Abstract
Introduction:
Endothelial dysfunction is a hallmark of sepsis pathophysiology, driven by inflammation, oxidative stress, and endoplasmic reticulum (ER) stress, leading to vascular abnormalities and organ failure. Niban, a multifunctional cellular stress response protein, regulates endothelial homeostasis, with its phosphorylation playing a critical role in mitigating ER stress. We hypothesized that sepsis-induced endothelial dysfunction in the aorta is driven by reduced Niban phosphorylation, which increases ER stress, but can be restored by NiPp, a cell-permeant phosphopeptide mimetic designed to replicate phosphorylated Niban.
Methods:
Rats were subjected to a cecal slurry model of polymicrobial sepsis. Molecular markers of ER stress and Niban phosphorylation were analyzed using Western blot, and the vascular reactivity of septic aortae was assessed in muscle baths. Ex vivo NiPp treatment on septic aortic dysfunction was also evaluated. The effects of ER stressors or circulating sepsis mediators, including lipopolysaccharides, interleukin-1β, and cell-free hemoglobin, on endothelial function in healthy aortic rings were tested with or without NiPp.
Results:
Septic aortas exhibited reduced Niban and endothelial nitric oxide synthase phosphorylation, as well as increased glucose-regulated protein 78 levels. Ex vivo treatment with NiPp improved endothelial dysfunction in septic rat aortas. Moreover, NiPp improved endothelium-dependent relaxation in aortic rings exposed to the ER stressor or any of the circulating sepsis mediators ex vivo.
Conclusion:
Together, these findings highlight reduced Niban phosphorylation as an important driver of vascular endothelial dysfunction during sepsis, and suggest a mechanistic link between sepsis, reduced Niban phosphorylation, and heightened ER stress.
Keywords
Introduction
Endothelial dysfunction is one of the defining features of sepsis pathophysiology. The endothelium is a monolayer of cells that lines the inner surface of the vasculature and is essential for maintaining vascular homeostasis, regulating blood flow, inflammation, as well as tissue oxygenation.1 –5 During sepsis, these critical functions become impaired and the resulting endothelial dysfunction, characterized by reduced vascular relaxation and increased permeability, triggers a cascade of tissue damage and organ dysfunction.1,2,5 Although inflammation and oxidative stress are widely recognized as key drivers of endothelial dysfunction in sepsis,3,6 the molecular mechanisms underlying endothelial resilience and repair remain poorly understood. This knowledge gap has hindered the development of targeted therapies aimed at preserving vascular function to prevent vasoplegia in sepsis. 5
There are several well-established mediators of endothelial injury in sepsis, including pro-inflammatory cytokines, bacterial endotoxins, and various damage-associated molecular patterns (DAMPs).1 –9 The pro-inflammatory cytokine, interleukin-1β (IL-1β), can drive endothelial activation and inflammation, and lipopolysaccharides (LPS) can promote oxidative stress and inflammation through Toll-like receptor pathways.1 –3 Additionally, cell-free hemoglobin (CFH), a DAMP released during red blood cell lysis, can exacerbate endothelial injury by generating reactive oxygen species (ROS) and amplifying inflammatory cascades.8 –10 These mediators likely work in combination during sepsis to cause vascular injury, but the cellular stress response mechanisms that dictate endothelial dysfunction and repair under septic conditions remain incompletely understood.
Recent evidence highlights the importance of the endoplasmic reticulum (ER) stress response in the development of endothelial dysfunction during sepsis.11 –13 The ER stress response is a cellular defense mechanism triggered by protein misfolding and metabolic disturbances, conditions frequently observed during sepsis.11,14 The unfolded protein response (UPR), a key arm of the ER stress pathway, aims to restore cellular homeostasis by upregulating molecular chaperones such as glucose-regulated protein 78 (GRP78) and inhibiting protein synthesis through phosphorylation of eukaryotic initiation factor 2 (eIF2α).11 –16 However, under prolonged or dysregulated ER stress, these protective mechanisms can fail, exacerbating cellular dysfunction and vascular injury.11,14 Notably, heightened ER stress, marked by increased blood levels of GRP78, has been observed in septic mice and patients.12,13 Yet, the precise relationship between aortic GRP78 levels and endothelial dysfunction during sepsis remains unclear.
A protein of growing interest in the context of ER stress is Niban, a multifunctional protein implicated in cellular stress adaptation.4,16,17 Niban is thought to play a role in the ER stress response by modulating protein folding and degradation pathways, which are critical for restoring cellular homeostasis under conditions of stress.4,16,17 Additionally, Niban has been associated with the regulation of mitogen-activated protein kinase (MAPK) signaling, plausibly acting as an endogenous kinase inhibitor. 4 Specifically, phosphorylation of Niban at serine 602 plays a protective role in mitigating the effects of ER stress in UV-induced stress responses and in various vascular injury models.4,18 Expression profiling has further demonstrated the presence of Niban in vascular tissues and endothelial cells, supporting its potential to regulate vascular function. 4 Vasoplegia is a hallmark of sepsis that is attributed to vascular dysfunction, notably ‘nonresponsive’ contractile agonists and endothelial impairment, at least in part due to oxidative and inflammatory stress. 5 Given that Niban is involved in ER stress responses 16 and that it plays a role in response to vascular injuries,4,18 there is a strong rationale for studying the specific role of Niban in sepsis-induced endothelial dysfunction. This represents a critical gap in our understanding of the cellular mechanisms by which endothelial responses to sepsis-induced stress occurs, and raises the possibility that modulating Niban phosphorylation offers a novel therapeutic strategy for mitigating vascular dysfunction in sepsis.
To address this knowledge gap, we developed NiPp, a novel cell-permeant phosphopeptide mimetic designed to replicate the functional effects of phosphorylated Niban. 4 As a cell permeant peptide, NiPp can cross cell membranes, allowing it to recapitulate the natural interaction with intracellular targets that are normally modulated by phosphorylated Niban.4,19 Thus, NiPp serves as an innovative tool to investigate the role of Niban phosphorylation in endothelial homeostasis under pathological conditions such as sepsis. To date, no studies have examined the changes in Niban activity or phosphorylation during sepsis, nor their impact on vascular endothelial dysfunction therein. We hypothesized that sepsis-induced vascular endothelial dysfunction is driven in part by reduced Niban phosphorylation, which increases ER stress, but can be restored by NiPp.
Methods
Materials
Unless otherwise specified, all reagents and chemicals were obtained from Sigma-Aldrich (St Louis, MO, USA). The synthesis and design of NiPp followed previously established protocols.
