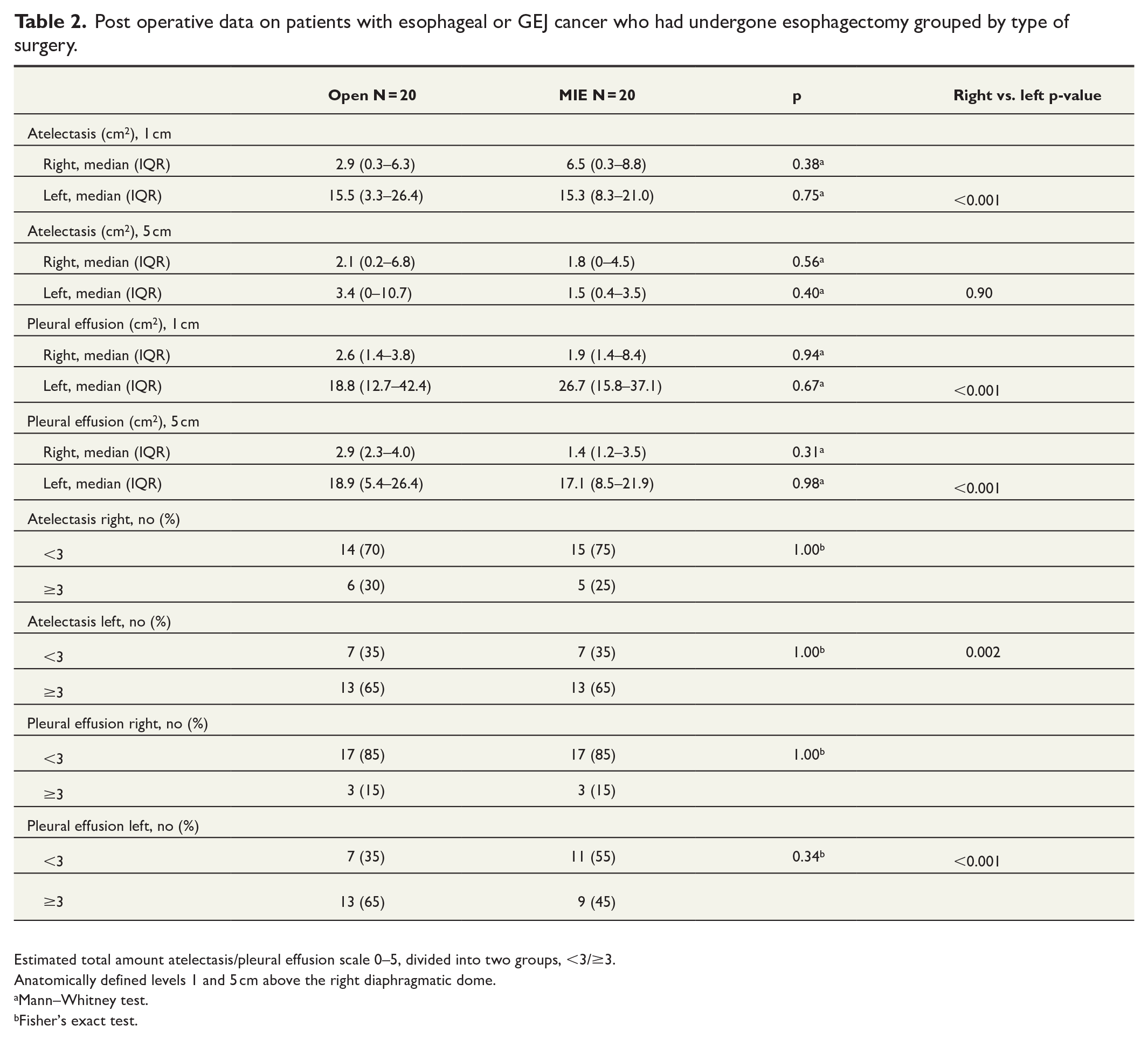
Estimated total amount atelectasis/pleural effusion scale 0–5, divided into two groups, <3/≥3.
Anatomically defined levels 1 and 5 cm above the right diaphragmatic dome.
Mann–Whitney test.
Fisher’s exact test.
Discussion
The postoperative radiologic appearance in esophagectomy is not previously described in detail. We present CT data, performed at three clear anatomical landmarks, after open and MIE. The possible clinical advantages with the minimally invasive approach were not reflected in the radiology appraisal in this study.
