Abstract
Ultrasound-accelerated thrombolysis (USAT) is advocated in pulmonary embolism (PE) based on the hypothesis that adjunctive ultrasound provides superior clinical efficacy compared to standard catheter-directed thrombolysis (CDT). This retrospective study was designed to compare outcomes between the two modalities. We analyzed patients with computed tomography-diagnosed PE at our institution treated with either USAT or standard CDT. Efficacy parameters assessed included invasive pulmonary artery systolic pressure (PASP; pre- and 24 hours post-treatment), non-invasive right-to-left ventricle (RV/LV) ratio (pre- and post-treatment), and general clinical outcomes (length-of-stay, significant bleeding, and mortality). We analyzed 98 cases (62 USAT and 36 CDT), in whom massive PE was diagnosed in 7%, intermediate/high risk in 81%, and intermediate/low risk in 12%. Overall, 92% had bilateral clot and 40% saddle embolus. At 24 hours, PASP decreased similarly in both groups (CDT Δ14.7 mmHg, USAT Δ10.8 mmHg; p = 0.14). Post-treatment, CDT showed similar improvement in the RV/LV ratio (CDT Δ0.58 vs USAT Δ0.45; p = 0.07), despite the baseline ratio being greater in the CDT group, indicating more severe RV strain (1.56 ± 0.36 vs 1.40 ± 0.29; p = 0.01). Intensive care unit and hospital length-of-stays were similar in both groups. A trend toward lesser significant bleeding rates in the CDT group (8.3% vs 12.9%, p = 0.74) as well as improved survival-to-discharge (97.2% vs 91.9%, p = 0.66) was observed. Compared to USAT, standard CDT achieves similar beneficial effects on hemodynamics, RV/LV ratios, and clinical outcomes. These observations suggest that salutary clinical results may be achieved without the need for very expensive devices.
Keywords
Introduction
Over the past decade, the field of acute pulmonary embolism (PE) has undergone notable change, characterized by increased detection attributable to more widespread use of computed tomography (CT) angiography. Management of PE has evolved as well as with the more frequent application of escalation of care strategies previously reserved for those in decompensated shock from PE. These strategies, such as systemic thrombolytic therapy, catheter-directed lytic (CDL) therapy, and mechanical thrombolysis, are now commonly applied to those with submassive PE. However, definitive data and consistent guidelines are lacking with respect to patient selection, as well as method and dose of drug delivery.1–3 CDL has been demonstrated to achieve improvement in pulmonary artery systolic pressure (PASP) and right heart strain as measured by the right-to-left ventricle (RV/LV) ratio.3–5 However, there is scant direct comparison of catheter delivery devices and pharmacologic regimens. Ultrasound-accelerated thrombolysis USAT) has been advocated, based on the hypothesis that adjunctive ultrasound would facilitate lysis and thereby provide superior clinical efficacy.6–8 However, this theory has not been adjudicated in a head-to-head study with less expensive standard catheters for use in the treatment of PE. The present retrospective analysis was designed to elucidate differences in hemodynamic and clinical outcomes between USAT and standard catheter-directed thrombolysis (CDT).
Methods
This retrospective, single-center study analyzed patients with CT-diagnosed PE treated with either USAT or standard CDT interventions from January 2010 to May 2018. Institutional Review Board approval was obtained. Patients were classified as high-risk (massive), intermediate/high-risk, or intermediate/low-risk (submassive) PE based on the European Society of Cardiology (ESC) guideline criteria 9 (Figure 1), which categorizes patients by clinical hemodynamics, imaging findings, and biomarkers. High-risk patients were defined as having a systolic blood pressure of < 90 mmHg for at least 15 minutes, unresponsive to intravenous fluid resuscitation, or requiring vasopressor support. Intermediate/high risk was defined as systolic blood pressure (SBP) > 90 mmHg, CT documentation of PE with a RV/LV ratio > 1, and positive biomarkers (troponin and brain natriuretic peptide (BNP)). Patients were clinically risk-stratified to determine the appropriateness for escalation of care according to standard clinical criteria via the HAS-BLED (Hypertension, Abnormal liver/renal function, Stroke history, Bleeding predisposition, Labile INR, Elderly, Drug/alcohol usage) score, 10 with a score > 2 considered a high bleeding risk.
