Abstract
Background:
Pulmonary embolism (PE) is the third-leading cause of cardiovascular mortality, accounting for 100,000 deaths per year in the United States. Although sex-based disparities have previously been described in this population, it is unclear if these differences have persisted with the expansion of PE evaluation and treatment approaches. The purpose of this study is to investigate sex-based differences in the evaluation, management, and outcomes of patients with acute PE.
Methods:
We performed a retrospective analysis of patients enrolled in the national Pulmonary Embolism Response Team (PERT) Consortium database between October 2015 and October 2022. We evaluated patient demographics, clinical characteristics, diagnostic imaging performed, treatment at several phases of care (pre-PERT, PERT recommendations, and post-PERT), and clinical outcomes.
Results:
A total of 5722 patients with acute PE (2838 [49.6%] women) from 35 centers were included. There were no differences in PE risk category between male and female patients. Women were less likely to undergo echocardiography (76.9% vs 73.8%) and more likely to receive no anticoagulation prior to PERT evaluation (35.5% vs 32.9%). PERT teams were more likely to recommend catheter-based interventions for men (26.6% vs 23.1%), and men were more likely to undergo these procedures (21.9% vs 19.3%). In a multivariable analysis, female sex was a predictor of in-hospital mortality (OR 1.53, 95% CI 1.06 to 2.21).
Conclusions:
In this analysis, we identified sex-based differences in the evaluation and management of patients presenting with acute PE. Subsequently, women presenting with acute PE were at higher risk of in-hospital mortality.
Keywords
Background
Acute pulmonary embolism (PE) is estimated to cause between 60,000 and 100,000 deaths per year in the United States and is associated with high rates of long-term complications. 1 Differences in the incidence, clinical presentation, treatment, and outcomes between male and female patients with PE have been described in the literature for decades.2–5 Women with acute PE are more likely to present with syncope and hemodynamic compromise.5–7 Women also have higher rates of clinically significant bleeding and blood transfusion.5,8,9 Historically, studies comparing mortality outcomes in male and female patients with acute PE have shown disparate results.5–8,10
Over the past several years, treatment options for patients with PE have grown substantially. New reperfusion strategies such as catheter-directed thrombolysis, surgical embolectomy, and endovascular pulmonary artery thrombectomy are being used with increasing frequency. 9 As approaches to acute PE management evolve, it is unknown whether there are differences in the use and outcomes of these various treatment modalities between male and female patients. Furthermore, it is unclear whether any sex-based differences in the management of these patients results in disparities in outcomes.
A recent analysis using the Nationwide Inpatient Sample database found that women who underwent percutaneous thrombectomy had higher rates of complications and mortality compared to men. 11 However, this study was limited by using claims-based data which lack the granularity of patient-level data. Conversely, a retrospective study of patients with PE by Pribhish et al. found no sex differences in the use of advanced therapies or survival to discharge after adjustment. 12 Although this study included detailed patient-level data, the results were limited to a single tertiary-care center.
This study aims to clarify if there are differences in the contemporary evaluation, management, and outcomes of male and female patients with acute PE using the multicenter Pulmonary Embolism Response Team (PERT) Consortium database.
Methods
We utilized the National PERT Consortium database for this analysis. The inclusion criteria were adult patients over the age of 18 years diagnosed with acute PE and has been previously described.13,14 Patients were excluded if information on patient sex was missing or incomplete. PE risk categories were defined according to European Society of Cardiology criteria 15 ; a detailed list of data elements can be found in the supplemental material. Covariates included in the multivariable analysis model were selected based on a thorough review of existing literature and statistical significance in univariate analysis. The study protocol was written and agreed upon by all authors prior to beginning statistical analyses. The study was approved by local ethics committees of all participating centers and was exempt from needing consent. A central institutional review board was utilized for this study to increase multicenter efficiency.
Endpoints
The primary outcomes were treatment type administered prior to PERT consultation/activation, treatment type recommended by PERT, and treatment type administered after PERT activation. Secondary outcomes included diagnostic imaging, in-hospital bleeding events, and in-hospital mortality.
Statistical analyses
Descriptive statistics were presented as mean and SDs if normally distributed or as median and IQR if not normally distributed. Categorical variables were presented as frequencies and percentages. For continuous variables, Student’s t-test or Mann–Whitney U-test, as appropriate, was performed for comparisons between groups. Pearson’s chi-squared test was utilized for analysis of categorical variables. Logistic regression models were constructed to evaluate the secondary clinical outcomes of in-hospital death and in-hospital bleeding stratified by sex and tertiles of age (< 45 years, 45–65 years, and > 65 years). Results were presented as odds ratio (OR) with 95% CI. Statistical significance was defined as p < 0.05. Multivariable logistic regression models were constructed to evaluate risk factors for in-hospital mortality and in-hospital major bleeding. Explanatory variables for these models can be found in the supplemental material. Data were analyzed using Stata/MP 16.1 (StataCorp LLC, College Station, TX, USA).
