Abstract
Background:
Peripheral artery disease (PAD) and end-stage kidney disease (ESKD) are independent risk factors affecting outcomes like in-hospital mortality. The role of a frailty index in prognosticating outcomes in patients with ESKD and PAD is unknown. We aim to assess the prognostic value of the Hospital Frailty Risk Score (HFRS) and its association with outcomes in these patients.
Methods:
We identified patients with PAD using data from the United States Renal Data System (USRDS) for the years 2015–2018. These patients were stratified into three categories of frailty risk based on their HFRS, a validated frailty assessment tool using ICD-10 codes: low (< 5), intermediate (5–10), and high risk (> 10) and based on revascularization or not. Primary outcomes included in-hospital mortality and composite of mortality or major amputation. Secondary outcomes encompassed postdischarge mortality and composite of mortality or major amputation at 1 year.
Results:
Out of 122,649 patients with PAD and ESKD, 4118 underwent revascularization and 118,531 did not. In-hospital outcomes demonstrated a nonlinear relationship and postdischarge outcomes displayed a nearly linear relationship with HFRS, regardless of revascularization status. In both cohorts, the high-risk group was associated with a significantly higher risk of in-hospital mortality/amputation (revascularization: odds ratio [OR] 4.6, 95% CI 3.3–6.2, p < 0.001; no revascularization: OR 3.1, 95% CI 2.8–3.3, p < 0.001) and mortality (revascularization: OR 5.5, 95% CI 3.4–8.7, p < 0.001; no revascularization: OR 5.1, 95% CI 4.6–5.6, p < 0.001) compared with the low-risk group.
Conclusion:
In patients with ESKD and PAD, the HFRS serves as a valuable predictor of mortality and amputation irrespective of revascularization. This information can support informed decision-making.
Keywords
Background
End-stage kidney disease (ESKD) and peripheral artery disease (PAD) are independently associated with higher mortality and morbidity. 1 The coexistence of these conditions presents a complex clinical scenario that significantly impacts the quality of life and overall prognosis of affected individuals. 2 Frailty, characterized by a state of diminished physical and physiological reserve, is highly prevalent in the ESKD population, with estimates of a greater than 60% prevalence in these patients. 3 The impact of frailty on mortality and morbidity in patients with ESKD and PAD has not been well studied. In this very high-risk population, frailty may help identify an even higher-risk patient group. Furthermore, a deeper understanding of the interplay between frailty, PAD, and ESRD can inform healthcare professionals about the most appropriate strategies for improving the quality of care and enhancing the overall outcomes in these patients.
Several validated frailty screening tools have been developed, such as the 5-item modified frailty index (mFI-5) and the Vascular Quality Initiative-derived risk analysis index (RAI). However, significant disparities in frailty detection persist, and frailty assessments remain nonstandardized and cumbersome.4–6 It remains unclear whether these tools accurately forecast outcomes in patients with ESKD, a patient group already burdened with elevated baseline risk. Furthermore, given the substantial number of patients with ESKD and PAD with impaired mobility compared to the general population, there are legitimate concerns regarding the clinical applicability of these objective frailty assessment tools within this specific patient cohort.
The Hospital Frailty Risk Score (HFRS) is a validated score calculated using various medical conditions based on the International Classification of Diseases (ICD) codes. It can be integrated into the electronic medical record to identify frail patients. 7 Despite the increasing interest in frailty assessment in various clinical contexts, there is limited information regarding the prognostic value of the newly developed HFRS and its association with clinical outcomes in patients with ESKD and PAD. Previous studies have investigated using the HFRS score in patients with chronic limb-threatening ischemia (CLTI) and have highlighted the potential positive implications in predicting outcomes.8,9 Hence, in this study, we aim to identify the utility of HFRS in predicting the impact of frailty in patients with ESKD on a stable, homogenous PAD group without CLTI requiring intervention or not.
Methods
Data source
The US Renal Data System (USRDS) serves as a prospective database encompassing most hemodialysis (HD) patients across the United States. It consolidates patient-specific information obtained from various sources, including the Centers for Medicare and Medicaid Services, the Centers for Disease Control and Prevention, the United Network for Organ Sharing, and end-stage renal disease networks. 10 We accessed deidentified patient-specific data for all individuals with ESKD and PAD from the USRDS database, covering the period from October 2015 to December 2018. This study was exempted by the University of Kansas Medical Center institutional review board due to the deidentification of patient data and prior approval by the ethical committee. Our study followed the Strengthening the Reporting of Observational Studies in Epidemiology reporting guidelines.
