Abstract
Background
Tenofovir alafenamide (TAF) is a new anti-hepatitis B virus nucleotide analogue that can cause dyslipidaemia in AIDS patients, but the effect of TAF on blood lipids in patients with chronic hepatitis B (CHB) is unknown. This study aimed to evaluate the effect of TAF on blood lipid levels in patients with CHB.
Methods
One hundred and twenty-one CHB patients were recruited as TAF group, including 69 treatment-naïve patients and 52 patients with nucleoside/nucleotide analogue experience before TAF treatment. All patients were followed up regularly for 48 weeks. Total cholesterol (TC), low-density lipoprotein cholesterol (LDL-C) and triglyceride (TG) levels and the incidence of dyslipidaemia before and after TAF treatment were compared.
Results
After 48 weeks of TAF treatment, the levels of TC, TGs and LDL-C in TAF group were significantly higher than those in control group. In TAF group, the TC and TG levels were significantly higher than that at baseline. Baseline TC and TGs levels had a significant effect on the incidence of abnormal TC and TG levels after 48 weeks treatment. The LDL-C decreased slightly but not significantly. The proportion of patients with TC abnormalities increased from 20.7% at baseline to 26.3% at week 48, LDL-C abnormalities decreased from 50.4% to 42.5% and TG abnormalities increased from 14.2% to 22.5%. There were no significant differences compared with control group, as well as compared with baseline.
Conclusions
Tenofovir alafenamide treatment mainly affects the TC and TG level in patients with CHB but has little effect on LDL-C.
Introduction
Hepatitis B virus (HBV) infection is currently a serious problem threatening human health. Approximately 257 million people worldwide have chronic HBV infection [1]. In recent years, the ageing trend of the chronic hepatitis B (CHB) population has been obvious, and comorbidities such as hypertension, diabetes mellitus and coronary heart disease have become more common [2–3]. Dyslipidaemia is a metabolic risk factor for hypermetabolism that can lead to an increase in the incidence of chronic diseases, thereby affecting the quality of life and survival rate of patients. Blood lipid elevation may lead to weight gain and obesity and produce long-term adverse outcomes, including metabolic syndrome, development and progression of type 2 diabetes mellitus (T2DM), and cardiovascular diseases. In addition, as a risk factor for hypermetabolism, dyslipidaemia results in an increased risk of liver cancer [4]. Among the current first-line nucleotide analogues against hepatitis B virus, tenofovir disoproxil fumarate (TDF) has been reported to have lipid-lowering effects [5], which is similar to statins [6], but the specific mechanism is unclear. As a modified drug of TDF, tenofovir alafenamide (TAF) has been increasingly applied in patients with CHB. Both TDF and TAF act through the active ingredient tenofovir (TFV). The plasma concentration of the active ingredient is much lower with TAF than with TDF administration; therefore, TAF has less nephrotoxicity than TDF. Unlike TDF, TAF does not have a lipid-lowering effect, instead, it elevates blood lipid levels in the AIDS population. In treatment-naïve patients with human immunodeficiency virus (HIV), compared with TDF, TAF is associated with higher blood lipid levels and a higher demand for lipid-lowering therapy [7]. In patients who have received TDF in a combined antiretroviral regimen, if TDF is replaced with TAF, lipid metabolism is affected due to the loss of the TDF-related statin effect, which leads to an increased incidence of dyslipidaemia [8–9], especially with an increase in low-density lipoprotein cholesterol (LDL-C) [10]. A cohort study suggested that the average total cholesterol (TC), high-density lipoprotein cholesterol (HDL-C), LDL-C and triglyceride (TG) levels were increased 18 months after switching from TDF to TAF [11]. In addition, TAF leads to a substantially greater weight gain than TDF [12–13].
The AIDS population and the hepatitis B population differ in many respects. In the AIDS population, patients lose weight due to the catabolism associated with viral infection, and effective antiviral treatment helps them regain weight [14]. Therefore, distinguishing whether weight recovery is attributable to health recovery or to drug action is difficult. In contrast, in patients with CHB, changes in blood lipids and body weight are not significant.
