Abstract

If patients with ominous symptoms of either ischemic rest pain, tissue loss, or gangrene are left untreated, then one-quarter of these patients will lose their leg and another quarter will die within 6 months. 1 This is the natural history of chronic limb-threatening ischemia (CLTI); a condition that is more fatal than most known malignant neoplasms. 2 Accordingly, all societal guidelines give a class I recommendation for lower extremity revascularization for these patients.1,3,4 This can be challenging and requires addressing severe multilevel disease, including heavily calcified infrapopliteal and inframalleolar arteries, which tend to be small with long and totally occlusive lesions. Since Drs Dotter and Judkins’s 1964 experimental percutaneous transluminal angioplasty (PTA) of the superficial femoral artery (SFA) to save a patient’s ischemic limb, there has been enormous progress in the endovascular tools and revascularization techniques. 5 These advances led to the doubling of endovascular revascularizations in the United States between 2003 and 2011, paralleled by a persistent decline in the rates of major amputation and in-hospital death. 6
Advances in femoropopliteal disease
Anatomically, the femoropopliteal segment has benefited most from advances in devices. Most notable are drug-coated balloons (DCBs), which were designed to eliminate the use of a metallic scaffold while maintaining antirestenotic properties. Despite early skepticism, DCBs proved invaluable in reducing restenosis compared to PTA. However, the momentum surrounding DCBs was quickly tempered in 2018 by a meta-analysis that reported an association between paclitaxel devices and long-term, all-cause mortality. 7 This was followed by three separate communications from the United States Food and Drug Administration (FDA) and the temporary halt of DCB utilization in many institutions. We, among others, identified the limitations of this meta-analysis early on, 8 and subsequent analyses have refuted those findings using patient-level data. 9 Most recently, the ‘Swedish Drug-elution Trial in Peripheral Arterial Disease’, a multicenter, randomized, open-label, registry-based trial, showed no difference in all-cause mortality among those treated with paclitaxel-coated versus uncoated devices up to 4 years. 10 As a result, DCBs have regained their momentum with promising future second- and third-generation devices that use alternative antiproliferative drugs and excipients.
Other advances in the femoropopliteal segment include the concept of atherectomy (debulking) plus DCB. Several analyses, including the recently presented ‘DiRectional AthErectomy + Drug-CoAted BaLloon to Treat Long, CalcifIed FemoropopliTeal ArterY Lesions’ (REALITY; NCT02850107) trial, have shown the potential of using this combination to minimize stent utilization and improve mid- to long-term patency; however, randomized data are lacking. 11 Another landmark study, known as ‘ELUVIA™ Drug-eluting Stent Versus Zilver® PTX® Stent’ (IMPERIAL), was a head-to-head randomized clinical trial (RCT) that demonstrated superior patency with Eluvia up to 2 years. 12 In all, the segmentalized approach to femoropopliteal disease, utilizing a combination of devices to address different challenges in the longest unbranched artery in the body, is probably the most remarkable endovascular accomplishment of the last decade. For example, a combination of DCB and interwoven Supera (Abbott Vascular, Abbott Park, IL, USA) stent in the popliteal and distal SFA segments, where there is significant tortuosity and extraordinary risk for stent under-expansion and fracture, could be considered, but additional data are necessary. More than ever, endovascular operators have the opportunity to select the proper tools for the appropriate patient and arterial segment (Figure 1A).
