Abstract
Introduction
Doravirine (DOR) is a novel non-nucleoside reverse transcriptase inhibitor (NNRTI) that retains activity against common NNRTI resistance mutations. In this study, we aimed to investigate the prevalence of DOR resistance mutations compared with that of resistance mutations for other NNRTIs among HIV-1-infected treatment‐experienced and -naïve patients from Poland.
Methods
Resistance to DOR and other NNRTIs was assessed in two datasets: 1760 antiretroviral treatment-naïve HIV-1 patients and 200 treatment‐experienced patients. All 1960 sequences were derived from the patients using bulk sequencing. For resistance analyses, Stanford HIV drug resistance database scores were used.
Results
Overall, DOR resistance was present in 32 patients (1.62%), of whom 13 (0.74%) were naïve and 19 (9.50%) were treatment-experienced. The most common DOR resistance mutations observed among the naïve patients were A98G and K101E (0.2% each), and those among cART-experienced patients were L100I (2.0%), K101E, V108I, H221Y, and P225H (1.5% each). Furthermore, among the naïve patients, less common resistance to DOR (0.7%) compared with that to nevirapine (NVP) (2.1%;
Conclusions
The frequency of transmitted drug resistance to DOR is low, allowing for effective treatment of antiretroviral treatment-naïve patients and rapid treatment initiation. In cART-experienced patients, this agent remains an attractive NNRTI option with a higher genetic barrier to resistance.
Keywords
Introduction
Currently, human immunodeficiency virus (HIV) infections can be effectively controlled using an array of antiretroviral regimens [1]. Antiretroviral drugs effectively reduce the viral load in blood serum to an undetectable level, which decreases the risk of disease progression and prevents HIV transmission [2,3].
In 2018, 86% (72–92%) of people who received treatment across 60 countries were reported to have suppressed viral loads. However, globally, it is estimated that only 53% (42–63%) of all people with HIV infection achieve this success [4]. Virological success rates differ from >90% for West and North European countries to ∼73.1% in Southern Africa [5]. In Europe and Central Asia, the highest virologic efficacy antiretroviral treatment rates of almost 99% was recorded in Denmark, Monaco, Switzerland, and the United Kingdom, whereas the lowest of 40–60% was observed in Albania, Azerbaijan, Bulgaria, Kazakhstan, and Ukraine [6].
Therapeutic success can be hampered by viral drug resistance, which is both a cause and consequence of antiretroviral therapy failure. Studies have estimated the prevalence of acquired and transmitted drug resistance (TDR) in naïve patients [7–10] as approximately 10% in low- and middle-income countries (LMICs) (due to late introduction and low availability of cART-combined antiretroviral therapy) [11,12]. Increasing frequency of TDR to first-line agents is especially important in the context of non-nucleoside reverse transcriptase inhibitors (NNRTIs), which are commonly used for therapy initiation, especially in LMICs [11,13,14]. Additionally, resistance mutations develop in 70–80% of patients with virological failure [15], with NNRTI resistance frequently observed in 50–97% of patients failing this drug class [11,16,17].
Non-nucleoside reverse transcriptase inhibitor–based therapies have long been associated with low genetic barrier to antiretroviral drug resistance with cross-resistance between first-generation NNRTIs such as efavirenz (EFV) and nevirapine (NVP). In antiretroviral treatment guidelines, susceptibility testing is recommended prior to NNRTI treatment initiation [18–21]. The major primary mutations known to affect NNRTI susceptibility include K103N/S, V106A/M, Y181C/I/V, Y188L/C/H, and G190A/S/E [22–24]. Each of these mutations causes intermediate or high resistance to NVP and EFV [25]. Additionally, Y181C/I/V and Y188L/C/H are associated with a 5-fold reduction in susceptibility to rilpivirine (RPV) [26–28].
Accessory mutations such as L100I, K101P, P225H, and M230L usually appear in conjunction with one of the primary NNRTI resistance mutations [29]. Sequences with multiple mutations, including K103N/L100I and K103N/P225H, are frequently observed [30,31]. To increase the NNRTI genetic barrier against resistance, novel drugs of this group have been developed against common NNRTI-resistant viruses (with mutations K101E, E138K, and Y181V). These drugs are suitable for single-daily dosing with improved safety profiles [32–34].
