Abstract
Introduction
BIC/FTC/TAF showed efficacy and tolerability in randomized trials as a switch strategy in virologically-suppressed people living with HIV. We evaluated its effectiveness in a real-life setting.
Methods
A retrospective monocentric cohort including 431 virologically-suppressed (HIV-RNA <50 copies/ml) people switching to BIC/FTC/TAF in the period 2018–2022 was evaluated. Probabilities of virological failure (VF, i.e.2 consecutive HIV-RNA ≥50 copies/ml or a single HIV-RNA ≥200 copies/ml) and of treatment discontinuation (TD) were estimated by Kaplan–Meier, and predictors of both outcomes were identified through multivariable Cox regression. Analysis-of-variance for repeated measures was used to examine changes in CD4 count and CD4-to-CD8 ratio.
Results
Overall, 16 VF occurred during 22 months of median follow-up time. Estimated probabilities of VF at 1, 2 and 3 years were 2.0% (95% CI 1.04.2%), 2.9% (95% CI 1.5%–5.6%) and 5.5% (95% CI 3.2%–9.2%), respectively. Caucasian ethnicity and a history of previous VF independently predicted VF. TD occurred in 42 cases, predominantly for simplification. One discontinuation due to VF was reported. No predictors of discontinuation were identified. An increase in CD4-to-CD8 ratio over 3 years was evidenced (
Conclusions
BIC/FTC/TAF demonstrated high effectiveness, tolerability and safety.
Introduction
Bictegravir is a new integrase strand transfer inhibitor (INSTI) with a high genetic barrier to the development of HIV-1 resistance, coformulated with the nucleos(t)ide reverse transcriptase inhibitors FTC and TAF in a single-tablet regimen (STR) for the once-daily treatment of HIV-1 infection in adults. 1 Results from randomized trials and real-world have ascertained the long-term effectiveness of BIC/FTC/TAF as first-line regimen in naive people living with HIV.2,3 The fundamental principle of regimen switching is to maintain viral suppression. 4 In randomized trials of virologically-suppressed people switching to BIC/FTC/TAF, a high percentage of participants maintained virological suppression, 5 despite the presence of archived NRTI resistance-associated mutations. 6 . Safety was also shown in different populations and clinical contexts.5,7 Observational studies have also confirmed the efficacy of switching to BIC/FTC/TAF in virologically-suppressed people in clinical practice but with limited follow-up times.8,9
We aimed to evaluate the effectiveness of this regimen in a real-life setting in a third level center in Rome.
Materials and methods
A retrospective, monocentric cohort including virologically-suppressed (HIV-RNA <50 copies/ml) people switching to BIC/FTC/TAF in the period 2018–2022 was evaluated. Patients with no follow-up visits were excluded. Primary endpoints were represented by time to VF (2 consecutive HIVRNA ≥50 copies/ml or a single HIV-RNA ≥200 copies/ml) and TD for any reason. Patients were followed-up from baseline (BL, i.e. time of switching to BIC/TAF/FTC) until both the endpoints and were censored at loss of follow-up (i.e. HIV-RNA determination not performed for at least 1 year) or death. In the analysis concerning time to VF, patients were also censored in case of TD.
Time to VF and TD were analyzed through Kaplan–Meier. Predictors of VF and TD were identified through multivariable Cox regression with stepwise backward selection of covariates (only variables showing an association at a significance level of <0.05 were retained in the final model).
Analysis-of-variance for repeated measures was used to examine secondary endpoints: changes in CD4 count, CD4-to-CD8 ratio, and metabolic profile from baseline up to 36 months. Predictors of variation in CD4, CD4-to-CD8 and metabolic profile at 12 months were analyzed through linear regression.
Results
We analyzed a cohort of 431 people living with HIV: 295 (68.5%) were men, mainly (84.9%), reporting heterosexual intercourses as the risk factor for HIV acquisition (44.3%), with a median age of 55 years and a median time from HIV diagnosis and cumulative ART exposure of 18 and 14 years, respectively. At baseline, more than 50% of study population had a nadir CD4 count <350 cell/μl and about 50% had a zenith HIV-RNA >100,000 copies/ml. At BL, median CD4 count was 615 cell/μl. Overall, 15% of them were people living with HIV and hepatitis C.
