Abstract
Objective
To explore the durability of multi-drug antiretroviral regimens in treatment-experienced PWH.
Design
This retrospective observational study including PWH who started mega-ART regimens between 1 January 2009 and 31 December 2019, selected from the ARCA cohort.
Methods
Time-dependent events were analysed by Kaplan–Meier methods, while Cox regression models were used to define the predictors of mega-ART discontinuation.
Results
A total of 1,514 ART-experienced PWH were included. Over a median follow-up of 47 weeks (IQR 15–127), 1,299 (83%) mega-ART were interrupted, with an incidence of 85.62 per 100 person-years of follow-up. In the multivariable analysis, predictors of higher risk of mega-ART discontinuation were a higher number of antiretroviral drugs included in baseline regimens (aHR 1.206, CI 95% 1.016–1.431, p = .032) and a higher baseline HIV RNA log10 (aHR 1.113, CI 95% 1.048–1.181, p < .001); otherwise, shorter duration of previous ART was associated with a lower risk of discontinuation (aHR 0.982, CI 95% 0.965–0.999, p = .037). When mega-ART was stopped, 299 PWH (23%) had HIV RNA levels above 50 copies/ml, 16/299 (1%) had HIV RNA levels >50 copies/ml but less than 200 copies/ml, 792 PWH (61%) had HIV RNA levels below 50 copies/ml, and 208 PWH (16%) had an undetermined HIV RNA load.
Conclusions
Mega-ART was characterized by limited durability and poor virological success.
Introduction
Current international recommendations for people with HIV (PWH) who have experienced virologic failure recommend a therapeutic regimen including at least two, and optimally three, completely active drugs based on resistance testing, keeping the genetic barrier to resistance high.1,2 In the early 2000s, achieving and maintaining HIV RNA suppression in PWH who have experienced multiple virological failures and side effects from antiretroviral drugs was a difficult target. Different strategies were developed to overcome this situation, including the use of multi-drug regimens comprising more than 3 and up to 9 drugs, often referred to as mega antiretroviral therapy (mega-ART).3,4 Different antiretroviral drug combinations were investigated in this setting, for example, raltegravir plus maraviroc plus etravirine, 5 raltegravir plus etravirine plus darunavir/ritonavir;6,7 or elvitegravir/cobicistat/emtricitabine/tenofovir alafenamide plus darunavir, 8 or different combinations based on multiple classes of antiretrovirals.9–13 The Mega-ART strategy was based on the expectation that more antiretrovirals could better reduce HIV RNA and delay the selection of drug resistance.3,14,15 However, mega-ART could inherently increase toxicity and intolerance, promoting lower adherence and the subsequent discontinuation of treatment.16,17 Before the arrival of darunavir (DRV) and dolutegravir (DTG), salvage therapy was a challenging mission. The combination of high genetic barrier drugs increased the opportunity of rescuing PWH with multi-class resistance.18–21
Several studies have explored the use of mega-ART without demonstrating a benefit,22,23 for example, the OPTIMA trial, did not observe the prevention of AIDS-defining events or death from any cause in PWH with poor retreatment options receiving a mega-ART.24,25
This study aimed to explore the durability of the mega-ART in a large cohort in a real-life setting.
Methods
The Antiviral Response Cohort Analysis (ARCA) is a large Italian database collecting demographics, ART history, HIV genotypes, HIV RNA load, CD4+ cell measurements, and AIDS-defining events from >40,000 PWH since the early 1990s. The study protocol was approved by the local institutional ethics committees, and all patients signed an informed consent for the use of their clinical and laboratory data. The study was performed in accordance with the ethical standard of the 1964 Declaration of Helsinki and with the Good Clinical Practice guidelines of the International Conference on Harmonization. We extracted data from PWH aged >18 who started a mega-ART between 1 January 2009 and 31 December 2019 to include only currently used antiretroviral drugs. Mega-ART was defined as an ART including more than three drugs belonging to more than two different classes of antiretroviral drugs among nucleoside reverse transcriptase inhibitors (NRTI), non-nucleoside reverse transcriptase inhibitors (NNRTI), integrase strand transfer inhibitors (INSTI) and protease inhibitors (PI), entry inhibitors, fusion inhibitors, and monoclonal antibodies. We classified mega-ART into four different options as follows: (1) NRTI plus INSTI plus PI ± others; (2) NRTI plus PI plus others; (3) NRTI plus INSTI plus others; and (4) others (treatments including maraviroc, enfuvirtide, and ibalizumab).