Considering the side effects of TAF in elevating blood lipids in AIDS patients, clinicians have focused on dyslipidaemia in patients with CHB. However, the effect of TAF on blood lipids in CHB has not been fully explored, and few real-world studies have been conducted. Therefore, whether TAF treatment affects blood lipid levels in CHB patients is unclear. The present real-world clinical study aims to evaluate the effect of TAF on blood lipid levels in patients with CHB.
Methods
Study protocol and study population
This was a prospective, real-world, observational clinical study (ClinicalTrials.gov registration number NCT04195074). The enrolled patients were all treated in the Department of Infectious Diseases of the Third Affiliated Hospital of Sun Yat-sen University between January 2019 and January 2021. The inclusion criteria were as follows: (1) positive serum hepatitis B surface antigen (HBsAg) for 6 or more months, (2) age older than 18 years and (3) agreement to use antiviral monotherapy with TAF. The exclusion criteria were as follows: (1) use of lipid-lowering drugs before the study; (2) decompensated cirrhosis, such as ascites, digestive tract haemorrhage and hepatic encephalopathy; (3) hepatocellular carcinoma (HCC) or other tumours; (4) pregnancy or lactation; (5) other concomitant liver diseases caused by hepatitis C virus (HCV) infection, autoimmune hepatitis, poisoning- or drug-induced liver damage, alcoholic liver disease, or genetic metabolic liver disease; (6) HIV infection or other immunodeficiency disease; (7) organ dysfunction or failure; (8) current glucocorticoid therapy or interferon therapy and (9) an inability to follow the protocol of this study, sign an informed consent form, or regularly return to the hospital for review and follow-up according to the study plan. Treatment-naïve patients using other first-line antiviral drugs were included as control group.
This study followed the ethical guidelines of the Declaration of Helsinki developed by the World Medical Association and the relevant norms and regulations of clinical research and was approved by the Medical Ethics Committee of the Third Affiliated Hospital of Sun Yat-sen University. All patients signed an informed consent form.
Treatment and follow-up
During the 48-week follow-up period, TAF group subjects continuously received oral TAF at the regular clinical dose (25 mg, once daily) for anti-HBV therapy. Subjects in control group received entecavir (ETV, 0.5 mg once daily) or TDF (300 mg once daily). During the follow-up period, no lipid-lowering agents were used unless absolutely necessary. In order to better evaluate the effect of TAF on blood lipids, patients requiring lipid-lowering drugs during the follow-up period were excluded from the statistical analysis. Other liver-protecting and enzyme-lowering drugs such as polyene glycyrrhizin diamine and phosphatidylcholine were used as appropriate. Antihypertensive drugs and hypoglycaemic agents were used normally. All participants returned to the hospital for follow-up visits every 3–6 months after initiation of the medication.
Data collection
Demographic data, body weight, body mass index (BMI) [body weight/height2 (kg/m2)], smoking status and metabolic diseases (cardiovascular disease, hypertension, diabetes mellitus, hyperuricaemia and fatty liver) were collected before TAF treatment. Fasting TC, LDL-C, HDL-C and TG levels were recorded at baseline and after 48 weeks of TAF treatment. Blood lipids were detected by an automatic biochemical analyser (7600 Series, HITACHI, JAPAN). The primary endpoints of our study were the changes in blood lipid levels and the proportion of patients with dyslipidaemia after 48 weeks of treatment. The secondary endpoint was risk factors for dyslipidaemia.