Results
Baseline patient demographics and clinic characteristics
A total of 5722 patients with acute PE (2838 women and 2884 men) were included in this study and came from 35 centers in the United States between October 2015 to October 2022. Baseline characteristics, risk factors for venous thromboembolism (VTE), and medication use stratified by patient sex are summarized in Table 1. Sixty-eight patients were excluded from the study due to missing data on patient sex. Women were older (mean ± SD: 62.4 ± 17.7 vs 60.9 ± 15.0 years), had a higher body mass index (BMI) (33.4 ± 8.5 vs 31.5 ± 7.7 kg/m2), and higher rates of hypertension (53.1% vs 50.3%), whereas men were more likely to have a history of diabetes mellitus (20.2% vs 18.0%), cigarette smoking (18.1% vs 11.6%), and hyperlipidemia (32.0% vs 29.1%). Although there were no differences in rates of prior personal history of VTE, women had more oral contraceptive use (6.7% vs 0.1%) and reduced mobility (31.6% vs 27.1%).
Baseline characteristics, vital signs, and laboratory studies.
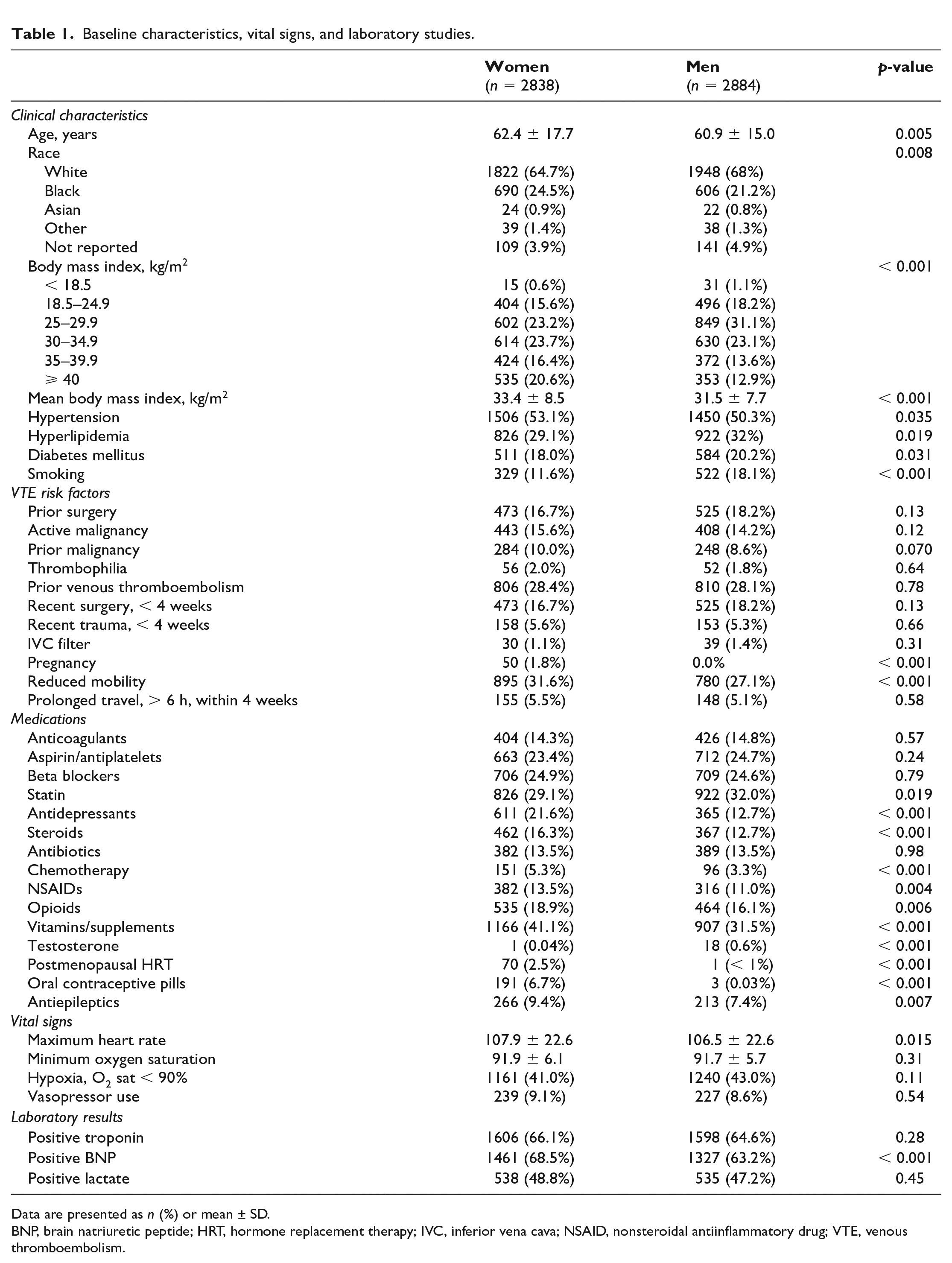
Data are presented as n (%) or mean ± SD.
BNP, brain natriuretic peptide; HRT, hormone replacement therapy; IVC, inferior vena cava; NSAID, nonsteroidal antiinflammatory drug; VTE, venous thromboembolism.
Vitals, laboratory studies, and PE severity
At presentation, heart rate was slightly higher in women (107.9 ± 22.6 vs 106.5 ± 22.6 bpm). There was no difference in minimum oxygen saturation, frequency of hypoxia (SpO2 < 90%), or vasopressor use between groups. The distribution of the PE risk category is shown in Figure 1. Most patients presented with intermediate-risk PE, followed by high-risk PE, and low-risk PE. There were no statistically significant differences in PE risk categories between men and women (Figure 1). A risk category was not assigned for 456 (8.0%) patients.
