Abstract

Keywords
Pulmonary embolism (PE) is among the most common cardiovascular causes of mortality, associated with up to 100,000 US deaths annually. 1 Mortality is linked to right ventricular (RV) failure. Therefore, risk stratification tools integrating clinical, imaging, and biochemical markers of RV stress and injury, particularly cardiac troponins, are foundational in guiding management decisions.2,3 The development of high-sensitivity troponin assays has enabled detection of myocardial injury at lower troponin concentrations. 4 However, the impact of institutional transition to high-sensitivity troponin assays on PE risk assessment and clinical decision making remains unclear.
At our center, standardized PE risk stratification is used to guide clinical decision making by a multispecialty pulmonary embolism response team (PERT). 5 Risk stratification is based on international guidelines 2 that incorporate troponin elevation. This approach has previously been used in prospective clinical trials to guide treatment decisions. 6 The PERT uses the Bova score, 7 which also considers troponin elevation to predict PE-related complications, to guide intensive care unit (ICU) admission recommendations. 5 Accordingly, we hypothesized that a recent institutional transition from a conventional to high-sensitivity troponin-I (TnI) assay would increase the proportion of PERT patients with an elevated troponin, altering risk stratification and increasing the frequency of ICU admissions.
We reviewed all PERT activations at two affiliated hospitals between September 2021 and January 2024 (approved by the local institutional review board). Hospital A implemented transition to high-sensitivity TnI assays on July 11, 2022, and hospital B on May 16, 2023. Both conventional and high-sensitivity assays used the Beckman Coulter Access 2 Immunoassay platform. Assay-specific thresholds for reporting elevated TnI were identical at both hospitals and defined a value exceeding the 99th percentile for a healthy reference population (conventional: > 40 ng/L; high-sensitivity: > 12 ng/L in men or > 20 ng/L in women).
The analysis included 316 unique patients (Table 1) divided into cohorts based on the TnI assay used at the time of PERT assessment: conventional (c-TnI, n = 158) versus high-sensitivity (hs-TnI, n = 158). Overall, the population has a median age of 64 years and 52.2% were men (Supplemental Table). Cohorts had similar proportions of patients with history of cancer, heart failure, and chronic lung disease, though c-TnI patients were more frequently managed at hospital B (93.0% vs 50.6%) due to later implementation of the hs-TnI assay (Supplemental Table).
Cardiac biomarkers, risk stratification, and clinical decision making.
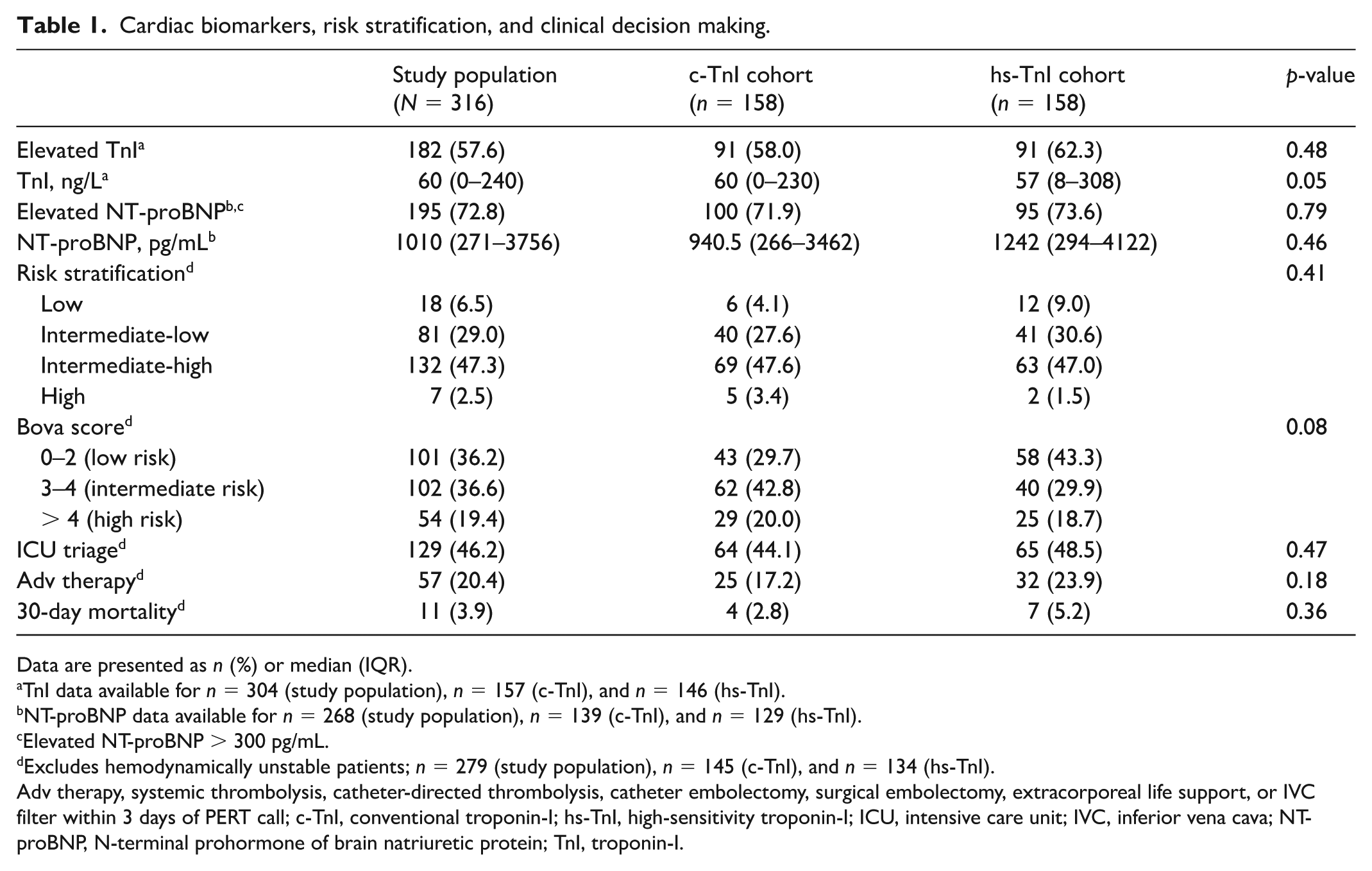
Data are presented as n (%) or median (IQR).