4
Briefly, NiPp (YARAAARQARA
Animals and aorta isolation
All animal procedures were conducted in accordance with the National Institutes of Health guidelines for the care and use of laboratory animals and were approved by the Vanderbilt Institutional Animal Care and Use Committee. Sprague-Dawley female rats (2–4 months old) were obtained from Charles River (Wilmington, MA, USA) and housed in the Vanderbilt University Medical Center (VUMC) animal facility under temperature-controlled conditions with a 12:12-hour light/dark cycle, with unrestricted access to standard chow and water for a minimum of 1 week prior to experimentation. Thoracic aortic tissue was immediately isolated for experimental use after CO2 euthanasia.
Measurement of vascular reactivity
Isolated rat aortae were cleaned of perivascular fat, cut into rings measuring 1–2 mm in thickness, and suspended in an organ bath containing bicarbonate buffer (120 mM sodium chloride, 4.7 mM potassium chloride, 1.0 mM magnesium sulfate, 1.0 mM monosodium phosphate, 10 mM glucose, 1.5 mM calcium chloride, 25 mM sodium bicarbonate, pH 7.4). The buffer was equilibrated with 95% O2/5% CO2 and maintained at 37°C.
Aortic rings were then set to a resting tension of 1 g for 1 hour, manually stretched to three times their resting tension, and returned to the initial tension for an additional hour to establish the optimal force–tension relationship, as previously described.3,11 Tissue viability was confirmed by exposing the rings to 110 mM potassium chloride (KCl; replacing sodium chloride in the buffer) before further experimentation.
Phenylephrine (PE; 1–5 × 10−7 M) was used to induce contraction, achieving approximately 60–70% of the maximal response to KCl. Endothelium-dependent and -independent relaxation responses were evaluated by exposing PE-precontracted rings to increasing concentrations of carbachol (CCH; 10−8 to 10−5 M) and sodium nitroprusside (SNP; 10−10 to 10−6 M), respectively. Force generation was recorded using a Radnoti force transducer (model 159901A; Radnoti LLC, Covina, CA, USA) integrated with a PowerLab data acquisition system and Chart software (AD Instruments Inc., Colorado Springs, CO, USA). Stress values were normalized to the tissue’s length and weight, 3 and relaxation responses were expressed as a percentage of the maximal PE-induced contraction, which was set to 100%.
Ex vivo treatment with circulating septic mediators
To assess the effects of individual sepsis mediators, aortic rings were incubated for different time periods as indicated in bicarbonate buffer alone or in the presence of IL-1β (50 ng/mL; LifeSpan Bioscience, Seattle, WA, USA), bacterial endotoxin LPS (1 µg/mL), or CFH (Hb3+; 20 1 mg/mL; Sigma) with or without NiPp (100 μM) or Scr3NiPp (100 μM). After incubation, endothelium-dependent and -independent relaxation responses were measured in PE-precontracted rings.
Cecal slurry (CS) polymicrobial sepsis model
To prepare cecal slurry (CS), female donor rats (2–4 months old; Charles River) were euthanized with CO2 and their cecal contents were collected, pooled, resuspended in 5% dextrose (330 mg/mL), and filtered through a 70-µm cell strainer. To minimize variability, a single large batch of CS was prepared, aliquoted, and stored at −80°C until use. Polymicrobial sepsis was induced in female rats (2–4 months old; Charles River) by intraperitoneal injection of CS (1.5 g/kg). Rats also received 30 mL/kg of 0.9% saline solution for fluid resuscitation and were euthanized via CO2 at 24 hours post-CS injection. Sham rats did not receive any intraperitoneal injections prior to euthanasia. Aortic tissues were immediately isolated for experimental use after euthanasia.
Western blot analysis
Aortic tissues were snap-frozen in liquid nitrogen and homogenized for protein extraction using modified RIPA buffer (Thermo Scientific, Waltham, MA, USA). Extracted proteins were separated by SDS-PAGE and transferred to nitrocellulose membranes. Membranes were blocked at room temperature for 1 hour using Intercept Blocking Buffer (Li-Cor Biotechnology, Lincoln, NE, USA), followed by overnight incubation at 4°C with primary antibodies targeting phosphorylated and total proteins: Niban (ProteinTech, Rabbit, 21333-1AP), GRP78 (ENZO Life Sciences, Rabbit, ADI-SPA-768), eIF2α (Cell Signaling, Rabbit, 2103), phosphorylated eIF2α (p-eIF2α) (Cell Signaling, Mouse, 9721), p38 MAPK (Cell Signaling, Mouse, 9228), phosphorylated p38 (p-p38) MAPK (Cell Signaling, Rabbit, 9211), endothelial nitric oxide synthase (eNOS) (Cell Signaling, Mouse, 5880), and phosphorylated eNOS (p-eNOS) (EMD Millipore, Burlington, MA, USA; Rabbit, 07-428). Polyclonal antibodies against phosphorylated Niban (p-Niban) were custom-generated by the Vanderbilt Antibody and Research Core. Rabbits were immunized with a synthetic peptide containing the phosphorylated serine 602 sequence conjugated to KLH, and antibodies were subsequently purified from rabbit serum. After primary antibody incubation, membranes were treated with IRDye-labeled secondary antibodies (Li-Cor Biotechnology) and visualized using the Odyssey Infrared Imaging System. Protein band intensities were normalized to glyceraldehyde 3-phosphate dehydrogenase (GAPDH) (Millipore, Mouse, MAB374), and phosphorylation levels were expressed as the ratio of phosphorylated protein to total protein.
Statistical analysis
Vascular reactivity data were obtained as the average of at least two rings per animal. Data were analyzed using GraphPad Prism 10 and presented either as individual values or as mean ± SD. Comparisons between two experimental groups were performed using the Mann–Whitney test, while paired t-tests were used for experiments with dependent (matched) pairs. Repeated two-way ANOVA with appropriate post hoc tests were used to determine differences among multiple unpaired or dependent (matched) samples from the same animal. Statistical significance was defined as p < 0.05.
Results
Polymicrobial sepsis causes vascular dysfunction
Induction of sepsis by injection of CS in rats impaired functional viability, PE-induced contraction, and endothelium-dependent relaxation of the aortas compared with sham rats (EC50 [CCH]: sham = 9.696 × 10−8 M vs CS = 1.851 × 10−7 M, p = 0.04) (Figure 1A–C). However, CS injections did not alter endothelium-independent relaxation compared to sham rat aortas (EC50 [SNP]: sham = 1.556 × 10−9 M vs CS = 4.374 × 10−9 M, p = 0.028) (Figure 1D).
