Abstract
Background/aim
Tenofovir amibufenamide (TMF) employs innovative ProTide technology and a methylation strategy to enhance the lipid solubility and plasma stability of the amide bond, providing advantages over tenofovir alafenamide (TAF). Despite promising Phase III clinical trial results demonstrating its antiviral efficacy, real-world data on TMF remains scarce. This study evaluates the antiviral efficacy and safety of TMF compared to TAF as the initial treatment in patients with high viral loads of chronic hepatitis B (CHB).
Methods
We retrospectively collected clinical data from March 1 2022 to June 30 2022 for highly viremic CHB patients who received either TMF (
Results
Baseline median HBV DNA levels were 7.85 (6.89, 8.36) IgIU/ml for TMF and 7.44 (6.89, 8.03) IgIU/ml for TAF. Median ALT levels were 102.0 (56.0, 210.0) U/L for TMF and 195.0 (73.5, 371.0) U/L for TAF, with HBeAg positivity rates of 70.7% and 75.0%, respectively. At 48 weeks, virological response rates (HBV DNA <10 IU/ml) were 43.5% (20/46) for TMF and 42.9% (12/28) for TAF (
Conclusion
TMF demonstrates comparable antiviral efficacy to TAF when used as initial therapy in highly viremic CHB patients, with similar impacts on renal function and lipid profiles.
Introduction
HBV remains a significant public health challenge worldwide. According to the World Health Organization (WHO); as of 2019, the global prevalence of hepatitis B surface antigen (HBsAg) was 3.8%, which translates to approximately 1.5 million new hepatitis B virus (HBV) infections, 296 million chronic cases and 820,000 deaths from liver failure, cirrhosis and hepatocellular carcinoma (HCC). 1 In China, the HBsAg prevalence was approximately 6.1%, contributing to 86 million chronic HBV infections and accounting for 47% of the global liver cancer burden, predominantly caused by HBV infection.2,3
Antiviral therapy plays a crucial role in managing chronic hepatitis B (CHB), significantly improving outcomes by reducing inflammation and necrosis of liver cells, decreasing hepatic fibrosis, lowering the risk of cirrhosis and liver cancer and slowing disease progression through the inhibition of HBV replication.4,5 Currently, entecavir (ETV), tenofovir disoproxil fumarate (TDF and tenofovir alafenamide (TAF) are recommended as first-line treatments in most CHB guidelines.6–8 With the WHO’s goal to eliminate viral hepatitis by 2030 and the introduction of the updated 2022 CHB guideline in China, 6 the demand for effective antiviral treatments has grown considerably, prompting the development of new antiviral agents.
Tenofovir amibufenamide (TMF, codename: HS-10234) was approved in mainland China in June 2021 by the National Medical Products Administration. Structurally, TMF includes an additional methylation compared to TAF, which enhances its fat solubility and stability in plasma. The half-life of TMF in vivo is longer than that of TAF, so that more TMF prototype drugs could be passively diffused into target cells, further increasing the concentration of active substance in hepatocytes.9,10 The efficacy and safety of TMF were demonstrated in a Phase III clinical trial. 11 However, real-world evidence on the safety and efficacy of TMF in patients with high viral loads is scarce. This study presents a retrospective analysis comparing the efficacy and safety of TMF versus TAF in treatment-naïve, highly viremic CHB patients.
Methods
Study population
This was a single-centre retrospective study conducted at Beijing YouAn Hospital, Capital Medical University. This is a large liver diseases specialized hospital treating patients deriving across the country. Patients with CHB meeting the following criteria were enrolled: age ≥18 years; eligible for antiviral treatment according to the China CHB Guidelines (2022 version) 6 ; HBV DNA level ≥2.0 E+5 IU/ml with positive or negative HBeAg; initial monotherapy with TMF or TAF from March 1 to June 30 2022. The exclusion criteria were as follows: acute hepatitis B; co-infection with hepatitis A virus, hepatitis C virus, hepatitis D virus, hepatitis E virus or human immunodeficiency virus; concomitant presence of cirrhosis, liver cancer or non-liver malignancy, autoimmune liver disease or genetic metabolic conditions; presence of other serious comorbid conditions. The study was approved by the Institutional Review Board of Beijing YouAn Hospital, Capital Medical University (IRB No. [2022]068) and performed in accordance with the principles of the Declaration of Helsinki.