Drug resistance-associated mutations at historical genotype (available on 235 patients, 54.5%) were mostly represented by M184 V/I alone (41 cases, 17.5%), TAMs (at least one: 37, 15.7%), both M184 V/I and at least one TAM (25, 10.7%), K65 R (4, 1.7%), Y115 F (1, 0.5%).
Patients mainly switched from another INSTI and NNRTI-based regimen, mostly (82.6% of cases) for simplification of the current regimen.
Patients’ characteristics at baseline.

Abbreviation: MSM men who have sex with men; HCV, hepatitis C; INSTI, integrase strand transfer inhibitor; TAF, tenofovir alafenamide fumarato; FTC, emtricitabine; NRTI, nucleoside reverse transcriptase inhibitor; PI, protease inhibitors; ATV/c, atazanavir/cobicistat; ATV/r, atazanavir/ritonavir; DRV/c, darunavir/cobicistat; DRV/r, darunavir/ritonavir; NNRTI, non-nucleoside reverse transcriptase inhibitor; RPV, rilpivirine; EFV, efavirenz; NVP, nevirapine; DTG, dolutegravir; EVG/c, elvitegravir/cobicistat; RGV, ralvitegravir; 3 TC, lamivudine; LPV, lopinavir.
Overall, 16 VF occurred during 22 months of median follow-up time (2.0 per 100 patients-years follow-up). Estimated probabilities of VF at 1, 2 and 3 years were 2.0% (95% CI 1.0%–4.2%), 2.9% (95% CI 1.5%–5.6%) and 5.5% (95% CI 3.2%–9.2%), respectively. Despite the number of protocol-defined VFs, only one VF led to treatment discontinuation: two people discontinued study treatment for dyslipidemia and subjective intolerance, whereas all other participants re-suppressed while continuing TAF/FTC/BIC (in 9 cases likely after reinforcing treatment adherence).
Caucasian ethnicity (vs others, aHR 0.32, 95% CI 0.11–0.94;
Details of virological failures.
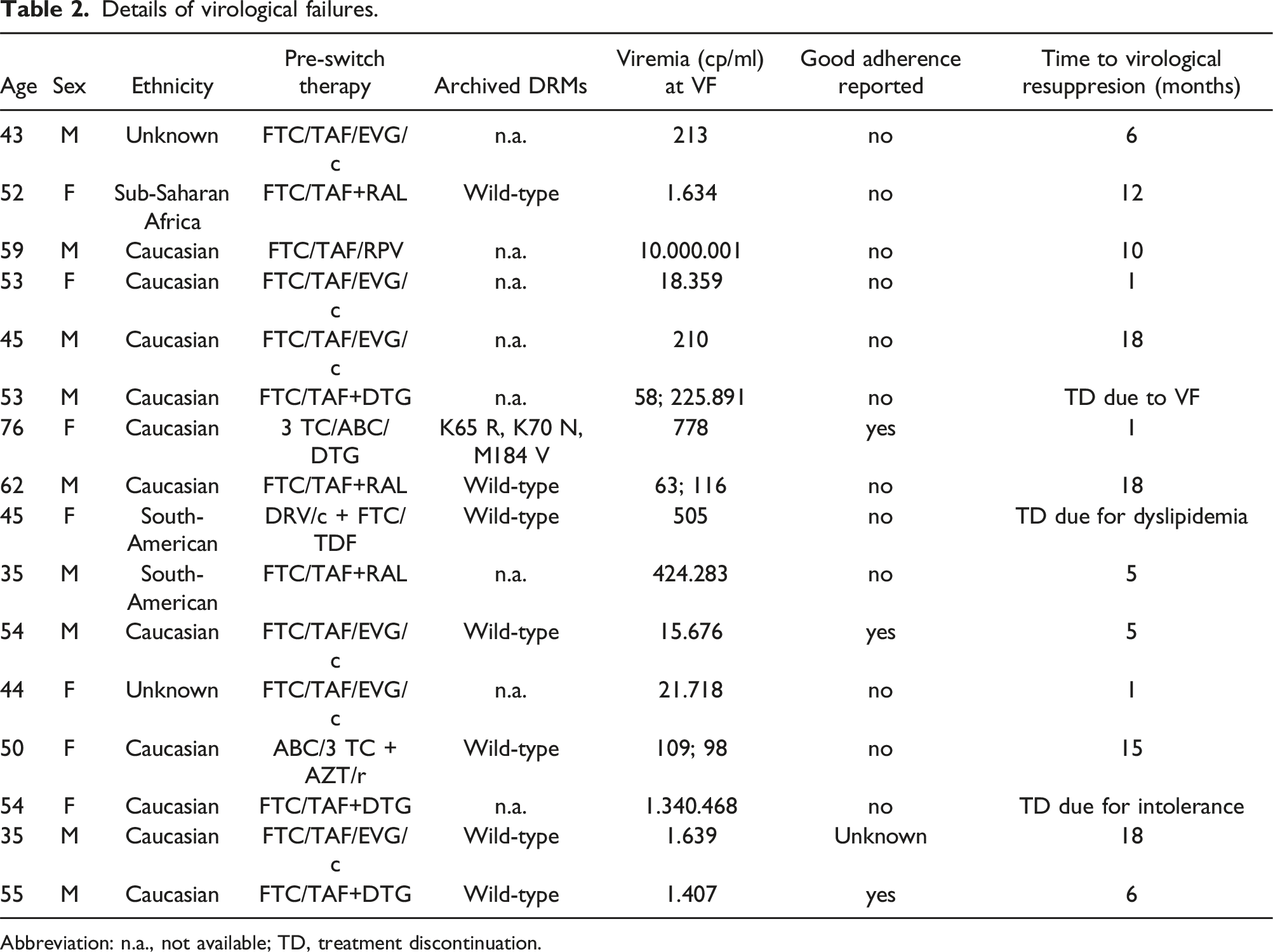