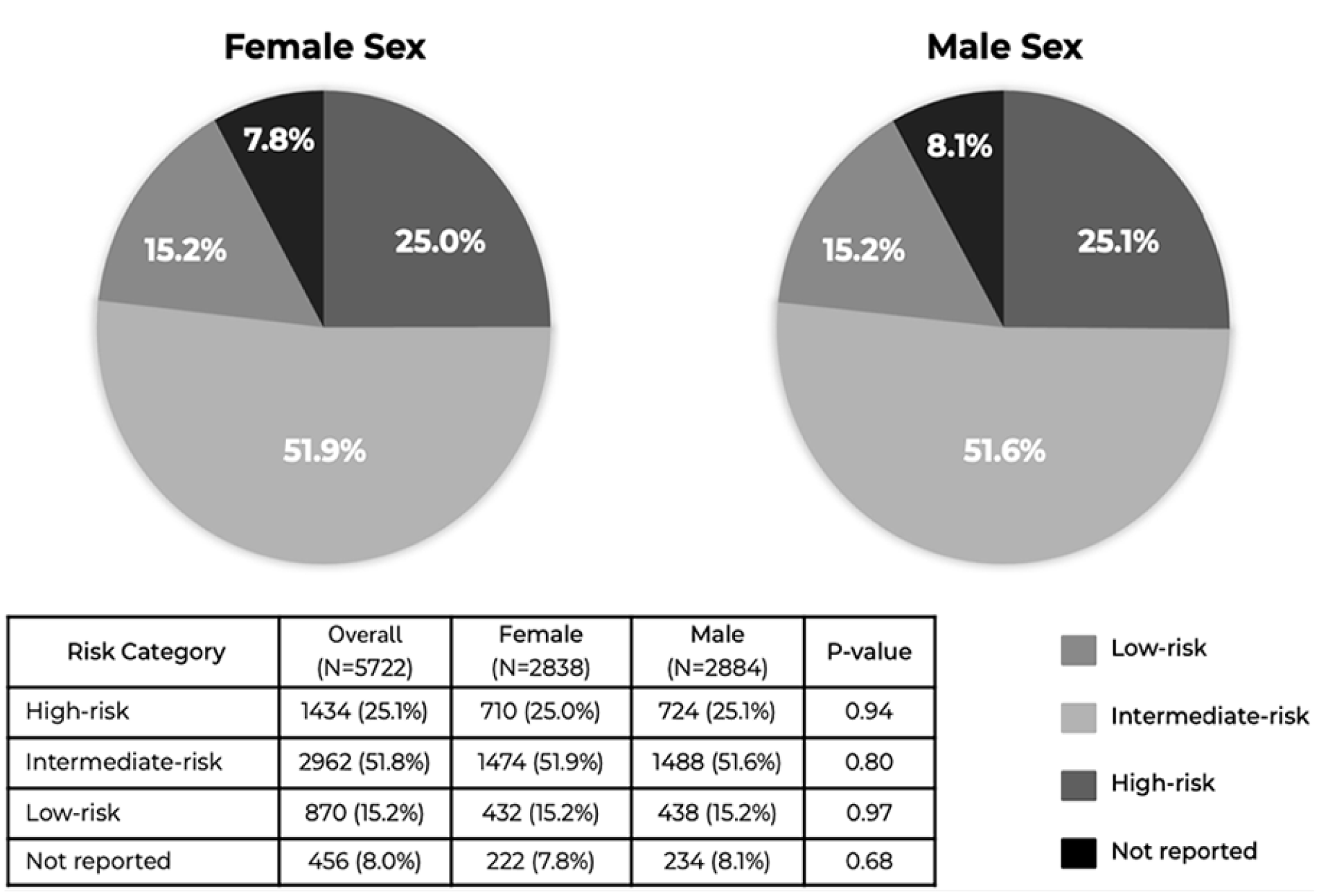
Distribution of pulmonary embolism risk category by patient sex. Pie charts demonstrating proportions of high-risk PE, intermediate-risk PE, and low-risk PE, and unknown risk category for patients of each sex. There were no differences in the distribution of PE risk category between women and men.
Diagnostic imaging
Imaging studies obtained are summarized in Table 2. Men were more likely to have computed tomography pulmonary angiogram (CTPA) as their diagnostic imaging modality (94.8% vs 93.4%). Ventilation-perfusion (VQ) scans were more common among women (2.5% vs 1.7%). After excluding pregnant patients, a nonsignificant trend in VQ scan frequency persisted (2.5% vs 1.7%). Men were more commonly diagnosed with saddle PE (21.2% vs 18.5%). The frequency of main pulmonary artery (51.3% and 52.4%), right atrium (RA) or right ventricle (RV) (6.2% and 6.7%), or distal pulmonary artery embolism (78.7% and 80.0%) location were similar between groups. Right heart strain and RV/LV ratio on CT did not differ between men and women.
Diagnostic imaging.