Data collection
Data were collected from the electronic medical record system. The records included demographic characteristics (age and sex), antiviral regimen and duration, alanine aminotransferase (ALT), aspartate aminotransferase (AST), HBV serological markers, HBV DNA level, blood creatinine (Cr), estimated glomerular filtration rate (eGFR), triglyceride (TG), total cholesterol (TC), low-density lipoprotein cholesterol (LDL-C) and abdominal ultrasound or CT results.
Outcomes
The primary efficacy endpoint was the virologic response, defined as the serum HBV DNA level <10 IU/mL at week 48. Other efficacy endpoints included the ALT normalization rate (ALT ≤40 U/L), HBeAg serological clearance (<1.0COI) and changes in HBeAg/HBsAg levels during antiviral therapy. Safety assessment included changes in renal function measured by serum Cr and eGFR, as well as changes in blood lipids measured by TG, TC and LDL-C.
Statistical analysis
Categorical data were analyzed by calculating frequencies (
Results
Patient characteristics
Baseline characteristics of the patients.

Virological response
The mean decrease in the HBV DNA level at week 4, week 12, week 24 and week 48 compared with the baseline was 3.65 ± 0.99 log10 IU/mL, 4.63 ± 0.84 log10 IU/mL, 5.82 ± 0.80 log10 IU/mL and 6.85 ± 0.86 log10 IU/mL VS 3.70 ± 0.96 log10 IU/mL, 4.83 ± 0.94 log10 IU/mL, 5.77 ± 0.86 log10 IU/mL and 6.38 ± 0.75 log10 IU/mL in the TMF and TAF group, respectively (Figure 1). There was no significant difference in HBV DNA decline between the two groups at each follow-up time point (Figure 1). The most substantial reduction in HBV DNA level occurred within the first 4 weeks of antiviral treatment. The virological response rate (HBV DNA <10 IU/ml) at 48 weeks was 43.5% (20/46) in the TMF group and 42.9% (12/28) in the TAF group Changes in HBV DNA levels at each follow-up time point in the TMF group and TAF group.
Biochemical response
Changes in clinical parameters at 24 and 48 weeks after antiviral therapy compared to the baseline.

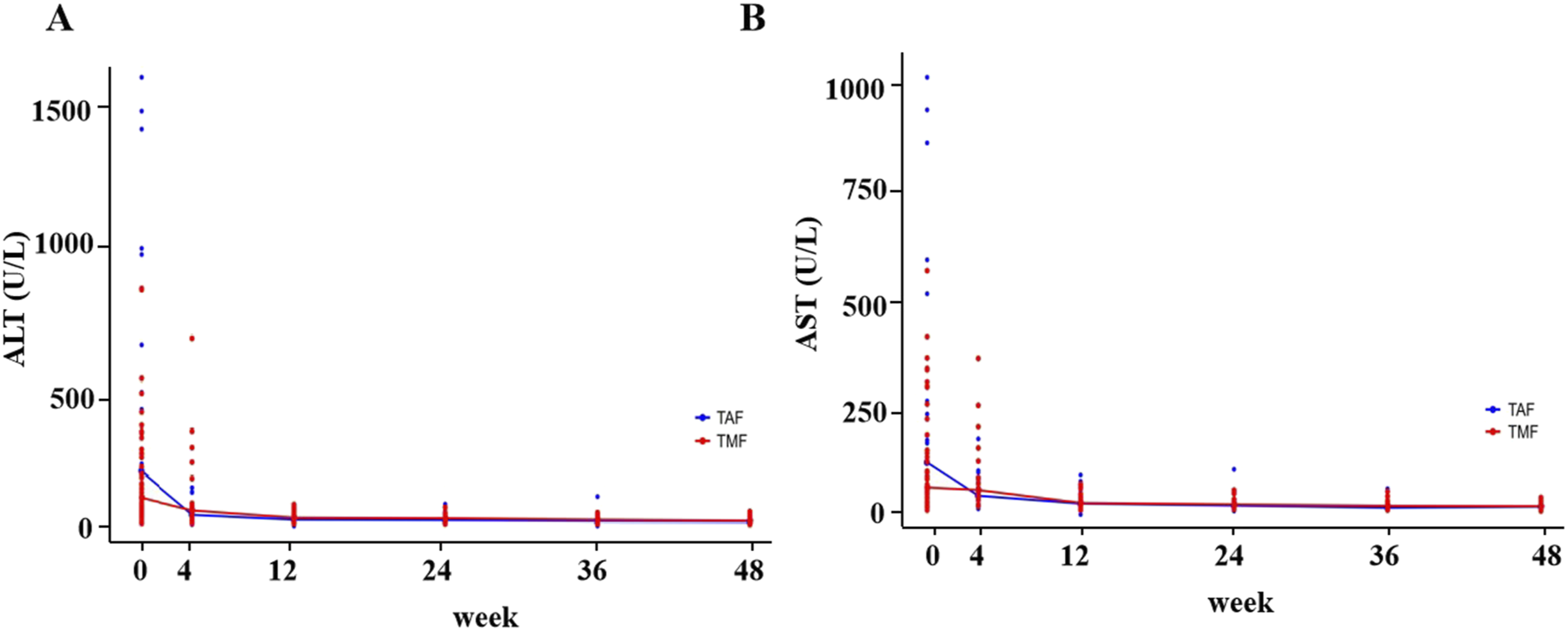
Changes in ALT (a) and AST (b) levels at each follow-up time point in the TMF group and TAF group.
HBsAg and HBeAg serological response