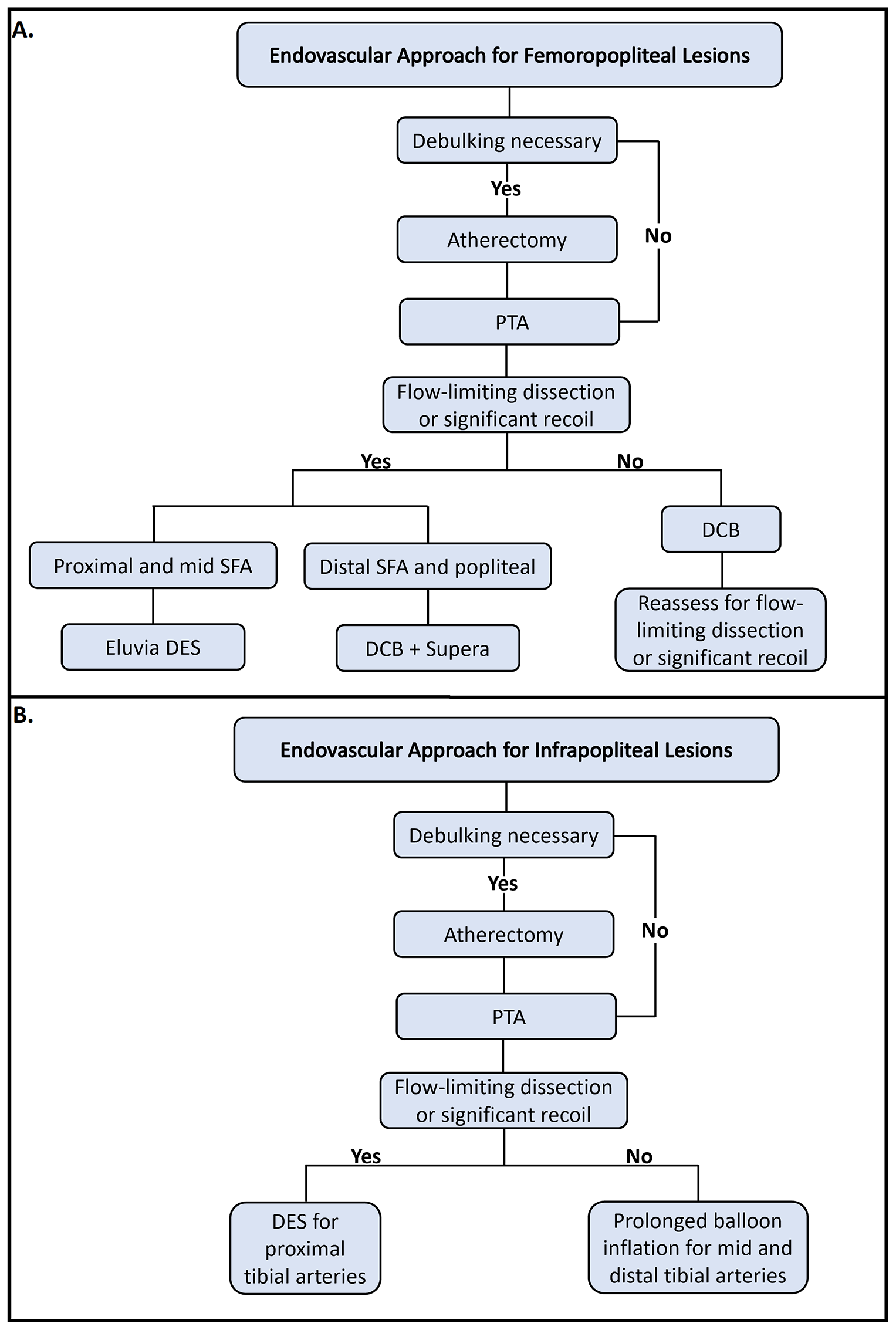
Endovascular revascularization algorithm for CLTI.
A few other advances in the femoropopliteal segment are on the horizon, but they will either need FDA approval or more significant real-world evidence for broader use. Of these, the Tack Endovascular System (Intact Vascular, Wayne, PA, USA) allows focal implantation of 6-mm long nitinol scaffold for post-angioplasty dissections. 13 Concerns about radial strength, cost-effectiveness, and re-crossing for future interventions have limited its broad applicability.
Heavy calcification may prevent full vessel preparation, result in dissections requiring stents, and lead to insufficient drug delivery. Intravascular lithotripsy (IVL; Shockwave Medical, Santa Clara, CA, USA), with its pulse therapy at high pressure (~50 ATM) and low balloon inflation, would theoretically fracture the superficial and deep calcium while avoiding soft tissue injury. The results of the randomized trial ‘Shockwave Medical Peripheral Lithoplasty System Study for PAD’ (DISRUPT PAD III) and the nonrandomized trial ‘Safety and Feasibility of the Shockwave Lithoplasty® System for the Treatment of Peripheral Vascular Stenosis’ (DISRUPT BTK) are promising.14,15 The IVL’s potential seems to be in the heavily calcified iliac and common femoral arteries; however, universal usage is likely not cost-effective.
