Abstract
Background:
Preclinical studies have demonstrated that therapeutic ultrasound (TUS) increases perfusion in peripheral artery disease (PAD). This pilot study assessed the safety and effectiveness of a noninvasive TUS device in patients with advanced PAD.
Methods:
A phased array of TUS transducers was fabricated on a wearable sleeve, designed to sonicate the posterior and anterior tibial arteries (and their collaterals) at the calf level. Twelve patients with PAD (Rutherford classes 3–5) were enrolled in a single-arm study in which they underwent 30–40 daily 90-minute TUS sessions to the diseased limb. Changes in pedal flow and tissue oxygenation (StO2) were measured by laser speckle and spatial frequency domain imaging, respectively. A subset of five patients underwent evaluation by laser Doppler, transcutaneous oximetry (TcPO2), and quality of life questionnaires (Vascular Quality of Life Questionnaire [VascuQoL] and the Walking Impairment Questionnaire [WIQ]).
Results:
Eleven out of 12 enrolled patients completed the study. During 90-minute TUS sessions pedal flow improved by 180% (p < 0.001) on laser speckle imaging, and 18% (p = 0.12) on laser Doppler. Tissue oxygenation improved by 18% (p = 0.43) on TcPO2 and by 6% (p = 0.097) on StO2. After all sessions, tissue oxygenation improved by 17% (p = 0.020) on StO2, without significant changes in laser Doppler (+39%, p = 0.41) or TcPO2 (–3%, p = 0.70), which was largely in the normal range (56 ± 15 mmHg) at baseline. VascuQoL improved by 2.4 points (14%, p = 0.080) and WIQ improved by 8.2 points (11%, p = 0.053).
Conclusions:
TUS for patients with symptomatic PAD was safe and well tolerated. Most metrics of tissue perfusion and oxygenation improved, but future sham-controlled studies are needed and planned.
Keywords
Background
Peripheral artery disease (PAD) affects over 200 million people worldwide. 1 Critical limb-threatening ischemia (CLTI) is the most advanced form of PAD and is estimated to affect 11% of such patients. 2 Despite advances in revascularization procedures, disease progression and amputation is still common.3–5 Furthermore, up to 20% of patients with CLTI have no revascularization options.6–8 This unmet need has driven the search for better CLTI therapies, most of which are still invasive and associated with significant disease progression, such as transcatheter deep venous arterialization which resulted in poor patency and only 66% amputation-free survival at 6 months. 9
In the cardiovascular system, therapeutic ultrasound (TUS) and related acoustic therapies have demonstrated increased perfusion and blood vessel growth (angiogenesis at the microvascular level and collateralogenesis at the macrovascular level) in animal models of coronary artery disease (CAD) 10 and PAD.11–13 Early human studies of extracorporeal TUS and other acoustic therapies for treatment of nonrevascularizable CAD patients showed promise, but these technologies failed to proliferate, likely due to the need for careful positioning in an acoustic window free of lung or bone by a sonographer, gating for electrocardiogram and cardiac motion, and thus frequent, in-person, labor-intensive treatments. In infrapopliteal PAD, treatment of the anterior and posterior tibial arteries, and gastrocnemius and soleus muscles has few acoustic window limitations, and no inherent motion limitations. Thus, a wearable, noninvasive TUS device (Vibrato, Newport Beach, CA, USA) for the treatment of infrapopliteal PAD in patients with or at risk for CLTI was developed and studied. The device includes a series of TUS transducers mounted on a wearable ‘sleeve’ sized and designed to be worn on the calf of patients with advanced PAD, in whom it sonicates the anterior and posterior tibial arteries, and gastrocnemius and soleus muscles with a goal of promoting acute vasodilation and chronic collateralogenesis. Herein, the results of the pilot first-in-human study using TUS are reported.
Methods
Device
Individual TUS transducers were designed with size and frequency optimized to generate an acoustic field that would encompass the anterior and posterior tibial arteries when placed on the posterior calf. Depending on patient calf size, a customizable array of these transducers (n = 10–16) were mounted on a wearable sleeve sized for the adult calf (Figure 1). Water-based ultrasound gel was used to optimize transducer skin coupling. The array was connected to a custom-built generator and user interface allowing sonication at four acoustic intensity doses, all within the range of intensities that had previously demonstrated efficacy in the aforementioned preclinical studies. The transducers are operated out of phase with each other to avoid interference, and the generator also monitors each transducer’s transmit pulse’s voltage and current continuously to calculate the coupling impedance to confirm appropriate transducer–tissue coupling (and thus compliance). If coupling impedance is outside the acceptable range, usually suggestive of air in the interface, the system pauses for the user to either correct the coupling issue (with additional gel or sleeve repositioning) or inactivate that individual transducer before continuing.