Doravirine (DOR) is an NNRTI most recently approved by the United States Food and Drug Administration in August 2018 and by the European Medicines Agency in November 2018 [35,36]. Efficacy studies showed similar efficacy for DOR (DRIVE-AHEAD, 77.5%; DRIVE-FORWARD, 73.0%), darunavir/ritonavir (66.0%), and EFV (73.6%) based on three drug regimen combinations [37–40]. DOR can be used as a TDF/3TC/DOR single tablet or separately with available NNRTIs. Additionally, DOR has no pre-treatment HIV load limitations, no food restrictions, a reduced number of drug–drug interactions, and a favorable neuropsychiatric profile compared with EFV. Compared with other NNRTIs, DOR retains activity against common NNRTI resistance mutations, including K103N, E138K, Y181C, and G190A [23,41,42]. To date, only few studies have published data on the prevalence of resistance to DOR, which ranged from 0.52% to 1.92% in antiretroviral treatment-naïve patients [37,39,43] and 5.60% (based on the ANRS algorithm) to 16.0% (based on the Stanford database) in treatment-experienced patients [44].
To address the knowledge gap in the frequency of DOR-associated mutations, we aimed to analyze the prevalence of DOR-associated mutations (and those associated with other NNRTIs) in a large dataset of antiretroviral treatment-naïve and -experienced patients with HIV-1 infection living in Poland.
Methods
Study groups and sequencing
The study included a dataset of 1960 sequences, of which 1760 were obtained from newly diagnosed antiretroviral treatment-naïve HIV-1 patients, linked to care in the years 2015–2019 at eight Polish centers (Białystok, Bydgoszcz, Chorzów, Gdańsk, Kraków, Szczecin, Wrocław, and Zielona Góra), and the other 200 were from treatment‐experienced patients treated at the Department of Infectious, Tropical Diseases and Immune Deficiency in Szczecin, Poland. All the sequences were derived from Polish patients of Caucasian origin. Plasma samples from the treatment-naïve patients were collected at care entry (first visit at HIV-treatment center or during in-hospital stay) prior to introduction of antiretroviral treatment and shipped to the genotyping laboratory.
Human immunodeficiency virus RNA extraction and reverse transcriptase (RT) and protease (PR) genotyping were performed using a genotyping assay (Viroseq 2.8 or 2.9, Abbott Molecular, Abbott Park, Illinois, USA) according to the manufacturer’s protocol. Amplicons were obtained using nested PCR and sequenced utilizing the standard BigDye™ technology on the ABI 3500 platform (Applied Biosystems, Foster City, CA). Sequencing was performed in a quality-controlled clinical laboratory at the Department of Infectious, Tropical Diseases and Acquired Immune Deficiency, Pomeranian Medical University in Szczecin, Poland.
Subtyping was performed using genotyping software (REGA genotyping 3.0 tool; http://dbpartners.stanford.edu:8080/RegaSubtyping/stanford-hiv/typingtool) based on the obtained PR and RT sequences.
The analyzed data included sex, age at diagnosis (age at first positive-confirmation test), HIV load at care entry, CD4 lymphocyte count at diagnosis, and subtype.
Drug resistance interpretation
DOR resistance-associated mutations were divided into the following key resistance mutations: V106A/M, Y188FL, P225H, F227L, and Y318F (based on the Stanford HIV drug resistance database, http://hivdb.stanford.edu). A mutation with a score >10–59 was considered to indicate intermediate resistance, whereas a score >60 indicated complete resistance. Variants with scores ≤10 are not shown.
Additionally, the frequency of A98G, L100I, K101E, V108I, Y181I, and H221Y was also investigated owing to their low resistance to DOR. Furthermore, to compare with resistance to other NNRTIs, the following mutations were also assessed: K101H/P, K103N, E138A/G/K/Q, V179L, Y181C, Y188C, G190A, and K238T.
Statistical analysis
Statistical comparisons were performed using the chi2 test for nominal variables. Continuous variables were analyzed using the Mann–Whitney U test for nonparametric statistics. Commercial software (Statistica™ 13.1, Statsoft, Warsaw, Poland) was used for the statistical calculations. Drug resistance in the treatment-experienced and -naïve groups was compared using Fisher’s exact test in R statistical platform, version 4.0.3.
Results
Baseline characteristics of patients
Treatment-naïve patients
Baseline characteristics for cART-unexposed patients based on NNRTI susceptibility.

* Total group size varies based on data availability. Viral load data for 1363 cases, CD4 lymphocyte counts for 955 cases, and age for 1736 cases.
Treatment-experienced patients
Baseline characteristics for cART-exposed patients based on NNRTI susceptibility.