Crossing long heavily calcified SFA chronic total occlusions (CTOs) can be difficult to accomplish. The PQ Bypass DETOUR System (PQ Bypass, Milpitas, CA, USA) is a percutaneous femoral-popliteal bypass using a stent graft deployed in an overlapping fashion from the SFA proximal to the occlusion into (anastomosis #1) the femoral vein, inside the vein and parallel to the arterial occlusion site, then out of the femoral vein into (anastomosis #2) the popliteal artery. The ‘Detour Endovascular Technique for Long OcclUsive Fem-pop Revascularization’ (DETOUR 2) investigational device exemption (IDE) single-arm trial will further evaluate this device (NCT03119233). Though promising, a few limitations exist, including the preclusion of future native artery revascularization and the risk of deep venous thrombosis. Importantly, the patency of this approach in patients with poor runoff remains unanswered.
Advances in below-the-knee (BTK) disease
In contrast, the below-the-knee (BTK) region has not experienced as much success in innovation as that of the femoropopliteal segment. Currently, PTA remains the main therapy for BTK disease, with balloon expandable DES use confined to bail-out strategies (Figure 1B); however, there have been some modifications in balloon designs such as tapered, cutting (AngioScultp; Philips, Amsterdam, Netherlands), and those with pillows and groves for atraumatic dilation (Chocolate PTA Balloon; Medtronic, Minneapolis, MN, USA). Unfortunately, DCB’s fate in the BTK territory has been discouraging. The ‘Study of IN.PACT Amphirion™ Drug Eluting Balloon vs. Standard PTA for the Treatment of Below the Knee Critical Limb Ischemia’ (INPACT-DEEP) failed to show a clinical benefit, with an observed unexplained numerical increase in major amputation in the DCB arm. 16 In addition, the ‘Lutonix DCB Versus Standard Balloon Angioplasty for Treatment of Below-The-Knee (BTK) Arteries’ (Lutonix BTK) trial failed to show superiority of DCB over PTA beyond 6 months. 17 However, the ongoing ‘Temsirolimus Adventitial Delivery to Improve Angiographic Outcomes Below the Knee’ (TANGO; NCT02908035) and ‘Lower-Limb Adventitial Infusion of DexaMethasone Via Bullfrog to Reduce Occurrence of Restenosis After Atherectomy (ATX)-Based Revascularization’ (LIMBO-ATX) trials are evaluating the efficacy and safety of different agents, temsirolimus and dexamethasone, respectively, when injected directly into the adventitia to bypass the questionable balloon-mediated drug delivery in the densely calcified BTK vessels.
In multiple RCTs, balloon expandable DESs have shown superiority to PTAs or bare-metal stents.18 –20 Self-expanding stents are not available for BKA; however, the ‘DES BTK Vascular Stent System vs PTA in Subjects With Critical Limb Ischemia’ (SAVAL; NCT03551496) trial is examining a self-expanding DES dedicated to address the long and compressible diseased BTK vessels. Having dedicated on-label DES for BTK lesions with longer length will better address this unmet need. Furthermore, the bioabsorbable vascular scaffold (BVS) with its temporary scaffold concept may have potential advantages for BTK disease with the results of the multicenter randomized ‘pivotaL Investigation of saFety and Efficacy of BRS Treatment-Below The Knee’ (LIFE-BTK; NCT04227899) trial anticipated in the near future.
Advances for treatment of ‘no option’ CLTI
A big challenge in the management of CLTI is the group of ‘no option’ patients. Depending on the case series, these range between 10% and 20% of all patients presenting with CLTI. 21 Deep vein arterialization is a novel therapy that uses the venous system to perfuse the foot by creating BTK artery-vein anastomosis using a stent graft. The ‘Early Feasibility Trial of the LimFlow Stent Graft System’ (PROMISE I; NCT03124875) data were recently presented, 22 and the large multicenter ‘Percutaneous Deep Vein Arterialization for the Treatment of Late-Stage Chronic Limb-Threatening Ischemia’ (PROMISE II, NCT03970538) trial is ongoing. Unfortunately, while there have been multiple studies to evaluate biologic therapy for CLTI and wound healing, 23 no such therapies have been approved by the FDA.