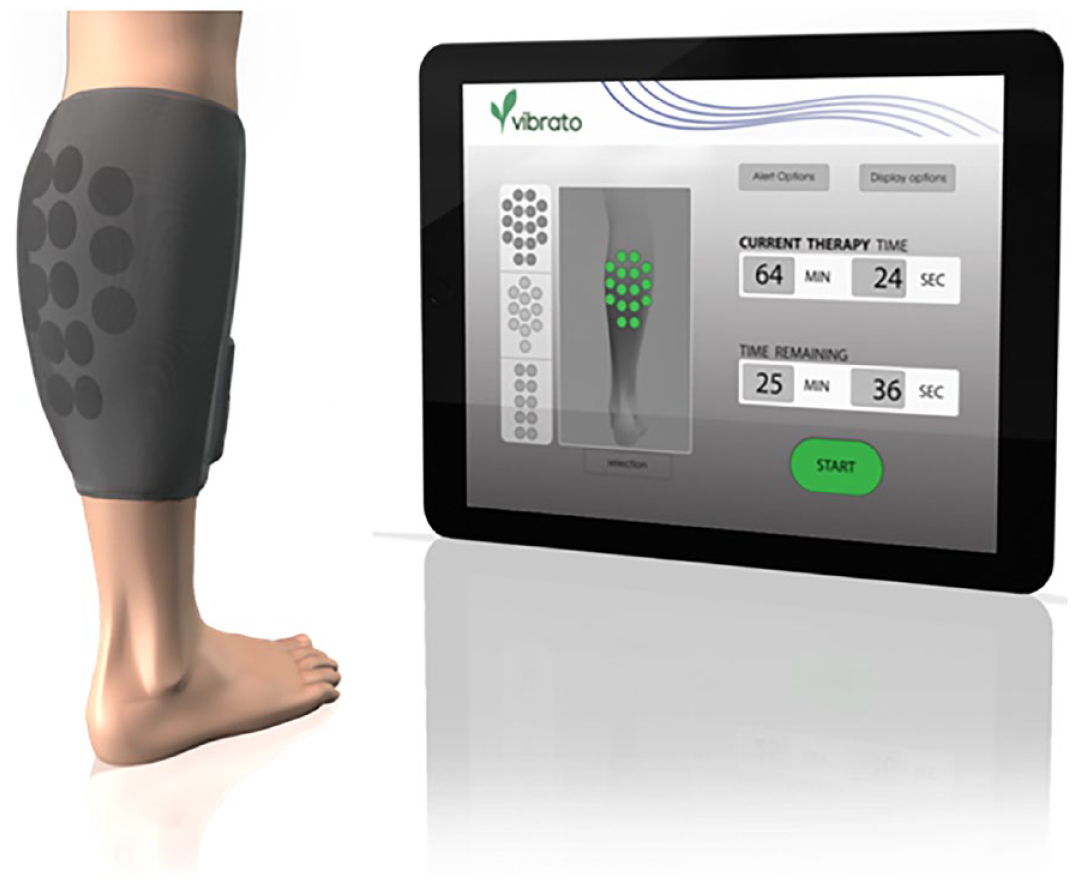
Therapeutic ultrasound device (image courtesy of Vibrato).
Patient selection and therapy
Patients with symptomatic PAD were recruited from one clinical site. The study protocol was approved by a centralized institutional review board. All participants provided written informed consent prior to enrollment in the study. The inclusion and exclusion criteria are listed in Table 1. Of note, patients in Rutherford classes 3–5 were included to assess differences in TUS efficacy at different stages of disease progression and endothelial function.
Inclusion and exclusion criteria.