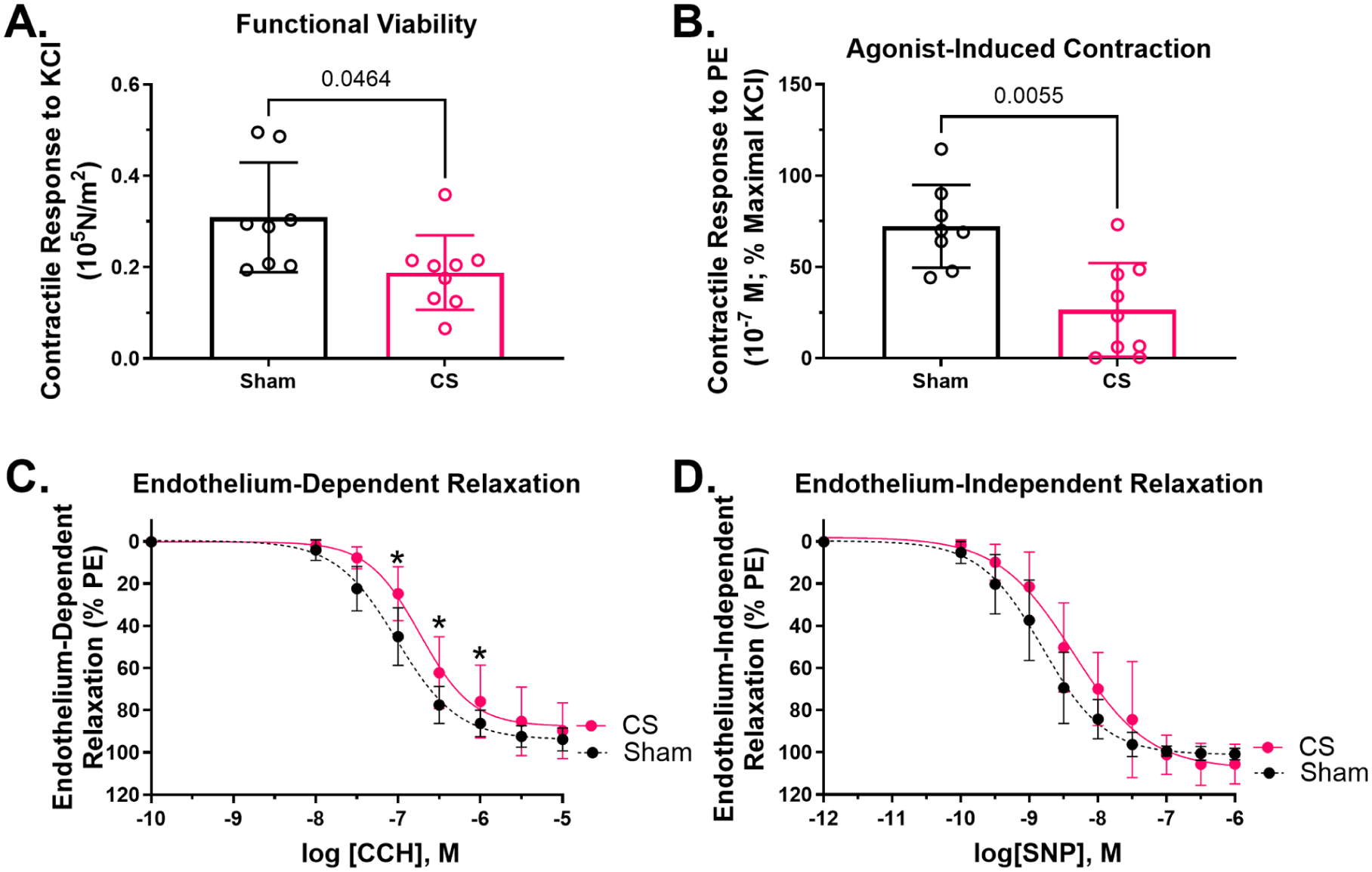
Impaired aortic functional viability, PE-induced contraction, endothelium-dependent relaxation, and endothelium-independent relaxation associated with a 24-hour septic insult (CS; 1.5 g/kg) in rats. Septic rat aortas had impaired functional viability
Sepsis reduces Niban phosphorylation but promotes endoplasmic reticulum (ER) stress in vivo
To determine if sepsis induced changes in Niban phosphorylation or ER stress in aortas, protein levels of p-Niban and ER stress markers were measured. Aortas isolated from CS-injected rats had lower levels of phosphorylated Niban compared to aortas isolated from sham rats (Figure 2D). In addition, GRP78 protein levels were higher in septic rat aortas compared to sham rat aortas (Figure 2E). Although not significant, septic rat aortas also had numerically higher p-eIF2α levels compared to sham rats (Figure 2F).
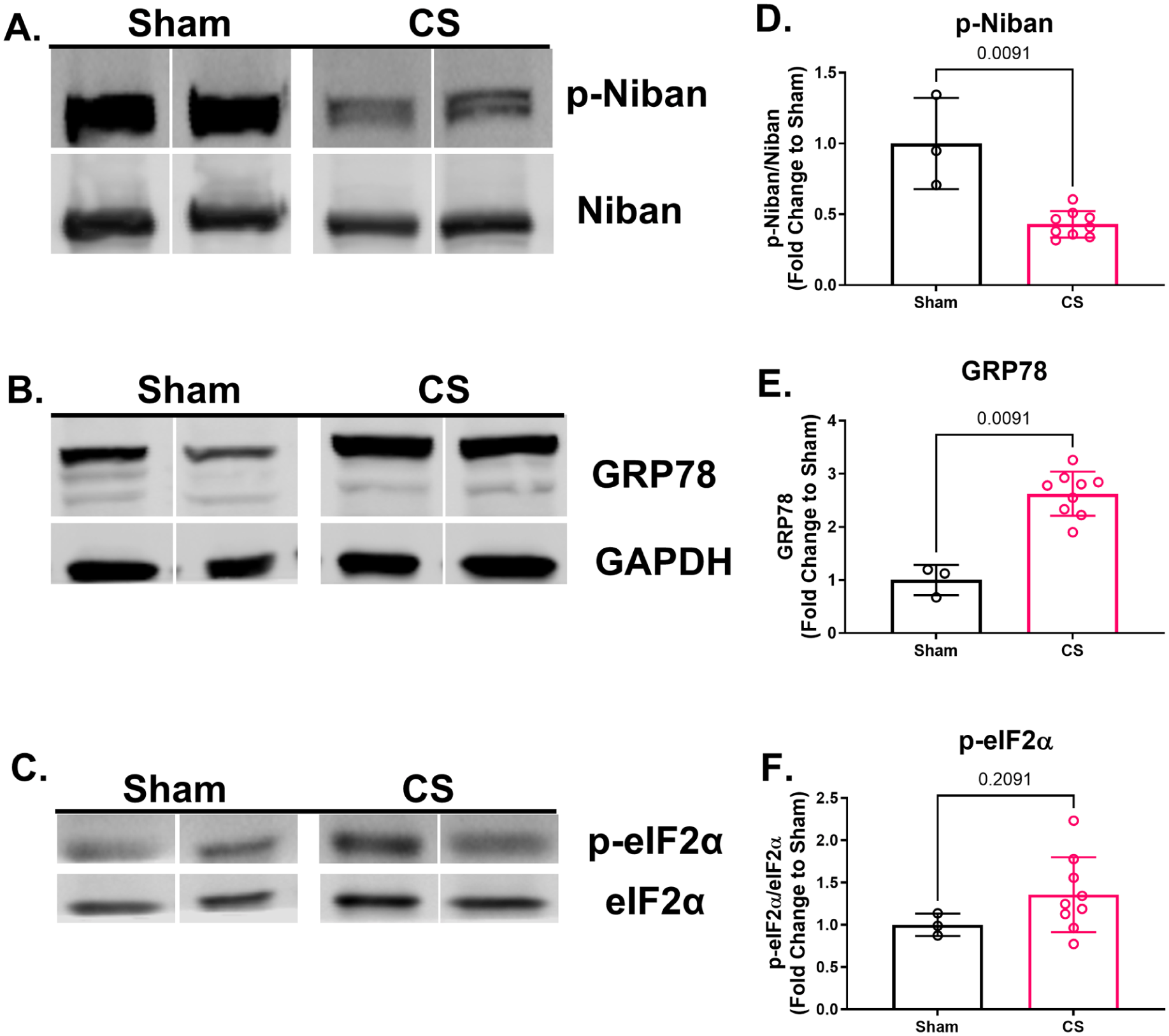
Reduced Niban phosphorylation and increased endoplasmic reticulum stress proteins associated with a 24-hour septic insult (CS; 1.5 g/kg) in rat aortas. Representative Western blots for p-Niban and Niban
Sepsis reduces phosphorylation of endothelial nitric oxide synthase (eNOS) but not p38 mitogen-activated protein kinase (MAPK) in vivo
To determine if CS-induced sepsis leads to alterations in p-eNOS or p38 MAPK, like other vascular injury models reported previously, 4 protein levels of p-eNOS and p-p38 MAPK were measured. Levels of p-eNOS were lower in septic rat aortas compared to sham rat aortas (Figure 3). However, no differences were observed in p-p38 MAPK levels between sham and septic rat aortas at 24 hours post-CS injection (Supplemental Figure S1).

Impaired phosphorylation of eNOS in rat aortas associated with a 24-hour septic insult (CS; 1.5 g/kg). Representative Western blots for p-eNOS and eNOS