Study population
We queried the USRDS using the International Classification of Diseases, 10th Revision, Clinical Modification (ICD-10-CM) codes to identify patients with a diagnosis of PAD, and selected their first hospitalization as the index hospitalization if they had multiple hospitalizations during the study period, ensuring that patients with multiple hospitalizations were only counted once (Supplemental Table S1). We excluded individuals who were under the age of 18 years, those who did not reside in the United States, and individuals whose primary payer during the index hospitalization was not Medicare Part A and Part B coverage. Additionally, patients who had undergone a renal transplant either before or during the index hospitalization were also excluded from our analysis. Furthermore, we considered the duration of dialysis treatment before the index hospitalization. To be included in our study, patients were required to have received continuous dialysis for a minimum of 3 months (equivalent to 90 days) leading up to their index hospitalization. This ensured that we focused on a population with a consistent dialysis history. Additionally, within the subset of patients with PAD, we implemented an additional exclusion criterion. We excluded individuals who had a diagnosis of CLTI to maintain a more homogenous and clinically stable PAD population. We divided patients with PAD into two groups: surgical or endovascular revascularization during index hospitalization and nonrevascularization. The ICD-10 codes used were standard as used in previous studies.8,11 For a visual representation of our selection and exclusion process, please refer to Figure 1.
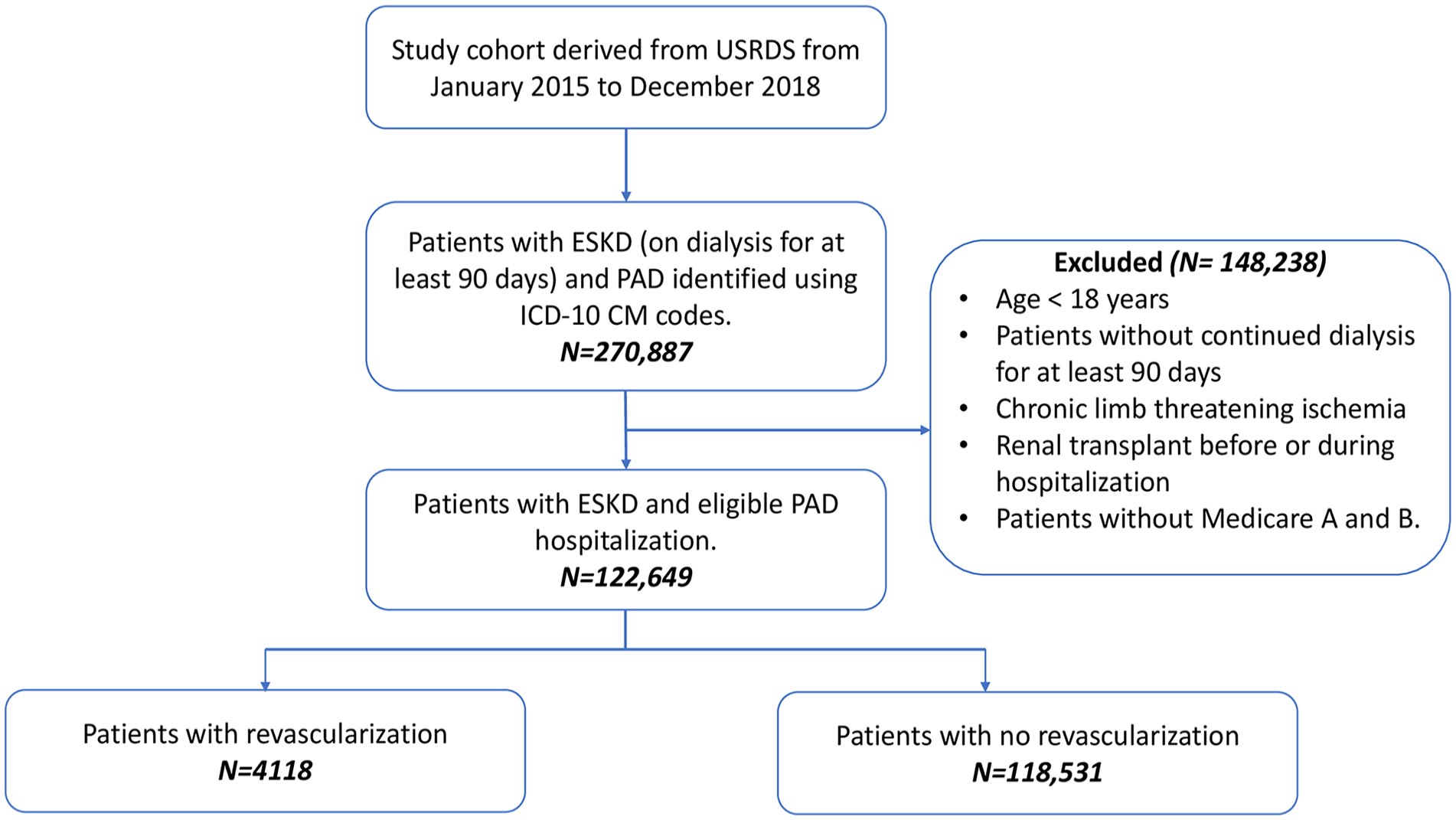
Patient selection flow chart.