Esophagectomy is a complex and high-risk procedure, resulting in postoperative complications for more than half of the patients. These can be fatal and can also adversely impact the quality of life as long as 10 years after the operation. 16 Pulmonary complications are the most frequent postoperative complications for this intervention. 17 Previous studies investigating open versus minimally invasive approaches have shown conflicting results regarding postoperative pulmonary complications. A study by Briez et al. showed a lower frequency of major pulmonary complications including acute respiratory distress syndrome (ARDS) in patients who had undergone a hybrid minimally invasive (laparoscopy and open thoracotomy) approach compared with open surgery. The authors also declared less postoperative atelectasis in the hybrid MIE group. 5 However, in a later prospective randomized controlled trial from the same group, there was no difference in respiratory failure or ARDS. 4 In a Japanese study by Sakamoto et al. 18 studying total MIE, the authors did not see any decrease in pulmonary complication, although a higher incidence of re-intubation was seen in the MIE group compared to open surgery. Finally, the multicenter randomized trial, the TIME trial, by Biere et al. 19 saw a reduction from 29% to 9% of patients affected by postoperative pneumonia in the MIE group compared to open surgery, although they did not see any difference in ARDS or respiratory failure. The results above have not been demonstrated outside of controlled trials, and lack of pre-defined registration of complication patterns has been suggested as an explanation for this. 20
Atelectasis is formed in dependent lung regions close to the diaphragm in most patients shortly after induction of general anesthesia.21,22 Factors related to the surgery procedure, such as patient positioning, single-lung ventilation, and postoperative pain, also contribute to lung collapse and the fact that atelectasis commonly persists postoperatively. 23 Atelectasis impairs blood oxygenation and constitute the pathophysiologic basis for the development of other postoperative complications.24,25
Patients undergoing open surgery with single-lung ventilation are known to be extra vulnerable to lung complications. 26 Single-lung ventilation increases the amount of atelectasis and pleural effusion on the side of the temporarily closed lung. 27 As single-lung ventilation was applied only in the present open surgery group, we were surprised not to see an increased amount of atelectasis and pleural effusion in this group. How can this be explained? It is possible that although patients undergoing MIE with thoracoscopy have both lungs ventilated during the whole operation, the right lung is still partially collapsed because of the contra-pressure by the capnothorax used in order to access the dorsal mediastinal structures. Another possible factor contributing to our results is that the positive end-expiratory pressure (PEEP), used in almost all types of thoracoabdominal surgery, often needs to be lowered to facilitate for the surgeon. In addition, a degree of lung manipulation and compression is inevitable during thoracoscopic surgery.
All patients in this study received one or two 28–32 Fr drains in the right hemithorax according to the operative routine. There was a significant difference between the right and left hemithorax in total atelectasis and pleural effusion. The difference affirms the efficacy of thoracic drainage, and the finding of more pleural fluid in the left hemithorax raises the question of routine postoperative drainage of the contralateral side. A recent meta-analysis by Bull et al. 28 discloses that most centers routinely place one or two drains on the right or left hemithorax, even though this might increase postoperative pain and has limited evidence for efficacy. In this study, there was a significant difference in the number of drains remaining on the postoperative day 5 CT, with two drains only appearing in the open surgery patients. This might affect the result but is unavoidable in a retrospective study, as both MIE and the use of one chest tubes have increased over time. 28 Finally, in patients undergoing esophagectomy, the use of thoracic drainage might also be purposeful for surveillance of anastomotic or chyle leakage, not examined in this study.
Patients with anastomotic leakage were excluded from this study. As the main aim was to investigate the difference between thoracotomy and thoracoscopy, and an anastomotic leak will undoubtedly obscure any such difference. Postoperative complications affecting the lungs, such as pneumonia, pneumothorax, ARDS, pleural effusion, and pulmonary oedema- or embolism are more common than anastomosis leakage, and we aimed to investigate as normal postoperative radiology as possible. Interestingly, mediastinal gas was found in one quarter of the patients at day 5 of a normal postoperative course, as was intra-abdominal gas in half of the patients. No significant differences were found between the groups in this regard.
The shorter operation time and length of stay after minimally invasive surgery compared to open was expected and in line with previous reports.29,30 As in many previous studies comparing open esophagectomy with MIE, the continuous strive to shorten postoperative length of stay can be a bias, as MIE was being practiced more in later years.
It would have been optimal to calculate the total volume of both atelectasis and pleural effusion. This was, however, very difficult due to the complicated geometry. Furthermore, the two entities influence each other, as increasing pleural effusion results in increased risk of atelectasis. The three anatomically defined landmarks, that is, 1 and 5 cm above the right diaphragm dome and the level of the carina, were, however, easy to use for delineation as well as in repeated measurements. We hope that this robust and simple approach can be of value for others.
Limitations
The sample size of 40 patients, 20 in each group, was chosen to investigate what we believed to be a clinically relevant reduction in atelectasis area of 10 percentage units for the MIE group. This sample size might, however, have been too small to reveal smaller differences between the open and minimally invasive approach. We do not believe that the non-significant difference in tumor stage between open and MIE (lower tumor stage in the open group) impacts the results, as the same resectional procedure was performed in all patients, without any extra-esophageal resections (such as lung or pericardial resections). Our study demonstrates reduced atelectasis and pleural effusion in the drained hemithorax compared to the other; however, we did not have access to data concerning postoperative pain and discomfort due to the thoracic drainages.
Conclusion
In this comparative study of open surgery and MIE, there were no differences in atelectasis or pleural effusion on postoperative (day 5) thoracic CT. The differences in postoperative lung complications between open esophagectomy and MIE, presented in other studies, must be explained by other factors. We believe that the defined levels of measurement used in this study, situated at clear anatomical landmarks, can be of value in future studies.
Footnotes
Data availability
The data supporting this study are not publicly available.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Swedish Cancer Society (CAN 2017/1086, J.H.) and Bengt Ihre Foundation (Ihre project support 2022, J.H.).
Informed consent
Individual consent was waived by the ethical review board (no. 2018/466 and 2019-04615).