Abbreviation: n.a., not available; TD, treatment discontinuation.
Discontinuation occurred in 42 cases (5.0 per 100 patients-year of follow-up): 21 for simplification to dual therapy (20 3 TC/DTG and 1 cabotegravir/rilpivirine long acting, CAB/RPV LA), 10 for toxicity (concerning central nervous system in 4 cases, including mood alterations, headache, depression, confusion, tremors, insomnia and nightmares, gastrointestinal in 4 cases, liver in 2 cases), 2 for myalgias, 1 for pregnancy, 4 for dyslipidemia, 2 for intolerance, 1 for drug unavailability and 1 for VF.
Regarding the 3 cases of TD among those who had VF: a 53-year-old man experienced VF, then resumed his previous therapy with FTC/TAF + DTG; a 45-year-old woman stopped BIC/FTC/TAF due to dyslipidemia and started doravirine/lamivudine/tenofovir disoproxil fumarate; and a 54-year-old woman discontinued therapy due to subjective intolerance and resumed FTC/TAF + DTG.
Estimated probability of TD at 1, 2 and 3 years were 6.4% (95% CI 4.3%–9.3%), 11.6% (95% CI 8.5%–15.6%) and 12.0% (95% CI 8.9%–16.2%), respectively.
No predictors of BIC/FTC/TAF discontinuation were identified. Particularly the regimen tolerability did not vary based on pre-switch regimen and reason for switch.
An increasing trend in CD4 count over 3 years was evidenced (
An increase in CD4-to-CD8 ratio over 3 years was evidenced (
Total cholesterol decreased over 3 years (
Discussion
The current real-life study is in line with trials’ result about the efficacy of BIC/FTC/TAF as a switch option in maintaining virologic suppression at 22 months of median follow-up time, also offering evidence of sustained immune reconstitution and metabolic safety over time.