The mega-ART discontinuation was defined as the simplification of a therapeutic regimen including three or fewer antiviral drugs, the intensification of a regimen with more antiretroviral drugs, death, or loss of follow-up (when HIV RNA load and CD4+ T cell count were missing for more than 1 year in the database). Boosted and non-boosted PI were considered the same drug. Intraclass switches, including changes between tenofovir disoproxil fumarate (TDF) and tenofovir alafenamide (TAF), abacavir (ABC), lamivudine (3 TC), emtricitabine (FTC), and between ritonavir and cobicistat, were not considered mega-ART discontinuations. The final dataset also included sociodemographic, epidemiology, behaviour, clinical and laboratory data, CDC stage, previous ART history, years of infection at the time of mega-ART start, viral subtype, nadir CD4+ T cell counts, zenith HIV RNA load, CD4+ T cell counts and HIV RNA load at the time of mega-ART start, cumulative exposition to different classes of antiretroviral drugs, last therapy before starting mega-ART, the time from viral load suppression (if viral load was undetectable at the time of starting mega-ART), HIV RNA genotype at the time of mega-ART start, and cumulative HIV RNA genotype, when multiple tests were available.
The cumulative genotypic sensitivity score (cGSS) was calculated individually only for the antiretrovirals used by each patient according to cumulative HIV RNA genotypes and based on the Stanford Inferred Drug Resistance Scores 26 A value of 1 was attributed to drugs with full activity, 0.75 for a low potential resistance level, 0.5 for a low resistance level, 0.25 for an intermediate resistance level, and 0 if the drug showed a lack of activity.
Statistical analysis
Descriptive statistics were used to describe the demographic characteristics of PWH. The durability of mega-ART was analysed by Kaplan–Meier (KM) curves up to 96 weeks from baseline, defined as the start of mega-ART. Univariable and multivariable analyses according to the Cox regression model were used to define the predictors of mega-ART discontinuation from a predefined list including age, gender, ethnicity, risk factor for HIV infection, CDC stage at baseline, time since first ART, time since HIV diagnosis, CD4+ T cell count nadir and HIV RNA load at zenith at the time of mega-ART start, HBsAg and HCV antibody status, type of mega-ART regimen and cGSS. The multivariable model included all factors retained from univariable analysis at a p-value of <0.05. Data were analysed using SPSS version 25 (IBM Corp., Armonk, NY).
Results
Patients’ characteristics at baseline.

ART: antiretroviral therapy; NRTI: nucleoside reverse transcriptase inhibitors; NNRTI: non-nucleoside reverse transcriptase inhibitors; PI: protease inhibitors; INSTI: integrase inhibitors; GSS: genotypic susceptibility score; AIDS: Acquired immune deficiency syndrome.

Antiretroviral combination in Mega-ART regimen. NRTI: nucleoside reverse transcriptase inhibitors; NNRTI: non-nucleoside reverse transcriptase inhibitors; PI: protease inhibitors; INSTI: integrase strand inhibitors.

Durability of Mega-ART regimens by Kaplan–Meier estimates (n = 1328).
Predictors of >3-drug-regimen interruption by univariate and multivariate Cox regression models.
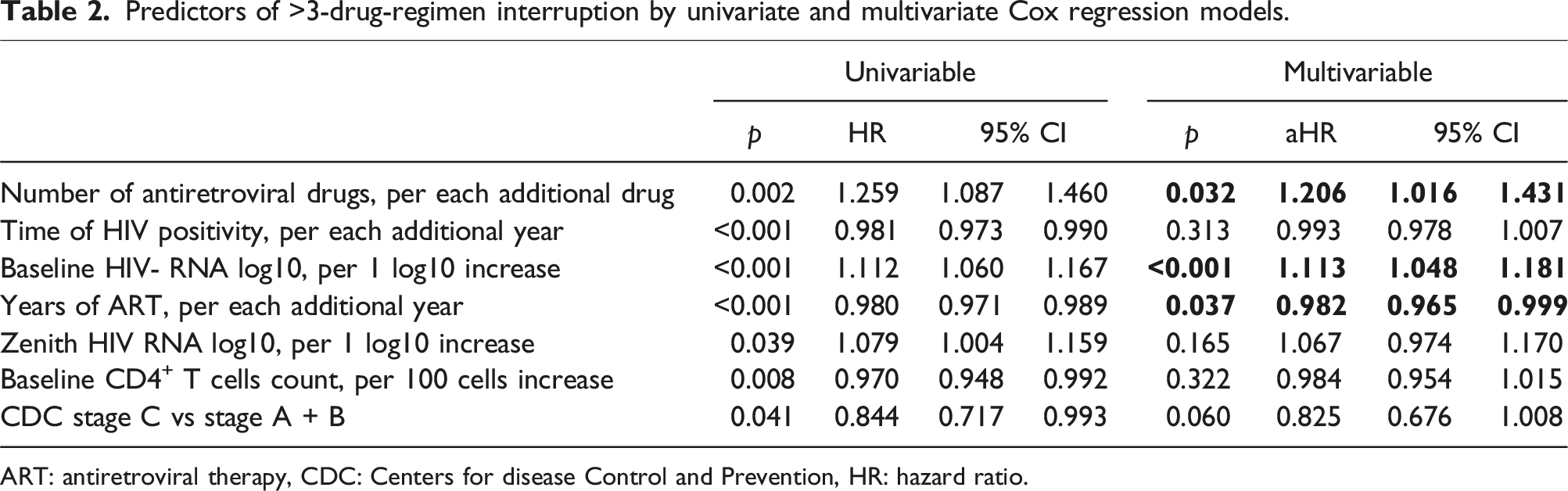
ART: antiretroviral therapy, CDC: Centers for disease Control and Prevention, HR: hazard ratio.