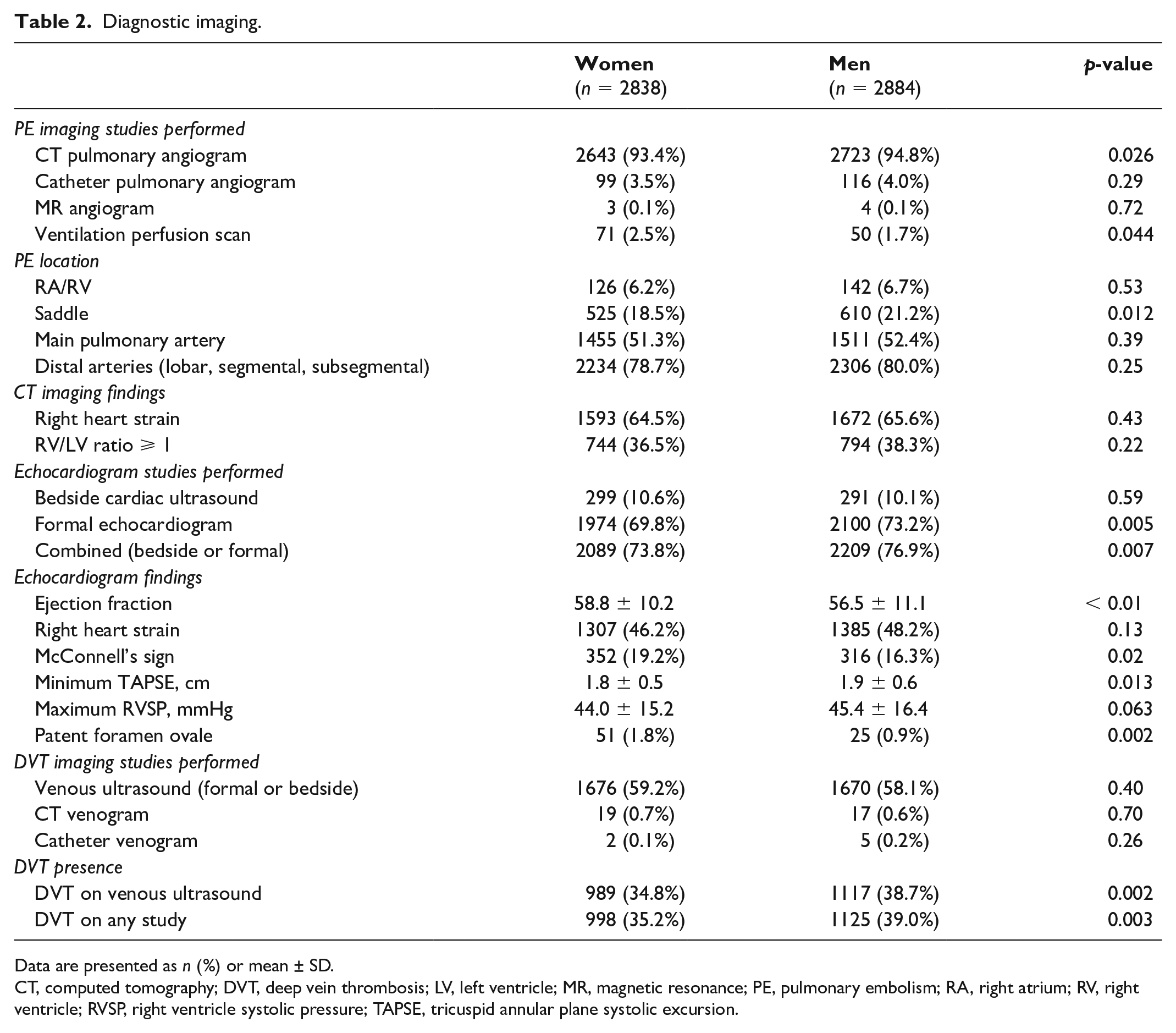
Data are presented as n (%) or mean ± SD.
CT, computed tomography; DVT, deep vein thrombosis; LV, left ventricle; MR, magnetic resonance; PE, pulmonary embolism; RA, right atrium; RV, right ventricle; RVSP, right ventricle systolic pressure; TAPSE, tricuspid annular plane systolic excursion.
We found that a higher proportion of formal echocardiograms were performed at the time of PE diagnosis in the male cohort (73.2% vs 69.8%). After multivariable analysis controlling for patient age, race, BMI, hypertension, diabetes, smoking, hyperlipidemia, and PE risk category, men remained more likely than women to receive a formal echocardiogram at diagnosis (OR 1.16, 95% CI 1.02 to 1.32, p = 0.021). Echocardiographic evidence of right heart strain did not differ between groups. However, women were more likely to demonstrate McConnell’s sign (19.2% vs 16.3%) and had a lower tricuspid annular plane systolic excursion (TAPSE; 1.8 ± 0.5 vs 1.9 ± 0.6 cm). There was no difference between maximum RV systolic pressure between groups.
Treatment modality
Treatment recommendations and treatments received are summarized in Tables S1–S4. Overall, men were more likely to receive intravenous (IV) heparin (54.1% vs 50.9%) and women were more likely to receive no anticoagulation (35.5% vs 32.9%) prior to PERT activation. Catheter-based interventions were more likely to be recommended by PERT for men (26.6% vs 23.1%) and more likely to be received by men (21.9% vs 19.3%). There were no differences in systemic thrombolysis or surgical embolectomy between the groups.