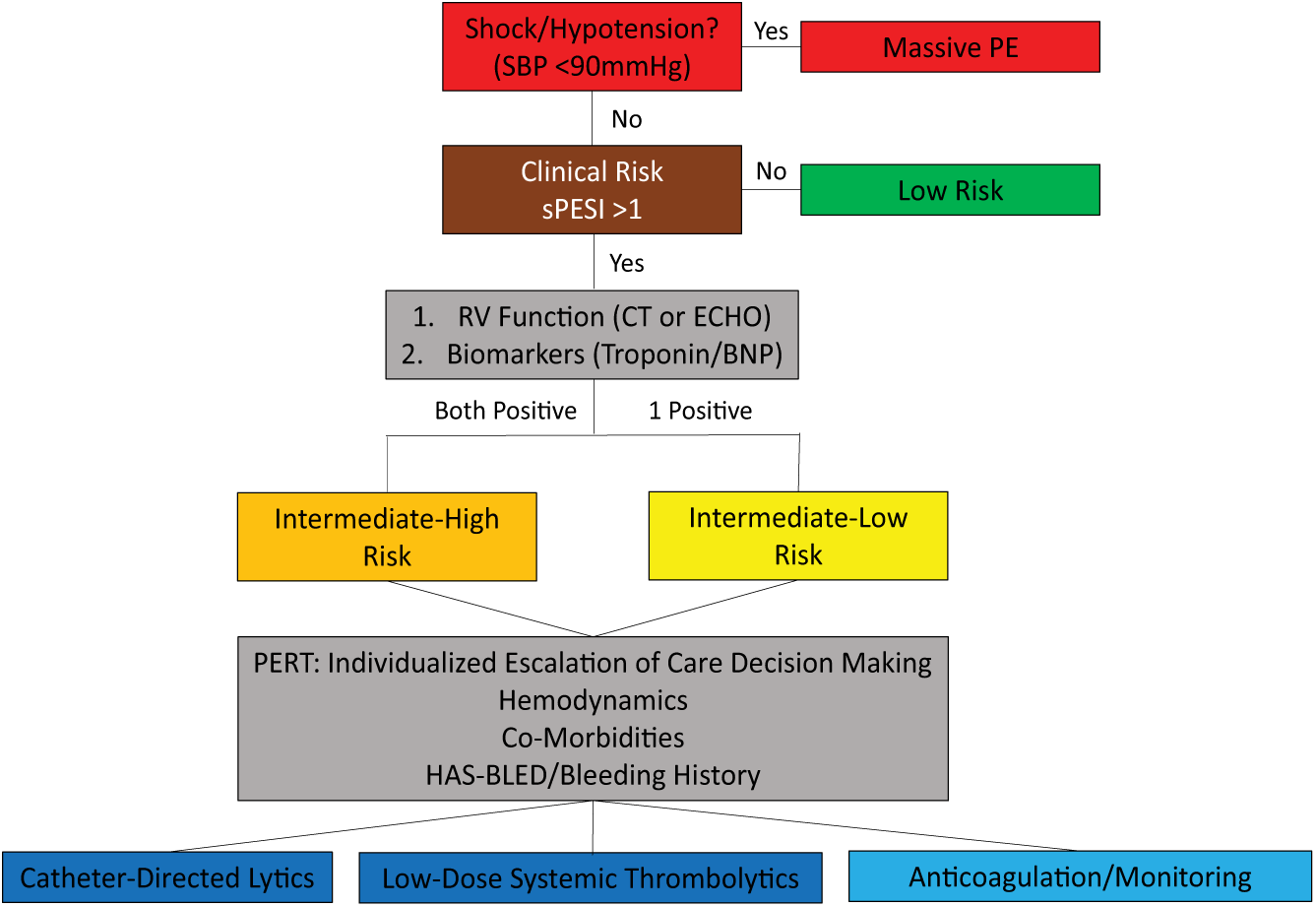
PE management algorithm.
Outcome parameters assessed included invasive PASP (pre- and 24 hours post-treatment), non-invasive RV/LV ratio (pre- and post-treatment), and general clinical outcomes (length-of-stay, significant bleeding, and mortality).
CT imaging
CT PE studies were performed according to standard procedures – thrombus location and distribution were derived from clinical radiology report and categorized as unilateral, bilateral, and saddle.
RV/LV ratio
At baseline, the RV/LV ratio was measured by a CT axial four-chamber view according to standard methods. 11 The post-treatment RV/LV ratio was measured at no earlier than 48 hours and no later than discharge from index hospitalization by 2D echocardiogram from the apical four-chamber view.12,13
Procedure
Patients underwent invasive therapy in the interventional radiology suite. Catheterization was performed according to established techniques, with standard CDT performed employing a multi-sidehole catheter (Cragg-McNamara; Boston Scientific, Marlbourogh, MA, USA) and USAT performed utilizing the EkoSonic Endovascular System (EKOS Corp., Bothwell, WA, USA). The technique employed and catheter chosen for a given patient was at the discretion of the interventionalist. Invasive PA pressure measurements were recorded prior to therapy. All patients received alteplase infusion ranging from 0.5 to 1 mg/catheter/hour. Upfront bolus dosing of 5 mg of tPA was administered at the operator’s discretion. Heparin infusion was also simultaneously administered at a rate of 500–1000 U/hour, which was fixed for the duration of the tissue plasminogen activator (tPA) infusion. Fibrinogen levels were monitored with set cut-offs for infusion termination if the nadir was reached.