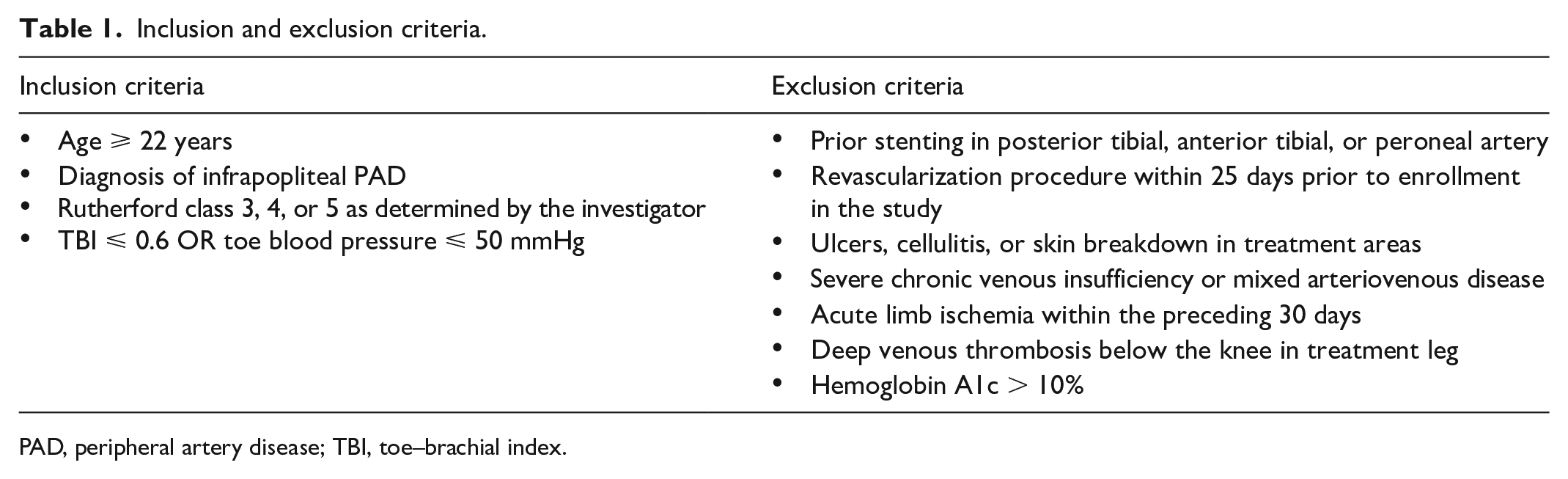
PAD, peripheral artery disease; TBI, toe–brachial index.
Patients underwent a total of 30–40 treatments (five per week) of the index limb lasting 90 minutes each. The first one to three treatments were performed in-clinic to allow a dose titration study of all three acoustic doses to confirm tolerability without significant paresthesia or cramping, and the highest tolerated dose was subsequently used for the remainder of the study. Additional treatments performed at the office included the 15th and 30th or 40th sessions (end of study) to allow for serial assessment of immediate posttherapy study endpoints. Patients were seated with the leg elevated and horizontal for all therapy sessions and endpoint assessments. The remaining sessions were administered at home. A trained study staff was present for all TUS home treatment sessions to ensure proper operation and protocol compliance.
No specific PAD medical therapy was prespecified, and utilization of specific medications at baseline is shown in Table 2. Changes in medical therapy were allowed at physician discretion.
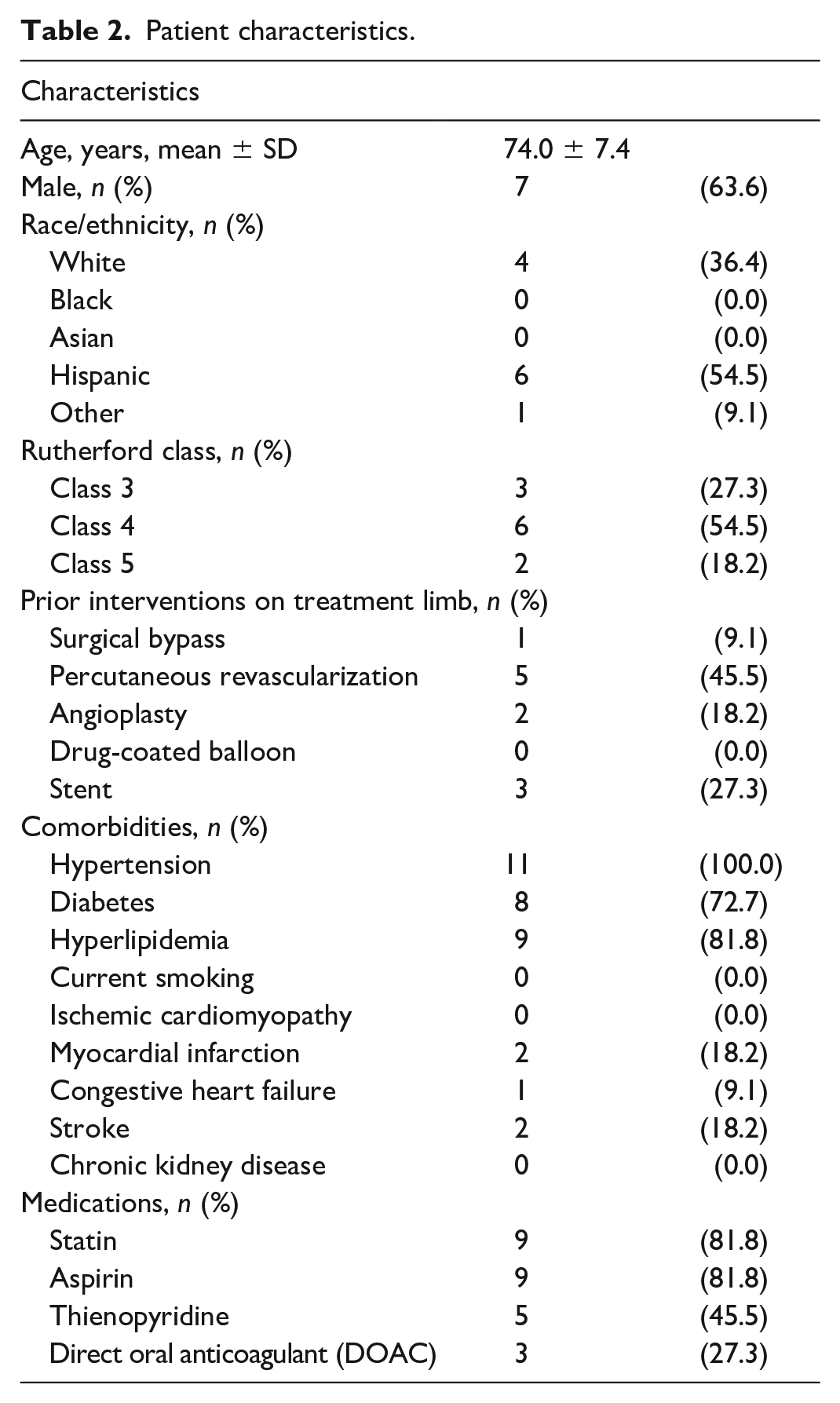
Patient characteristics.
Endpoints
To assess acute perfusion and flow increases during therapy, patients were monitored at the second digit on the treatment limb with a laser speckle tracking probe (FlowMet; Medtronic, Minneapolis, MN, USA) throughout the 90-minute therapy and for 15 minutes post-TUS. FlowMet is designed to assess real-time changes in flow. 14 At every in-office session, FlowMet values were obtained for 5 minutes prior to TUS as the baseline measurement. Subsequent values were normalized to that baseline mean.