The results of treatment recommendations and treatments received stratified by PE risk category are shown in Tables S2–S4. Among patients with high-risk PE (Table S2), PERT was more likely to recommend catheter-based interventions for men (33.1% vs 25.5%) and men were more likely to receive catheter-based interventions (26.5% vs 20.0%). Men remained more likely to receive catheter-based interventions after adjusting for several baseline covariates (age, race, BMI, hypertension, diabetes, smoking, and hyperlipidemia, and PE risk category) (OR 1.19, 95% CI 1.04 to 1.38, p = 0.010). For patients with intermediate-risk PE (Table S3), women were more likely to receive no anticoagulation (35.5% vs 32.0%) prior to PERT activation. Surgical thrombectomy was recommended more frequently for men (2.4% vs 1.3%) although there was no significant difference in the proportion of patients who received this treatment (1.5% men vs 0.7% women). Men were also more likely to receive an IVC filter (6.2% vs 4.4%) than women. There were no differences in rates of systemic thrombolysis or catheter-based therapies. For patients with low-risk PE (Table S4), no anticoagulation was more likely to be recommended by PERT for men (7.5% vs 3.9%). There were no differences in the treatments received prior to or after PERT activation.
Clinical outcomes
There were no differences in the incidence of in-hospital major bleeding between women and men (5.3% and 4.9%, respectively, OR 0.93, 95% CI 0.74 to 1.18) (Table S5). When stratified by age, bleeding events were higher among women aged 45–65 years (5.8% vs 3.9%, OR 1.51, 95% CI 