Definition of frailty
We determined each individual’s HFRS by analyzing ICD-10-CM diagnostic codes associated with 108 specific medical conditions, whether used as primary or secondary diagnoses. This HFRS system has been validated in a large population in the United Kingdom and is based on diagnoses linked to high resource utilization. 7 The assigned weight for each ICD-10-CM code ranged from 0.1 to 4.4, reflecting the strength of the association with frailty, as established in the study by Gilbert et al. 7 To calculate the HFRS for each patient, we summed the weighted values as previously described. One of the medical conditions in the HFRS is chronic kidney disease (CKD), which was excluded as the study population is of ESKD. For comprehensive information on the creation and validation of the HFRS, please consult the study by Gilbert et al. 7 A list detailing the 109 medical conditions along with their corresponding ICD-10-CM diagnostic codes can be found in Table 1 (and Tables S1 and S5). We classified individuals into one of three categories based on their calculated HFRS, employing cut-off points: low risk (< 5), intermediate risk (5–10), and high risk (> 10) for frailty. In this study, individuals falling into the intermediate-risk and high-risk categories were designated as frail.
Prevalence, scoring, and ICD-10-CM codes of medical conditions used in HFRS composition for the study population in peripheral artery disease.
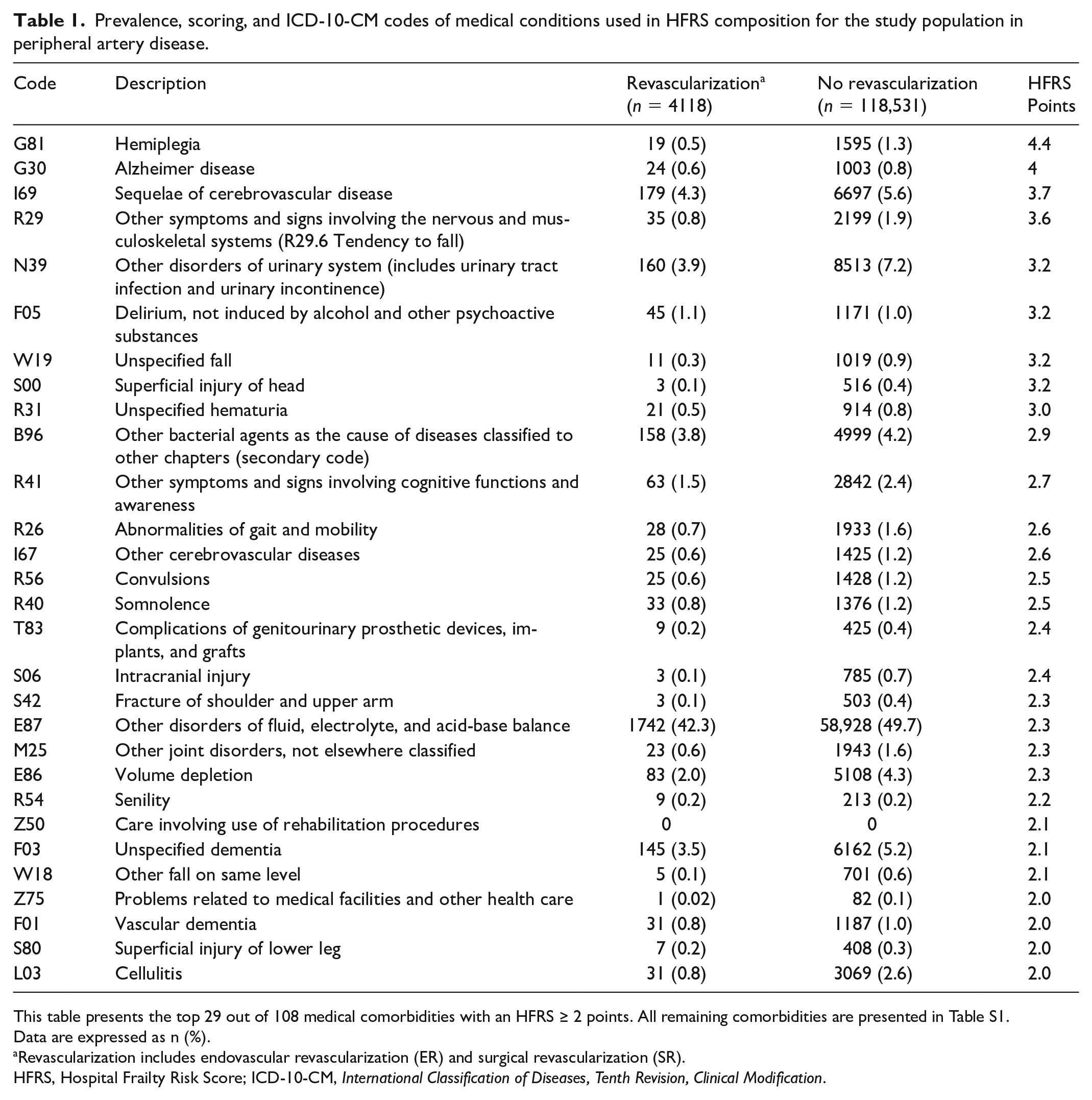
This table presents the top 29 out of 108 medical comorbidities with an HFRS ≥ 2 points. All remaining comorbidities are presented in Table S1.
