Abstract
Background
This study aims to evaluate the cost-effectiveness of tenofovir alafenamide fumarate (TAF) versus tenofovir disoproxil fumarate (TDF) for chronic hepatitis B treatment from a payer’s perspective in a limited-income context like Vietnam.
Methods
A Markov model was developed to estimate the lifetime cost and effectiveness (measured in quality-adjusted life year, QALY) of TAF compared to TDF in the HbeAg+ patient population. Efficacy data came from clinical trials, and costs were based on 2023 data from an exit survey of 94 inpatients and 464 outpatients in Bach Mai hospital. Other clinical data were also sourced from CHB patients at Bach Mai hospital. Along with deterministic analysis, two-way sensitivity analysis, probabilistic sensitivity analysis, threshold, and budget impact analysis were performed.
Results
Compared to TDF, TAF yielded an additional cost of USD 3,983 and an additional QALY gained of 0.14, resulting in the incremental cost-effectiveness ratio (ICER) of USD 32,090 per QALY gained. The ICER exceeds the cost-effective threshold of three-time gross domestic product (GDP) per capita, that is, USD 11,348, by 2.8 times. According to one-way sensitivity analysis, ICERs were driven mainly by transition probabilities and TDF/ TAF prices. TAF would be cost-effective compared to TDF at the three-time GDP per capital threshold if TAF price were reduced by 33.4%.
Conclusions
TAF is not cost-effective compared to TDF for treating chronic hepatitis B in HBeAg+ patients. The study offers relevant evidence for policymakers to consider including TAF in the social health insurance package, with a focus on price negotiation. Future updates are needed as new evidence on the effectiveness and costs of treating chronic hepatitis B emerges.
Keywords
Introduction
Hepatitis B is a widespread infectious disease worldwide, caused by the hepatitis B virus. Vietnam is one of the countries with a high rate of hepatitis B virus infection in the general population and a large burden of hepatitis B, with an estimated prevalence of over 8% in the general adult population.1,2 In 2017, the World Health Organization estimated around 7.8 million people in Vietnam were living with chronic hepatitis B (CHB). 3 The treatment burden significantly impacts patients, families, the healthcare system, and society as a whole, especially in a limited-income country like Vietnam.
The Liver Research Association has issued global guidelines for first-line treatment of CHB, recommending oral nucleot(s)ide analogues (NAs) and pegylated interferon (Peg-IFN) as treatment options.4–7 NAs work by inhibiting HBV replication through binding to DNA polymerase, while Peg-IFN is a protein with antiviral, antitumour, and immunomodulatory effects. In line with these international guidelines, the national protocol for CHB treatment suggests that therapy is long-term, often lifelong, with first-line use of oral NAs such as tenofovir alafenamide fumarate (TDF), tenofovir disoproxil fumarate (TAF), and entecavir (ETV). Peg-IFN regimens are reserved for specific cases. 8
TDF and TAF are both derived from tenofovir and work similarly by inhibiting HBV reverse transcriptase. TDF has been used for HBV treatment since 2008, 9 showing strong antiviral activity in CHB patients, though long-term use has been linked to impaired kidney function and reduced bone mineral density. 10 TAF, a newer version, offers greater stability in plasma and requires a lower dose while maintaining the same effectiveness as TDF in inhibiting HBV, with fewer side effects on the kidneys and bones during prolonged treatment. 11 Studies also indicate lower rates of hepatocellular carcinoma and mortality with TAF compared to TDF. 12 Both national and international guidelines recommend TAF and TDF as first-line treatments for CHB.4–8 However, in Vietnam, TAF is priced at around USD 1.23 (ranging from USD 0.98 to 1.48) per tablet of 25 mg, higher than TDF at approximately USD 0.65 (ranging USD 0.17 to 0.78) per tablet of 300 mg. While TDF is fully covered by the Social Health Insurance (SHI) scheme, TAF has been officially included in the Ministry of Health’s treatment guideline in Vietnam since 2019 but has not yet been included in the SHI benefit package. 8 The SHI scheme, which covers around 91.6% of the population, is the main financial source for treatment services in Vietnam, and policymakers are evaluating whether adding TAF to the SHI benefit package offers good value for money. 13
To address this issue, evidence on the cost-effectiveness of TAF versus TDF in treating CHB has been crucial, particularly in low-income countries like Vietnam. Globally, several cost-effectiveness studies have examined TAF and TDF, but most were conducted in developed countries and produced inconsistent results (please see Appendix Table S1 for our quick review of relevant cost-effectiveness studies on PubMed up to December 2023). For instance, in a study by Zonglin Dai et al. (2018), compared with the baseline strategy, TDF was the most cost-effective therapy for HBeAg-negative CHB patients (USD 1,418 per QALY gained) achieving a satisfactory goal. Among nucleotide analogues, TAF was the most cost-effective strategy and had a higher acceptability to achieve an optimal goal in the Eastern region of China (under the one-time GDP per capita threshold). 14 In contrast, a 2019 study by Feng Tian et al. from the Canadian health system perspective concluded that TAF was not cost-effective compared to TDF and ETV. To become cost-effective, the price of TAF would need to decrease by 33.4%, assuming a willingness-to-pay threshold of 50,000 Canadian dollars. 15 The inconsistent findings, combined with variations in healthcare systems, make it challenging to directly apply these results to Vietnam. Therefore, this study aims to assess the cost-effectiveness of TAF vs TDF within the context of Vietnam from a payer’s perspective. The findings will offer critical evidence to local authorities in determining whether TAF should be included in the SHI benefit package.