Durability of four drug regimens according to calendar period (n = 1328).
The proportion of prescribed mega-ART regimens compared to less than 3-drug regimens decreased from 6 to 1.3% from 2009 to 2019, respectively (data not shown). After discontinuation of mega-ART, data on transition to a new regimen were available in 619 (48%) of the cases: 205 (33%) had switched to a PI-based regimen, 143 (23%) to an INI-based regimen, 92 (15%) to both PI- and INI-based therapy, and 179 (29%) to other regimens.
Discussion
In our large database analysis, mega-ART represented a limited and decreasing percentage of treatments over the years. Interestingly, the ARCA database preferentially included PWH with available genotypes, providing the study with PWH more exposed to ART. However, the significant sample size allows us to study this strategy extensively. First, mega-ART includes a very large number of combinations of antiretroviral therapies. Often, these rescue combinations are not supported by adequate clinical trials.1,4,27 In the ARCA cohort, the choice of optimal ART for each PWH was at the physician’s discretion based on drug availability. Another interesting aspect observed was the duration of mega-ART. The use of this strategy was time limited. In part, as clearly evidenced by the poor virological success, but also because of the development of side effects to antiretrovirals, of which, unfortunately, we cannot specify the cause because ARCA does not accurately collect causes of discontinuation. In past years, obtaining and maintaining virologic suppression in particular cases of heavily treated persons or with multiple virologic failures has been a challenge for physicians, who were forced to use a mega-ART regimen. 28
Better performance of mega-ART regimens has been observed in recent years, with a significant decrease in their discontinuation. In particular, the combination of DRV/ritonavir plus DTG with a high genetic barrier and better affinity for target enzymes allows for simplified rescue therapy while maintaining high levels of efficacy. 29
Above all, the introduction of the second generation of INSTIs has also made it possible to reduce side effects and limit discontinuations. 30
Regarding the most widely used combination, it appears evident that PI and INSTI, administered with at least a nucleoside/nucleotide backbone, are the preferred choice. NRTIs are frequently included in mega-ART regimens, and there is some evidence that certain NRTIs retain activity against HIV even in the presence of resistance mutations.31–35 A further aspect concerns the virological success of mega-ART strategies. Virologic response to ART is sometimes limited in PWH with previous treatment and in whom viral suppression has not been achieved. This lack of efficacy is partly due to the extensive cross-resistance commonly observed within antiretroviral drug classes or adherence issues, which is particularly important in a population exposed to a high pill burden. 36
The goal of ART should always be to achieve undetectable HIV RNA load; if this cannot be done, the treatment plan should focus on maintaining immunologic function, preventing clinical progression, and minimizing the development of resistance that might jeopardize future treatments. 1
We cannot compare the results with other studies conducted in similar populations. Despite the heavy drug exposure, notably, over 40% of PWH had a GSS lower than 2, dramatically reducing the potential efficacy of the strategy of interest. In PWH with failure of the three classes of antiretrovirals, it is unlikely that long-term virological suppression can be achieved; therefore, achieving immunological success by increasing CD4+ T-cell counts is considered an important goal in these PWH, with the long-term goal of maintaining low mortality. Treatment with more drugs has been associated with better CD4+ T-cell count trends in some studies.37,38
Recovery of CD4+ counts is also a phenomenon highlighted in recent studies in highly treatment-experienced (HTE) populations.39,40 In our experience, CD4+ recovery occurs even in people with less CD4+ and as expected in people newly diagnosed with HIV.