Data are expressed as n (%).
Revascularization includes endovascular revascularization (ER) and surgical revascularization (SR).
HFRS, Hospital Frailty Risk Score; ICD-10-CM, International Classification of Diseases, Tenth Revision, Clinical Modification.
Study outcomes
Primary outcomes included in-hospital mortality and a composite of mortality or major amputation. Secondary outcomes encompassed in-hospital morbidity, length of stay, postdischarge mortality, composite of mortality or major amputation (involves complete or partial removal of a limb above ankle or wrist joint), major adverse limb events (MALE) and major adverse cardiovascular events (MACE) at 1 year. MALE comprised postdischarge major amputation, postdischarge mortality, and reintervention; MACE encompassed stroke, postdischarge death, or myocardial infarction. Follow up for the 1-year outcomes extended until the occurrence of death, the termination of Medicare AB coverage, kidney transplant, 1 year after discharge from the index hospitalization, or the study’s endpoint on December 31, 2018, whichever came first. Outcomes were identified using relevant ICD-10-CM codes (Table S2).
Statistical analysis
Continuous variables are reported as means ± SD or medians with IQR; categorical variables are presented as percentages. For each clinical outcome, we constructed categorical models using the low-risk group as a reference. In-hospital outcomes were analyzed with logistic regression, evaluating the impact of HFRS. One-year outcomes were assessed with cumulative incidence function (CIF) curves, considering death as a competing outcome. We employed the Fine and Gray model for competing-risk regression for 1-year outcomes. Multivariable models were adjusted for age, sex, and the Elixhauser Comorbidity Index. Additionally, we used HFRS as a continuous variable to create restricted cubic spline curves, and logistic regression was used to assess nonlinear relationships. Spline curves were adjusted for age, sex, and Elixhauser Comorbidity Index. Model performance was assessed using receiver operating characteristic (ROC) curves and the area under the curve (AUC), as shown in Tables S3 and S4. All p-values were two-sided, with statistical significance set at p < 0.05. Statistical analyses were performed using SAS 9.4 software (SAS Institute Inc., Cary, NC, USA) and STATA, version 16.1 (StataCorp LLC, College Station, TX, USA).
Results
Study population
A total of 122,649 patients with ESKD and PAD were identified from October 2015 to December 2018. Among them, 4118 patients underwent revascularization and 118,531 did not. The mean (SD) age of the revascularization cohort was 67.1 (11.6) years, involving 57.9% male patients. The mean (SD) age of the no revascularization cohort was 66.0 (12.6) years, including 54.2% male patients. The median follow-up time for postdischarge outcomes was 365 days.
Frailty and baseline characteristics
Table 1 shows the prevalence of the 29 ICD-10-CM codes that contribute the most points (at least 2 points) to the HFRS. All other relevant data can be found in Table S5. In both the revascularization and no revascularization cohorts, the most frequently diagnosed codes were ‘Other disorders of fluid, electrolyte, and acid-base balance’ (42.3% and 49.7%, respectively). Among the patients with ESKD, 86.5% in the revascularization group and 90.9% in the no revascularization group were receiving hemodialysis (HD), and the rest received peritoneal dialysis (PD), as shown in Table S6. The prevalence of diabetes mellitus in the revascularization group was 74.7% and 86.0% in the no revascularization group (Table S7). Table 1 also shows the prevalence of common comorbid baseline conditions in both groups.
Table 2 shows the baseline characteristics of the patients. In total, there were 1749 (42.5%) and 32,574 (27.5%) patients in the revascularization and no revascularization groups, respectively, categorized as low risk with a cut-off point score of < 5. Additionally, 1848 (44.9%) and 60,972 (51.4%) patients were classified as intermediate risk (5–10), and 521 (12.7%) and 24,985 (21.1%) patients fell into the high-risk category (> 10) in the revascularization and no revascularization cohorts, respectively. The median (IQR) HFRS was 5.6 (3.6–8.0) for the revascularization cohort and 6.8 (4.8–9.4) for the no revascularization cohort. Furthermore, 57.5% of patients in the revascularization cohort and 72.5% in the no revascularization cohort were classified as frail, based on an HFRS of ≥ 5.
Patient characteristics included in the study.