Materials and methods
We conducted a model-based economic evaluation in compliance with the Vietnam health technology assessment guideline. 16 The study protocol was developed and consulted with an expert panel from Hanoi University of Public Health. A Markov model over a lifetime horizon (with a one-year cycle) was constructed from payer perspectives to compare the cost and outcome of the two interventions. This perspective not only covers the impact of the intervention on SHI but also on patients since the out-of-pocket payment among SHI card holders remains very high. 13 We followed the Consolidated Health Economic Evaluation Reporting Standards 2022 (CHEERS 2022) for reporting the study 17 (see Appendix Table S2 for the completed CHEERS checklist).
Study population and setting
The hypothetical population comprises CHB patients with an average age of 35 years and a hepatitis B envelope antigen positive (HBeAg+) status, reflecting the typical profile of CHB patients who started to be treated with TAF/TDF at Bach Mai Hospital in Vietnam. This starting age aligns with the average age of participants in various randomized controlled trials assessing the effectiveness of TAF and TDF. Bach Mai Hospital, located in Hanoi, Vietnam, is one of the largest and most well-known medical facilities in the country, with over 3,600 hospitalization beds, and provides services for about 2.5 million outpatient visits annually. Bach Mai Hospital is a leading facility in applying the most up-to-date approved health technologies, including TAF for CHB patients. The hospital is also the forefront of implementing an electronic medical record database, which facilitates the collection of context-specific data for this study in the absence of important national-level data. We retrieved data for 148 CHB outpatients receiving TAF/TDF who visited the hospital from June 2022 until June 2023. Their average age at their first visit was 35.8 years old and only two were categorized as HbeAg-negative. Thus, our focus is solely on HBeAg+ patients due to their high proportion in this current CHB population, with treatment priorities emphasizing this group because of the active replication of HBV.
Intervention and comparator
We compared the lifetime costs and effectiveness of TAF versus TDF for treating CHB in HBeAg+ patients, with both treatments taken for life. TAF is administered at 25 mg once daily, while TDF is given at 300 mg once daily, both in tablet form.
We do not consider the do-nothing scenario since all CHB patients who are eligible for NAs will receive at least TDF or ETV since both TDF and TEV are included in the SHI benefit package, making them available for all eligible patients. Within this cost-effectiveness study, we do not take into account ETV as an alternative due to a small proportion of patients receiving ETV in this specific setting. The dominant use of TDF makes it unfeasible to gather reliable information on the annual cost for patients using ETV.
Model structure
A Markov model was developed to simulate the transition of disease states in CHB patients with HbeAg + treated with TAF or TDF. The model was initially built by reviewing the current national treatment guidelines, relevant economic evaluations, and related RCTs on treatment outcomes.14,15,18 Then, the model structure was verified based on experts’ opinions from a scientific panel of two health economics experts and in-depth interviews with three hepatologists to see if the model is relevant to reflect current management approaches for CHB in the Vietnam healthcare setting. The final model includes eight health states: CHB, HBeAg seroconversion, HBsAg seroclearance, compensated cirrhosis (CC), decompensated cirrhosis (DC), hepatocellular carcinoma (HCC), liver transplantation, and death (see Figure 1). The patient cohort enters the model at the CHB state. Each year, the patient could move between different health states with a certain probability, as indicated by the arrows in Figure 1. The model was constructed using Microsoft Excel. The model was checked for programming internally by the last author and externally by one modelling expert. Any issues raised were discussed among the authors to finalize the model. Markov model.