Similar results were observed in our study. As observed in past studies, the factors that affected the discontinuation of the mega-ART regimen were adherence and the development of resistance, both of which were influenced by accumulated toxicity, ageing, comorbidities, polypharmacy, and high levels of baseline HIV RNA.41–43
Although the factors that led to the discontinuation of the ART regimen were not accurately stated, based on the reported HIV RNA load in our database, we can assume that adherence and the development of resistance affect the durability of mega-ART regimens.
An important feature was that the percentual of PWH with HBsAg positivity in this cohort was higher than observed in literature (17% vs 4%), so this could allow the need of an NRTI based regimen.44–46
Limitations
Our study had several limitations. First, due to the structure of the ARCA database, we are unable to define the cause of the discontinuation of antiretroviral therapy. Second, we had so much data missing due to the retrospective nature of the study and the fact that data on ARCA were entered voluntarily by different centers in Italy. Third, we had 44.8% of an ‘unconventional’ regimen that was not codified and not classifiable in the regimen described in the methods. Finally, we lacked data on mortality and AIDS-related events during the follow-up period. So, it is important to underline how difficult it was to classify the different therapeutic regimens, especially those used in an era when the association of antiretroviral drugs was often used without relying on registered studies but on clinical experience.
Strengths
The strengths of the study are mainly the large sample size analysed, the large number of centers involved nationwide in the real-life setting, and the wide calendar time span analysed. A further aspect concerns the virological success of mega-ART strategies. Virologic response to ART is sometimes limited in PWH with previous treatment and in whom viral suppression has not been achieved. This lack of efficacy is partly due to the widespread cross-resistance shown in antiretroviral drug classes or adherence issues, which is particularly important in a population exposed to a high number of antiretroviral regimens. 36
Conclusions
Mega-ART was characterized by limited durability and poor virological success that nowadays limit its use in PWH who have failed multiple ART regimens.
Although heavily treatment-experienced PWH are decreasing in prevalence, the construction of a tailored antiretroviral regimen remains one of the major challenges for HIV providers.
Footnotes
Acknowledgements
We thank all PWH who have signed the informed consent and the ARCA site investigator: Luca Butini (ANCONA-Immunologia Clinica); Romana del Gobbo (ANCONA-Malattie Infettive); Andrea Giacometti (ANCONA-Clinica di Malattie Infettive); Patrizia Bagnarelli (ANCONA-Virologia); Danilo Tacconi (AREZZO-Malattie Infettive); Annalisa Saracino (BARI-Clinica Malattie Infettive Università); Paola Laghetti (BARI-Virologia); Annapaola Callegaro (BERGAMO-Microbiologia e Virologia); Laura Comi (BERGAMO-Malattie Infettive); Alessia Zoncada (CREMONA-Malattie Infettive); Massimo Crotti (CREMONA-Immunoematologia e Medicina Trasfusionale); Vincenzo Mellace (CATANZARO-SERT Soverato); Surace/Latella (CATANZARO-Centro Malattie Epatiche e Trapianti); Lucio Cosco (CATANZARO-U.O. Malattie Infettive Ospedale Pugliese Ciaccio); Alfredo Focà (CATANZARO-U.O. di Microbiologia Clinica); Daniela Segala (FERRARA-Malattie Infettive AOU S. Anna); Sergio Ferrara (FOGGIA-SC Malattie Infettive Universitarie AOU Ospedali Riuniti di Foggia); Maria Rosaria Lipsi (FOGGIA-SSD Microbiologia e Virologia AOU Ospedali Riuniti di Foggia); Paola Corsi (FIRENZE-Malattie Infettive CAREGGI); Massimo Di Pietro (FIRENZE-Malattie Infettive SM Annunziata); Luisa Galli (FIRENZE-Malattie