All patients were monitored in the Cardiac Care Unit (CCU) during the duration of the infusion therapy and underwent repeat invasive PA pressure measurement at 12–24 hours. It is important to consider changes in PE management approaches over the time period of the present study at our institution. From the onset of patient enrollment in January 2010 until August 2016, CDL protocol and management was made at the discretion of the interventional radiologist. Their typical approach employed CDL with repeat pulmonary angiography and pulmonary pressures to determine timing of treatment termination. Use of bolus dosing of tPA at treatment onset also occurred at their discretion. This non-protocolized management, during which time CDL was becoming increasingly utilized, was based on the available literature, although then, as now, no clear guidelines existed. Publication of the SEATTLE II study in 2015, the largest trial of CDL to date, popularized a treatment protocol which limited dose and duration of CDL. In August 2016, our institution, along with other peer institutions, adopted the concept of a PE Response Team 14 and began routinely applying a multidisciplinary, shared decision-making approach to management decisions of all patients presenting with acute PE. At this point, the treatment protocol for CDL transitioned to the widely utilized SEATTLE II protocol. 5
Clinical outcomes
The efficacy parameters assessed included baseline and post-therapy invasive PASP, RV/LV ratios, intensive care unit (ICU) and hospital length-of-stays, significant bleeding events defined by both International Society on Thrombosis and Hemostasis (ISTH) and Global Utilization of Streptokinase and tPA For Occluded Arteries (GUSTO) criteria,15,16 and survival-to-discharge.
Statistical analysis