Key assumptions
The following assumptions were made in the analysis, similar to previous studies.14,18–20 All assumptions were consulted with experts to see if they were acceptable in the Vietnam context. The efficacy of TAF was assumed to be similar to that of TDF in subsequent years of treatment (after the first 12 months) due to the lack of evidence on the long-term efficacy of TAF compared to TDF. This is a common assumption applied in cost-effectiveness studies of TAF versus TDF that have been conducted to date. 14 Patients in all health states could die due to background mortality. Mortality rates from HbsAg seroclearance and HBeAg seroconversion states are assumed to equal background mortality. We assumed patients in both arms, that is, treated with TDF and TAF, were equally likely to receive all other medical services.
We do not include health states related to adverse events from TAF/TDF treatment due to the lack of specific data on their occurrence among the study population. However, by using actual data from Bach Mai Hospital for separate cohorts of patients using TDF or TAF, the costing of health states captures the resource consumption for treating adverse events (e.g. cost for para-clinical tests to access the adverse event, infusion cost, and medication cost for treatment).
Input parameters
Clinical data
Model inputs.

Notes: USD: United State dollar; CC: compensated cirrhosis; DC: decompensated cirrhosis; HCC: hepatocellular carcinoma; TAF: tenofovir alafenamide fumarate; TDF: tenofovir disoproxil fumarate; CHB: chronic hepatitis B; HbeAg: hepatitis B envelope antigen; HbsAg: hepatitis B surface antigen.
Utility data
Table 1 presents utility values used for different health states. The utility values were collected from studies conducted by Adrian R. Levy (2008). 31
Costs data
The cost of interventions and cost of health states were estimated from the payer perspective, covering all direct medical costs, including the cost of TAF/TDF tablets, hospitalization bed, para-clinical examinations (e.g. diagnostic imaging, functional tests, and blood test), surgical procedures, other supportive medicine and infusions other than TAF/TDF, medical supplies, and other medical services.
It is assumed that CHB patients use TAF/TDF as prescribed for their lifetime. The prices of TAF/TDF tablets were collected from Bach Mai Hospital’s database on medicine and pharmaceutical products in 2022. Annual costs of outpatient treatment and hospitalization were estimated using the average number of outpatient treatments and hospitalizations multiplied by the corresponding unit costs. The average number of outpatient visits and hospitalizations for each health status were collected from a cost of illness study in Vietnam 32 and the Bach Mai Hospital’s database.
To estimate the unit cost of outpatient visits and hospitalizations among CHB patients, we extracted data from 94 hospitalized CHB patients and 461 CHB outpatient visits between June 2022 and June 2023 using outpatient and inpatient databases from Bach Mai Hospital. Among the outpatients, 30.8% were female, with an average age of 48.4, and 40.6% of patients were using TAF. For inpatients, 25.5% were female, with an average age of 44.7, and none were using TAF. Therefore, we assumed that inpatients treated with TAF would likely receive inpatient treatment services similar to those treated with TDF. The cost of liver transplantation was collected from a local study published in 2022. 33 The study complied with the principles of the Ethics Council, Hanoi University of Public Health, and was approved in Decision No. 387/2023/YTCC-HD3 dated August 18, 2023. Data collected from Bach Mai Hospital database on inpatient and outpatient treatment received formal approval from the hospital’s director board. Detailed information on cost parameters is presented in Appendix Table S3. Appendix Table S4 also presents the estimation of the annual cost of CHB health state, categorized by cost items. Indeed, the annual total cost of the CHB health state for patients treated with TAF is USD 370. The annual outpatient cost is USD 298, accounting for 36.4% of the total cost. For patients treated with TDF, the annual total cost of the CHB health state is USD 335, with an annual outpatient cost of USD 264, accounting for 47.5% of the total cost. The annual cost of TAF and TDF drugs accounts for 54.8% and 40.4% of the total annual cost, respectively.