TnI data available for n = 304 (study population), n = 157 (c-TnI), and n = 146 (hs-TnI).
NT-proBNP data available for n = 268 (study population), n = 139 (c-TnI), and n = 129 (hs-TnI).
Elevated NT-proBNP > 300 pg/mL.
Excludes hemodynamically unstable patients; n = 279 (study population), n = 145 (c-TnI), and n = 134 (hs-TnI).
Adv therapy, systemic thrombolysis, catheter-directed thrombolysis, catheter embolectomy, surgical embolectomy, extracorporeal life support, or IVC filter within 3 days of PERT call; c-TnI, conventional troponin-I; hs-TnI, high-sensitivity troponin-I; ICU, intensive care unit; IVC, inferior vena cava; NT-proBNP, N-terminal prohormone of brain natriuretic protein; TnI, troponin-I.
Contrary to our hypothesis, the proportion of patients with TnI elevation did not differ between cohorts (c-TnI = 58.0% vs hs-TnI = 62.3%; p = 0.48; Table 1). Median TnI levels were comparable between groups (c-TnI = 60 ng/L vs hs-TnI = 57 ng/L; p = 0.05) and above assay thresholds for elevated TnI designation, reflecting the severity of illness in this PERT referral population. As the primary advantage of the high-sensitivity assay is a lower limit of detection, 4 the relatively high TnI levels observed in both groups likely explain the lack of difference in proportion with elevated TnI.
Because hemodynamic instability is an independent determinant of PE risk stratification, 2 we excluded 37 hemodynamically unstable patients from analyses of risk stratification and clinical decision making. Table 1 shows there was no difference in PERT-designated risk stratification between groups. Similarly, there was no difference in Bova score distribution and no difference in the frequency of PERT-recommended ICU admission (c-TnI = 44.1% vs hs-TnI = 48.5%; p = 0.47) between groups. We observed no differences in the use of advanced therapies for PE (c-TnI = 17.2% vs hs-TnI = 23.9%; p = 0.18) or in-hospital mortality at 30 days (c-TnI = 2.8% vs hs-TnI = 5.2%; p = 0.36) between cohorts.
Our findings differ from previous analyses. Bikdeli et al. 8 recently reported differences in risk stratification and clinical outcomes based on TnI assay sensitivity. In a cohort of patients with both conventional and hs-TnI testing, high-sensitivity measures were associated with a higher proportion of patients classified as having an elevated TnI (31.7% vs 16.7%) and fewer patients classified as having low-risk disease. Clinical outcomes were worse among patients with an elevated TnI measured by the conventional but not the high-sensitivity assay, suggesting that the latter may have overestimated risk. 9 Importantly, disease severity was lower in this study population (nearly 90% low- and intermediate-low-risk disease) than our predominantly intermediate-high-risk PERT population. The discriminatory power of the high-sensitivity assay may be lost among the sicker patient population frequently served by PERTs. Indeed, median TnI values in our population exceed the hs-TnI threshold (16 ng/L) previously shown to optimally predict adverse outcomes and mortality in a study similarly derived from patients with lower disease severity. 9
We acknowledge several limitations to this retrospective analysis. The population includes patients evaluated by a single PERT at an academic medical center, potentially limiting generalizability. Cohorts were not matched, introducing potential unmeasured confounders between groups. More patients in the hs-TnI cohort had unmeasured troponin levels at PERT activation (0.6% vs 7.6%; p = 0.004; not shown) but these missing data are unlikely to explain our observation. If all missing TnI values in both groups are designated elevated, the proportion of patients with TnI elevation between groups remains unchanged (58.2% vs 65.2%; p = 0.20). The retrospective study design precludes causal inference, and the relatively low event rates limit power for outcomes like mortality.
Despite these limitations, we found no effects of institutional transition to a hs-TnI assay on PERT risk stratification or clinical decision making. When compared to previous studies, our findings suggest that transition to the hs-TnI assay may be more consequential for risk assessment and decision making in patients with less severe PE. We believe that analysis of large, multicenter registry datasets will help identify optimal troponin thresholds to guide clinical decision making for patients with PE of all disease severity.
Supplemental Material
sj-pdf-1-vmj-10.1177_1358863X251393230 – Supplemental material for No effect of transition from conventional to high-sensitivity troponin assay on pulmonary embolism risk stratification and decision making: A single-center experience
Supplemental material, sj-pdf-1-vmj-10.1177_1358863X251393230 for No effect of transition from conventional to high-sensitivity troponin assay on pulmonary embolism risk stratification and decision making: A single-center experience by Meghan J Price, Caya McFalls, Minh Phan, Jennifer Yui and Todd M Kolb in Vascular Medicine
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