Ex vivo NiPp mitigates vascular endothelial dysfunction induced in vivo by CS
To examine the effects of NiPp on septic aorta with endogenously impaired endothelial function and ER stress, aortas from septic rats were treated with NiPp in the muscle bath for 2 hours prior to measurement of vascular reactivity. NiPp treatment improved endothelium-dependent relaxation in aortas isolated from CS-injected rats (EC50 [CCH]: CS = 1.858 × 10−7 M vs CS + NiPp = 1.381 × 10−7 M, p = 0.15; Figure 4A). Post hoc paired comparisons were statistically significant at 5 × 10−8, 10−7, and 5 × 10−7 M (p = 0.0007, 0.0002, and 0.003, respectively; Figure 4B). However, NiPp treatment did not affect functional viability, PE-induced contraction, or endothelium-independent relaxation in aortas isolated from CS-injected rats (EC50 [SNP]: CS = 4.374 × 10−9 M vs CS + NiPp = 4.285 × 10−9 M, p = 0.23; Figure S2).
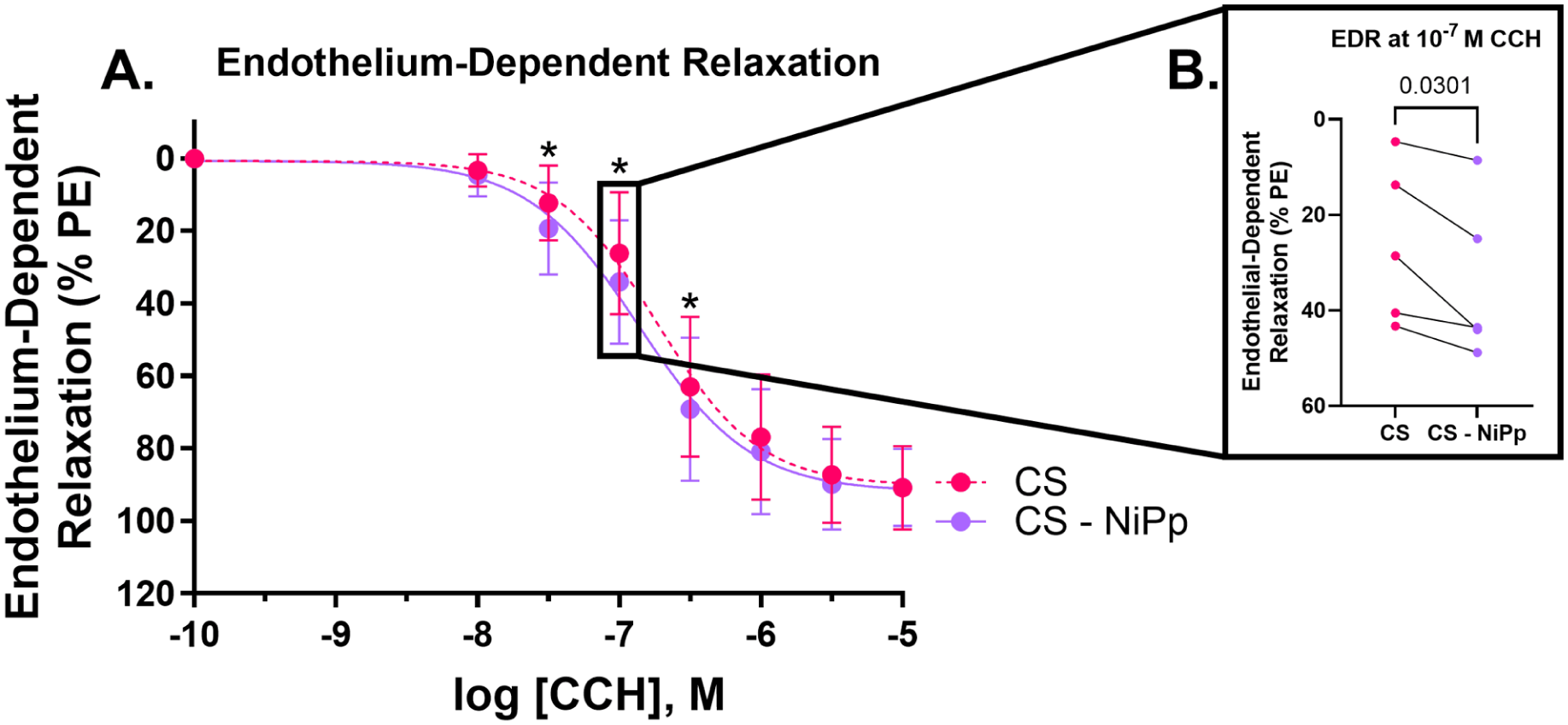
Effect of ex vivo NiPp treatment on impaired EDR of isolated aortas following a 24-hour septic insult (CS; 1.5 g/kg) in rats. Although treatment of PE-contracted aortas isolated from septic rats with NiPp did not alter the average endothelium-dependent relaxation (%) across concentrations of CCH
Circulating septic mediators induced vascular endothelial dysfunction ex vivo
Exposure of isolated rat aortas to a bacterial endotoxin, LPS, impaired endothelium-dependent relaxation compared to untreated control aortas (Figure 5A). The exposure of isolated rat aortas to CFH, a DAMP and oxidative stress mediator, also impaired endothelium-dependent relaxation compared to untreated control aortas (Figure 5B). Similarly, exposing isolated rat aortas to an inflammatory insult via IL-1β impaired endothelium-dependent relaxation compared to untreated control aortas (Figure 5C & 5D).