Technical advances
Procedurally, techniques such as retrograde (pedal, tibial, or femoral) access enables operators to recanalize CTOs that were thought to be noncrossable once before. Use of radial artery access has its own indications and may decrease access site complications, facilitate same-day discharge, and result in a better patient satisfaction. Treating CTOs has become even more feasible because of the introduction and adoption of many novel techniques, such as: controlled antegrade and retrograde subintimal tracking (CART), reversed CART, parallel balloon inflation, rendezvous (subintimal wiring with externalization of the wire), and PIERCE (percutaneous direct needle puncture of calcified plaque), just to name a few. Revascularizing the specific infrapopliteal vessel that directly feeds the nonhealing wound territory, known as the ‘angiosome concept’, and recanalization of the pedal arch, are additional advances. 24 There has been a renewed pledge to understand below-the-ankle arterial disease, perfusion, and revascularization. Importantly, the operators’ flexibility and commitment to limb salvage have helped fuel the field of CLTI.
Future opportunities for optimization of care delivery
Despite the advancements in devices and techniques, many legs are unnecessarily amputated and disparities in revascularization and amputation persist among patients with CLTI. We identified the shortcomings of hemodynamic testing in diagnosing CLTI and showed that over 30% of patients with three-vessel significant arterial disease or occlusion have a normal or noncompressible ABI. 25 Additionally, there is a significant delay in seeking revascularization and care for most patients with CLTI. If passed, the recent ‘Amputation Reduction and Compassion’ (ARC) Act introduced by six congressional representatives (H.R.8615) will likely improve diagnosis and access to these patients but a global effort on education is likely necessary. 26 Moreover, studies have shown that a good portion of these patients are deprived from the guidelines-directed appropriate medications. 27 Once patients have been identified, a multidisciplinary approach that will address all aspects of CLTI and limb salvage is crucial. This includes timely revascularization and expeditious wound care through prompt referrals, and rigorous follow-ups by vascular medicine experts to ensure proper primary and secondary preventions.
The ultimate frontier is to address reimbursement and promote value-based care for patients with CLTI. Many of the above revascularization treatments are not reimbursed appropriately. For example, DCB in the femoropopliteal segment is reimbursed as a conventional PTA despite the high-level evidence for its efficacy. Furthermore, the current reimbursement system does not take into consideration the complexity of the procedure. The impact of this is beyond reimbursement; it is discouraging to skillful operators who dedicate their practices to the care of patients with the most complex vascular disease and could have negative consequences on patients.
Collectively, the field of CLTI can learn from those of coronary artery disease and transcutaneous aortic valve replacement (TAVR). National registries, like the National Cardiovascular Data Registry (NCDR) and Society of Thoracic Surgeons (STS) national database, have elevated quality of care by mandating reporting and public availability of outcomes data. Measures to maximize multidisciplinary limb salvage evaluation, track outcomes, and report data are desperately needed for CLTI to ensure timely revascularization, analogous to heart teams for TAVR and left main disease and the door-to-balloon time for ST-segment elevation myocardial infarction (STEMI).
CLTI is a complex disease which occurs in patients with complex comorbidities and with high stakes for limb loss and death. Many providers from different backgrounds treat this condition; however, until we reach collective consensus and optimize multispecialty collaboration, the direction towards a future state will be painfully slow. It is no surprise that in some centers all patients with CLTI receive open bypass, whereas they would only receive endovascular revascularization in other centers. The future state should allow faster diagnosis and identification of CLTI, prompt referral, multidisciplinary evaluation, skilled CLTI operators, personalized revascularization, close follow-up, and ultimately aggressive primary and secondary preventive therapies We must, collectively, mandate advanced training for both endovascular and open procedures and advocate that complex CLTI cases only be treated at ‘Centers of Excellence’ that can offer most, if not all aspects of CLTI care. As part of this, improving access to rural and underserved communities and addressing disparities in amputation and revascularization should be part of our future state. Together we will reach the future state of CLTI care, where countless limbs and lives can be saved (Figure 2).
