Abstract
Background and Aims:
Surgical site infections are relatively common after spinal deformity surgery. Early detection of deep wound infections is important, since it may allow retention of spinal instrumentation. However, serum C-reactive protein and erythrocyte sedimentation rate may remain elevated for almost 6 weeks, making differential diagnosis of systemic inflammatory response and acute deep bacterial wound infection difficult. Plasma procalcitonin has been suggested to be a useful indicator for bacterial infection. However, there are no studies evaluating behavior of procalcitonin in patients undergoing major spine surgery with instrumentation.
Materials and Methods:
A total of 50 consecutive adolescents (37 idiopathic scoliosis and 13 neuromuscular scoliosis, mean age = 15 years at surgery and follow-up time = 21 months (range = 12–29 months)) undergoing scoliosis surgery participated in this prospective follow-up study. White blood cell count, serum C-reactive protein, and plasma procalcitonin levels were measured on the day before surgery, on the day of surgery, and daily thereafter for 1 week. None of the patients developed signs of acute or delayed wound infection during the follow-up period; however, two neuromuscular scoliosis patients developed severe postoperative pneumonia, and their inflammatory parameter data will be reported separately.
Results:
Plasma procalcitonin levels peaked on the first postoperative day (mean = 0.19 ng/mL, range = 0.04–1.29 ng/mL), and mean values were less than 0.5 ng/mL during the whole first postoperative week, while C-reactive protein remained elevated during the whole first postoperative week (highest mean value = 63.8 mg/L (range = 5–248 mg/L) on third postoperative day). Patients with idiopathic scoliosis had lower C-reactive protein levels (p < 0.05 from first to sixth postoperative day) and lower procalcitonin levels (p < 0.05 from third to seventh postoperative day) than neuromuscular scoliosis patients. Two patients with postoperative pneumonia showed elevated procalcitonin values over the whole postoperative week (22.34 ng/mL and 0.72 ng/mL highest values, respectively).
Conclusions:
Elevated plasma procalcitonin levels seem useful when excluding acute deep wound infection from systemic inflammatory response.
Keywords
Introduction
Surgical site infections are relatively common after spinal deformity surgery affecting from 0.9% to 11.9% of pediatric patients (1, 2). Similarly, systemic inflammatory response is common after major surgery, and it can be difficult to differentiate between bacterial infection and inflammation (3). The clinical signs of acute deep wound infections (within 6 weeks) after spinal instrumentation include fever, pain, swelling, erythema, and wound drainage or discharge (4). It is important to detect deep wound infections as soon as possible because early, aggressive debridement and irrigation with appropriate antibiotics may allow preservation of instrumentation and successful fusion in most of the cases when the delay from the initial surgery is less than 90 days (5, 6). Biomarkers, such as elevated serum C-reactive protein (CRP), white blood cell (WBC) counts, and erythrocyte sedimentation rate (ESR) can assist in diagnosing deep subfascial infection, but often are not diagnostic. In adults with instrumented spinal fusion, CRP peaks at second postoperative day, and it may remain elevated for almost 6 weeks making differential diagnosis of systemic inflammatory response and deep surgical wound infection difficult (7–10). Similarly, ESR peaks on fourth postoperative day and may remain elevated for 6 weeks after instrumented spine surgery.
Plasma procalcitonin (PCT) has been suggested to be a useful indicator for bacterial infection. The normal level of PCT in noninfected persons is under 0.05 ng/mL, and a cutoff point of 0.5 ng/mL has been proposed to distinguish between viral and bacterial infection (11–14). This substance increases within 6–12 h after onset of bacterial infection and halves daily when infection is controlled (13). In children and adult patients undergoing cardiac surgery, PCT has been reported to peak within 24 h and return to normal levels within the first postoperative week (3, 14). PCT has been reported to identify patients developing infectious complications with a high negative predictive value (15). Bottner et al. found that PCT has high specificity (0.98) identifying peri-prosthetic infection (16). PCT has also been reported to guide antibiotic treatment in patients with cardiac surgery when having PCT values less than 0.5 ng/mL (17–19). Thus, PCT might be suitable for differentiating the systemic inflammatory response from acute deep bacterial wound infection in patients undergoing instrumented spinal fusion.