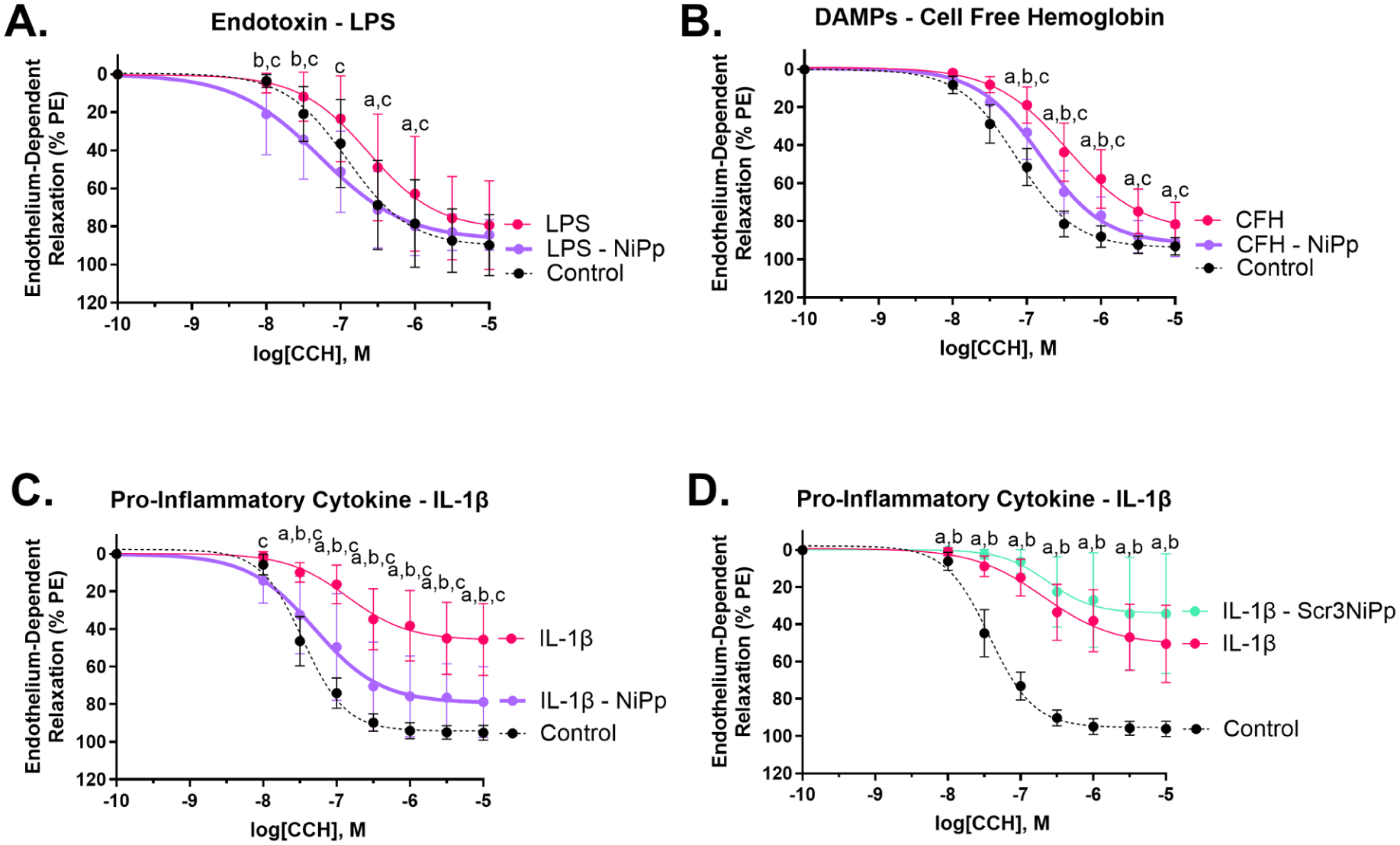
Effect of NiPp and Scr3NiPp on endothelium-dependent relaxation impaired by individual septic mediators. Treatment of PE-exposed rat aortas with LPS (1 μg/mL)
NiPp mitigates vascular endothelial dysfunction induced by sepsis mediators ex vivo
To determine whether increasing Niban phosphorylation mitigates endothelial injury induced by circulating septic mediators, isolated rat aortas were treated with LPS, IL-1β, and CFH in the presence of NiPp. Cotreatment of NiPp mitigated the effects of all tested mediators on endothelium-dependent relaxation (EC50 [CCH]: LPS = 2.356 × 10−7 M vs LPS + NiPp = 5.344 × 10−8 M, p = 0.007; IL-1β = 1.411 × 10−7 M vs IL-1β + NiPp = 4.986 × 10−8 M, p = 0.018; CFH = 3.526 × 10−7 M vs CFH + NiPp = 1.547 × 10−7 M, p = 0.002; Figure 5A–C). However, cotreatment with Scr3NiPp did not alter the effects of IL-1β on endothelium-dependent relaxation (EC50 [CCH]: IL-1β = 2.066 × 10−7 M vs IL-1β + Scr3NiPp = 2.359 × 10−7 M, p = 0.24; Figure 5D).