The cardiovascular disease risk level was calculated for each patient according to the guidelines for the prevention and treatment of dyslipidemia in Chinese adults [15]. The specific definitions of risk populations were as follows. The very high-risk population was defined as patients who had been diagnosed with atherosclerotic cardiovascular and cerebrovascular diseases. The high-risk population was defined as (1) patients with LDL-C ≥ 4.9 mmol/L or TC ≥ 7.2 mmol/L; (2) patients with diabetes mellitus, 1.8 mmol/L ≤ LDL-C ≤ 4.9 mmol/L or 3.1 mmol/L ≤ TC ≤ 7.2 mmol/L, and age ≥40 years; (3) Patients with hypertension have three risk factors (smoking, HDL-C < 1.0 mmol/L, male ≥45 years or female ≥55 years), or have two risk factors and 2.6 ≤ LDL-C < 4.9 mmol/L or 4.1 ≤ TC < 7.2 mmol/L; or (4) patients with any two or more of the following risk factors: (a) systolic blood pressure ≥160 mmHg or diastolic blood pressure ≥100 mmHg, (b) non-HDL-C ≥ 5.2 mmol/L, (c) HDL-C <1.0 mmol/L, (d) BMI ≥28 kg/m2 and (e) smoking. The low- and moderate-risk population was defined as patients with hypertension but fewer than two risk factors or no hypertension. For each risk category, guidelines were used to determine the optimal LDL-C targets, which were 1.8 mmol/L for the very high-risk population, 2.6 mmol/L for the high-risk population and 3.4 mmol/L for the low- and medium-risk populations. The upper limits of the reference values of TC and TG in this study were 5.70 mmol/L and 1.92 mmol/L, respectively. A blood lipid level above the reference value was defined as dyslipidaemia.
Statistical analysis
SPSS 21.0 (SPSS, Chicago, IL, USA) was used to analyse the data. Normally distributed data are described as the mean ± standard deviation (x ± s), while nonnormally distributed data are described as the median (M) and quartiles. Count data are expressed as frequencies and percentages (%). Independent sample T test or Wilcoxon signed rank sum test was used to compare quantitative data between TAF group and control group, while Chi-squared test or Fisher's exact text (if an expected value of ≤5 was found) was used to compare categorical data. Changes in lipids were assessed by comparing the blood lipid levels at baseline and after 48 weeks of TAF treatment. Normally distributed data were analysed using the paired t-test, and nonnormally distributed data were analysed using the Wilcoxon signed rank sum test. Categorical variables were analysed with the McNemar test. If the changes in blood lipids were statistically significant, logistic regression analysis was performed with the variables age, sex, BMI, smoking, baseline blood lipids and cardiovascular disease risk level to find the correlates of abnormal changes in blood lipids. All tests were two-sided, and
Results
Baseline characteristics of the patients
In TAF group, a total of 126 eligible patients with CHB (18 patients with compensated cirrhosis) were enrolled, including 72 treatment-naïve patients who received TAF as the initial treatment and 54 nucleoside/nucleotide analogue (Nuc)-experienced patients who received another treatment before TAF treatment. All patients completed 48 weeks of follow-up. In TAF group, five patients with baseline dyslipidemia were treated with lipid-lowering drugs during the follow-up period, including three treatment-naïve patients and two Nuc-experienced patients. In control group, 33 patients were enrolled, including 13 patients treated with ETV and 17 patients treated with TDF, and three patients with baseline dyslipidemia adopted lipid-lowering drugs during the follow-up period. Therefore, 121 cases in TAF group and 30 cases in control group were finally included in the analysis (Figure 1). Among them, 39 (32.2%) patients in TAF group and 10 (33.3%) in control group had one or more metabolic diseases other than dyslipidaemia, including diabetes mellitus, hypertension, hyperuricaemia and non-alcoholic fatty liver disease. In TAF group, 99 (81.8%) were male and 22 (18.2%) were female, and the average age was 41.3 ± 9.4 years. The cardiovascular disease risk level in each patient was calculated according to the ‘2016 Chinese Guidelines for the Management of Dyslipidaemia in Adults’ [15]. A total of 81.0% of the patients were in the low- and medium-risk population, 19.0% were in the high-risk population, and no patients were in the very high-risk population. The demographic and baseline clinical characteristics of the patients are summarized in Table 1. The median blood lipid levels at baseline are shown in Table 2. All baseline characteristics were comparable with those of control group. Flow chart of patient inclusion Baseline clinical characteristics BMI: body mass index; TC: total cholesterol; LDL-C: low-density lipoprotein cholesterol; TGs: triglycerides; ETV: entecavir; TDF: tenofovir disoproxil fumarate; LDT: telbivudine. Changes in blood lipid levels after TAF treatment (mmol/L, median [IQR]) TC: total cholesterol; LDL-C: low-density lipoprotein cholesterol; TGs: triglycerides.