Method of analysis and presentation of results
Results are presented as the incremental cost per QALY gained with TAF compared to TDF. A discount rate of 3% was applied to both costs and outcomes, as recommended by Vietnam’s health technology assessment guidelines. Costs were measured in Vietnamese dong (VND) and converted to 2020 United States dollars (USD) using the exchange rate of USD 1 = 25,380 VND. We utilized the current cost-effectiveness threshold values in Vietnam of USD 3782 and USD 11,348, which are widely accepted based on one and three times the Gross Domestic Product (GDP) per capita in Vietnam in 2022. If TAF was found to be not cost-effective compared to TDF, a threshold analysis was conducted to determine the cost-effective price of TAF.
Both one-way and probabilistic sensitivity analyses (PSA) using Monte Carlo simulations were conducted, with 10,000 iterations to address parametric uncertainties. Clinical values were varied within the 95% confidence intervals reported in the literature. According to experts, utility and cost values were adjusted by 10% around the base case value or within the upper and lower limits (see Table 1). We assigned a beta distribution for transition probabilities and utility parameters, and a gamma distribution for cost parameters. The results of the one-way sensitivity analysis were presented in a tornado diagram. Meanwhile, the PSA results were reported as mean ICER and 95% certain intervals (95% CIs), illustrated through incremental cost-effectiveness planes and cost-effectiveness acceptability curves.
For budget impact analysis, the target population (i.e. CHB patients eligible for using TAF) is estimated based on the prevalence of CHB cases in Vietnam in 2021. According to the national official plan for hepatitis disease treatment and control for the period 2021–2025, 34 50% of CHB cases are diagnosed, and 60% of diagnosed CHB cases received treatment. To estimate the required budget from the SHI scheme, we applied the SHI coverage rate of 91.6%. 13 The proportion of insured patients for whom SHI fund covered 100%, 90%, and 80% of the cost (corresponding to co-payment rates of 0%, 10%, and 20%) was 21.7%; 23.0%, and 55.3%, respectively, based on an analysis of direct medical costs of 1,395,204 insured diabetes patients in Vietnam in 2017. 35
Result