Compared with the baseline, the median decrease in HBsAg level was 0.30 (0.18, 1.29) log10 IU/ml ( Changes in HBsAg (a) and HBeAg (b) levels at each follow-up time point in the TMF group and TAF group.
Safety: renal function and blood lipids
In this study, serum Cr level and eGFR were analyzed to evaluate renal function, and TG, LDL-C and TC represented blood lipid levels. Data on bone metabolism were not available. Compared with the baseline, the median serum Cr level and eGFR decrease was −3.00 (−7.00, 1.00) μmol/L (
Discussion
The virologic response is the most fundamental endpoint during antiviral therapy for CHB patients. In the two Phase Ⅲ clinical trials of TAF,12,13 virological response rates (HBV DNA <29 IU/ml) were 64% versus 67% in HBeAg-positive patients and 94% versus 93% in HBeAg-negative patients compared with TDF at week 48. Similarly, the Phase Ⅲ clinical trial of TMF 14 was also compared with TDF. 50.2% versus 53.7% of HBeAg-positive patients and 88.9% versus 87.8% of HBeAg-negative patients achieved HBV DNA less than 20 IU/ml in the TMF and TDF groups, respectively. Currently, there is no head-to-head prospective clinical study between TAF and TMF.
The results of this retrospective study showed that 43.5% (20/46) and 42.9% (12/28) of patients in the TMF and TAF groups achieved a virological response (HBV DNA <10 IU/ml) at 48 weeks, and the decrease in HBV DNA level at week 4, week 12, week 24 and week 48 between the two groups was similar, with the greatest reduction within the first 4 weeks of antiviral treatment. From this study, TMF demonstrates comparable antiviral efficacy to TAF when used as initial therapy in highly viremic CHB patients. However, the virological response rate of this study at 48 weeks was lower than that of the Phase III clinical studies above, and in another similar real-world study
15
36.3% of patients had HBV DNA below lower limit of detection (<10IU/ml) at baseline. It is speculated that this difference may result from a higher baseline HBV DNA level and highly sensitive DNA detection methods. Changes in renal function and lipid metabolism indexes at each follow-up time point in the TMF group and TAF group. (a) Serum Cr level; (b) eGFR level; (c) TC level; (d) TG level; (e) LDL level.