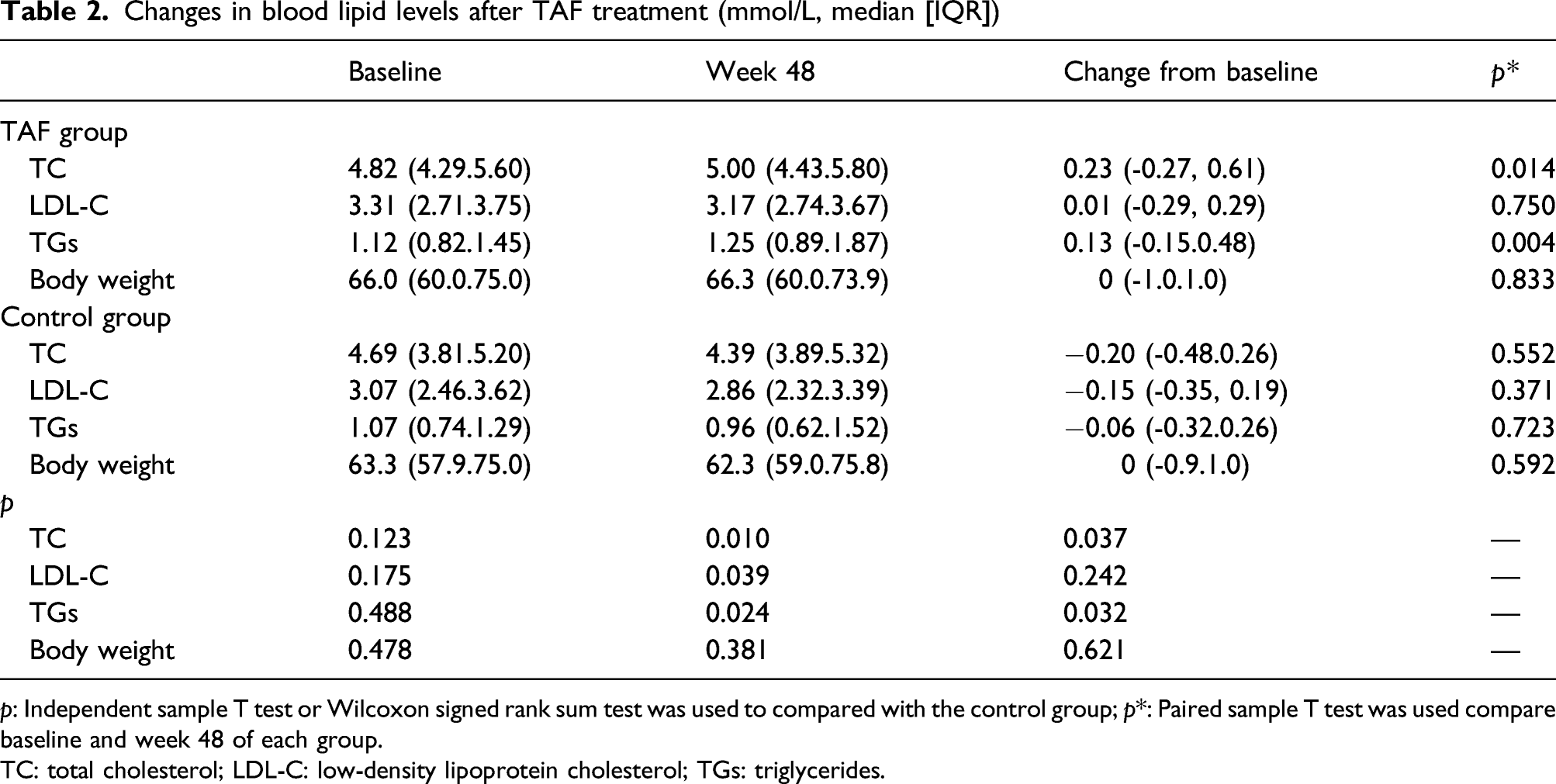
Changes in blood lipid levels and body weight
After 48 weeks of TAF treatment, the levels of TC, TGs, and LDL-C in TAF group were significantly higher than those in control group. In TAF group, the TC and TG levels were significantly higher than that at baseline, increasing by 0.23 (−0.27, 0.61) mmol/L and 0.13 (−0.15, 0.48) mmol/L, respectively. However, no significant change from baseline in the LDL-C concentration was noted, it was decreased but not significantly lower than that at baseline. There were no statistical differences in body weight between the two groups, and no statistical difference was found in each group before and after treatment (Table 2).