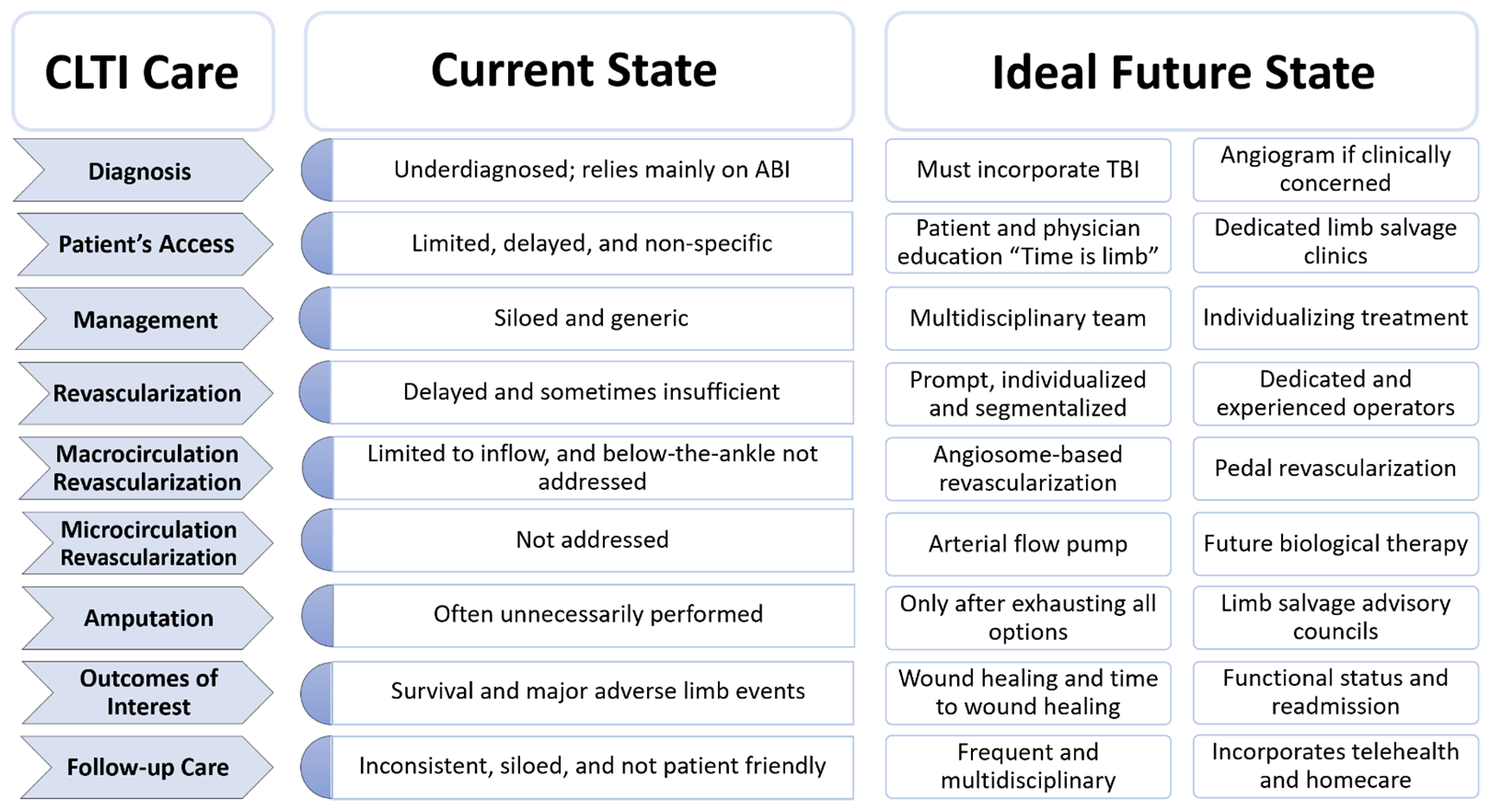
Current versus future state of CLTI care.
Footnotes
Declaration of conflicting interests
Mehdi H Shishehbor serves as an advisory board member and consultant for Abbott Vascular, Medtronics, Boston Scientific, Philips, and Terumo and as a National or Site Principal Investigator for LimFlow, Terumo R2P, and Abbott LIFE-BTK. Tarek A. Hammad has no disclosures.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