No studies thus far have evaluated behavior of plasma PCT in patients undergoing spine surgery with or without instrumentation. We aimed to describe the immediate postoperative behavior of PCT in children and adolescents undergoing spinal deformity surgery and to compare it with the traditional markers of infection and inflammatory reaction (CRP, WBC, and fever).
Materials and Methods
A total of 50 consecutive children and adolescents (mean age at surgery = 15 years; 37 with adolescent idiopathic scoliosis (AIS) and 13 with neuromuscular scoliosis (NMS)) undergoing major spine surgery with instrumentation participated into this prospective follow-up study (Table 1). The mean follow-up time was 21 months (range = 12–29 months). None of the patients developed signs of acute or delayed wound infection during the follow-up period, but two patients with NMS developed postoperative pneumonia, and their results for inflammatory parameters will be reported separately. A total of 45 patients underwent all posterior surgery with pedicle screws, and 5 patients with idiopathic thoracolumbar Lenke 5C curves had anterior approach and anterior instrumentation only.
Clinical characteristics and perioperative data of the study groups.
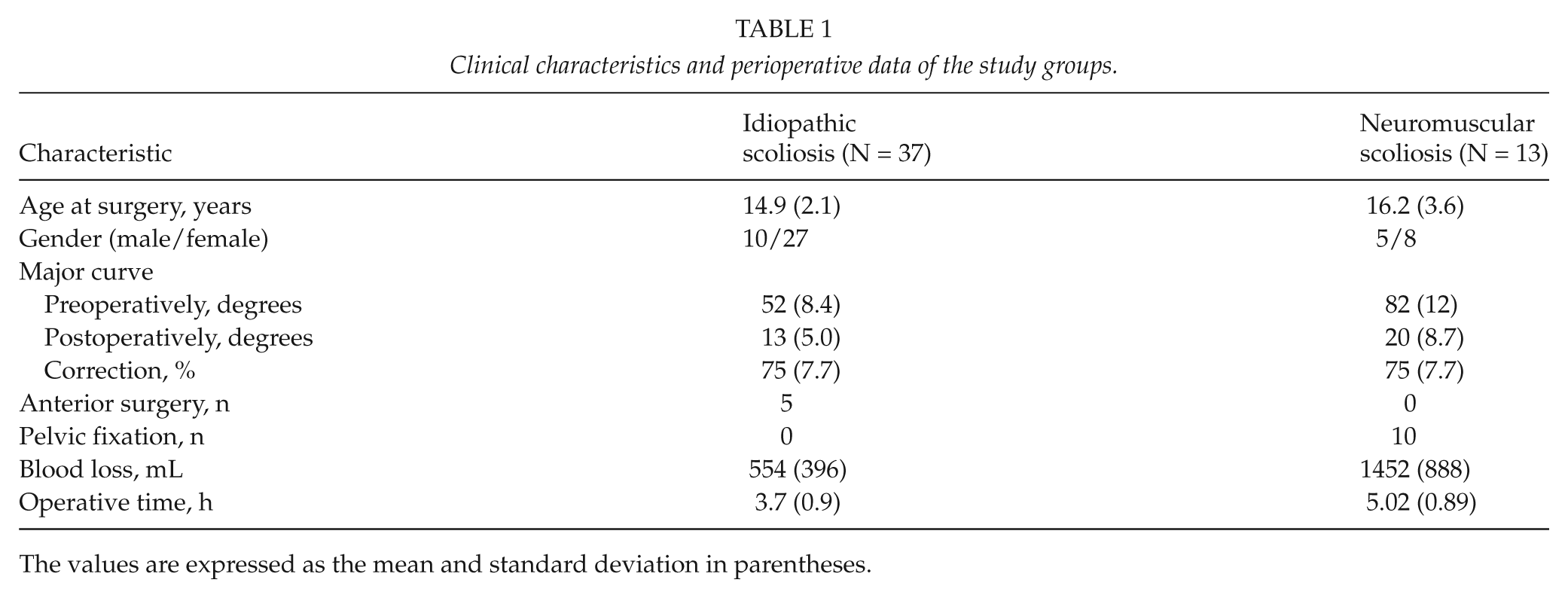
The values are expressed as the mean and standard deviation in parentheses.
