Abstract

Keywords
Drug-coated endovascular therapies for femoropopliteal artery lesions are intended to prevent or delay restenosis relative to uncoated therapies. 1 Paclitaxel-based drug-coated balloons have consistently demonstrated 1-year patency benefits compared with uncoated percutaneous transluminal angioplasty (PTA) in clinical trials1,2 and continually expanding evidence has overcome recent controversy regarding paclitaxel safety. 3 The low-dose paclitaxel Ranger drug-coated balloon (DCB) (Boston Scientific, Marlborough, MA, USA) demonstrated short-term clinical benefit in the RANGER II SFA randomized trial, with 1-year primary endpoint analysis showing primary patency superior to standard PTA; 4 long-term results have not yet been reported.
This 5-year follow up to the RANGER II SFA trial (
Statistical analyses beyond the 12-month primary endpoints were considered exploratory and not powered for statistically based conclusions. The Kaplan–Meier product-limit method was used to estimate the primary patency and freedom from clinically driven TLR (CD-TLR) rates. CD-TLR was defined as the need for a repeat procedure due to Rutherford category or worsening ABI. Statistical analyses were performed with SAS, version 9.4 (SAS Institute, Inc.).
As previously described, this trial collected informed consent and enrolled 376 patients: mean age 70.2 years, 36.2% women, and 42.8% with diabetes mellitus (no significant differences between groups). 4 Overall, the mean lesion length was 83.4 mm and the mean ABI at baseline was 0.8 ± 0.2, with 28.2% of patients having moderate or severe calcification (no significant differences between groups). 4 At baseline, chronic limb-threatening ischemia (CLTI) with ischemic rest pain (Rutherford score of 4) existed in 9.4% (26/278) and 7.1% (7/98) of patients in the Ranger DCB and PTA groups, respectively, and the remaining patients had moderate or severe claudication (Rutherford score of 2 or 3). Follow-up visits at 36 months were completed by 212 (76.3%) and 75 (76.5%) patients, and a total of 213 (76.6%) and 73 (74.5%) patients in the respective groups were evaluable for the 5-year safety endpoint analysis. Long-term acetylsalicylic acid use was similar between groups at both 36 months (77.0% vs 79.5%, p = 0.66) and 60 months (76.4% vs 80.0%, p = 0.57).