1.01 to 2.26). In a multivariable analysis, predictors of in-hospital major bleeding included high-risk acute PE (OR 4.05, 95% CI 2.44 to 6.73), recent surgery (OR 1.59, 95% CI 1.19 to 2.12), and systemic thrombolysis (OR 1.90, 95% CI 1.27 to 2.84). White race was associated with a lower rate of in-hospital major bleeding (Table S6). There were no differences in in-hospital mortality rates in the univariate analysis (7.7% vs 8.2%, OR 1.07, 95% CI 0.89 to 1.30; Table S7). In a multivariable analysis that incorporated blood lactate levels, female sex was a predictor for in-hospital mortality (OR 1.53, 95% CI 1.06 to 2.21). Female sex remained a predictor of mortality in a multivariable analysis that excluded whole blood lactate measurements (OR 1.35, 95% CI 1.00 to 1.81) (Tables 3 and 4; model covariates found in supplemental material). In addition to female sex, other predictors of in-hospital PE-related mortality included malignancy (OR 1.60, 95% CI 1.09 to 2.35), high-risk PE category (OR 2.86, 95% CI 1.20 to 6.81), elevated lactate (OR 2.35, 95% CI 1.58 to 3.50), and vasopressor use (OR 2.38, 95% CI 1.49 to 3.80) (Table 3).
Regression analysis evaluating the association between baseline variables and in-hospital mortality (with lactate).
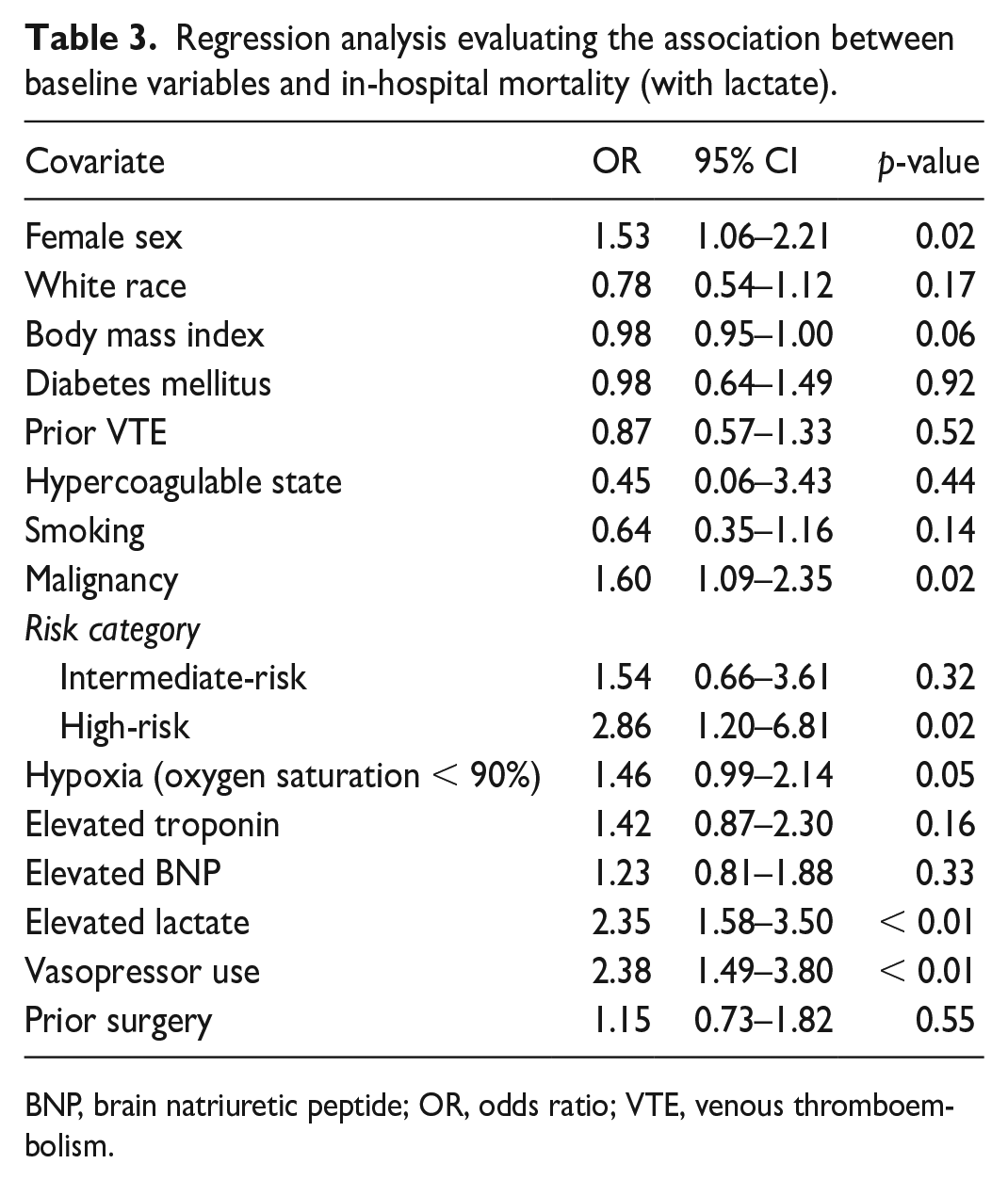
BNP, brain natriuretic peptide; OR, odds ratio; VTE, venous thromboembolism.
Regression analysis evaluating the association between baseline variables and in-hospital mortality (without lactate).
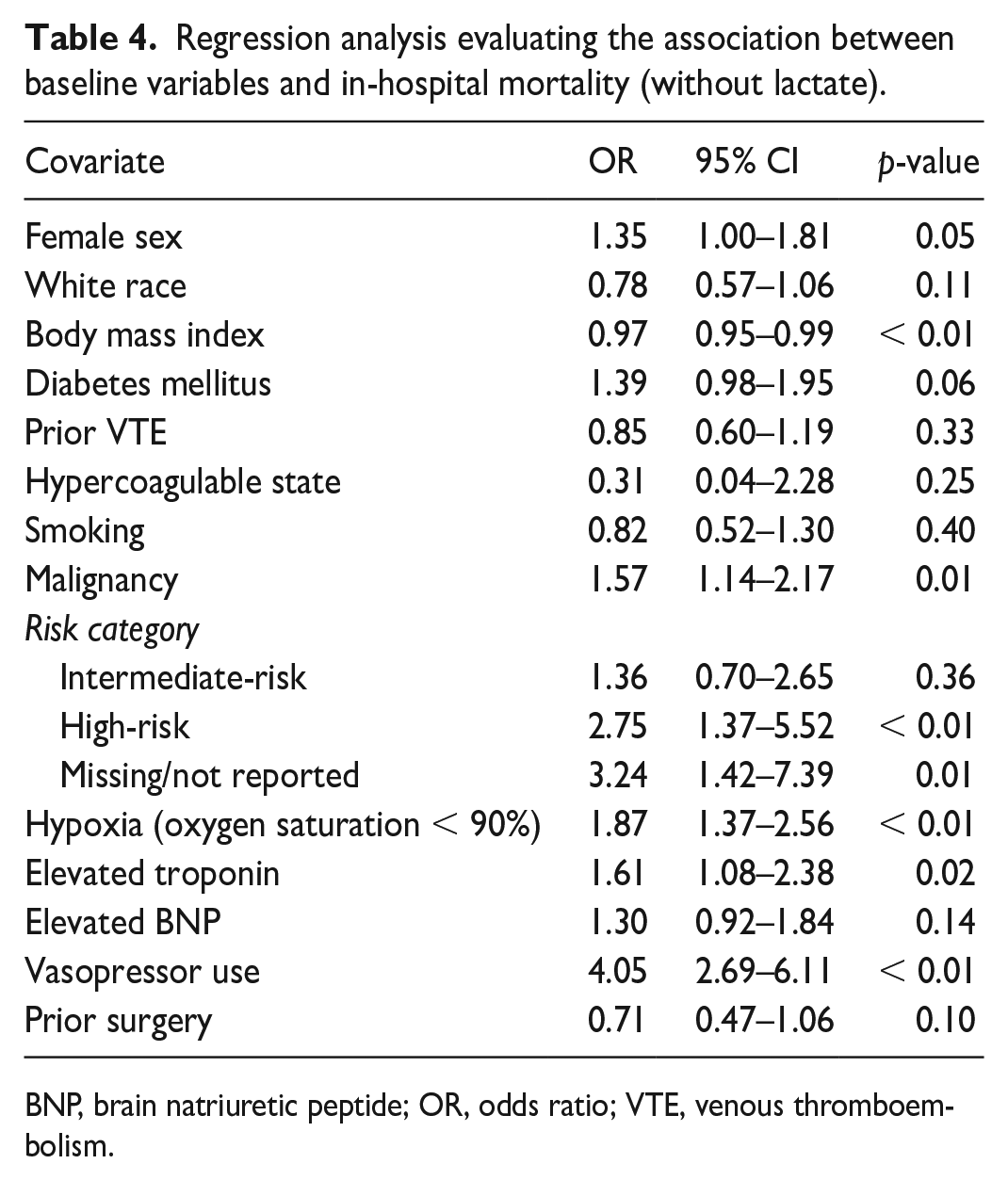
BNP, brain natriuretic peptide; OR, odds ratio; VTE, venous thromboembolism.