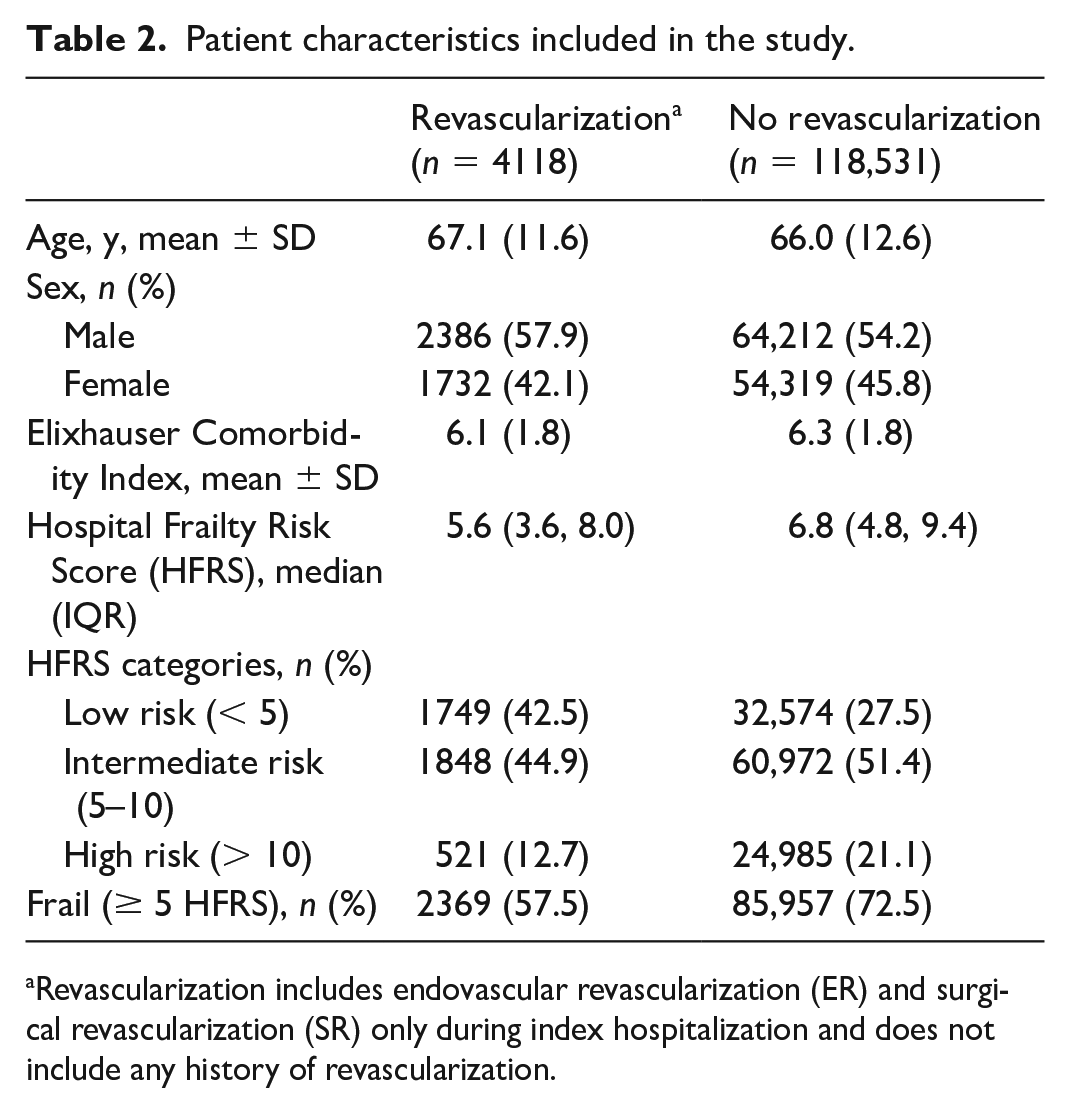
Revascularization includes endovascular revascularization (ER) and surgical revascularization (SR) only during index hospitalization and does not include any history of revascularization.
Primary outcomes
The intermediate and high-risk HFRS groups exhibited a greater risk of in-hospital mortality and complications, including mortality and the combined outcome of mortality and/or major amputation. In the revascularization cohort, these groups showed a 2.6-fold and 4.6-fold increase in the risk of in-hospital mortality and/or major amputation compared to the low-risk HFRS group. Likewise, in the no revascularization cohort, they had a 1.9-fold and 3.1-fold higher risk of these outcomes (Figure 2 and Table 3). The relevant c-statistic for in-hospital mortality and/or major amputation without considering the HFRS was 0.69 for the revascularization cohort and 0.63 for the no revascularization cohort. However, when the HFRS was included, the c-statistics improved to 0.74 and 0.67, respectively (p < 0.001), as shown in Table S3.