Patients treated with the Ranger DCB maintained a high primary patency rate of 77.4% at 36 months (day 1095) as estimated from Kaplan–Meier analysis, with patients treated with PTA at 73.5% (log-rank p = 0.26; Figure 1A). Most patients in the Ranger DCB group demonstrated hemodynamic and primary sustained clinical improvement at both 24 and 36 months, with the between-group difference diminishing over time (Supplemental Table S1).
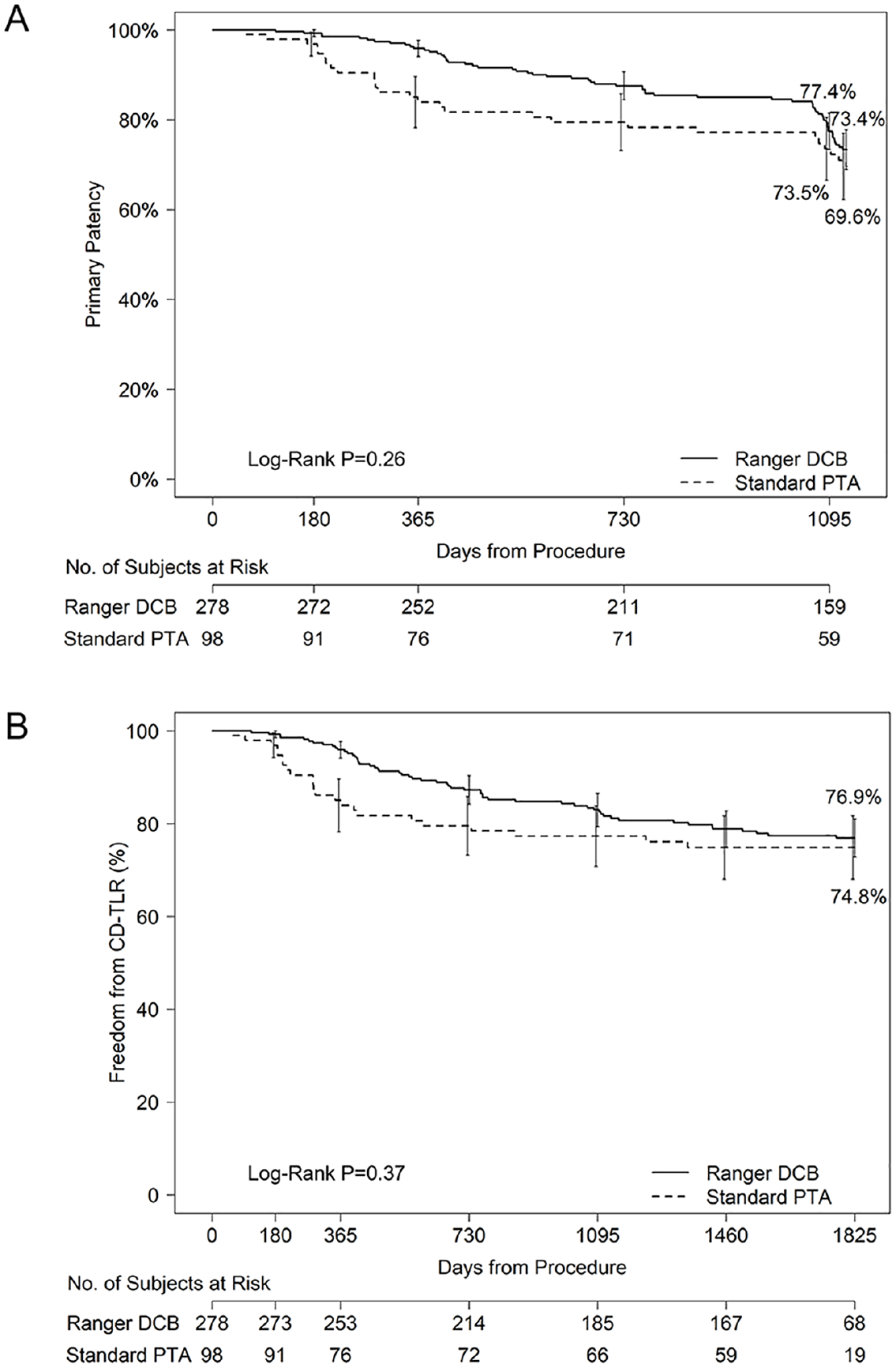
Three-year primary patency and 5-year freedom from CD-TLR.
The 5-year incidence of crude mortality, target limb major amputation, and CD-TLR are shown in Table S1; rates did not differ significantly between the Ranger DCB and PTA arms at 5 years. The Kaplan–Meier estimates of freedom from CD-TLR at 5 years were 76.9% vs 74.8% for the Ranger DCB and PTA, respectively (log-rank p = 0.37; Figure 1B).
These long-term RANGER II SFA trial results reinforce previous short-term findings, 4 with the Ranger DCB maintaining a high 36-month primary patency rate of approximately 77% with clinical improvement observed from baseline. At 5 years, the treatment group continued to demonstrate a high freedom from the CD-TLR rate. 5 Safety outcomes were comparable to standard PTA through 5 years, with no significant difference in mortality observed. Ranger DCB performance in this study is consistent with results from the randomized COMPARE study. 6
Though the primary patency and freedom from TLR rates were high for the DCB group, statistically significant differences from the PTA arm were not observed at 36- and 60-month follow up due to unexpectedly high rates in the PTA arm. Successful predilatation before randomization was required in this study and may partly explain the reduced need for reintervention in the PTA arm relative to prior randomized studies.5,7 Additionally, the RANGER II trial was powered to detect differences through 12 months, which may have also impacted the ability of this long-term follow up to detect statistical differences. Paclitaxel from this device has previously been shown to diminish after 60 days postprocedure, 8 which may partly explain why the clinical benefit is most strongly observed at earlier stages after treatment in this progressive disease.