The remainder of the endpoints were assessed pre- and posttreatment of the first and last (in-office) TUS sessions. Tissue oxygenation (StO2) was assessed using spatial frequency domain imaging of the plantar surface of the foot using the Clarifi system (Modulim, Irvine, CA, USA). Ankle and toe pressures were measured in standard fashion to record ankle and toe brachial indexes (ABI/TBI).
For the last five patients, additional exploratory endpoints were introduced. Transcutaneous oximetry (TcPO2) was assessed at the level of the dorsal foot (Perimed, Järfälla, Sweden). Local microvascular blood perfusion of the plantar foot was assessed using laser Doppler (Perimed). Finally, we assessed patient-reported outcome measures (PROM) with the Vascular Quality of Life Questionnaire-6 (VascuQoL-6; scores range from 6 to 24, with higher scores suggesting improved health status) and the Walking Impairment Questionnaire (WIQ; scores range from 21 to 105, with higher scores suggesting less impairment).
Patients subsequently returned at 1, 3, and 6 months for repeat endpoint measurements (StO2 and ABI/TBI) to evaluate the durability of TUS effects. PROMs were also assessed at 6 months. Though this was not a study of claudicants, the TUS device sonicates the gastrocnemius and soleus muscles in parallel with the anterior and posterior tibial arteries, so WIQ tracked changes in claudication in an exploratory manner.
Safety endpoints studied included severe pain at time of TUS, ecchymoses, new skin breakdown or ulceration beneath the TUS transducers and device, acute limb ischemia, and deep venous thrombosis.