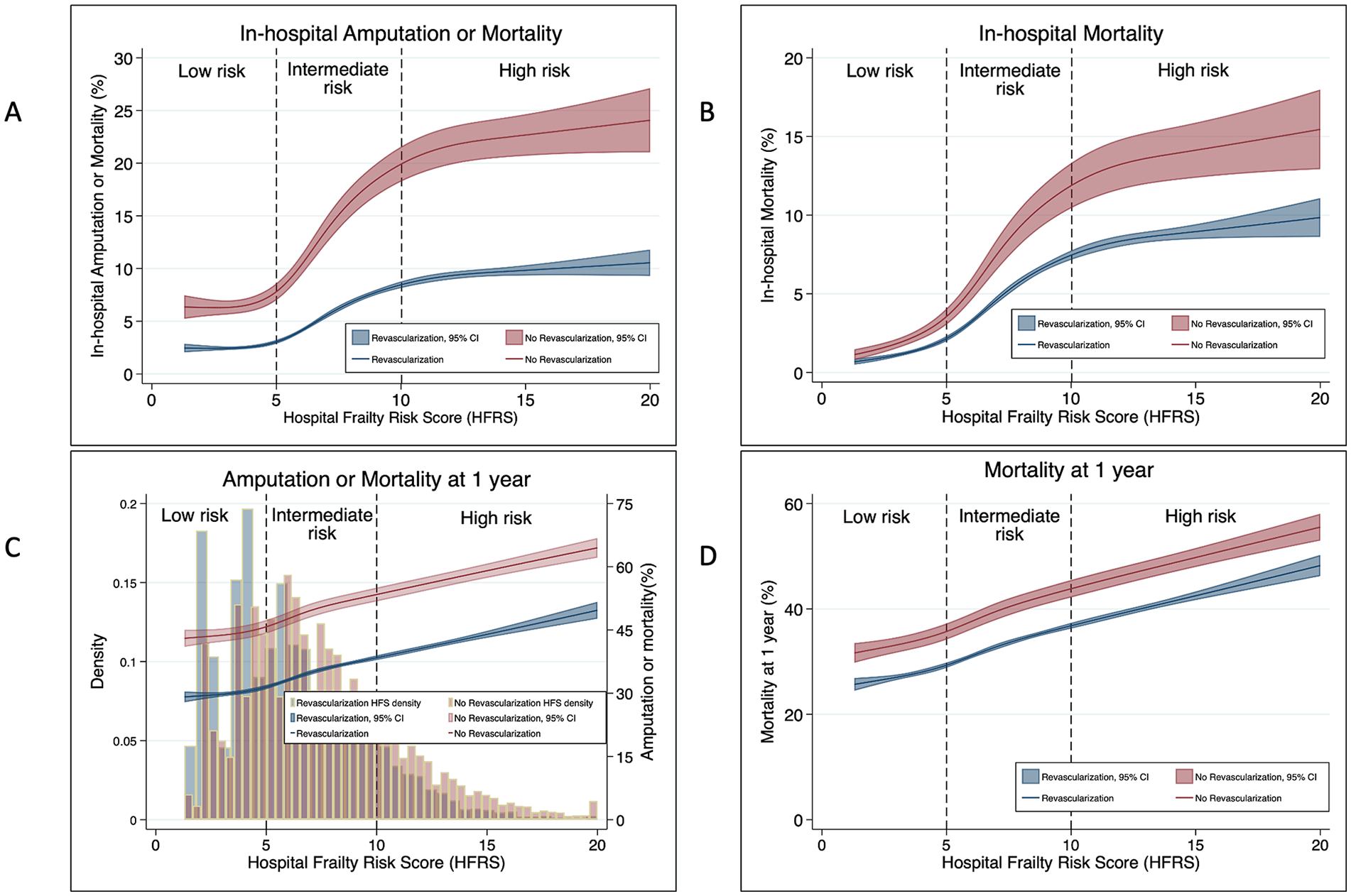
Relationship of the Hospital Frailty Risk Score (HFRS) with in-hospital amputation and/or mortality (A), in-hospital mortality (B), 1-year amputation and/or mortality (C), and 1-year mortality (D) in patients with ESKD undergoing revascularization or no revascularization for peripheral artery disease. The figure illustrates nonlinear upward trends in these outcomes relative to HFRS, depicted using restricted cubic spline curves.
In-hospital and 1-year outcomes for the study population based on frailty risk categories.