Base-case results
Base-case analysis and probabilistic sensitivity analysis results.

Notes: USD: United State dollar; QALY: Quality-Adjusted Life Year; ICER: incremental cost-effectiveness ratio; discount rate of 3% was applied for both cost and outcomes.
Sensitivity analysis
The results of the one-way sensitivity analysis are presented as a tornado diagram (Figure 2). The ICER results were most sensitive to variations in transition probabilities from CHB to HBsAg seroclearance (first year, treated with TAF/TDF), from CHB to HbeAg seroclearance (first years, treated with TDF), and price of TAF/TDF (see details in Appendix Table S3). Tornado diagram showing the effect of input parameters on the ICER.
The results from the Monte Carlo simulation with 100,000 replications were displayed in a cost-effectiveness plane (Figure 3). Almost all ICER plots were located in the upper-right quadrant of the plane. The single black square indicates the mean ICER, while the dotted black line and solid black line represent the cost-effectiveness thresholds of one-time GDP per capita and three-time GDP per capita, respectively, with estimates below these lines considered cost-effective. The PSA results were also presented as mean ICERs and their 95% CI from the 100,000 simulations (see Table S2). Scatter plot of probabilistic results.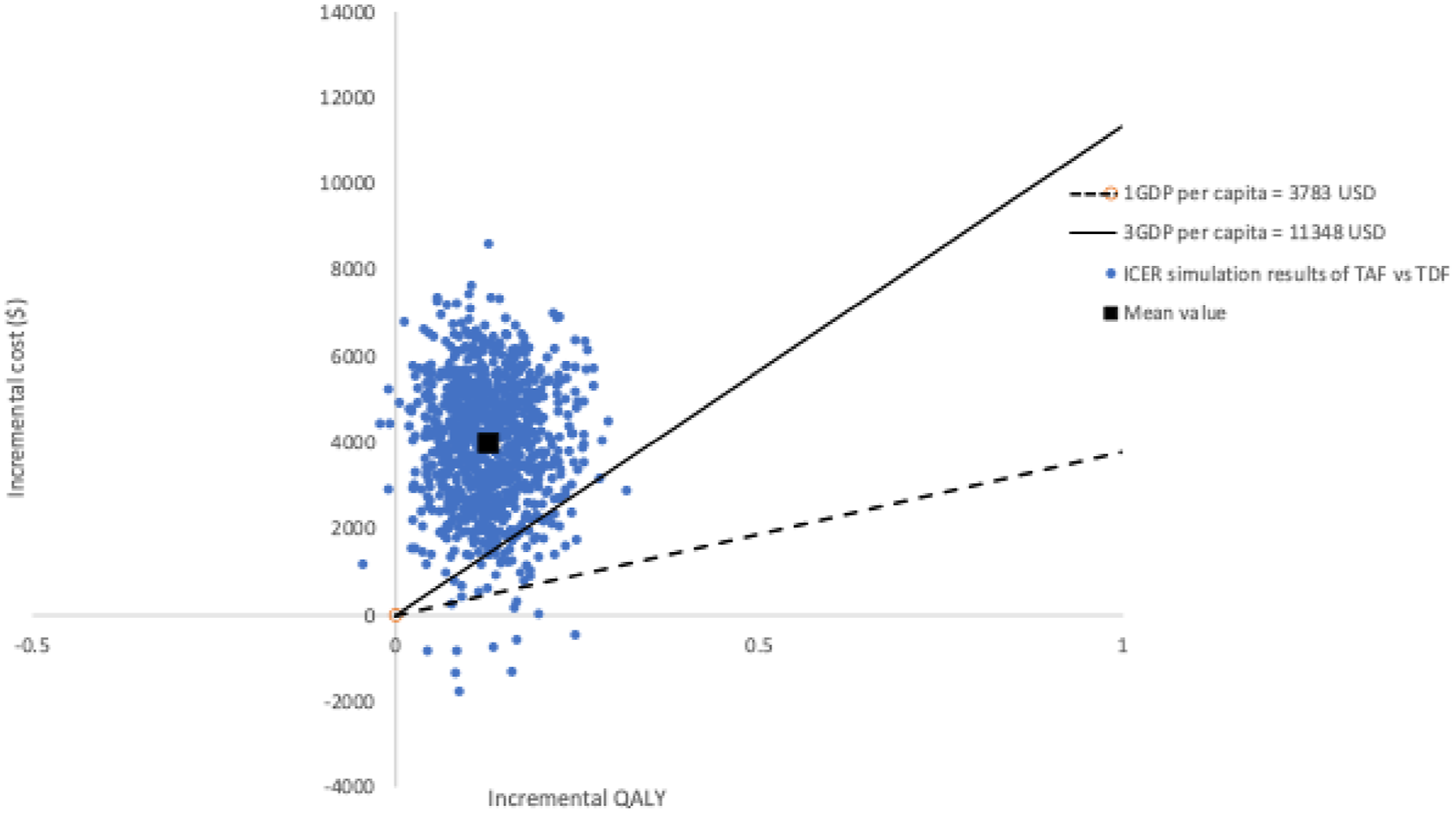
The cost-effectiveness acceptability curve was also constructed (see Appendix Figure S1, showing that at the cost-effectiveness thresholds of one-time and three-time GDP per capita (i.e. USD 3,783 and 11,348) per QALY gained, the probability of TAF being cost-effective compared to TDF is 0% and 2%, respectively). The probability of the TAF intervention being cost-effective rises to approximately 50% when the willingness-to-pay threshold increases to about USD 31,521.
Threshold analysis
Threshold analysis results.

Notes: USD: United State dollar; QALY: Quality-Adjusted Life Year; ICER: incremental cost-effectiveness ratio; discount rate of 3% was applied for both cost and outcomes; * cost-effective or not compared to the current threshold of 1xGDP per capita = USD 3.782 and 3xGDP per capita= USD 11.348; ** price at week ICER equals 1x GDP per capita; *** price at week ICER equals 3x GDP per capita.
Budget impact analysis
Budget impact analysis results.