Discussion
In this large cohort of patients from a prospective, multicenter quality assurance database, we found significant differences in the evaluation, treatment recommendations, treatments received, and outcomes between men and women presenting with acute PE. Specifically, we found that (1) women were less likely to undergo echocardiography, (2) men were more likely to receive IV heparin prior to PERT activation and more likely to receive catheter-based interventions despite presenting with a similar severity of illness, and (3) female sex was a predictor of in-hospital mortality after accounting for baseline variables.
Echocardiography can be used to identify RV dysfunction and further risk stratify patients with acute PE. Women were less likely to receive an echocardiogram despite presenting with similar PE-risk severity and being less likely to have had a CTPA, which may also provide information on RV function. Of note, male patients in our study were more likely to have saddle PE. It is possible that the greater proximal clot burden visualized among men pushed providers to get additional imaging to assess RV function by echocardiography.
The optimal treatment of acute PE is complex and should be individualized based on a patient’s comorbidities and risk of decompensation.16–19 Initiation of timely anticoagulation is imperative in all patients who do not have contraindications. In our study, men were more likely to receive IV heparin prior to PERT activation. Women were more likely to receive no anticoagulation prior to activation of PERT and had lower usage of IV heparin and catheter-based therapies. After PERT activation, women were equally likely to receive IV heparin. However, female patients remained less likely to undergo catheter-based interventions. These findings were driven by differences in recommendations and management in the high-risk PE group. Although small differences in baseline characteristics were present, there were no sex differences in the PE risk categories. Thus, differences in management are not fully accounted for by the severity of illness at presentation.
There are several plausible explanations for the sex-based disparities in PE evaluation and treatment modalities identified in our analysis. First, female patients may have presented with atypical symptoms that confounded the diagnosis and delayed treatment. In a recent study evaluating patients presenting with PE at a single center in Tokyo, female patients were more likely to report symptoms of dyspnea and disturbance of consciousness whereas male patients were more likely to report chest pain and leg symptoms. 5 Second, sex- and gender-related sociocultural differences may have influenced patient preferences and provider behavior. In a prior study, patients presenting to the emergency room with potential acute coronary syndrome were surveyed on their preferences for noninvasive versus invasive testing. Investigators found that patients identified as women were less likely to accept the physician’s recommendation for cardiac catheterization and more likely to select medical therapy over percutaneous coronary intervention. 20 However, preferences for invasive procedures would not explain the differences we identified in diagnostic imaging, anticoagulation use prior to PERT activation, or differences in recommendations for catheter-based interventions. Alternatively, implicit bias among providers may play a role. In a 2017 study exploring the impact of implicit gender bias, cardiologists were more likely to rate the utility of invasive testing higher in men compared to women with similar cardiovascular risk and abnormal stress tests. 21
It is well established that female patients may be at increased risk for bleeding events due to various hormone-related hematologic, pharmacokinetic, and anatomic factors.8,9,22-24 Though there were no differences in rates of in-hospital major bleeding in the overall cohort, there was an increased risk among women aged 45–65. Similarly, the Pulmonary Embolism Thrombolysis (PEITHO) study found that female patients were more likely to experience major extracranial bleeding after systemic thrombolysis. 25 Of note, the women in our study were taking more NSAIDs and steroids, putting them at a greater risk for bleeding compared to men.
After accounting for multiple baseline differences in patient characteristics and presentation, we found that female sex was a predictor for in-hospital mortality. These findings suggest that the differences identified in PE evaluation and management in this study may have led to worse outcomes in female patients. Prior studies examining mortality differences in PE based on patient sex have yielded discrepant results. Goldhaber et al. analyzed data from the International Cooperative Pulmonary Embolism Registry (ICOPER) and found that men were at an increased risk of PE-related mortality. Importantly, this study included patients presenting with PE between 1995 and 1996 and thus predates contemporary risk classification systems and catheter-directed therapies. Mortality rates were nearly twice as high in the ICOPER registry despite only 4.2% of these patients presenting with hemodynamic instability. 23 By contrast, 9% of the patients in the current study required vasopressors at the time of presentation. The differences between the ICOPER data and our study highlight the advancements made in PE diagnosis and management over the last two decades.