* Total group size varies based on data availability. Viral load data for 170 cases, CD4 lymphocyte counts for 167 cases, and age for 160 cases.
Prevalence of NNRTI DRMs, including DOR
Overall, resistance to any NNRTI was detected in 165 patients (8.42%), of whom 105 (5.97%) were treatment-naïve and 60 (30.0%), treatment-experienced. DOR resistance (at least one DOR DRM) in the total dataset was detected in 32 (1.62%) individuals, of whom 13 (0.74%) were treatment-naïve and 19 (9.50%) treatment-experienced.
The number of DOR resistance sequences with one, two, three, or four mutations among the treatment-naïve patients was four (0.23%), six (0.34%), two (0.11%), and one (0.06%), respectively (Supplementary Table 1). For cART-experienced patients, the numbers were one (0.50%), five (2.50%), nine (4.50%), and three (1.50%), respectively. Notably, one (0.50%) treatment-experienced patient had five mutations (Supplementary Table 2).
The number of sequences with DRMs for other NNRTIs is presented in the next section.
Considering all the mutations for both low and high resistance to DOR, the most prevalent among the antiretroviral-naïve patients were A98G and K101E ( Frequency of doravirine resistance-associated mutations per codon position.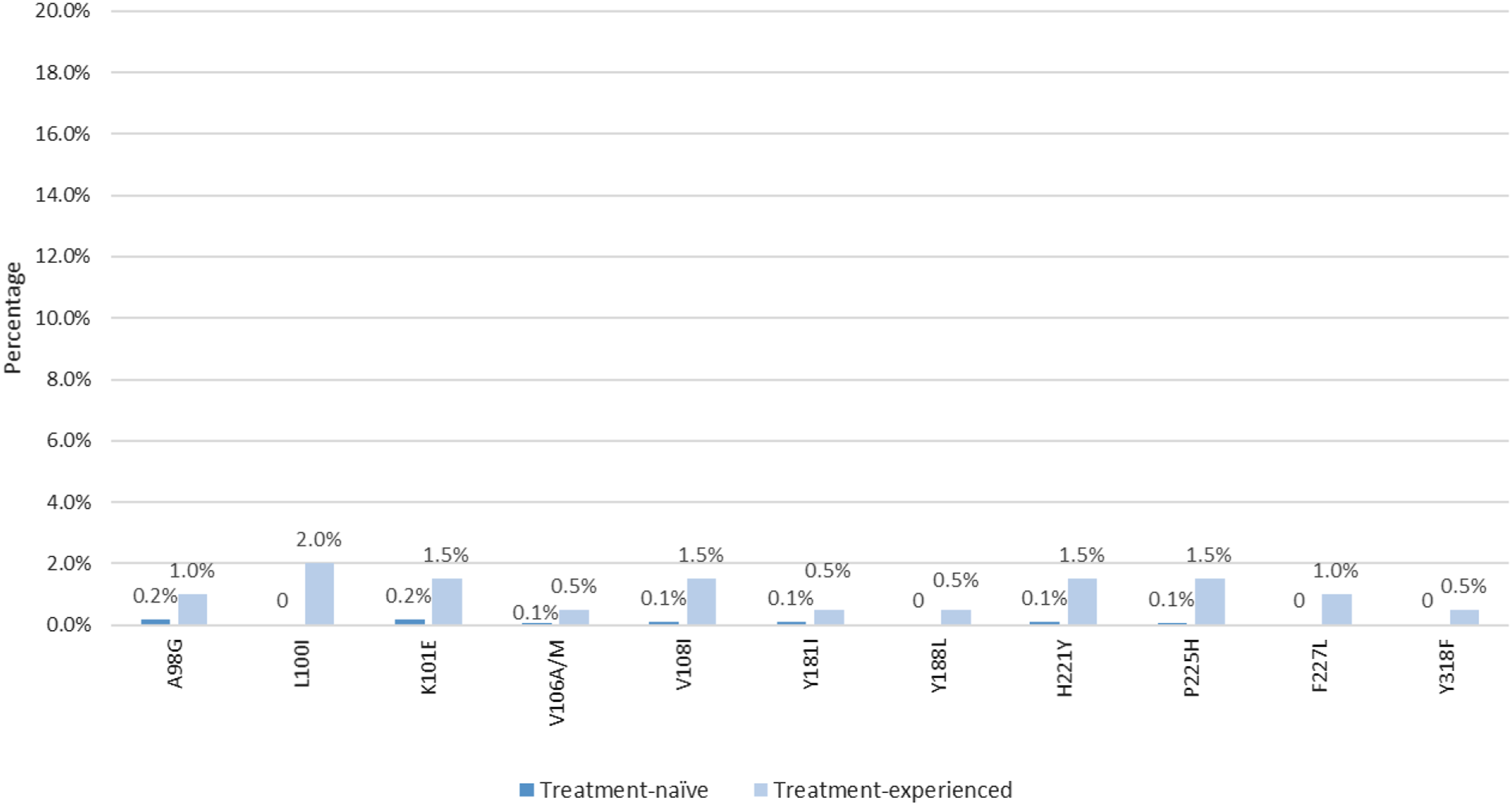
The most common mutations associated with resistance to other NNRTIs in both the treatment-naïve and -experienced patients were E138A/G/K/Q (83 [4.7%] and 15 [7.5%] sequences, respectively), K103N (10 [0.6%] and 36 [18.0%] sequences, respectively), and Y181C/I (5 [0.3%] and 13 [6.5%] sequences, respectively). DRM frequencies for the individual NNRTIs, among antiretroviral-naïve and -experienced individuals, are shown in Supplementary Figures 1–4.