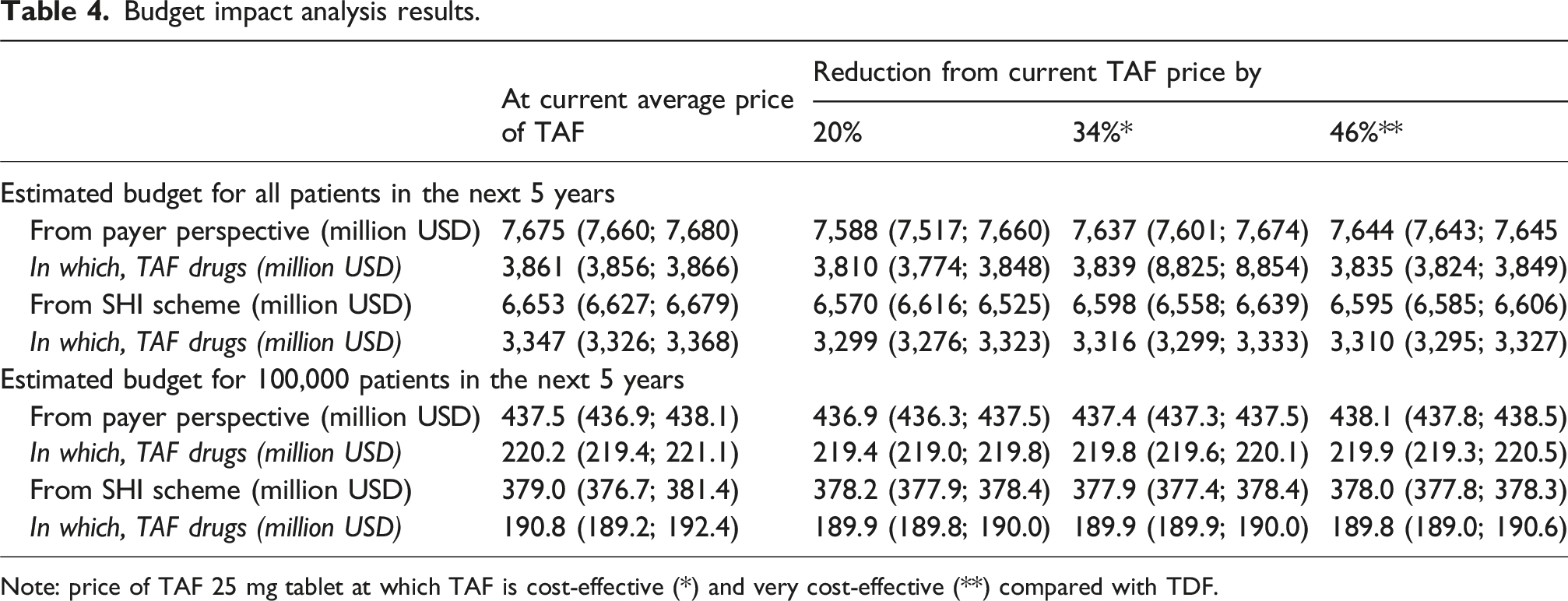
Note: price of TAF 25 mg tablet at which TAF is cost-effective (*) and very cost-effective (**) compared with TDF.
Discussion
Our finding from deterministic and sensitivity analysis confirmed that lifetime treatment with TAF compared to TDF among CHB patients with HbeAg+ is not cost-effective at its current price of TAF in Vietnam in 2022 from the payer perspective. Replacing TDF with TAF for lifetime treatment of CHB among HbeAg + patients incurred USD 32,090 per QALY gained, which exceeded the cost-effectiveness threshold of one-time GDP per capita (i.e. USD 3783) by 8.5 times.
The ICERs reported in our study are lower than those in similar studies. For example, Feng Tian (2020) reported an ICER of 94,142 Canadian dollars per QALY gained. 15 However, compared to another study by Zonglin Dai (2022) that modelled an ICER of USD 10,710 per QALY gained in China, 14 our study reported a higher ICER. It is important to note that the thresholds used in other studies are significantly higher than in ours, ranging from USD 7,176 in Zonglin Dai’s study to CAD 50,000 in Feng Tian’s study, compared to USD 3,783 in our study. Besides, it is difficult to compare the ICERs among countries due to the high heterogeneity in the economic background, healthcare systems, burden of CHB, and study methodology. Our study enriches the current literature on the cost-effectiveness of TAF in a lower-income country. Our study confirms that TAF is not cost-effective due to its high cost in a resource-constrain context like Vietnam and other low-middle-income countries.