In the overall analysis, 42/431 subjects discontinued BIC/FTC/TAF (mainly for simplification), with only one case attributable to VF. In fact, even if an adherence as low as 70% may still sustain virological suppression with BIC/FTC/TAF, 10 most patient experiencing VF in our cohort reported a reduction in pill intake and likely re-suppressed after appropriate physician’s counselling.
Importantly, resistance mutations did not alter the risk of VF. At BL, 17.5% of participants had M184 V/I mutation and 15.7% had at least one thymidine analogue mutation, thus harbouring at least one RAM associated with lower susceptibility to the nucleos(t)ides in the BIC/FTC/TAF regimen. Only one of these showed VF, in line with the analysis conducted by Andreatta et al. 7 which emphasized the regimen’s efficacy in people living with HIV with virologic control, including those with archived NRTI RAMs. Moreover, a recent retrospective review 6 investigating the safety and efficacy of switching to BIC/FTC/TAF, found a prevalence of M184 V of about 10%, with no relevant evidence of impairment of the virologic efficacy.
In recent years, simplified two drug regimens (2DR) have become popular as switch strategies in virologically-suppressed patients. Few data are available on adherence required to assure success of 2DR. 11 A model in vitro showed higher forgiveness of TAF-based triple therapy compared to 2DR, and consistent protection against emergence of drug resistance during simulations of short lapses in adherence, 12 supporting the choice of this strategy in patients with suboptimal adherence. 10 Additionally, findings from the SOLAR study 13 showed that a bi-monthly regimen of CAB/RPV LA was non-inferior compared to daily oral BIC/FTC/TAF but patients on dual therapy with LA experienced virological failure with presence of mutations, unlike the BIC/FTC/TAF group.
An immune reconstitution effects of BIC/FTC/TAF14,15 have been confirmed over 144 weeks, showing notable improvements in CD4+ T cell count and CD4+/CD8 + ratio over time, essential for the long-term health of people living with HIV, along with a balanced metabolic profile. 16 A recent meta-analysis indicated an association between TAF use and weight gain, especially when combined with dolutegravir, with no relevant differences to BIC. 17 In our cohort, an improvement in total cholesterol suggested metabolic benefits over the study duration.
The study has some limitations, including its retrospective design and lack of uniform data collection on important variables, especially treatment adherence. Furthermore, the absence of a control group hampers to get definitive conclusions about treatment efficacy and safety.
Despite its limitations, the study presents a real-life evidence of the durability of BIC/FTC/TAF over 144 weeks in a large cohort of heavily-treatment experienced patients, supporting the robustness of this regimen in the medium term. Studies with longer follow-up will help to confirm these data and to evidence the durability of the regimen over the long term.
Footnotes
Author contributions
Conceptualization R.A.P., F.L., C.T., A.B. and SD.G.; methodology, R.A.P., F.L., D.F., P.F.S., G.B., A.C., SD.G., C.T. and A.B.; software, R.A.P., V.I., R.J.S., A.D.A., F.L., D.F., G.B., A.C., SD.G. and A.B.; validation, R.A.P., F.L. and A.B.; formal analysis, R.A.P., F.L. and A.B.; investigation, R.A.P., F.L., D.F., V.I., R.J.S., A.D.A., P.F.S., G.B., A.C., SD.G., C.T. and A.B.; resources, R.A.P., F.L., SD.G. and A.B.; data curation, R.A.P., F.L. and A.B.; writing—original draft preparation, R.A.P., F.L. and A.B.; writing—review and editing, R.A.P., F.L. and A.B.; visualization, R.A.P., F.L. and A.B.; supervision, SD.G.; project administration, C.T. and SD.G.; funding acquisition, C.T. and SD.G. All authors have read and agreed to the published version of the manuscript.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: A.C. received support for travel to meetings from ViiV Healthcare, A.B. received speakers’ honoraria from ViiV Healthcare, and fees for attending advisory boards from Janssen-Cilag. SD.G. was a paid consultant or member of advisory boards for Gilead Sciences, ViiV Healthcare, Janssen-Cilag, Merck Sharp & Dohme and Bristol-Myers Squibb. All other authors: none to declare.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Medical Grant from Gilead Sciences.