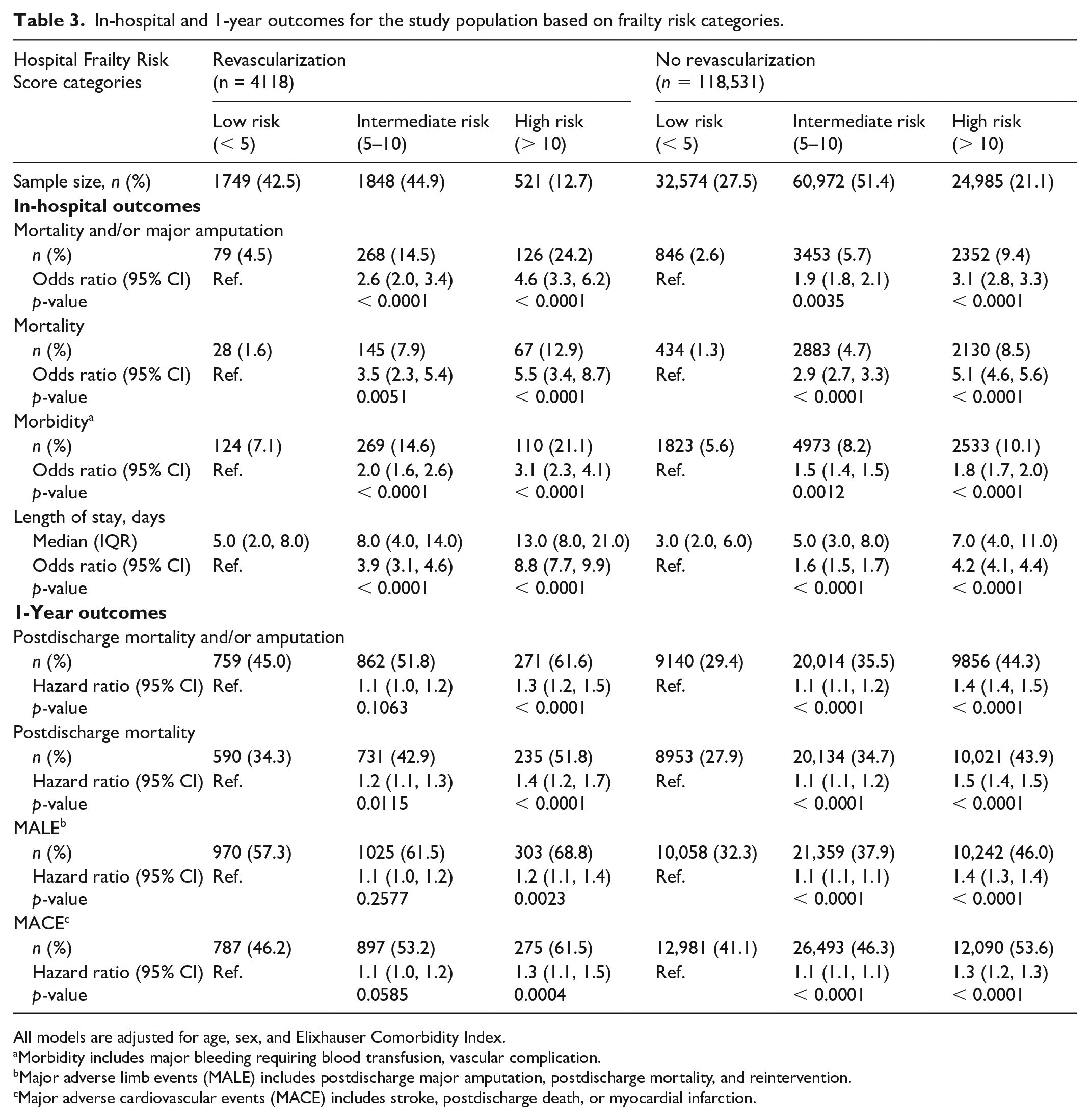
All models are adjusted for age, sex, and Elixhauser Comorbidity Index.
Morbidity includes major bleeding requiring blood transfusion, vascular complication.
Major adverse limb events (MALE) includes postdischarge major amputation, postdischarge mortality, and reintervention.
Major adverse cardiovascular events (MACE) includes stroke, postdischarge death, or myocardial infarction.
Secondary outcomes
Intermediate and high-risk HFRS groups exhibited higher in-hospital morbidity, length or stay (LOS), and 1-year postdischarge outcomes such as mortality, a composite of mortality or major amputation, MALE, and MACE compared with the low-risk HFRS group for both the revascularization and the no revascularization cohorts (Table 3).
The high-risk HFRS group was associated with a 1.3-fold higher risk of postdischarge mortality and/or major amputation at 1 year in the revascularization cohort and a 1.4-fold higher risk of postdischarge mortality and/or major amputation at 1 year in the no revascularization cohort compared with the low-risk HFRS group (Table 3 and Figure 2).
Discussion
Using a large nationwide dataset, our study validates the prognostic utility of the electronic medical record generated frailty score (the HFRS) in patients with ESKD. The main findings of the study are that compared to the low-risk HFRS group, an intermediate or high-risk HFRS group is associated with significantly higher in-hospital mortality and a composite of mortality or major amputation. Additionally, an intermediate or high-risk HFRS group was associated with worse 1-year outcomes such as mortality, a composite of mortality or major amputation, MALE, and MACE events irrespective of revascularization status compared to a low-risk HFRS group.
Frailty has been studied as an independent prognostic factor in patients with CKD/ESKD. López-Montes et al. used the Fried phenotype and short physical performance battery risk score to study outcomes of frailty in 117 elderly patients (age > 69 years) on hemodialysis. Frail patients had a 2.6 times higher mortality risk compared to nonfrail patients at 12 months. 12 Overall, patients with PAD and ESKD have significantly worse outcomes than PAD without ESKD. 13 The original frailty index (FI) included about 70 items as a part of the comprehensive geriatric assessment (CGA). 14 However, newer FI tools were formed due to the burdensome clinical application of CGA. Around 16 different frailty assessment tools have been identified in vascular surgery patients.15,16 Chen et al. performed a meta-analysis involving around 24 studies and 1,886,611 participants, studying the association of frailty scores and postoperative outcomes in patients undergoing vascular surgeries which involved all types of lower-extremity revascularization and major lower-extremity amputation. 17 This study noted that several scores, including the mFI-5, Rockwood frailty index, Edmonton frail index scale, and Fried criteria, detected the prognostic implications of these scores.18–22 Patients with higher frailty scores had significantly higher in-hospital mortality than the group with a lower frailty. These studies did not specifically focus on patients with ESKD. In our study, we used the HFRS to identify frail patients with PAD and ESKD and found higher mortality, amputation, MACE, and MALE among frail patients compared with nonfrail patients irrespective of revascularization.