Statistical analysis of baseline characteristics and outcomes were performed using the chi-squared test and Fischer’s exact test, depending on the nature of the data. Paired t-testing was performed on repeated measures data (Microsoft Excel, Redmond, WA, USA; 16.16.2).
Results
Patient characteristics
Of the 98 patients analyzed, standard CDT was employed in 36 (37%), whereas 62 (63%) underwent USAT. General demographics and clinical parameters pertinent to PE severity for the two groups are summarized in Table 1. It is noteworthy that the standard CDT group appeared to suffer on the more severe end of the PE spectrum. Specifically, compared to the USAT group, those undergoing standard CDT were more frequently categorized as high-risk (massive) (11.1% vs 4.8%, p = 0.42) and had greater baseline RV strain (RV/LV ratio 1.56 ± 0.36 vs 1.40 ± 0.29; p = 0.01).
Baseline characteristics.
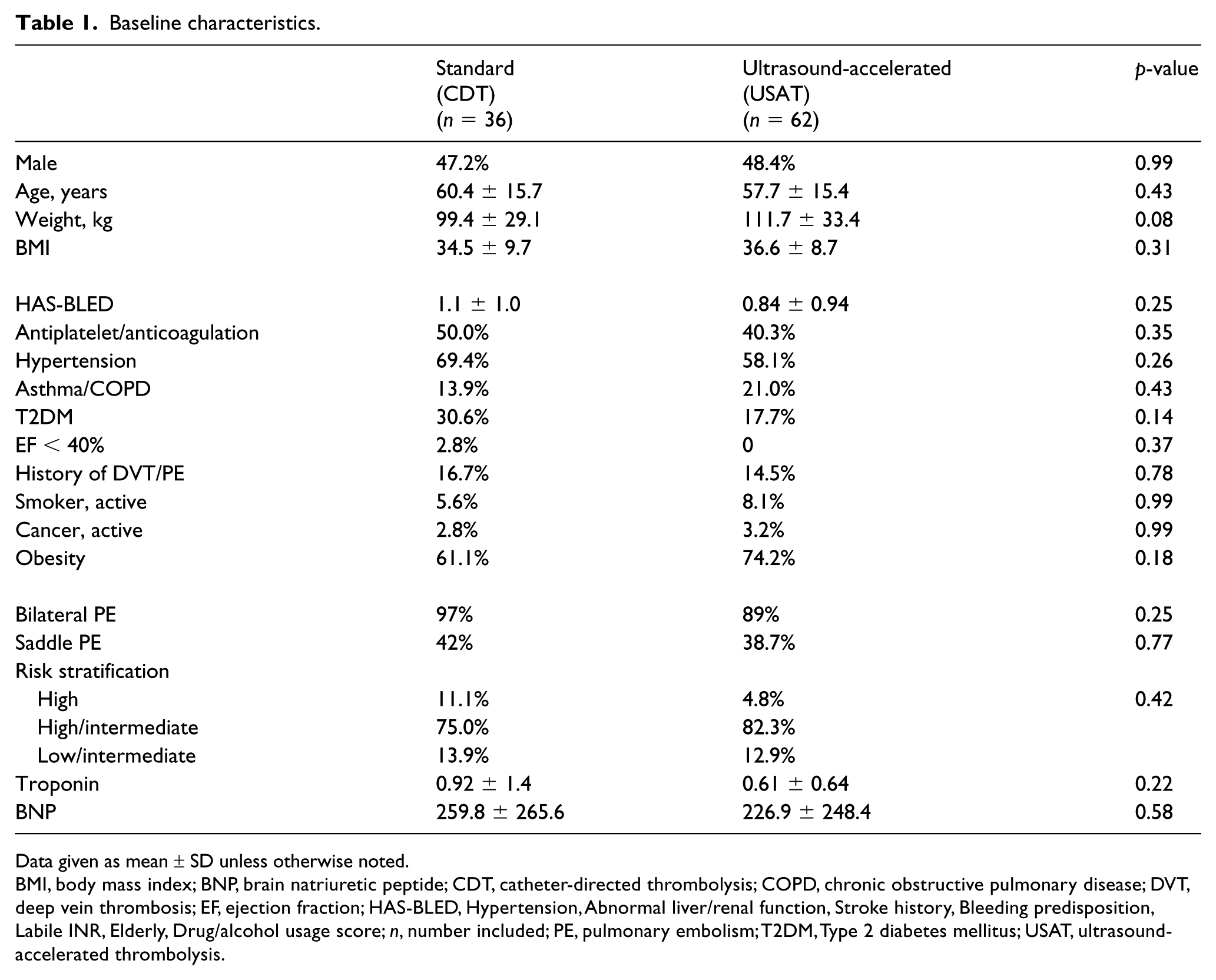
Data given as mean ± SD unless otherwise noted.
BMI, body mass index; BNP, brain natriuretic peptide; CDT, catheter-directed thrombolysis; COPD, chronic obstructive pulmonary disease; DVT, deep vein thrombosis; EF, ejection fraction; HAS-BLED, Hypertension, Abnormal liver/renal function, Stroke history, Bleeding predisposition, Labile INR, Elderly, Drug/alcohol usage score; n, number included; PE, pulmonary embolism; T2DM, Type 2 diabetes mellitus; USAT, ultrasound-accelerated thrombolysis.
Procedural details
The standard CDT group was treated for a shorter duration overall (22.9 vs 31.5 hours, p = 0.01), as well as during the first 24 hours (15.7 vs 19.7 hours, p = 0.002) compared to the USAT group. The average tPA dose administered during the first 24-hour period, including boluses, was also significantly less than the USAT group (19.8 vs 25.7 mg, p = 0.001). Bolus dosing of tPA was equally prevalent in both groups (16.7% vs 17.7%, p = 0.89) (Table 2).
Procedural characteristics.
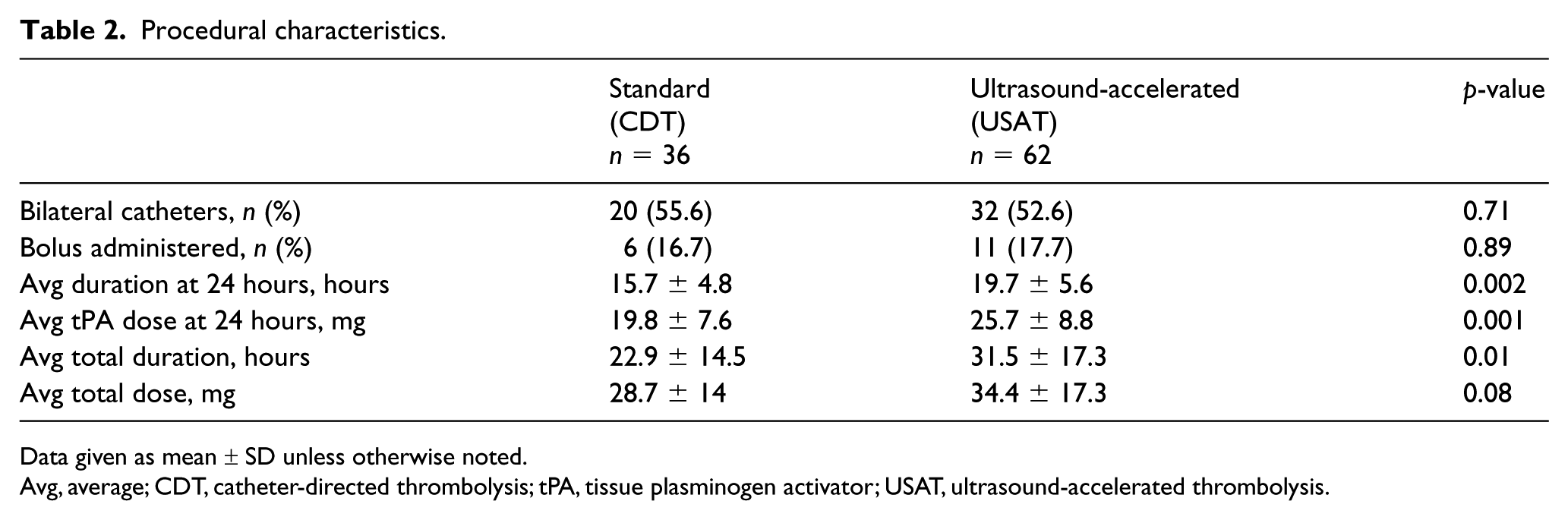
Data given as mean ± SD unless otherwise noted.