ER stress induces vascular endothelial dysfunction ex vivo
Sepsis has been linked to increased ER stress.12,13 To determine the effect of heightened ER stress on endothelial function, isolated rat aortas were exposed to tunicamycin (TM), an inhibitor of glycosylation that disrupts the protein folding machinery. 16 Exposure to TM impaired endothelium-dependent relaxation of the aortas compared to untreated control rat aortas (EC50 [CCH]: control = 1.868 × 10−7 M vs TM = 2.744 × 10−7 M, p = 0.03; Figure 6).
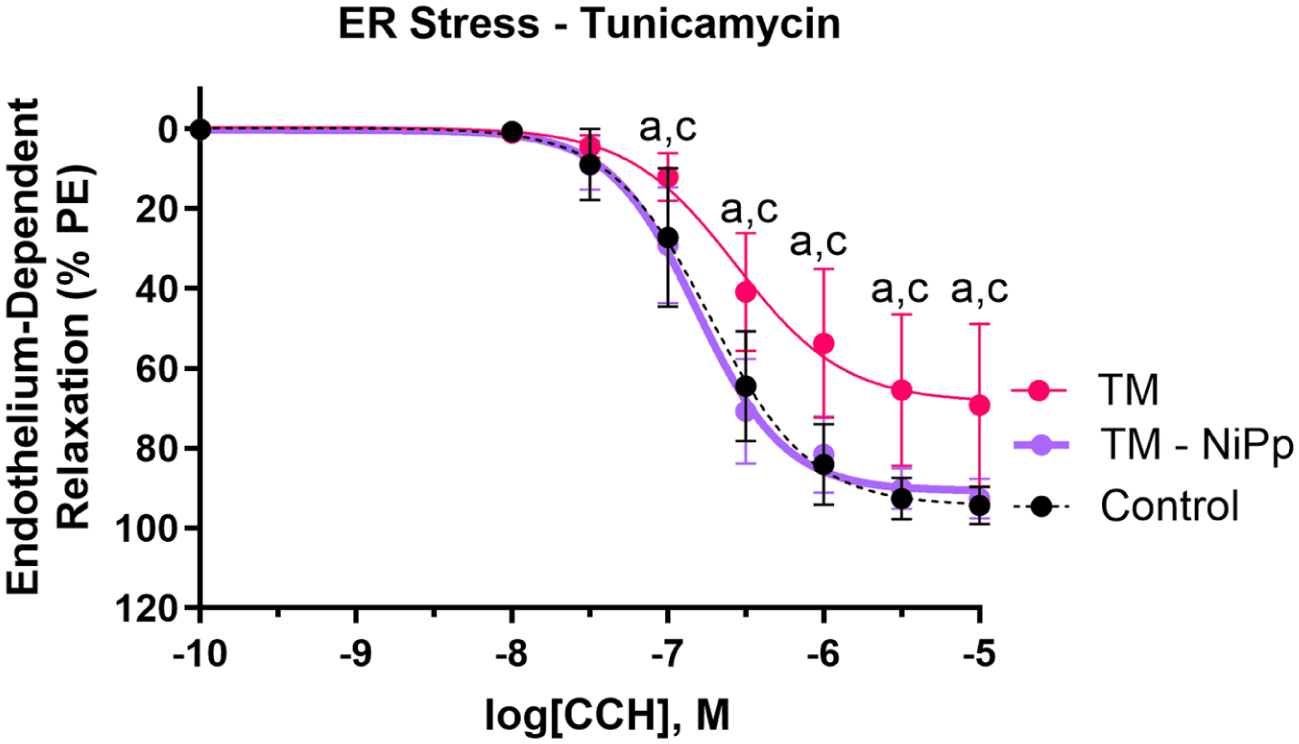
Effect of NiPp on endothelium-dependent relaxation impaired by ER stress. Treatment of PE-exposed rat aortas with TM (50 ng/mL) induced a greater loss of endothelium-dependent relaxation (% PE) compared with untreated control aortas across increasing concentrations of CCH. Cotreatment of rat aortas with NiPp mitigated the TM-induced loss in endothelium-dependent relaxation.
NiPp mitigates vascular endothelial dysfunction induced by an ER stressor ex vivo
Reduced Niban phosphorylation has also been associated with heightened ER stress. 16 To determine if increasing Niban phosphorylation would mitigate endothelial dysfunction induced by ER stress, isolated rat aortas were treated with TM in the presence of NiPp. NiPp treatment mitigated the effects of TM on endothelium-dependent relaxation (EC50 [CCH]: TM = 2.744 × 10−7 M vs TM + NiPp = 1.546 × 10−7 M, p = 0.05; Figure 6).
Discussion
Sepsis-induced vascular dysfunction arises from the complex interplay of inflammation and oxidative stress, which can both influence and be exacerbated by ER stress, collectively contributing to endothelial dysfunction.3,5,8,11,13,14 Niban is an ER stress-inducible protein that may be a regulator of the ER stress pathways with pro-survival functions.4,16,17 Previous findings indicating that the downregulation of Niban phosphorylation is associated with vascular injuries 4 suggest a potential link between ER stress and Niban regulation in sepsis, highlighting the possibility that restoring Niban phosphorylation could improve sepsis-induced endothelial dysfunction. Using a polymicrobial CS sepsis model, the current study identified an association between impaired vascular function, reduced Niban phosphorylation, and heightened ER stress during sepsis. Moreover, we demonstrated that increasing Niban phosphorylation attenuated vascular endothelial dysfunction in septic aorta associated with the CS model of sepsis, as well as in ex vivo endothelial dysfunction induced by individual mediators of sepsis or ER stress.