Changes in dyslipidaemia rate
After TAF treatment, the proportion of patients with TC abnormalities increased from 20.7% (25/121) at baseline to 26.3% (30/114) at week 48 (an increase of 5.6%), the proportion of patients with LDL-C abnormalities decreased from 50.4% (61/121) at baseline to 42.5% (51/120) (a change of −7.9%), and the proportion of patients with TG abnormalities increased from 14.2% (17/120) at baseline to 22.5% (27/120) (an increase of 8.3%). McNemar’s exact test indicated that the incidence of dyslipidaemia at baseline was not significantly different from that after TAF treatment, Chi-square test or Fisher’s exact test suggested that there were no statistically significant differences when compared with control group (Additional Supplemental Table 1).
After 48 weeks treatment, among patients with normal blood lipid levels at baseline, the incidences of dyslipidaemia were TC 15.2%, LDL-C 11.9% and TGs 15.7% in TAF group, compared with 0%, 11.1% and 7.7% in control group, respectively, the differences were not statistically significant (all
Blood lipid changes after 48 weeks of TAF treatment in patients who received ETV or TDF before TAF treatment
Changes in blood lipid levels from baseline to 48 weeks of TAF treatment in patients who received ETV or TDF before TAF
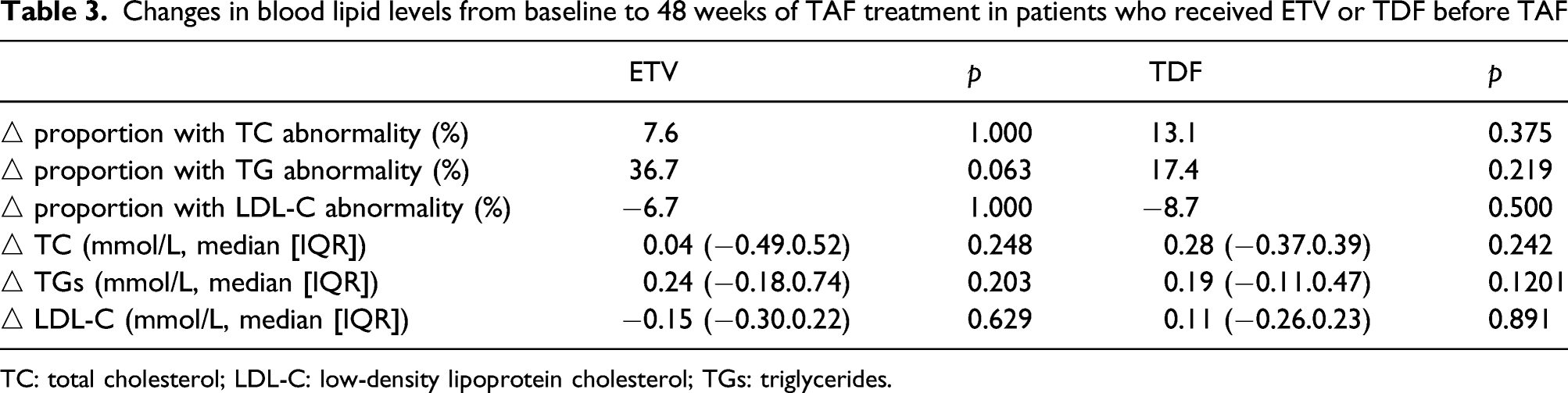
TC: total cholesterol; LDL-C: low-density lipoprotein cholesterol; TGs: triglycerides.