One limitation of this study is leveraging a 3:1 clinical trial design, which puts greater weight on individuals in the PTA arm. Additionally, having the comparison group of standard PTA shows the effect of paclitaxel but does not address how this device compares to other DCBs as in other studies. 6
Overall, the long-term results from this study support the safety profile of the Ranger DCB beyond 12 months. Compared to the control group, patients treated with the Ranger DCB showed a continuation of higher patency at 36 months and a higher freedom from the CD-TLR rate at 60 months, indicating delayed need for additional interventions during long-term follow up. However, the differences between the two treatments diminished with time and did not reach statistical significance.
Supplemental Material
sj-pdf-1-vmj-10.1177_1358863X251349782 – Supplemental material for Long-term results of the RANGER II SFA randomized trial of the Ranger drug-coated balloon vs standard percutaneous transluminal angioplasty for femoropopliteal artery treatment
Supplemental material, sj-pdf-1-vmj-10.1177_1358863X251349782 for Long-term results of the RANGER II SFA randomized trial of the Ranger drug-coated balloon vs standard percutaneous transluminal angioplasty for femoropopliteal artery treatment by Ravish Sachar, Yoshimitsu Soga, Louis Lopez, Amane Kozuki, Marianne Brodmann, Vijay S Ramanath, Masahiko Fujihara, Anna M Chavez, Mohammad M Ansari and Thomas Zeller in Vascular Medicine
Footnotes
Acknowledgements
The authors thank the following Boston Scientific employees for their assistance: Lei Zhang, ScM, study statistician; Cynthia Peterson, MS, clinical trial management; Gregory M Sindberg, PhD and Elizabeth Davis, PhD, for medical writing support.
Data availability statement
Declaration of conflicting interests
Dr Sachar has served as consultant for Boston Scientific and Medtronic; has received research funding from Boston Scientific, Medtronic, and Terumo in the last 24 months; and is a shareholder of Contego Medical. Dr Soga serves as advisor to Boston Scientific. Dr Brodmann has served as consultant for Medtronic, BD BARD, Philipps, Biotronik, Boston Scientific, Cagent, Shockwave, Cook Medical, Bayer Healthcare, Daiichi Sankyo, MedAlliance, Reflow Medical; Speakers’ Bureau for Medtronic, BD BARD, Philips, Biotronik, Boston Scientific, Cagent, Shockwave, Cook Medical, Bayer Healthcare, Daiichi Sankyo, MedAlliance, Reflow Medical. Dr Ramanath has received honoraria from Shockwave Medical, Cardiovascular Systems, Inc., Boston Scientific Corp.; has received compensation for educational programs from Boston Scientific Corp., Terumo Medical Corp. Dr Fujihara has served as consultant for Boston Scientific. Dr Chavez is an employee of and holds stock in Boston Scientific. Dr Ansari has served as advisory board member for Boston Scientific, Medtronic, and Cordis; a steering committee member for Philips; has received compensation for educational programs from Edwards, Gore, Bard, and Boston Scientific; and has received funds for research or clinical trials from Abbott, Boston Scientific, and Bard. Dr Zeller reports receiving honoraria from Abbott Vascular, Veryan, Biotronik, Boston Scientific Corp., Cook Medical, Gore & Associates, Medtronic, Philips-Spectranetics, TriReme, Veryan, Shockwave, Biotronik, B Braun, Efemoral; has consulted for Boston Scientific Corp., Cook Medical, Gore & Associates, Medtronic, Spectranetics, Veryan, Intact Vascular, Veryan; holds common stock in QT Medical. The remaining authors have no conflicting interests to disclose.
Funding
The RANGER II SFA trial was funded by Boston Scientific, who also provided trial management, biostatistical analysis, and medical writing services.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