Replacing TDF by TAF for lifetime treatment of CHB would be very cost-effective at the threshold of one-time GDP per capita if the cost of TAF 25 mg tablet were reduced by 46%. This result could support the policymakers in price negotiation with the pharmaceutical companies to make this effective drug more affordable. It is important to note that even with this discounted price of TAF, the budget impact of including TAF in the SHI benefit package could be significant. With the current number of approximately 6.4 million CHB patients (assuming 50% diagnosed and 60% diagnosed cases receiving TAF) and an SHI coverage rate of 91.6%, 13 the total SHI budget required for TAF over the next five years could reach USD 3,310 million (95% CI 3,295–3,327). This translates to an estimated SHI budget of USD 189.8 million (95% CI: 189.0–190.6) per 100,000 CHB patients who are eligible for TAF treatment. For comparison, the total SHI expenditure in 2021 was about USD 4.18 billion. 36 A recently published study using the SHI reimbursement database showed that among 1,395,204 insured patients with type 2 diabetes mellitus, the actual annual SHI payment for antihyperglycemic medications was only USD 56.6 million. 35 These figures highlight the substantial budget impact of including TAF in the SHI benefit package. Given this financial burden, careful consideration of reimbursement policies and feasibility is crucial in the current context. It is necessary to re-perform this cost-effectiveness analysis of TAF in the near future, as the prices of selected brands of TAF tablets appear to have decreased from 2022 to 2024, potentially offering a more promising ICER in the future.
This study is the first cost-effectiveness analysis of TAF for CHB patients with HBeAg+ in Vietnam. Our research has several notable strengths. First, regarding the model structure, we adapted the Markov model from international studies, combined with expert consultation, and maximized the use of context-specific cost data. Our Markov model also employs a one-year cycle length, consistent with other cost-effectiveness models on the same topic. In the future, this model can be updated to assess the cost-effectiveness of TAF, providing valuable insights for policymakers and evaluating intervention cost-effectiveness in Vietnam.
Second, the input parameters were sourced from the most reliable and trustworthy data. This is the first study to collect detailed cost component data for each health state in CHB treatment. The cost data in our study was gathered from Bach Mai Hospital, Vietnam’s first fully comprehensive national general hospital and one of the largest hospitals in the country. To estimate transition probabilities for the model, we used evidence from studies conducted in China, a neighbouring country in Asia, ensuring no significant differences in HBV epidemiology. Since TAF was only approved for use in Vietnam in 2019, there is no available cost data related to its use among CHB patients. We collected data from 94 hospitalized CHB patients and 461 outpatient visits between June 2022 and June 2023 using Bach Mai Hospital’s outpatient and inpatient databases. Thanks to the hospital’s comprehensive patient databases, we could track the direct medical costs for all patients, including those reimbursed by the SHI scheme and out-of-pocket payments from patients and their households.
Although our study has notable strengths and innovations, it also has certain limitations. First, regarding the model structure, we did not include a model for CHB patients with HBeAg- due to the lack of crucial input parameters, particularly the current status of diagnosis of CHB patients with HBeAg- in Vietnam. Therefore, we recommend expanding future research to develop a model for CHB patients with HBeAg- in Vietnam. Besides, the inclusion of HbeAg seroconversion may be debated, as it is no longer the primary endpoint for treatment cessation in the international literature. However, it remains a criterion for treatment cessation in the national treatment guideline for CHB patients 8 (effective date from July 29, 2019). This appears to be similar to certain contexts, so some similar studies published recently still include the HBeAg seroconversion state in the Markov model.14,37,38 Based on consultations with experts during the model development, we believe that retaining this health state remains relevant in our context. However, future modellers should carefully consider this to ensure alignment with updated treatment guidelines.
Second, we do not include health states related to adverse events from TAF/TDF treatment, despite this being a key advantage of TAF over TDF, due to the lack of specific data on their occurrence among the study population. However, by using actual data from Bach Mai Hospital for separate cohorts of patients using TDF or TAF, the costing of health states captures resource consumption for treating adverse events (e.g. costs for para-clinical tests to assess adverse events, infusion costs, and medication costs for treatment). The results of the cost analysis further support this approach, showing that the annual cost for CHB patients receiving TDF is higher than for those receiving TAF in terms of para-clinical tests, diagnostic imaging, and other medications outside TDF/TAF drugs. This reflects current clinical practice, where the management of adverse events is integrated into overall patient care costs. Introducing a separate health state for adverse events in the context of unreliable data on its treatment among the target population in Vietnam may overestimate the cost-effectiveness of TAF compared to TDF.