Statistical analysis
Repeated measures models were used to assess the overall relative difference across study visits between pretreatment and posttreatment measurements of perfusion by laser Doppler and TcPO2 and StO2. FlowMet measurements (which are taken once per second) were analyzed using measurements at 4-second intervals with a covariate for measurement prior to treatment and during treatment. The overall difference between pretreatment and during treatment was estimated using a repeated measures model.
The Shapiro–Wilk test was used to assess normality of data. Data are reported as mean ± SD when normally distributed, and median (IQR or range) when nonnormally distributed. Two-tailed, paired t-tests were used to compare normally distributed data, and Wilcoxon matched-pairs signed-rank test used to compare nonnormally distributed data. One-way ANOVA was used for chronic trends, and for comparing across multiple Rutherford classes.
Results
Patient characteristics
Eleven of the 12 patients enrolled completed the 90-minute TUS sessions (seven men, four women). Their mean age was 74.0 years (range 59–83 years) at the time of enrollment. One patient withdrew due to exacerbation of unrelated medical conditions that prevented him from completing TUS sessions and was not included in the final analysis. There were three, six, and two patients in Rutherford classes 3, 4, and 5, respectively. Five patients had undergone prior revascularization at least 25 days prior to study enrollment. Other baseline characteristics are listed in Table 2.
Immediate posttreatment assessment
Over the course of each in-office 90-minute TUS session (n = 44), there was a mean 180% increase in pedal blood flow (95% CI 113–247, p < 0.001) as measured by FlowMet compared with the 5-minute pre-TUS baseline. Flow remained stably elevated for the 15-minute post-TUS period (Figure 2). Each patient demonstrated a statistically significant increase in perfusion during TUS therapy. There was a trend toward patients in earlier stages of PAD/CLTI having a greater acute vasodilatory response: Rutherford class 3 with 328% improvement (p = 0.0003 compared with pre-TUS baseline), class 4 with 157% improvement (p < 0.0001), and class 5 with 69% improvement (p = 0.0012; ANOVA comparison across Rutherford classes p = 0.054).
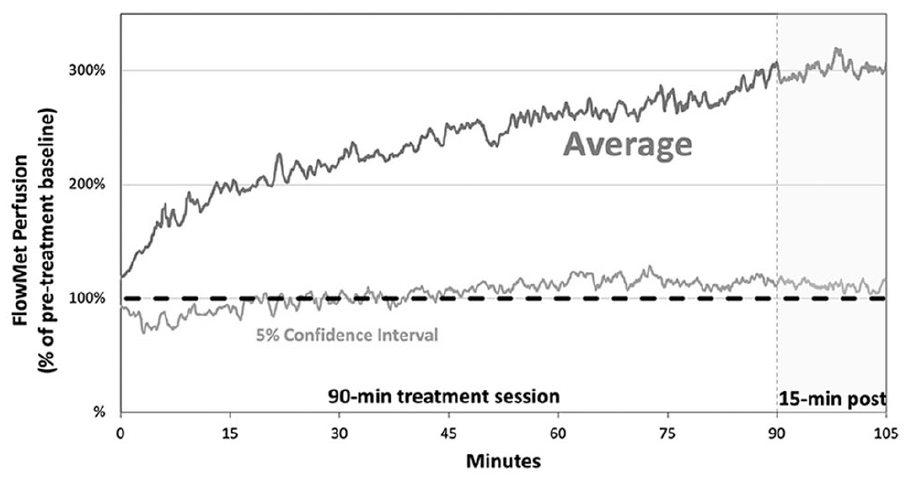
Normalized real-time acute perfusion measurements by FlowMet during therapeutic ultrasound.
When assessed acutely during in-office TUS sessions, there was a StO2 mean increase of 6% (95% CI −1 to 12, p = 0.097) after 90 minutes of TUS, as measured by Clarifi spatial frequency domain imaging (Figure 3).