Although it has been reported that TAF and TMF were superior to TDF in ALT normalization in different studies,12–14,16 different biochemical criteria may yield different results in ALT normalization rates between different drugs. In our study, the ALT normalization (<40 U/L) rates were 87.9% and 90.6% in the TMF and TAF groups at week 48, respectively, which were numerically higher than that in previous studies. The decrease in ALT/AST levels from baseline was also not statistically different between the two groups at each visit time. Li et al. reported that there was no significant difference in ALT normalization between TAF and TMF after 24 weeks of treatment, 17 which was consistent with our study. So far, there has been no consensus on which antiviral drugs have better ALT normalization rates. Under normal circumstances, in real-world studies, the simultaneous use of liver-protecting drugs may affect biochemical results.
HBeAg clearance or seroconversion is an important endpoint for HBeAg-positive CHB patients. Previous studies12,14,18,19 reported that the HBeAg clearance rate was approximately 12% to 22% after 48 weeks of treatment with first-line antiviral drugs. Similarly, the HBeAg clearance rate was 21.1% and 18.2% in the TMF and TAF groups, respectively, after 48 weeks in our study. None of the patients achieved HBsAg clearance or seroconversion. Although there was a significant difference in the decrease of HBsAg levels at week 48 compared to baseline, the decrease was relatively small, and it was speculated that it was more due to the activation of the body’s immune function (baseline ALT/AST levels elevated obviously in most patients).
Regarding renal safety, both Phase III12,13,16 and post-marketing clinical experience showed that TAF was better than TDF. As a new prodrug of tenofovir, both TMF and TAF had good renal safety with a slight increase in serum Cr and a decrease in eGFR at 48 weeks from our study. However, the decrease in eGFR was statistically significant, suggesting that the long-term effects of TMF and TAF on kidney function still need to be paid attention to clinically. In lipid metabolism, as previously reported,12,13,20 TAF treatment was related to a higher rate of dyslipidemia and weight gain in HBV and HIV patients. The rates of ≥grade 3 fasting LDL levels were 4% at week 48 and 6% at week 96 in patients with TAF treatment, respectively. 17 Also, it was reported 15 that TMF had a higher incidence of dyslipidemia and more weight gain than TDF. Similarly, in our study, compared with baseline, TMF and TAF treatment led to a statistically significant increase in LDL-C level, while TC and TC showed no significant changes at 48 weeks. The influence of TMF and TAF treatment on blood lipids was similar at each study visit time point.
In conclusion, from our study, TMF demonstrates comparable antiviral efficacy to TAF as initial therapy in highly viremic CHB patients, with similar impacts on renal function and lipid profiles, and provides more options for antiviral treatment in CHB patients. Due to the limitations of a single centre and a retrospective study with a small sample size, further studies with larger sample sizes and longer follow-up are needed to confirm its antiviral efficacy and safety.
Footnotes
Author Contributions
Conceptualization: L.Q. and J.F.L; data curation: D.L., L.L.W. and S.R.; formal analysis: Q.Z. and J.H.X.; funding acquisition: J.F.L.; investigation: S.J.Z. and X.Y.C.; project administration: Q.Z., L.Q. and J.F.L.; supervision: S.J.Z. and X.Y.C.; writing—original draft: Q.Z. and J.F.L; writing—review and editing: L.Q. and J.F.L. All authors have read and agreed to the published version of the manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Beijing Natural Science Foundation (7232080); Capital Clinical Diagnostic Techniques and Translational Application Projects (No. Z221100007422063); Capital’s Funds for Health Improvement and Research (2022-1-2172).