Third, the alternatives being compared in our study may not be representative of the entire country. We only compare the cost-effectiveness of TAF versus TDF, while other alternatives, such as no treatment or treatment with ETV, may be available. However, due to the lack of national-level data on intervention costs and the costs of health states associated with these alternatives, we relied on the Bach Mai Hospital database to collect not only cost information but also clinical data. Within the context of Bach Mai Hospital, all CHB patients eligible for NAs receive either TDF or ETV, making no treatment an unfeasible comparator. Additionally, the small proportion of patients treated with ETV limits our ability to gather sufficient data to include it as a comparator in the current analysis. Therefore, future research should address this limitation by identifying all possible comparators relevant to their policy context.
Forth, our study was conducted from the payer’s perspective, covering direct medical costs incurred by both patients and the SHI. We recognize that health technology assessments are ideally conducted from a societal perspective to better support policymakers. However, given the current circumstances—specifically, the limited use of TAF—our analysis from the payer’s perspective provides a comprehensive view, already accounting for a significant portion of direct healthcare costs.
Fifth, the lack of TAF-treated inpatients due to the new introduction of TAF limited our ability to collect data through patient interviews to estimate the annual cost of TAF-related treatment. Instead, in estimating the annual cost of health states for patients treated with TAF, we assumed that inpatient treatment among TAF-treated patients is similar to TDF-treated patients. Thus, future researchers could aim to calculate the annual cost of TAF/TDF treatment based on patient surveys (once TAF use becomes more common among inpatients) and explore additional direct non-medical costs and indirect costs.
Sixth, there were certain gaps in estimating costs for each health state due to a lack of disease data in Vietnam. For example, most medical records included in our study only report patients having cirrhosis or not, without specifying whether it was compensated or decompensated cirrhosis. Additionally, during data collection, many medical records lacked information on patients’ HBeAg status, with no record for HBeAg. Due to the inability to collect data on HBeAg- patients, our study therefore only considered the Markov model for CHB patients with HBeAg+. We recognize this as a limitation of the available data for HTA studies that should be addressed in the future. We recommend Bach Mai Hospital for future updates in their clinical diagnosis practice to include complete information on disease stages and patient characteristics.
Lastly, since TAF was incorporated into the Ministry of Health’s treatment guidelines in 2019, its use remains limited across the country. Therefore, Bach Mai Hospital provides a greater opportunity to select CHB patients treated with TAF to gather cost data related to TAF, as it is Vietnam’s first fully comprehensive national general hospital and one of the largest in the country. While the study offers valuable insights into TAF costs for hospitals at a similar level, its limited use means that the findings do not reflect outcomes for lower-tier hospitals. As TAF becomes more widely adopted, future research will have the opportunity to assess this on a larger scale to more accurately represent costs across the broader population.
Conclusion
In the current context, replacing TDF with TAF for lifetime treatment of CHB in HBeAg + patients is not cost-effective from a payer’s perspective, with an ICER of USD 32,090 per QALY gained, which is 8.5 times higher than the current threshold of one-time GDP per capita (USD 3,783). Policymakers should carefully evaluate the inclusion of TAF in the SHI benefit package under these circumstances. Only by reducing the price of TAF by at least 33% could it become a cost-effective option compared to TDF for lifelong treatment of CHB. The cost-effectiveness evidence of TAF versus TDF should be revisited in the near future, once there is updated evidence on the effectiveness of both drugs and more comprehensive cost data related to CHB treatment, especially as TAF usage expands nationwide.
Supplemental Material
Supplemental Material - Cost-effectiveness of tenofovir alafenamide fumarate for treatment of chronic hepatitis B: Evidence from a tertiary hospital in Vietnam
Supplemental Material for Cost-effectiveness of tenofovir alafenamide fumarate for treatment of chronic hepatitis B: Evidence from a tertiary hospital in Vietnam by Tran Dieu Thuy, Nguyen Thu Ha, Do Duy Cuong, Le Van Long, and Nguyen Quynh Anh in Antiviral Therapy
Footnotes
Acknowledgements
The authors extend their sincere gratitude to the Board of Directors at Bach Mai Hospital for facilitating data collection for this research. We are especially grateful to the professors, lecturers, and teaching assistants from the Master’s Program in Public Health – Health Technology track at Hanoi University of Public Health for their insightful feedback and unwavering support, which significantly enhanced the quality of our study.
Ethical Statement
The study complied with the principles of the Ethics Council, Hanoi University of Public Health, and was approved in Decision No. 387/2023/YTCC-HD3 dated August 18, 2023.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
All data supporting the findings of this study are available within the paper and its Supplementary Information.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