Study Design
The study was a prospective follow-up study. Clinical medical records and radiographs of the spine were prospectively recorded. Follow-up examinations were performed by one of the two orthopedic spine surgeons (I.H. and O.P.) before surgery and on the day when the patient was discharged from the hospital. All patients were operated by one of the two orthopedic spine surgeons. The indication for scoliosis surgery was an idiopathic scoliosis of 45° or more or a progressive NMS of 50° or more. Pelvic fixation for NMS was performed when L5 tilt over S1 endplate was over 10°.
The WBC (/mm3), serum CRP (mg/L), and plasma PCT levels (ng/mL) were measured on the day before surgery, on the day of surgery, and daily thereafter for 1 week postoperatively. CRP and PCT concentrations and WBC counts were determined by the central laboratory of our hospital. WBC counts were determined by Sysmex automated analyzer (Sysmex Corporation, Kobe, Japan). CRP levels were determined by immunoturbidimetric assay by Modular P800 automatic analyzer (Roche Diagnostics GmbH, Mannheim, Germany). PCT levels were determined by immunochemiluminometric assay (ECLIA) by Modular E180 automatic analyzer (Roche Diagnostics GmbH, Mannheim, Germany).
Standing or sitting posteroanterior and side radiographs of the whole spine (scoliosis) were taken preoperatively, immediately after surgery on table, and during ward period. One independent observer (educated research nurse K.M.) measured all radiographs. For idiopathic scoliosis patients bending radiographs and for NMS patients spinal radiographs under traction were obtained. Additionally, NMS patients had a full medical evaluation by a consultant pediatrician.
Prophylactic Antibiotics and Operative Techniques
All patients received a single dose of intravenous cefuroxime (60 mg/kg, maximum dose 3 g), which was continued for the next 48 h at a dose of 30 mg/kg (max. dose 1.5 g) three times daily. Additionally, a single dose of intravenous vancomycin (10 mg/kg, max. dose 1 g) was given for all patients.
In the posteriorly operated patients, the patient is turned prone, and posterior elements of the spine are exposed carefully with electrocautery. A total of 11 (30%) idiopathic patients and 11 (85%) neuromuscular patients underwent apical Smith-Petersen osteotomies (Ponte procedure) (p = 0.0073). A total of 12 NMS patients received morselized allogeneic bone graft.
Pedicle screws were inserted with the free hand technique based on posterior bony elements according to Kim et al. (20). Multiaxial reduction screws were used at the apical concave side. Vertebral column derotation was performed using the vertebral column manipulator device. Iliac fixation (N = 10) was performed with long iliac screws and connectors. Spinal deformity correction is obtained by double rod cantilever maneuver or concave rod derotation. A closed suction drain was used in all patients for the first 24 h.
The anterior approach was a thoraco-abdominal for the Th11-L3 area. The patient is laid in a lateral decubitus position. Segmental vessels in the anterior fusion area were ligated unilaterally to allow wide and safe exposure of the spine including anterior longitudinal ligament. Multilevel discectomies were performed. Anterior spinal fusion was obtained through autologous rib grafting of the disk spaces. Then anterior instrumentation with plates and two screws for each vertebra from proximal end to distal end vertebra were carried out.
All patients were carefully followed up at the pediatric intensive care unit with a mean arterial pressure of 70 mmHg or more during the first 24 h. Lower leg movement was checked every 4 h. All patients were mobilized within the first postoperative week.
Statistical Analysis
Values are given as means, standard deviations (SD), and ranges. A two-tailed independent T test was used to calculate the level of significance for continuous variables with Bonferroni correction and χ2-test for categorical variables. P values equal to or below 0.05 were considered statistically significant.
Ethical Aspects
We obtained permission to perform this study from the Ethics Committee of the hospital where the study was conducted. Since all data gathered were part of the patients’ normal care, informed consent was not obtained.
Results
Radiographic correction and perioperative data
Major curve (MC) averaged preoperatively 59° (40°–105°) and 14° (2°–38°) at postoperative radiographs (Table 1). MC correction averaged 75% (58%−96%). Blood loss averaged 792 mL (range = 95–3000 mL) with significantly higher blood loss for NMS than AIS patients (p = 0.002). The mean operative time was 4 h and 2 min, with significantly longer operation room (OR) time for NMS than AIS (p < 0.001). None of these patients developed an acute (within 6 weeks of surgery) or delayed superficial or deep wound infection.