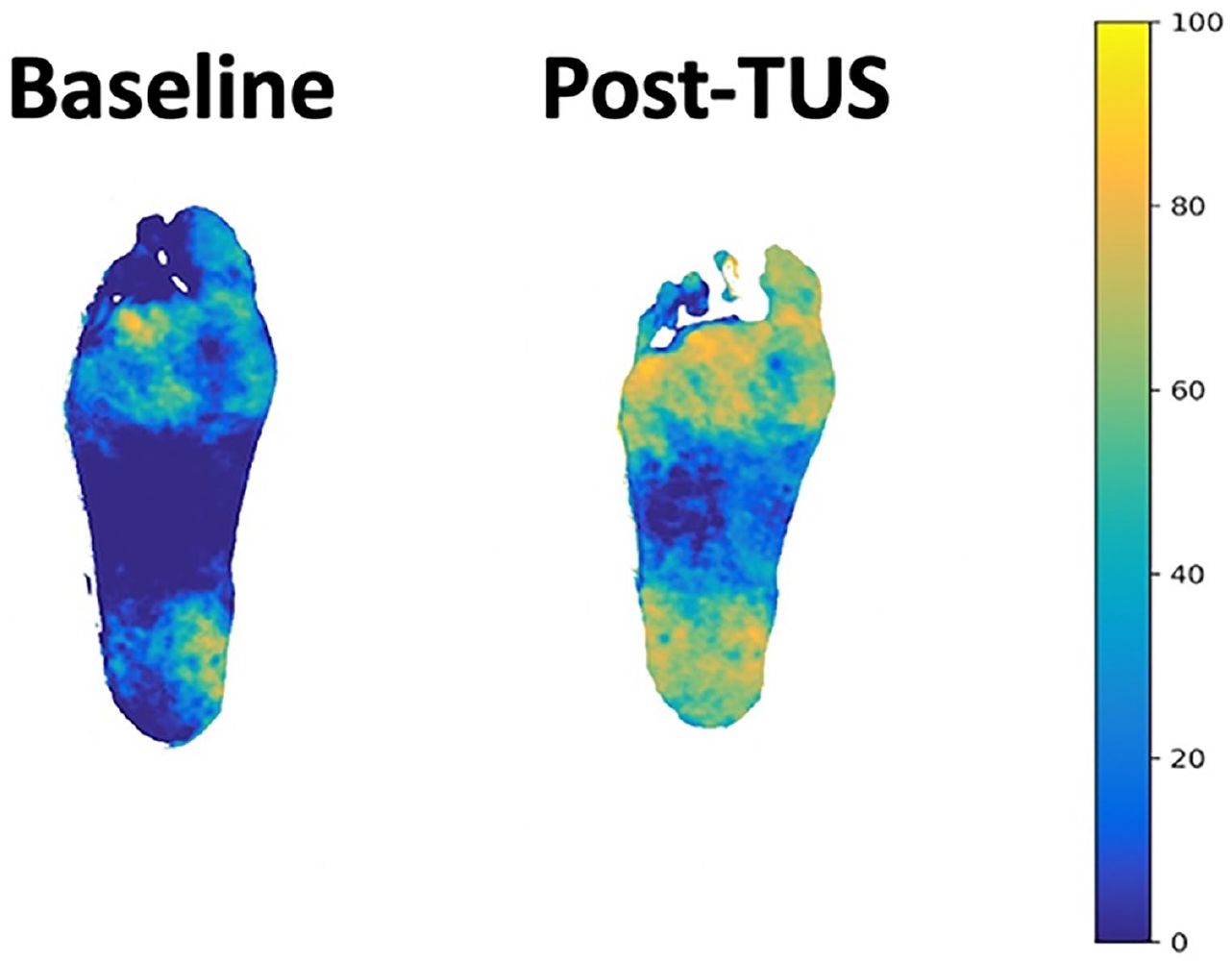
Improvement in tissue oxygenation by Clarifi spatial frequency domain imaging (StO2). TUS, therapeutic ultrasound.
Both ABI and TBI increased nonsignificantly over the course of 90 minutes of TUS from 1.05 ± 0.36 to 1.07 ± 0.37 (p = 0.81) and from 0.37 ± 0.15 to 0.41 ± 0.16 (p = 0.34), respectively. The ABI measurements in the normal range likely reflect the prevalence of calcified, noncompressible vessels in our study population, for whom TBI is a better reflection of their Rutherford class 3–5 PAD severity.
Laser Doppler measurements demonstrated an 18% increase in perfusion, from 16.6 ± 4.4 flux units at baseline to 19.6 ± 7.20 (p = 0.12) after 90 minutes of TUS. TcPO2 increased from 49.4 ± 18.8 to 58.3 ± 15.4 (p = 0.43) over the course of 90 minutes of TUS.