Another study by Tanabe et al. analyzed data from a Japanese registry and found that women had a significantly higher risk of 30-day PE-related mortality (5.0% vs 2.8%). However, women in that study also presented with greater severity of illness than men. 5 In a study of the National Inpatient Sample database by Agarwal et al., female patients admitted with PE had a significantly higher risk of in-hospital mortality. Owing to limitations of the data sample, however, this study could not account for severity of illness on presentation, laboratory data, or echocardiographic findings. 9 Another study using a Spanish registry found that 30-day mortality was similar between sexes. However, when restricting the analysis to patients who were hemodynamically stable at the time of presentation, female patients had a higher risk of death. 8 A recent meta-analysis incorporating many of these studies reported no overall difference in mortality between male and female patients. Nevertheless, variations in study methods and length made the results difficult to synthesize and interpret. 26
Our study adds to the body of literature, providing valuable insight into the differences in the evaluation, management, and outcomes between men and women presenting with PE. Our findings suggest that sex-based differences in PE evaluation and management may be changing alongside treatment advancements in the field. Furthermore, it is possible that these differences may lead to disparities in clinical outcomes between male and female patients. This study highlights the importance of future research to elucidate the sex-based factors that affect the care of patients with acute PE.
Strengths and limitations
The strength of the present analysis is the large number of patients included from a multicenter, prospective PE-specific registry from diverse settings around the United States. To date, this registry is the largest PE-specific database. The registry allows evaluation of several different PE treatment modalities throughout all phases of care. There are, however, multiple limitations to this study. First, our analyses did not account for all the baseline differences in the male and female cohorts. In addition, the PERT registry did not provide data on PE-specific mortality. Lastly, we categorized patients according to the sex category recorded in the registry. However, information on patients’ gender identities and how each site defined patient sex (assigned at birth, legal, etc.) was not available. Owing to this limitation, we were unable to explore disparities between cisgender and transgender individuals. Future registry data should include these key variables in order to understand the complex manner in which biological sex characteristics, gender-specific and gender-affirming hormone treatments, gender identity, and gender expression can impact patient care and outcomes.
Conclusion
In this large cohort of patients from a prospective, multicenter quality assurance database, we found significant sex-based differences in the evaluation and management of patients with acute PE. After accounting for baseline differences, female sex is associated with higher risk of in-hospital mortality.
Supplemental Material
sj-pdf-1-vmj-10.1177_1358863X241281872 – Supplemental material for The influence of patient sex on pulmonary embolism evaluation, treatment modality, and outcomes
Supplemental material, sj-pdf-1-vmj-10.1177_1358863X241281872 for The influence of patient sex on pulmonary embolism evaluation, treatment modality, and outcomes by Joshua Newman, Elizabeth Bruno, Sorcha Allen, Jonathan Moore, Robert Zilinyi, Asma Khaliq, Fahad Alkhafan, Clara Vitarello, Robert Lookstein, Brent Keeling, C Michael Gibson, Kenneth Rosenfield, Eric A Secemsky, Rachel P Rosovsky and Amir Darki in Vascular Medicine
Footnotes
Acknowledgements
A commentary by Javanmardi et al. 27 will accompany this article.
Declaration of conflicting interest
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Dr Lookstein is a member of the scientific advisory board for Boston Scientific and Medtronic; is a consultant for Penumbra, Abbott Vascular, Neptune Medical, Bard Vascular, Cordis, Biosense Webster, Abbott Vascular; receives research support from Philips Healthcare, Spectranetics, Terumo, Boston Scientific, INARI, Penumbra, Ethicon, Vesper, and Black Swan; and is an equity shareholder for Imperative Vascular, Summa Vascular, Innova Vascular, and Thrombolex. Dr Gibson has received research grant support from Johnson and Johnson, Bristol Myers Squibb, and Daiichi Sankyo. Dr Rosenfield is a member of the scientific advisory board for Abbott Vascular, Althea Medical, Angiodynamics, Auxetics, Becton-Dickinson, Boston Scientific, Contego, Crossliner, Innova Vascular, Inspire MD, Janssen/Johnson and Johnson, Magneto, Mayo Clinic, MedAlliance, Medtronic, Neptune Medical, Penumbra, Philips, Surmodics, Terumo, Thrombolex, Truvic, Vasorum, and Vumedi; has received research grant support from the National Institutes of Health, Abiomed, Boston Scientific, Novo Nordisk, Penumbra, and Gettinge-Atrium; has equity in Accolade, Access Vascular, Aerami, Althea Medical, Auxetics, Contego, Crossliner, Cruzar Systems, Embolitech, Endospan, Imperative Care/Truvic, Innova Vascular, InspireMD, JanaCare, Magneto, MedAlliance, Neptune Medical, Orchestra, Prosomnus, Shockwave, Skydance, Summa Therapeutics, Thrombolex, Valcare, Vasorum, and Vumedi; and is a board member for the National PERT Consortium, a not-for-profit 501(c)(3) organization. Dr Secemsky has received funding from the National Institutes of Health, Food and Drug Administration and the University of California San Francisco; receives institutional research support from Becton-Dickinson, Boston Scientific, Cook, Cardiovascular Systems Incorporated, Laminate Medical, Medtronic, and Philips; and is a consultant for Abbott, Becton-Dickinson, Bayer, Boston Scientific, Cook, Cardiovascular Systems Incorporated, Medtronics, Philips and VentureMed. Dr Rosovsky receives institutional research support from Bristol Myers Squibb and Janssen; is a consultant for Abbott, Bristol Myers Squibb, Dova, Inari, Janssen, Penumbra; is a national lead investigator for Storm-PE and Penumbra; and is a board member of the National PERT Consortium, a not-for-profit 501(c)(3) organization. Dr Darki has received research grant support from Boston Scientific. All remaining authors have reported that they have no relationships to disclose.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