Infettive Pediatria Meyer); Grazia Colao (FIRENZE-Virologia CAREGGI); Andrea Tosti (FOLIGNO-Malattie Infettive/SERT); Emanuele Pontali (GENOVA-Malattie Infettive Ospedali Galliera); Antonio Di Biagio (GENOVA-Malattie Infettive Ospedale S. Martino); Bianca Bruzzone (GENOVA-Laboratorio di Igiene Ospedale S. Martino); Cesira Nencioni (GROSSETO-Malattie Infettive); Barbara Rossetti (GROSSETO-Malattie Infettive); Stefano Rusconi (LEGNANO-Malattie Infettive); Riccardo Pardelli (LIVORNO-Malattie Infettive); Angelo Regazzetti (LODI-Malattie Infettive); Alberto Degiuli (LODI-Virologia Lodi); Michele De Gennaro (LUCCA-Malattie Infettive); Paolo Bonfanti (MONZA-Malattie Infettive); Sergio Malandrin (MONZA-UO Microbiologia AO S. Gerardo); Eleonora Gabrielli (MACERATA-Malattie Infettive); Giovanni Pellicano (MESSINA-Malattie Infettive A.O.U. Policlinico G. Martino); Paola Cicconi (MILANO-Clinica di Malattie Infettive Ospedale S. Paolo); Andrea Gori (MILANO-Fondazione IRCCS Ca’ Granda Ospedale Maggiore Policlinico, UOC Malattie Infettive); Valeria Micheli (MILANO-Laboratorio Microbiologia Ospedale L. Sacco [Seconda Divisione Malattie Infettive]); Paola Meraviglia (MILANO-Seconda Divisione Malattie Infettive Ospedale L. Sacco); Amedeo Capetti (MILANO-Prima Divisione Malattie Infettive Ospedale L. Sacco); Maria Luisa Biondi (MILANO-Laboratorio di diagnostica molecolare infettivologica AO S. Paolo); Cristina Mussini (MODENA-Clinica Malattie Infettive); Monica Pecorari (MODENA-Virologia); Nicola Gianotti (HSR-Studio MUSA); Antonio Cascio (PALERMO-Malattie Infettive Policlinico ‘P. Giaccone’); Daria Sacchini (PIACENZA-Malattie Infettive); Giustino Parruti (PESCARA-Malattie Infettive); Ennio Polilli (PESCARA-Virologia Pescara); Daniela Francisci (PERUGIA-Malattie Infettive); Stefania Zanussi (AVIANO-Centro di Riferimento Oncologico); Alessandro Nerli (PRATO-Malattie Infettive); Lucia Lenzi (PRATO-Virologia); Carlo Calzetti (PARMA-Divisione Malattie Infettive ed. Epatologia Azienda Ospedaliera); Angela Vivarelli (PISTOIA-Malattie Infettive); Roberto Gulminetti (PAVIA-Ambulatorio Clinica Malattie Infettive S. Matteo); Raffaele Bruno (PAVIA-Clinica Malattie Infettive e Tropicali); Fausto Baldanti (PAVIA-Virologia S. Matteo); Claudia Lazzaretti (REGGIO EMILIA-Malattie Infettive); Alessandro Zerbini (REGGIO EMILIA-S.S Autoimmunità, Allergologia e Biotecnologie Innovative); Francesca Lombardi (ROMA-Laboratorio virologia Cattolica); Simona Di Giambenedetto (ROMA-Università Cattolica del Sacro Cuore, Roma Italia, Istituto di Clinica Malattie Infettive); Loredana Sarmati (ROMA-Cattedra Malattie Infettive Tor Vergata); Marco Montano (ROMA-Virologia per Malattie Infettive Tor Vergata); Vincenzo Vullo (ROMA-Malattie Infettive e Tropicali La Sapienza-Umberto I); Ombretta Turriziani (ROMA-Medicina Sperimentale e Patologia-Sezione Virologia-La Sapienza); Andrea Antinori (ROMA-Malattie Infettive INMI Spallanzani); Maurizio Zazzi (SIENA-Virologia); Enzo Boeri (MILANO-Virologia HSR); Stefano Bonora (TORINO-Malattie Infettive Amedeo di Savoia); Valeria Ghisetti (TORINO-Laboratorio di Virologia, Ospedale Amedeo di Savoia); Paolo Grossi (VARESE-Clinica Malattie Infettive e Tropicali); Federica Poletti (VERBANIA-Malattie Infettive VERBANIA); Federica Poletti (VERBANIA-Virologia); Stefano Nardi (VERONA-Centro di Medicina Preventiva-ULSS 20).
Author contributions
Conceptualization: L.L., B.R., A.D.B. Data curation: L.L., B.R., C.R., C.C., M.B., R.P., I.R., F.B., L.G., M.T., C.M., S.M., A.B., A.S., M.Z., A.D.B. Formal analysis, B.R., A.S., S.M. Draft of the manuscript: L.L., B.R., A.D.B., M.Z. Review of the article and critical revision: all the Authors.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Transparency declarations
ARCA was sustained by unconditional educational grants from ViiV Healthcare, GILEAD Sciences, MSD, Janssen.