Specifically, we employed NiPp, a cell-permeant phosphopeptide mimetic designed to recapitulate the functional effects of phosphorylated Niban, to establish the physiologic relevance of Niban in vascular homeostasis. NiPp has been shown to improve endothelial function after P2X7R activation, 18 a DAMP-responsive receptor, and acidosis, 4 both of which are associated with sepsis. 21 In this study, we demonstrated that NiPp not only improved endothelium-dependent relaxation in septic aortic rings but also in normal aortic rings exposed to LPS, IL-1β, and CFH, key drivers of endothelial injury in sepsis.3,5,22,23 These mediators represent the inflammatory and oxidative stress conditions characteristic of sepsis,2,6 and our findings suggest that modulating Niban phosphorylation may be a relevant therapeutic target. In addition, these data demonstrate the broad applicability of NiPp in addressing endothelial dysfunction in sepsis. Moreover, the elderly population is particularly susceptible to sepsis, in part due to vascular complications associated with age-related endothelial dysfunction.24 –27 In a previous publication, we demonstrated that NiPp can restore endothelial function in aged human saphenous veins, 4 highlighting the potential importance of Niban regulation in this vulnerable population. The concentration of NiPp chosen for the current study was based on the dose–response study in which 50 μM did not show salutary effects on IL-1β-induced impairments of endothelium-dependent function (Figure S3). Together with our previous studies using a higher NiPp dose (500 µM) on different vascular injuries, it suggests that there is a range of ex vivo pharmacologic efficacy for NiPp that is likely to be context-dependent. Furthermore, the Scr3NiPp control peptide, which contains a scrambled cargo sequence of NiPp, failed to confer protection against inflammatory insult, reinforcing the specificity of NiPp. Scr3NiPp also failed to affect P2X7R-induced endothelium dysfunction (data not shown), further corroborating the specificity of NiPp. Collectively, these results underscore the therapeutic relevance of Niban phosphorylation in mitigating sepsis-induced vascular endothelial dysfunction and the utilization of NiPp for exploring new molecular mechanisms underlying vascular dysfunction.
The current study also demonstrated impaired vascular reactivity to KCl and PE in septic rat aortas, reflecting hyporesponsiveness to vasoconstrictors characteristic of sepsis-induced vasoplegia.2,5 Briefly, KCl-induced contraction relies on smooth muscle depolarization and calcium influx, while PE-induced contraction depends more on adrenergic receptor activation and downstream calcium signaling. 28 However, both processes are modulated by endothelium-derived vasoconstrictors or vasodilators. During sepsis, endothelial damage caused by inflammatory mediators and oxidative stress disrupts these endothelial signals.2,5 Previous rat studies have demonstrated similar reductions in vasoreactivity to KCl and PE during sepsis, linking the impairments to endothelial dysfunction.29 –32 Sepsis-induced endothelial cell death may also further impair endothelial signaling, calcium uptake, and release of various vasoconstrictive factors. 2 Notably, the preserved SNP-induced relaxation in septic aortas indicates that nitric oxide-mediated smooth muscle responsiveness remains intact in this sepsis model despite a reduction in eNOS phosphorylation. This suggests that the impairments in KCl- or PE-induced contraction are likely due to a loss of endothelium-derived vasoconstrictors, adrenergic receptor dysfunction, or impaired calcium handling rather than altered nitric oxide signaling in smooth muscle cells. Alternatively, reduced responses to KCl may be a result of cell death caused by the complex circulating septic milieu in vivo.7,10,12,14 The observation that acute treatment of septic aorta with NiPp did not restore responses to KCl or PE suggests that Niban regulation does not interfere with PE signaling pathways, or that NiPp may selectively impact endothelial vasodilatory signaling rather than the molecular mechanisms that drive endothelium-derived vasoconstriction or smooth muscle-mediated vasoreactivity in the septic rat aorta. Nevertheless, the improvement in endothelial responses implicates a role for Niban in endothelial homeostasis, a crucial aspect of sepsis-induced vascular dysfunction and the development of vasoplegia.
Building on established knowledge that sepsis impairs vascular homeostasis through inflammation and oxidative stress,3,21,23,33,34 our study highlights the role of ER stress as a key mediator of the pathologic endothelial response in sepsis, with Niban activation emerging as a potential regulator of ER stress responses in this context. Specifically, we observed elevated protein levels of GRP78, a well-established marker of ER stress linked to both sepsis 13 and vascular dysfunction, 35 along with a trend toward increased phosphorylation of eIF2α, a critical component of one of the three UPR branches.11,14 –16 These findings suggest that sepsis induced by CS promotes ER stress, which is accompanied by reduced Niban phosphorylation in septic aortas. The observed increase in GRP78, without corresponding changes in the transcriptional regulation of eIF2α, provides new mechanistic insights into our sepsis model. Together, these results suggest that vascular ER stress in sepsis may primarily result from dysregulation of the other branches of UPR signaling and impaired degradation of misfolded proteins.11,14,15 Alternatively, this discrepancy may reflect the timing and sequential activation of ER stress pathways, wherein GRP78 upregulation occurs in the earlier phase, and eIF2α phosphorylation occurs later.11,14,15 This latter interpretation would align more favorably with previous findings linking decreased Niban levels to increased eIF2α phosphorylation in models of ER stress. 16 Whereas GRP78 levels were not directly measured following NiPp treatment, the observed improvements in endothelium-dependent relaxation for septic aortas and those treated with the ER stressor, TM, suggest that modulating Niban phosphorylation could mitigate the downstream effects of sepsis-induced ER stress. These findings raise important questions about the time course and mechanisms through which Niban phosphorylation influences endothelial stress responses, including whether Niban directly regulates ER stress pathways like the UPR, or acts indirectly through upstream signaling cascades. Further elucidation of how Niban affects ER stress and vascular function will be critical for understanding its broader role in maintaining vascular homeostasis.
Sepsis progression involves multiple cellular and molecular mechanisms that contribute to vascular dysfunction, including dysregulated inflammatory responses, oxidative injury, and altered endothelial-derived vasoactive mediators such as nitric oxide, prostanoids, endothelin, and endothelium-derived hyperpolarizing factor (EDHF). These mechanisms collectively respond to the surrounding physiological environment and may compensate for the absence of endothelial nitric oxide and prostacyclins.36,37 Various signaling pathways, such as nuclear factor-κB, MAPKs, and hypoxia-induced factor 1α, are activated and the interplay can trigger detrimental effects. 38 For example, ER stress plays a pivotal role in downregulating eNOS, leading to impaired NO synthesis, which is vital for maintaining endothelial function. Similarly, ER stress, excessive inflammation, and oxidative stress activate p38 MAPK, further reducing eNOS activity. 39 In the CS-induced septic aortae, we found reduced eNOS phosphorylation, which likely contributes to the impaired endothelium-dependent relaxation. Although p38 MAPK activation is often anticipated during cellular stress in sepsis, because many circulating mediators (including those used in our ex vivo model) activate p38 MAPK,40 –43 no difference in p38 MAPK phosphorylation was observed 24 hours after sepsis induction. This may be explained by the timing of vascular p38 MAPK regulation, either through direct activation by sepsis-associated mediators or as downstream ER stress signaling. Previous studies have reported reduced p38 MAPK phosphorylation in skeletal muscle in a chronic intra-abdominal septic abscess rat model, 44 whereas increased phosphorylation has been observed in the lungs and brain after cecal ligation puncture-induced sepsis in mice.45,46 These findings highlight the complexity of the septic environment, where various pathological stimuli elicit dynamic and organ-specific responses during the progression of endothelial dysfunction. Further research into the temporospatial regulation and interplay between Niban and ER stress responses in vascular function will be critical to understanding their broader role in maintaining vascular homeostasis.