Plasma PCT
Plasma PCT levels (mean = 0.14 ng/mL, range = 0.04–0.79 ng/mL) peaked on the first postoperative day in patients with AIS, and only one patient with AIS had a PCT level of 0.5 ng/mL or higher during the first postoperative week (Fig. 1). In patients with NMS without infection, PCT values (mean = 0.38 ng/mL, range = 0.04–1.29 ng/mL) peaked on the first postoperative day, and one patient had value 0.5 ng/mL or more during the first postoperative week. AIS patients had significantly lower PCT levels (p < 0.05 from third to seventh postoperative day) than NMS patients. The highest individual PCT value was 0.79 ng/mL for AIS patients and 1.4 ng/mL for NMS patients without clinical signs of any bacterial infection or prolonged intubation. PCT remained elevated (22.34 ng/mL and 0.72 ng/mL highest values) on the two NMS patients, who developed a severe postoperative pneumonia (Fig. 1).
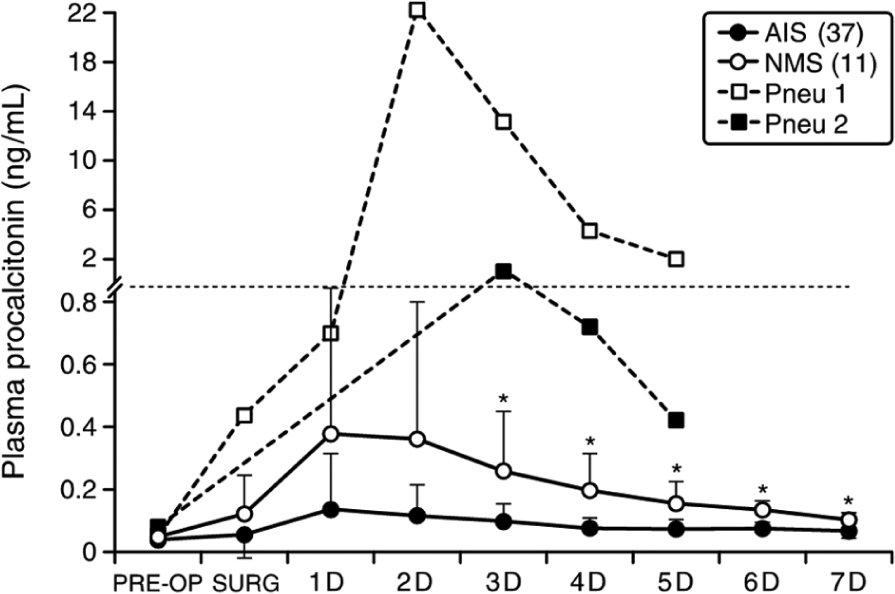
Plasma procalcitonin levels (mean ± SD) in patients with idiopathic scoliosis and in patients with neuromuscular scoliosis.
Serum CRP
In contrast, serum CRP remained elevated during the whole first postoperative week with mean peak values on the third postoperative day (in AIS, mean = 44 mg/L, range = 5–135 mg/L; in NMS, 123 mg/L, range = 11–248 mg/L) (Fig. 2). In two neuromuscular patients with severe postoperative pneumonia, the highest CRP value was also noticed on the third postoperative day (149 mg/L or 395 mg/L). Patients with AIS had a significantly lower CRP levels than patients with NMS (p < 0.05 from first to sixth postoperative day). The highest individual CRP value was 135 mg/L for AIS patients and 248 mg/L for NMS patients, without clinical signs of any bacterial infection or prolonged intubation.