Analysis of risk factors for dyslipidaemia
Analysis of risk factors related to total cholesterol and triglycerides abnormalities after 48 weeks of TAF treatment

*represents the control.
BMI: body mass index; TC: total cholesterol; TGs: triglycerides.
Discussion
Increasing evidence indicates that HBV infection is inversely associated with lipid metabolism and is not conclusively associated with insulin resistance or T2DM and atherosclerosis risk [16]. Therefore, whether serum lipids will increase with HBV elimination after antiviral therapy is worth considering. Moreover, due to the effect of TAF in elevating blood lipids in AIDS patients, this issue warrants greater attention. Our real-world study analysed changes in blood lipid levels in patients with CHB after antiviral therapy. The results showed that compared with control group, serum lipids in TAF group were indeed significantly increased, and 48 weeks of TAF treatment caused some metabolic abnormalities in blood lipids, namely, an increase in the average TC and TG level, but had little effect on LDL-C levels. Our results are partially consistent with those of previous studies suggesting that TAF has little effect on blood lipids, especially TGs. In patients with CHB, the results of a phase 3 clinical trial before the commercialization of TAF [17] suggested that compared with the decrease in the median fasting blood lipid level after TDF treatment, the change in the median fasting blood lipid level after TAF treatment was smaller. Although the proportion of patients with a fasting LDL-C level of grade 3 or above (≥300 mg/dl) was higher after TAF treatment than after TDF treatment, the proportion of patients with an LDL-C level of grade 3 or above did not markedly change from 48 weeks to 96 weeks. In addition, most patients with dyslipidaemia after TAF treatment for CHB had dyslipidaemia at baseline [17–19]. Therefore, compared with that of TDF, the effect of TAF on lipids has been considered to be neutral [17]. In a study of patients with CHB and low-level viremia who switched from ETV treatment to TAF treatment, although TAF treatment resulted in increased fasting LDL-C, fasting glucose, and urinary glucose levels, the increases were mild and temporary and usually occurred in patients with CHB who already had elevated baseline blood lipid concentrations [20]. Some studies have found that TAF can affect blood lipids, but TGs changes are rarely mentioned. In a randomized controlled study of patients with drug-resistant CHB, the average increase in TC, LDL-C and HDL-C levels after 48 weeks of treatment relative to baseline was significantly greater in patients who switched from TDF treatment to TAF treatment than in patients who continued TDF treatment, and the TC to HDL-C ratio slightly but non-significantly decreased in the two groups over time [21]. The decrease in lipid levels in HIV or CHB patients taking TDF is attributed to the TDF-induced increase in plasma TFV [22–23]. In our study, the fasting TC and TG level increased after TAF treatment, and logistic regression analysis showed that this elevation was correlated with the baseline TC and TG level, consistent with previous studies [17–20]. The mechanism and clinical relevance of this TG elevation is still unclear, and further research with longer follow-up times should be conducted to determine whether this effect will increase the risk of cardiovascular diseases. However, the effect of TAF on TGs deserves more attention.