Prior studies have evaluated the impact of frailty on LOS and found variable results.17,23 It is important to note that these studies were mostly among cases undergoing abdominal aortic repair and not revascularization of PAD. We found that there was a significant and incremental increase in the LOS with an increasing degree of frailty. This was seen for both groups of patients (with and without revascularization).
In a prospective, single-center study involving 643 patients (not specifically with ESKD) with CLTI treated with endovascular or surgical repair, the 2-year survival rate and amputation-free survival were independently associated with frailty. This study used a 9-level clinical frailty score (CFS) to assess frailty. 24 In the low, intermediate, and high-risk clinical frailty groups, the 2-year overall survival rates were 80.5%, 63.1%, and 49.3% (p < 0.001) and the amputation-free survival rates were 77.9%, 60.5%, and 46.2% (p < 0.001), respectively. 24 Similarly, Morisaki et al. studied the impact of frailty on 266 patients undergoing infrapopliteal revascularization. The amputation-free survival rates 1 and 2 years after revascularization were 81.8% and 72.9% for nonfrail patients and 45.8% and 34% for clinically frail patients (p < 0.001). 25 Further multivariate analysis in the same study showed that patients with ESKD on hemodialysis had a twofold higher risk of 2-year amputation compared with patients with normal kidney function. Similarly, our study also showed that patients with ESKD with high-risk HFRS undergoing revascularization had a significantly higher 1-year postdischarge mortality and/or amputation rate compared to the low-risk HFRS group.
The HFRS was initially studied and validated by previous studies in vascular surgery patients.8,26,27 Gouda et al. validated the use of HFRS in all noncardiac surgery patients including about 3500 patients undergoing aorto-femoral or femoral-popliteal bypass. The results of this study concluded that patients with an intermediate to high HFRS had two to three times higher odds of 30-day mortality and 7–10 times higher odds of in-hospital mortality compared with the low-risk HFRS group. The long-term 1-year mortality and MACE were also worse in patients with a higher HFRS. 28 Thus, using the HFRS and employing ICD diagnosis codes within the readily accessible administrative claims database presents a cost-effective alternative clinical risk scoring system for hospitals to proactively assess frailty risk, eliminating the implementation burden associated with other manual scoring systems. This study establishes a robust association between the HFRS and 1-year mortality and/or amputation, MALE, and MACE. Therefore, assessing the risk levels using the HFRS enables physicians to educate patients and families about expected outcomes during the follow-up period in patients with ESKD. Patients with a higher HFRS may also need intensive follow up.
Limitations
Our study has several limitations. The USRDS is a national database that depends on the accuracy of medical records and ICD-10 coding. Hence, an under/over coding error could be possible. This study limits our ability to define the anatomic extent of PAD lesions. This is important in defining the lesion and selecting procedure and even prognostic outcomes. We lack data on the categorization of patient type (ambulatory vs nonambulatory) and timing of revascularization (i.e., elective vs urgent), which might affect the postoperative outcomes, and we only have data available until December 2018 to analyze from the database. We also acknowledge that the management of PAD and ESKD has advanced and changed in the last few years due to the rise of new techniques and medications, which may limit our ability to extrapolate the results to the current date directly. The original HFRS excludes patients aged less than 75 years or with mental health disorders and certain components like polypharmacy or poor functional ability, which are important contributors to frailty. Despite these limitations, we believe the analysis provides data on national outcomes from a well-recognized database, making our results generalizable.
Conclusion
In patients with ESKD and PAD, the Hospital Frailty Risk Score (HFRS), derived from the electronic medical record, serves as a valuable predictor of mortality and amputation, irrespective of revascularization. HFRS can enhance preoperative risk assessment and support more informed decision-making processes.
Supplemental Material
sj-pdf-1-vmj-10.1177_1358863X251316837 – Supplemental material for Prognostic value of the Hospital Frailty Risk Score (HFRS) and outcomes in peripheral artery disease and end-stage kidney disease
Supplemental material, sj-pdf-1-vmj-10.1177_1358863X251316837 for Prognostic value of the Hospital Frailty Risk Score (HFRS) and outcomes in peripheral artery disease and end-stage kidney disease by Monil Majmundar, Chan Wan-Chi, Kunal N Patel, Vidit Majmundar, Rhythm Vasudeva, Kirk A Hance, Adam Ali, George Hajj, Axel Thors, Jinxiang Hu and Kamal Gupta in Vascular Medicine
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