The inclusion of multiple mediators of sepsis-induced endothelial injury, alongside a polymicrobial rat model of sepsis, strengthens the translational relevance of this study. LPS, IL-1β, CFH, and TM represent distinct but interconnected drivers of vascular dysfunction during sepsis.3,6,10,23,33 By evaluating NiPp across these mediators, our study demonstrates its ability to target multiple pathways relevant to sepsis pathology. 6 Furthermore, the protective effects of NiPp in septic rat aortae indicate that Niban phosphorylation has the potential to modulate systemic vascular dysfunction in sepsis, extending beyond its effects on individual mediators. Importantly, this finding suggests that restoring Niban phosphorylation after a septic insult counteracts the complex, synergistic vascular insults present in polymicrobial sepsis and provides clinical relevance to further interrogation of the potential of NiPp as a rescue therapy in humans. This multitargeted approach offers a promising avenue for addressing the vascular complications of sepsis and potentially other conditions characterized by endothelial injury. Moreover, ex vivo examination of septic aorta also allows for the investigation of endogenous cellular and molecular defects and the discovery of salutary therapeutic mechanisms that improve vascular functions.
Despite these advances, there are several limitations in this study that warrant consideration. The ex vivo nature of our experiments using intact aortic tissues in a muscle bath system provided a controlled environment to isolate vascular effects and evaluate endothelial dysfunction induced by sepsis or its mediators. This approach eliminated systemic influences such as inflammatory mediators, hemodynamics, or multiorgan interactions,47 –50 as well as confounding variables related to targeted drug delivery, metabolism, cellular uptake, or optimized dosing. 51 However, it does not allow for assessment of the systemic impact of NiPp, including its effects on survival and multiorgan dysfunction in sepsis. Though the aortic ring model enabled a focused evaluation of endothelial function and the differentiation between endothelium-dependent and endothelium-independent relaxation, it does not account for the heterogeneity of vascular beds throughout the body (i.e., the complexity and differences between macrovascular and microvascular dysfunction), both of which are critical for tissue oxygenation and perfusion during sepsis,1,2 potentially limiting its applicability to broader physiological contexts. Further, only female rats were utilized in this study, which may limit the generalizability of the findings, as sex differences in vascular reactivity and endothelial function during sepsis have been well documented.52 –55 The use of a single sex was intended to provide initial insights into the cellular mechanisms associated with Niban regulation in a controlled setting. Investigating the systemic effects of NiPp on vascular function across different vascular beds, in both sexes, as well as in relation to broader physiological outcomes during a septic insult, will be essential for understanding its therapeutic potential and will be the focus of follow-on studies. This will also help identify potential sex-specific differences in endothelium- or smooth muscle-derived factors that influence Niban’s role in vascular responses to sepsis.
Additionally, although lower concentrations of NiPp 50 µM did not elicit detectable effects in IL-1β-treated aortas, concentrations between 100 and 500 µM restored endothelium-dependent relaxation in ex vivo models. This highlights the importance of optimizing peptide concentrations based on experimental context, and future studies will need to establish the dose–response profile of NiPp in vivo to determine its range of pharmacological efficacy, therapeutic window, and potency in septic models. Furthermore, vascular responses to sepsis involved multiple cellular and molecular mechanisms that vary across temporal and spatial contexts. Our findings suggest that reduced Niban phosphorylation contributes to ER stress, but the precise molecular mechanisms and time course remain unclear. Future studies will need to focus on defining these dynamics and exploring potential interactions between Niban phosphorylation and other stress response pathways, such as those activated by inflammatory cytokines and oxidative stress mediators. Finally, examining whether Niban phosphorylation influences other arms of the ER stress response could provide further insights into its mechanism of action and its broader role in the adaptation of vascular cells to stress during sepsis.
Conclusion
This study identifies Niban phosphorylation as a critical regulator of vascular function in sepsis and demonstrates the therapeutic potential of NiPp for restoring endothelial function in septic vasculatures. By mitigating endothelial dysfunction, NiPp addresses a key mechanism of sepsis-induced vascular injury, offering a novel approach for maintaining vascular homeostasis. These findings pave the way for further research into the molecular mechanisms underlying Niban-mediated stress adaptation and the development of NiPp as a targeted therapy for sepsis. Future investigations should focus on validating these results in vivo, optimizing NiPp administration strategies, and expanding our understanding of Niban in vascular homeostasis under pathological conditions.
Supplemental Material
sj-tif-1-vmj-10.1177_1358863X251412772 – Supplemental material for Mitigating sepsis-induced vascular endothelial dysfunction through Niban phosphorylation
Supplemental material, sj-tif-1-vmj-10.1177_1358863X251412772 for Mitigating sepsis-induced vascular endothelial dysfunction through Niban phosphorylation by Brandon Baer, Madeleine Morelli, Brian Wadzinski, Anish Katta, Amanda J Sisung, Colleen Brophy, Julie A Bastarache and Joyce Cheung-Flynn in Vascular Medicine
Supplemental Material
sj-tif-2-vmj-10.1177_1358863X251412772 – Supplemental material for Mitigating sepsis-induced vascular endothelial dysfunction through Niban phosphorylation
Supplemental material, sj-tif-2-vmj-10.1177_1358863X251412772 for Mitigating sepsis-induced vascular endothelial dysfunction through Niban phosphorylation by Brandon Baer, Madeleine Morelli, Brian Wadzinski, Anish Katta, Amanda J Sisung, Colleen Brophy, Julie A Bastarache and Joyce Cheung-Flynn in Vascular Medicine
Supplemental Material
sj-tif-3-vmj-10.1177_1358863X251412772 – Supplemental material for Mitigating sepsis-induced vascular endothelial dysfunction through Niban phosphorylation
Supplemental material, sj-tif-3-vmj-10.1177_1358863X251412772 for Mitigating sepsis-induced vascular endothelial dysfunction through Niban phosphorylation by Brandon Baer, Madeleine Morelli, Brian Wadzinski, Anish Katta, Amanda J Sisung, Colleen Brophy, Julie A Bastarache and Joyce Cheung-Flynn in Vascular Medicine
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This work was supported by grants from the National Heart, Lung, and Blood Institute (R01HL070715 to Colleen Brophy) and National Institute on Aging (RF1AG075341 to Julie A Bastarache). Funders had no role in the study design, data collection and analysis, decision to publish, or preparation of the manuscript.
Supplemental material
Supplemental material for this article is available online.