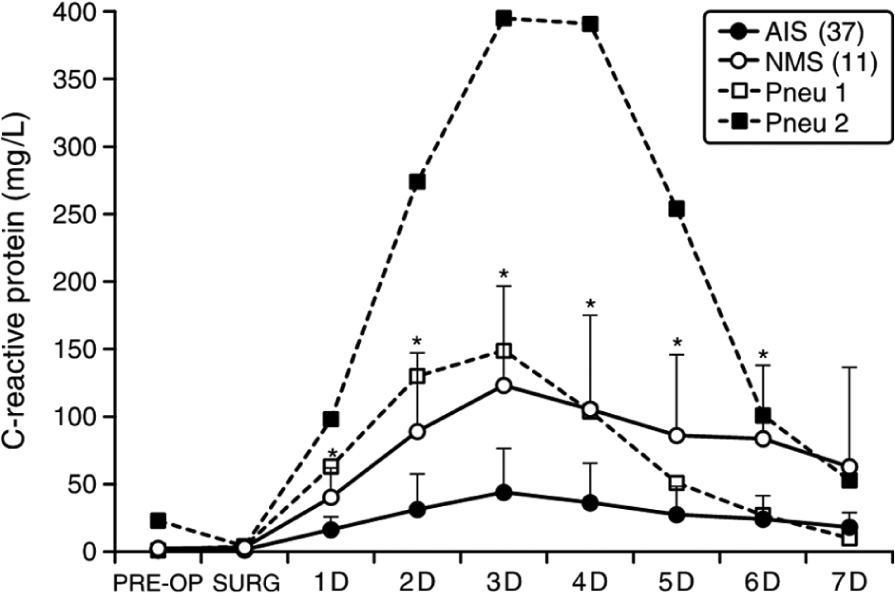
Serum C-reactive protein (mean ± SD) in patients with idiopathic scoliosis and in patients with neuromuscular scoliosis.
WBC Counts
WBC elevated with peak values on the day of surgery (NMS) (mean = 12,980/mm3 (range = 8200–20,700/mm3)) or first postoperative day (AIS) (mean = 16,646/mm3 (range = 9700–26,900/mm3)) and remained slightly elevated during the first postoperative week (Fig. 3). In two neuromuscular patients with postoperative pneumonia, the WBC count was similar compared to other NMS patients.
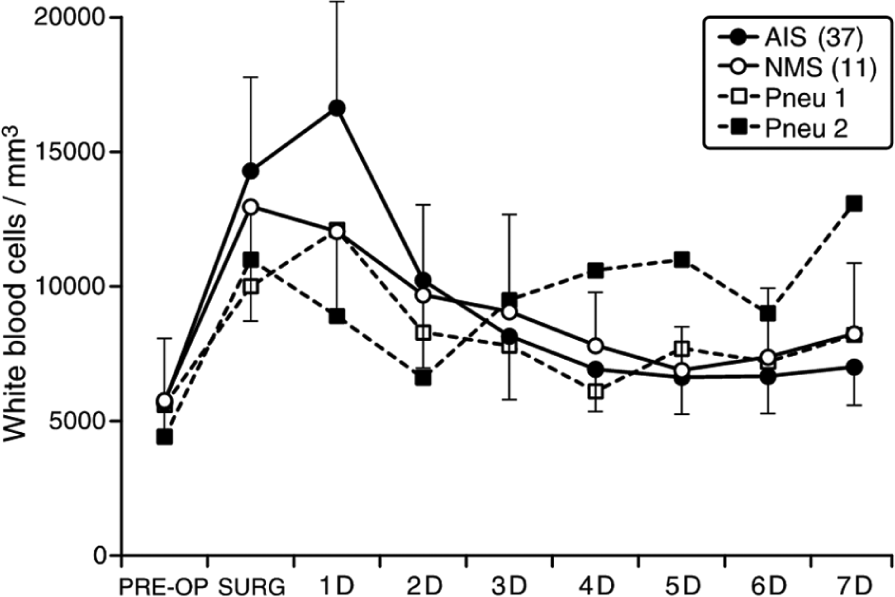
White blood cell count (mean ± SD) in patients with idiopathic scoliosis and in patients with neuromuscular scoliosis.
Patients with idiopathic scoliosis became afebrile during first postoperative week, while patients with NMS had slightly elevated body temperature during the whole first postoperative week.
Discussion
Deep wound infection is a serious complication of spinal deformity surgery affecting up to 12% of children with NMS (1, 2). Acute wound infections are classified as infections detected within 6 weeks of surgery. Early detection of deep wound infection is clinically of outmost importance, since it may allow retention of spinal instrumentation with irrigation and debridement only, and patients can expect similar clinical outcome compared to patients without infection (5, 6). Major spine surgery induces a systemic inflammatory response as evidenced by fever and highly elevated acute phase proteins such as CRP and PCT. Based on clinical findings, it is often impossible to discriminate between acute bacterial infection and systemic inflammatory reaction. This may result into inadvertent long-term (over 48 h) intravenous antibiotic treatment, although studies have suggested that 24-h prophylactic antibiotic treatment should be enough for spinal deformity surgery (21).