End-of-therapy and follow-up perfusion and tissue oxygenation
After completion of all TUS sessions in the study, Clarifi spatial frequency domain imaging demonstrated an increase in StO2 from 62.8 ± 17.1 (pre-TUS baseline) to 73.6 ± 16.6 (p = 0.020). Tissue oxygenation remained elevated in follow up 1 month after the end of TUS (73.3 ± 16.6, p = 0.18), but not at 3 months (64.6 ± 19.1, p = 0.61) or at 6 months (68.2 ± 18.5, p = 0.21), with a significant temporal trend (Figure 4). Unlike the FlowMet findings, there was no between-Rutherford class difference in chronic tissue oxygenation increase: increases above baseline in Rutherford classes 3, 4, and 5 were 13%, 29%, and 22%, respectively (ANOVA comparison across Rutherford classes p = 0.85).
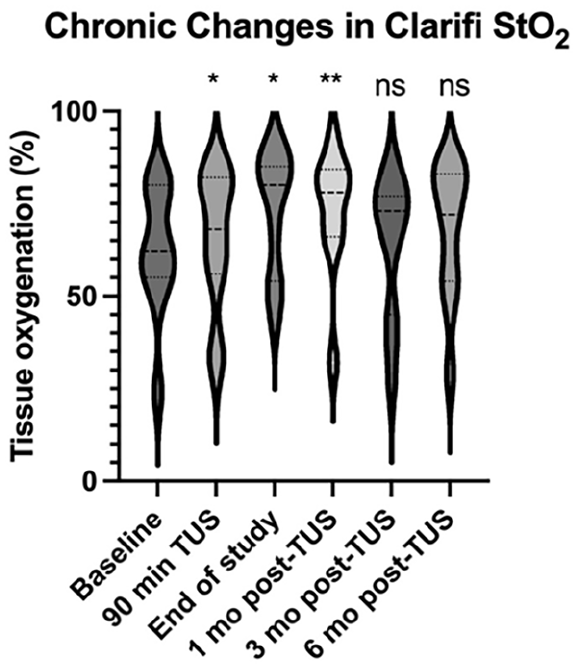
Chronic changes in tissue oxygenation by Clarifi spatial frequency domain imaging (StO2).
Laser Doppler measurements increased nonsignificantly from 14.6 ± 9.6 to 20.3 ± 8.4 flux units (p = 0.41) over the course of all TUS treatments in the study (Table 3). TcPO2 remained stable without improvement over the study period (56.2 ± 15.1% to 54.3 ± 18.7%, p = 0.70). Similarly, there were no significant changes in TBI with stable mean value (0.41 ± 0.12 to 0.39 ± 0.12, p = 0.55). ABI increased nonsignificantly by 11% (0.95 ± 0.27 to 1.05 ± 0.24, p = 0.43) at the end of TUS therapy and remained stable 1 month later (1.01 ± 0.29, p = 0.69). ABI decreased nonsignificantly at 3 months (0.87 ± 0.19, p = 0.50) and at 6 months (0.84 ± 0.21, p = 0.40) following the completion of TUS therapy.
Therapeutic ultrasound results.
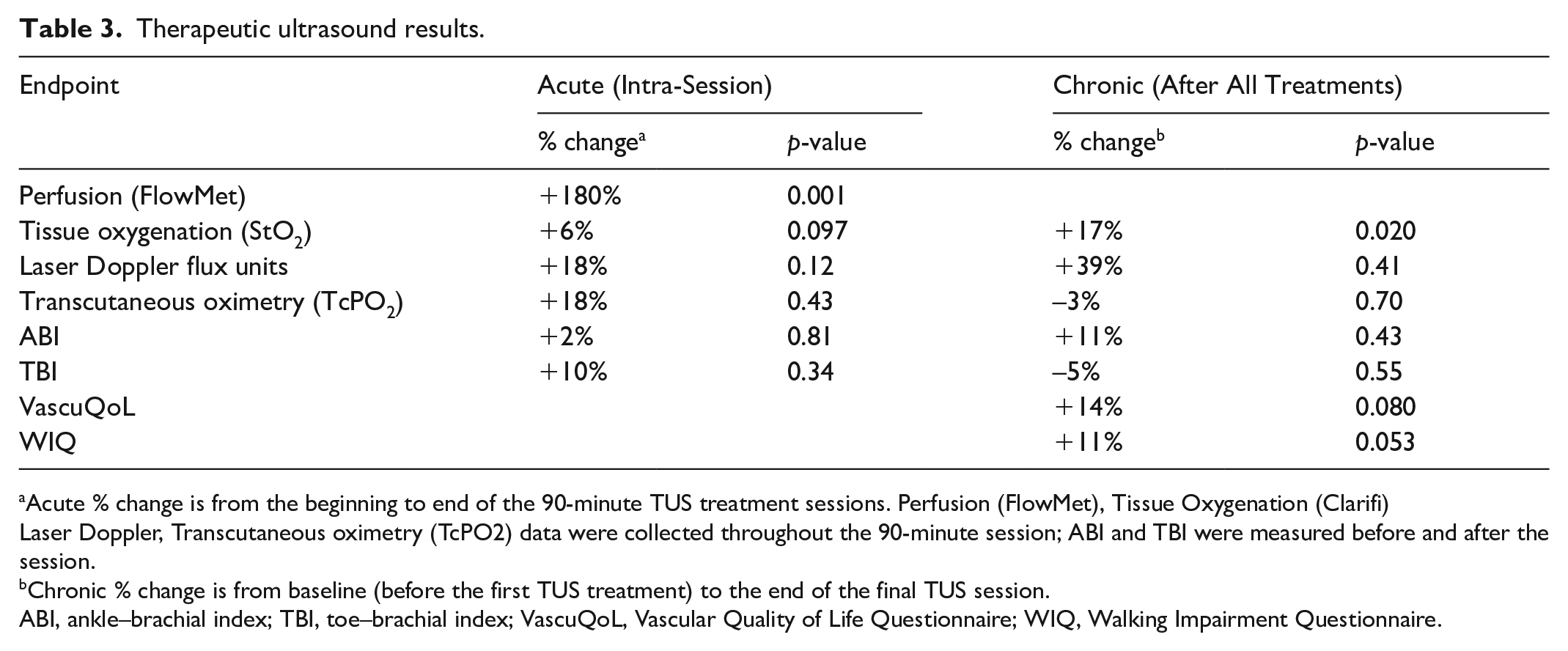
aAcute % change is from the beginning to end of the 90-minute TUS treatment sessions. Perfusion (FlowMet), Tissue Oxygenation (Clarifi)Laser Doppler, Transcutaneous oximetry (TcPO2) data were collected throughout the 90-minute session; ABI and TBI were measured before and after the session. bChronic % change is from baseline (before the first TUS treatment) to the end of the final TUS session.
ABI, ankle–brachial index; TBI, toe–brachial index; VascuQoL, Vascular Quality of Life Questionnaire; WIQ, Walking Impairment Questionnaire.
Tolerability and adverse events