Avg, average; CDT, catheter-directed thrombolysis; tPA, tissue plasminogen activator; USAT, ultrasound-accelerated thrombolysis.
Clinical outcomes
Baseline PASP did not differ between the groups (Table 3). Following therapy, PASP improved to a similar magnitude in those undergoing standard CDT as in those in the USAT group (CDT Δ–14.7 vs USAT Δ–10.8; p = 0.14). Despite the fact that at baseline the standard CDT group showed greater RV strain (RV/LV ratio 1.56 ± 0.36 vs 1.40 ± 0.29; p = 0.01), both groups showed equivalent post-therapy normalization of RV/LV ratios (CDT Δ0.58 vs USAT Δ0.45; p = 0.07) (Figure 2). ICU and hospital length-of-stays were similar in both groups and there was a trend toward lesser significant bleeding rates in the CDT group (Table 4; 8.3% vs 12.9%, p = 0.74), as well as better survival-to-discharge (97.2% vs 91.9%, p = 0.66).
Pre/post-treatment hemodynamics, imaging.
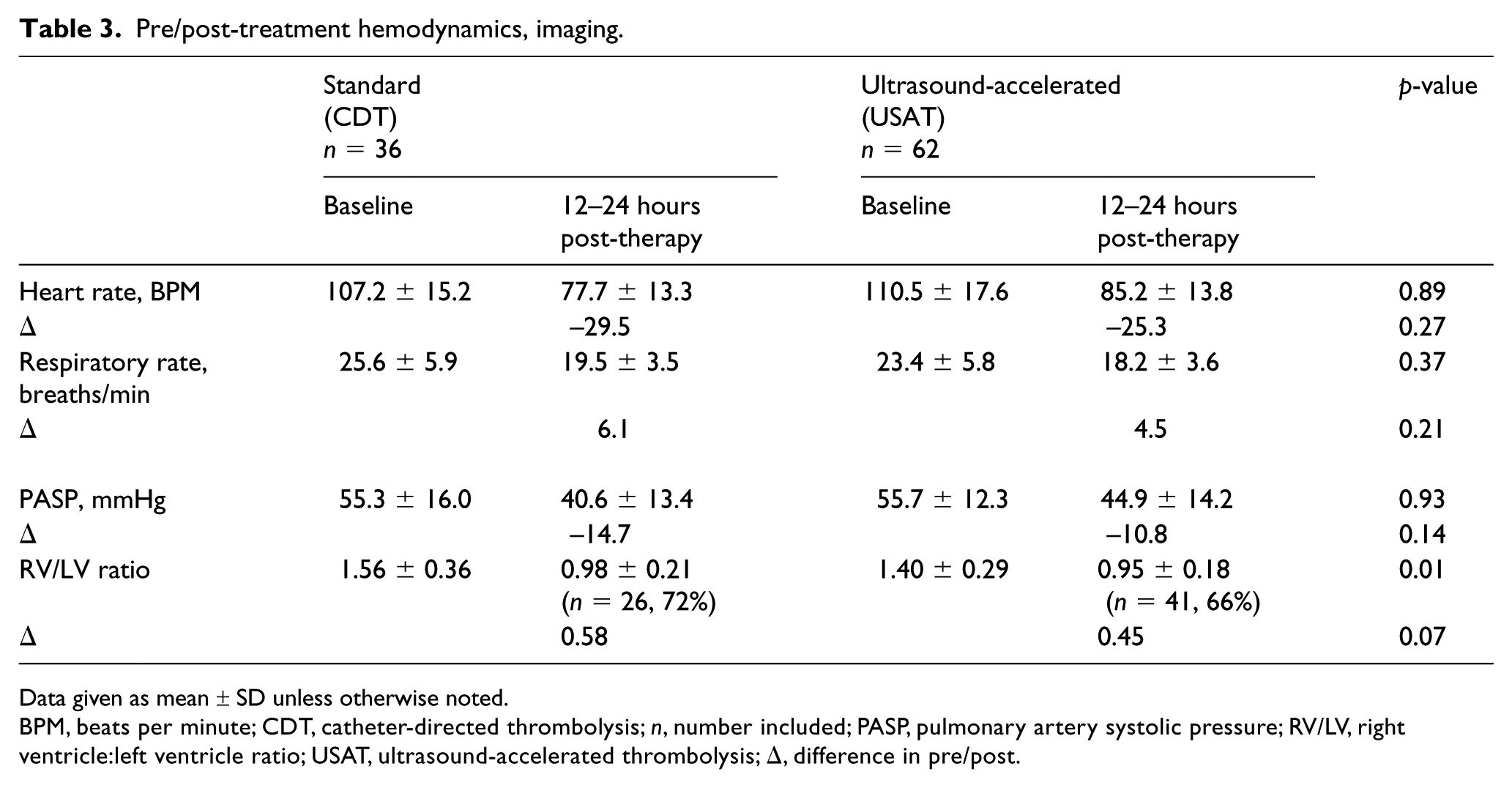
Data given as mean ± SD unless otherwise noted.