CRP has been reported to be elevated up to 6 weeks after surgery, with peak values after second or third postoperative day and with higher values after lumbar spine instrumented fusion than after minimally invasive surgery (7–10). The longevity of elevated CRP values means that usefulness of CRP in the detection for acute deep wound infection remains poor. To the best of the authors’ knowledge, no previous study has evaluated the behavior of plasma PCT in patients undergoing spinal deformity surgery. In patients undergoing cardiac surgery, peak PCT levels are reached within 24 h (3, 11). In this patient population, PCT values have been used to stop postoperative antibiotic treatment, when PCT values have been less than 0.5 ng/mL. Arkader et al. reported that PCT was able to differentiate between cardiopulmonary bypass-initiated systemic inflammatory response syndrome and confirmed bacterial sepsis in children (14). Bottner et al. compared interleukin-6 (IL-6), PCT, and tumor necrosis factor (TNF)-α as markers of peri-prosthetic infection. They found that the specificity of PCT was 0.98. They concluded that PCT may be useful in further assessment of patients with positive CRP and elevated levels of IL-6 (16). PCT has been studied also in critically ill sepsis patients. In a recent meta-analysis, PCT was found to have a mean sensitivity of 0.77 and specificity of 0.79 for sepsis patients, and it was concluded that it is a helpful biomarker for early diagnosis of sepsis, but must be interpreted carefully in the context of medical history, physical examination, and microbiological assessment (22). In this study, 50 consecutive adolescents undergoing scoliosis surgery were evaluated. PCT levels peaked on the first postoperative day, with only one AIS patient having a mildly elevated value of 0.79 ng/mL during the first postoperative week. In patients with NMS, the postoperative course of PCT is slightly more elevated, but also the surgical trauma (blood loss, levels fused, and time of surgery) is more severe. The 24-h half-life of PCT is also clinically very important because it allows the clinician to recognize whether antibacterial treatment is effective (13). If PCT value will not reduce by 50% during the first 24-h treatment period, the effectiveness of antimicrobial treatment must be reevaluated. It remains to be seen whether PCT is a useful marker also for delayed deep wound infections caused by low-virulent bacteria. None of our patients developed delayed deep wound infection during the follow-up period.
The weakness of this study is that none of the patients developed deep wound infection; therefore, comparison between infection and noninfection groups could not be performed. PCT remained elevated only on the two NMS patients, who developed a severe postoperative pneumonia. To observe a clinically significant effect size (1.0), it would require a sample size of 16 patients per group. In our hospital, this would require all pediatric spine patients over 8 years given the relatively low deep wound infection rate in our unit.
In conclusion, we assessed the normal postoperative course of PCT after spinal deformity surgery. Our findings support the hypothesis that significantly elevated PCT levels (0.5 ng/mL or more) would be a useful marker for bacterial infection, and additionally the favorable kinetics of this prohormone supports its use as an diagnostic aid for acute deep wound or other bacterial infections after spinal deformity surgery. Elevated PCT levels could, therefore, be used in differentiating inflammatory response from acute deep wound infection clinically significantly earlier than using CRP values: If plasma PCT is elevated postoperatively, bacterial infection (pneumonia, urinary tract infection, and deep wound infection) should actively be searched for. On the contrary, repeatedly low plasma PCT reliably excludes bacterial infection after spinal deformity surgery in adolescents. However, the values of PCT must be interpreted in the context of patient’s medical history and physical examination.
Footnotes
Acknowledgements
The authors thank Sanna Vilo for her help planning this study.
Declaration of Conflicting Interests
The present study was funded by grants from the University Hospital, Foundations, and Industry. For this particular study, none of these had a role in study design, writing or conclusions. The funds were used only for salaries of the researchers and research nurses.
Funding
J.S. has been supported by the Turku University Central Hospital foundation. J.S. is a research fellow, O.P. is a member, and I.H. is a leader of the Finnish Pediatric Orthopaedic Study Group, which has received funding from the Finnish Pediatric Research Foundation and Medtronic International.