Our study also found that after 48 weeks of TAF treatment, some patients with normal baseline blood lipid levels developed dyslipidaemia, while some patients with dyslipidaemia at baseline became normolipidaemic. Overall, the proportions of hypertriglyceridaemia and hypercholesterolaemia were higher after treatment than at baseline, while the proportion of LDL-C abnormalities was 7.9% lower than that at baseline, although none of these differences was significant. LDL-C or TC has an independent predictive effect on the risk of cardiovascular disease in individuals or populations [24]. In our real-world study, TAF was less likely to be associated with hyper-LDL-C. In particular, the median LDL-C level showed a decreasing trend compared with the baseline level, and the LDL-C level returned to normal in 27.9% of patients with high baseline LDL-C. Therefore, we suspect that the risk of TAF-induced dyslipidaemia and associated cardiovascular disease is low.
We further compared the proportion of patients with dyslipidaemia and blood lipid levels before versus after TAF treatment in patients who received ETV or TDF before TAF treatment. No significant change in blood lipid levels was observed after TAF treatment compared with baseline in patients who received ETV or TDF before TAF, which also differs from the results of a past study [21]. These results further confirm that TAF has little effect on blood lipids in patients who have been treated with nucleoside (nucleotide) analogues. In our study, except for five patients with baseline dyslipidemia who were excluded from treatment with lipid-lowering agents, none of the other patients in the analysis received hypolipidemic therapy, implying that the changes in blood lipids caused by lipid-lowering agents were excluded; therefore, our results better reflect the effect of TAF on blood lipids.
Our study has the following limitations. First, the sample size of our study was not large. Second, the follow-up time was not long. A multicentre, long-term, prospective study is needed. However, as a real-world study exploring TAF treatment in CHB patients and changes in blood lipids, our study is the most detailed thus far. Only further studies with longer follow-up periods can clarify the long-term effect of TAF on the blood lipid profile and the risk of cardiovascular disease in patients with CHB. Clinicians should consider the risk of dyslipidaemia when prescribing TAF and focus on the risk factors for cardiovascular diseases, including BMI, smoking, hypertension, and dyslipidaemia, to reduce the risk of cardiovascular diseases in the elderly population.
In summary, TAF treatment in patients with CHB mainly affects TC and TGs among blood lipids and has little effect on LDL-C. The long-term effect of TAF awaits clarification.
Supplemental Material
sj-pdf-1-avt-10.1177_13596535221082399 – Supplemental Material for Changes in blood lipids in patients with chronic hepatitis B after 48 weeks of tenofovir alafenamide treatment: A prospective real-world clinical study
Supplemental Material, sj-pdf-1-avt-10.1177_13596535221082399 for Changes in blood lipids in patients with chronic hepatitis B after 48 weeks of tenofovir alafenamide treatment: A prospective real-world clinical study by Yeqiong Zhang, Zhipeng Li, Qiumin Luo, Wenxiong Xu, Lu Wang, Shu Zhu, Liang Peng and Chan Xie in Antiviral Therapy
Footnotes
Authors contributions
Liang Peng, Yeqiong, Zhang, Chan Xie designed the study. Yeqiong Zhang, Zhipeng Li, Qiumin Luo, Wenxiong Xu, Lu Wang, Shu Zhu collected the data. Yeqiong Zhang, Zhipeng Li, Qiumin Luo contributed to the statistical analysis. Yeqiong Zhang wrote the first draft of the manuscript. All authors commented on previous versions of the manuscript and read and approved the final manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by grants from the National major science and technology project for the prevention and treatment of AIDS and viral hepatitis (2018ZX10302204-002, 2018ZX10302205-002), Natural Science Foundation of China (No.82070611 and 81873572), Guangzhou Science and Technology Plan Projects (201904010442), Sun Yat-Sen University Clinical Research 5010 Program (2020007 and 2018009), Research project on degree and postgraduate education reform in guangdong province (2018JGXM04), Young teacher training program of sun yat-sen university (16ykpy40), and the Five-Year Plan of Third Affiliated Hospital of Sun Yat-sen University (K00006).
Trial registration
This trial was registered on the Clinical Trials.gov Protocol Registration and Results System (registration number: NCT04195074).
Data availability
The data used to support the findings of this study are available from the corresponding author upon request.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