Comparison between number of patients with intermediate and high resistance to DOR and other NNRTIs
Among the antiretroviral-naïve patients, DRM to DOR was less common ( Comparison between number of patients with intermediate and high-level resistance to doravirine and other non-nucleoside reverse transcriptase inhibitors. * 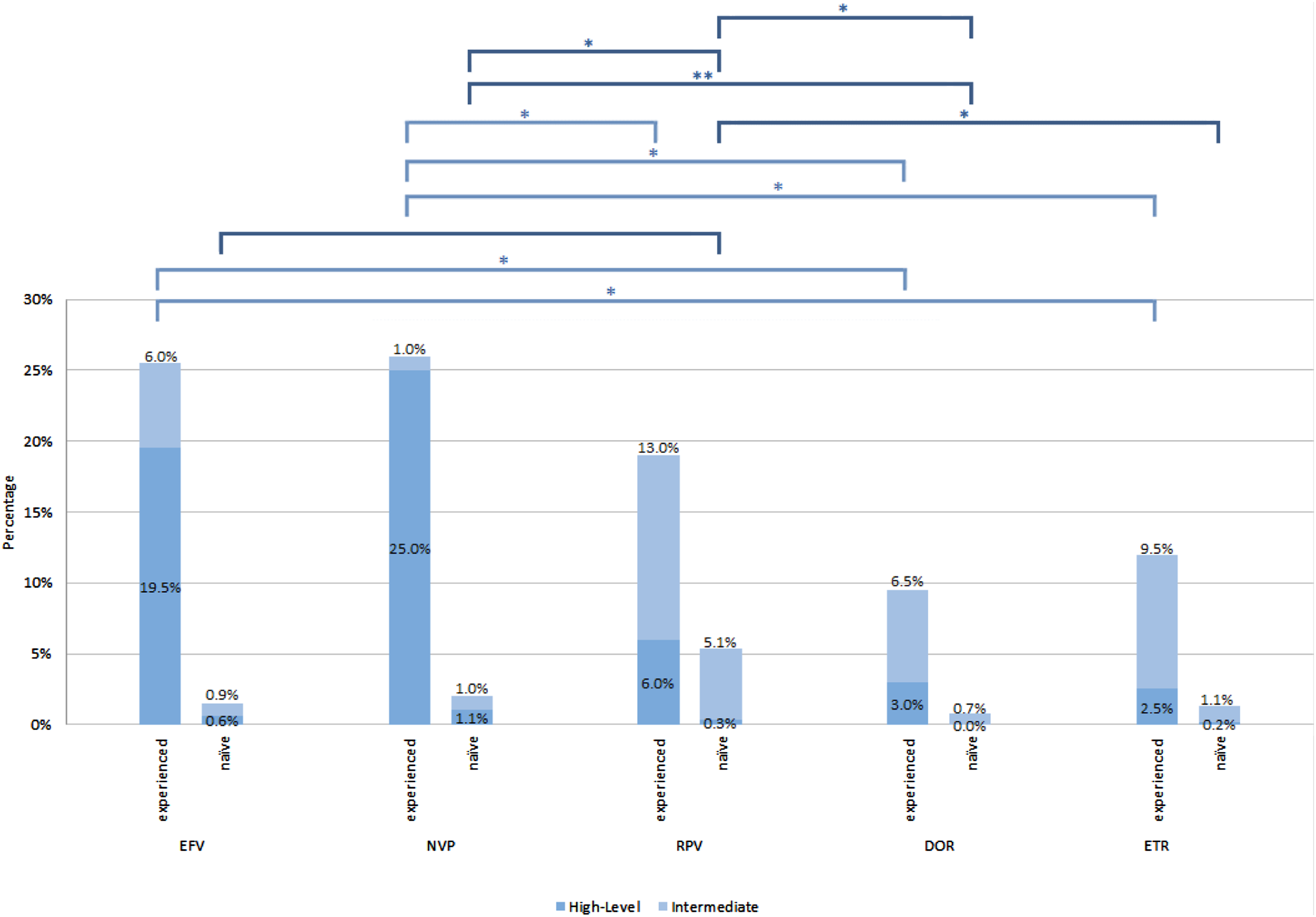
Discussion
The data presented in this paper outline the frequency of DRMs associated with DOR compared with those for other NNRTIs among both treatment-naïve and -experienced individuals from Poland, in the years 2015–2019. This is the first Polish study on the frequency of DOR resistance, with the inclusion of a large dataset of sequences. Analyses were based on the Stanford HIV resistance database, as DOR resistance mutations were not included in the surveillance algorithm [45] and confirmed that the frequency of mutations reducing DOR susceptibility was exceptionally low (<2%), especially among treatment-naïve patients (0.74%), being 9.5% for treatment-experienced patients. This favorable resistance profile observed from real-world data confirms the possibility of rapidly introducing DOR as the initial antiretroviral regimen and eliminating the necessity of baseline genotyping prior to cART.
In treated patients, the occurrence of resistance to DOR is infrequent. In the DRIVE-FORWARD clinical trial that included 382 naïve patients, only two (0.52%) patients failed treatment owing to the emergence of NNRTI resistance mutations, with selection of the V106I/H221Y/F227C and V106A/P225Y/H variants [46]. Moreover, in the DRIVE-AHEAD study, among 364 patients, seven patients (1.92%) with protocol-defined virological failure had DOR resistance mutations (Y188L, Y318F, V106I+F227C, V106I+H221Y+F227C, F227C, V106A+P225H+Y318F, and V106M/T+F227C/R) [39]. Similar to the data shown in our study, these DOR resistance mutations were irregularly present with the most common V106A mutation in the DRIVE trials, present in only one (0.06%; V106A) treatment-experienced patient (the V106I mutation was present in 71 naïve [4.03%] and five cART-experienced patients [2.50%], but owing to its low scoring in the Stanford database, it was not analyzed in the current study).