BPM, beats per minute; CDT, catheter-directed thrombolysis; n, number included; PASP, pulmonary artery systolic pressure; RV/LV, right ventricle:left ventricle ratio; USAT, ultrasound-accelerated thrombolysis; Δ, difference in pre/post.
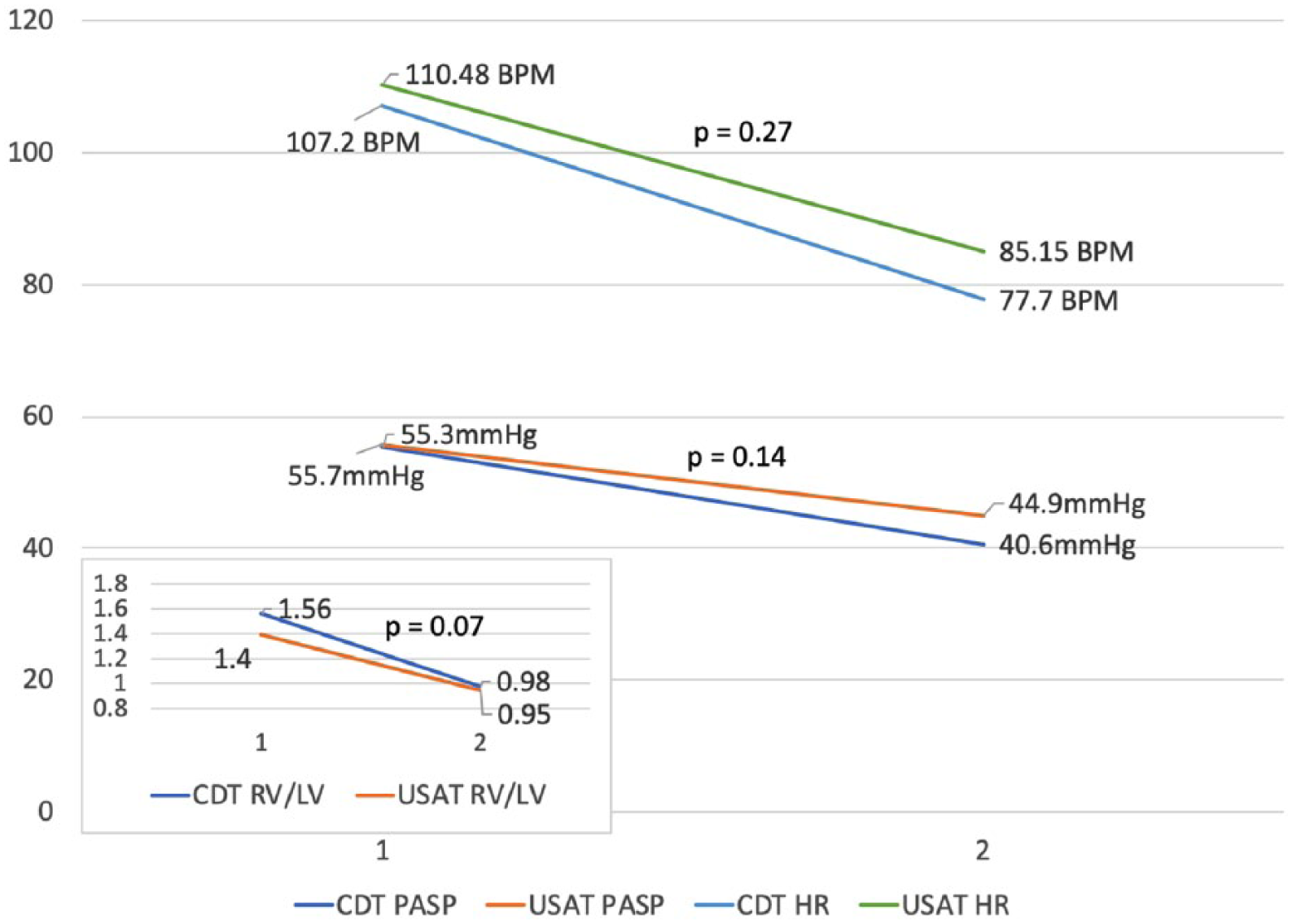
Standard CDT versus USAT heart rate, PASP and RV/LV ratio comparison pre- and post-therapy.
Outcomes.
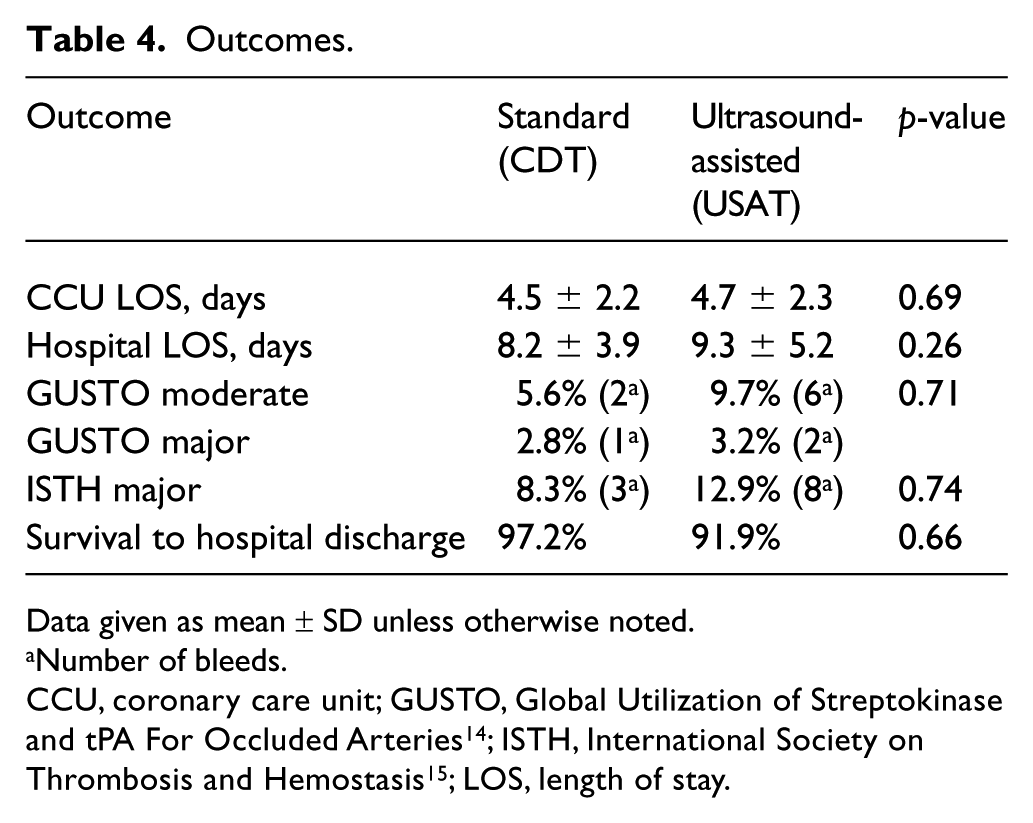
Data given as mean ± SD unless otherwise noted.
Number of bleeds.
Discussion
The present findings suggest that standard CDT achieves similar improvements in hemodynamics, RV/LV ratios, and clinical outcomes compared to USAT when analyzed in a retrospective, non-randomized fashion. These hypothesis-generating observations are consistent with and extend those of prior retrospective studies and may have clinical implications, particularly given the additional cost of USAT equipment.
Acute PE induces substantial clinical morbidity and is the third leading cause of cardiovascular mortality in the United States. These statistics have led to increased attention on improving these outcomes, with particular focus on more aggressive utilization of escalation of care processes with various thrombolytic strategies. 17 Unfortunately, definitive data and consistent guidelines with respect to patient selection, as well as method and dose of drug delivery, are lacking.
Full-dose systemic thrombolytic therapy (TT) (alteplase 100 mg) is established front-line management for patients suffering high-risk (massive) PE, and low-dose systemic TT (alteplase 50 mg) is now also increasingly applied in selected intermediate-risk (submassive) cases.18–20 However, systemic TT has been associated with major bleeding events of 10%, including catastrophic intracranial hemorrhage (ICH; rates of 2% 18 ). Therefore, alternative strategies have been designed to achieve efficacy but mitigate bleeding complications.