Moreover, Soulie et al. (2018) reported that DOR resistance was present in 1.4% (
In a different study by Soulie et al. [44] conducted on a dataset of 9199 sequences, among treatment-experienced patients, the prevalence of sequences associated with DOR resistance was 16.0% (
To conclude, DOR resistance mutations associated with virological failure were infrequent among antiretroviral treatment-naïve (99.3% of patients with complete DOR susceptibility) and -experienced (90.5% of sequences with DOR susceptibility) patients. Both groups also reported lower frequency of resistance to DOR than to other NNRTI drugs, which justifies the use of DOR in treatment-naïve groups and represents the attractive NNRTI option related to the higher genetic barrier to resistance in cART-experienced patients.
The limitation of this study is related to the fact that analysis of the DOR resistance mutations was based only on the Stanford HIV drug resistance database; as a new agent, it was not included in the resistance interpretation algorithms; also for the treatment-experienced group, we did not collect the full history of antiretroviral drug use—such analysis is in progress.
Supplemental Material
sj-pdf-1-avt-10.1177_13596535211043044 – Supplemental Material for Low prevalence of doravirine-associated resistance mutations among polish human immunodeficiency-1 (HIV-1)–infected patients
Supplemental Material, sj-pdf-1-avt-10.1177_13596535211043044 for Low prevalence of doravirine-associated resistance mutations among polish human immunodeficiency-1 (HIV-1)–infected patients by Kaja Scheibe, Anna Urbańska, Paweł Jakubowski, Maria Hlebowicz, Monika Bociąga-Jasik, Aleksandra Raczyńska, Aleksandra Szymczak, Bartosz Szetela, Władysław Łojewski and Miłosz Parczewski in Antiviral Therapy
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The study was funded by the National Science Centre project UMO-2018/30/E/NZ6/00696
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