CDL therapy (PA infusion of alteplase 10–23 mg) has been demonstrated to improve hemodynamics with reduced bleeding rates. The ULTIMA study, comparing CDL versus heparin alone, showed zero mortality and no major bleeding events. 3 These findings were not validated in the subsequent larger non-randomized SEATTLE II study, which demonstrated CDL achieved clinical benefit, but with an 11.4% GUSTO moderate bleeding rate. 5 Both studies employed facilitated ultrasound catheters (EKOS Corp.), a strategy predicated on the hypothesis that adjunctive ultrasound enhances thrombolysis by inducing reversible disaggregation of un-crosslinked fibrin, thereby augmenting binding sites and promoting thrombolysis.6–8 Standard CDT alone (without adjunctive ultrasound) delivers lytic via a simple multi-sidehole PA catheter. Unfortunately, there is a paucity of prospective randomized data comparing CDT to USAT, with prior retrospective uncontrolled studies yielding conflicting results without conclusive guidelines.
One comparison in a very small cohort showed USAT achieved more complete thrombus removal with lower hemorrhagic complications compared to standard CDT of longer duration and with higher tPA dose. 21 Mortality was lower in the USAT group (although not significantly so), suggesting its overall superiority. Conversely, the prospective PERFECT registry 4 showed no difference in magnitude of improvement in PASP in those treated with USAT versus standard CDT, similar to the findings of the present study. A recent retrospective review also showed no statistically significant difference in hemodynamic, clinical outcomes or procedural complications between the two approaches. 22
The present observations support the concept that standard CDT achieves similar magnitudes in improvement of hemodynamics, RV/LV ratios and clinical outcomes compared to USAT. The lack of superior improvement occurred despite USAT being associated with longer indwelling catheter durations and a higher total lytic dose. A trend toward greater significant bleeding rates was noted in the USAT group, which is possibly related to the observed difference in catheter-dwell times and lytic dose. Importantly, ICU and hospital length-of-stays were similar in both groups and there was no difference in survival-to-discharge. However, these observations must be taken in the context of this retrospective, non-randomized analysis.
It is also important to consider differences in financial costs between the two treatment strategies. A rudimentary cost-savings analysis would estimate that for each 100 patients treated, the USAT treatment approach would incur approximately $350,000 of additional cost compared to use of standard CDT alone (USAT catheter $2250, 23 standard multi-sidehole catheter $150), assuming non-inferiority of standard CDT catheters. The catheter console capital equipment cost of $14,500 would also need to be amortized according to the volume of patients treated. It is beyond the scope of the present study to delineate the exact financial cost–benefit analysis. Further investigation must be performed to determine whether these additional costs are justified by improved outcomes.
Limitations
It is important to emphasize the limitations pertinent to the methods of this study. First, the non-randomized nature of this study and the small number of patients included pose intrinsic limitations as the two groups differ with respect to key variables such as body weight and tPA dose. While not statistically significant, these and other such differences limit interpretation and extrapolation of the present findings. Second, during the study period, changes occurred in the therapeutic regimen after our institution’s establishment of a PE Response Team in August 2016 and transition to use of a standard protocol for CDL, popularized by observations from the SEATTLE II study. Prior to this, fewer patients received lytic therapy in general and the method and dose varied greatly according to physician preference. While this change likely impacted both arms of the present study equally, such a transition in method should be noted as a possible confounder and results should be interpreted accordingly. Third, the present cohort represents a selected group from the overall institutional PE population, which may create bias. A significant number of low HAS-BLED bleeding risk patients (n = 45) 24 were preferentially managed by low-dose systemic TT (rather than CDL), thereby potentially creating a selection bias for a cohort with a higher bleeding risk that was treated with CDL. Therefore, absolute bleeding rates in both groups should be viewed in that context.
Conclusion
In summary, the present retrospective study found no significant difference in clinical outcomes between ultrasound-accelerated thrombolytic catheters (USAT) and standard multi-sidehole catheters (CDT) when used for catheter-directed lysis of acute PE. This observation suggests that standard CDT may achieve similar hemodynamic and clinical benefits to USAT without the need for very expensive devices. Future prospective controlled studies will be necessary to firmly establish the optimal catheter technique and pharmacologic regimen for management of acute PE.
Footnotes
Acknowledgements
The authors would like to acknowledge the members of the Beaumont PE Response Team for their invaluable contributions: Vicki McNally, PERT Coordinator, and Drs Amr Abbas, Steven Ajluni, Aaron Berman, David Berger, Abhay Bilolikar, Abigail Brackney, Joseph Ciacci, Carol Clark, Prurshottam Dixit, Simon Dixon, Bradley Fields, Michael Gallagher, Akhil Gulati, Jeremy Handel, Ivan Hanson, Herman Kado, Matthias Kirsch, Robert Safian, Michael Savin, Mazen Shoukfeh, Robert Swor, Steven Timmis, and Stephen Vartanian.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