All patients were able to tolerate the therapy, with eight patients completing the highest acoustic dose, two the medium dose, and one the low dose. Compliance was excellent, with 11 of 12 enrolled patients completing all of the recommended treatments.
There were no serious adverse events. One patient reported transient mild calf discomfort during the dose titration period that resolved with dose reduction without sequelae.
Patient-reported quality of life outcomes and wounds
VascuQoL-6 improved from a baseline of 17.2 ± 2.6 to 19.6 ± 4.3 (p = 0.080) at the end of all TUS treatments (6 weeks to 2 months), and WIQ improved from 77.2 ± 13.8 to 85.4 ± 12.5 (p = 0.053). VascuQoL-6 remained higher than baseline (18.8 ± 2.5, p = 0.21) and while WIQ returned to baseline (76.8 ± 7.7, p = 0.93) 6 months after the end of treatment. Two of the patients in the study had toe ulcers (both on the dorsal surface of the digits: one 4 mm and the other 5 mm in maximal dimension) in the treatment limb, all of which healed over the course of the TUS (one from the Wound, Ischemia, and foot Infection [WIfI] classification 3 to 1 and the other from 2 to 1 over the course of 30 TUS treatments). Both patients had histories of hypertension, diabetes (well-controlled with oral agents), and were Rutherford class 5 by definition. One patient (TBI 0.38) had no prior revascularization procedures, and the other (TBI 0.26) had undergone right popliteal artery balloon angioplasty within the prior 6 months.
Discussion
This first-in-human, early feasibility study of TUS in patients with or at risk of CLTI demonstrated acute increases in digital blood flow and pedal perfusion. Tissue oxygenation was also significantly increased as measured by spatial frequency domain imaging. These findings are consistent with prior studies of TUS.12–15 The treatments were well tolerated by enrollees without any serious adverse events, confirming the safety of the therapy.
TUS has historically been used at high acoustic intensities and in continuous wave mode for thermal ablation. However, pulsed ultrasound at low duty cycles at intensities in between those of diagnostic imaging and thermal ablation can generate diverse nonthermal bioeffects. 15 These bioeffects are likely mediated through TUS activation of the Pannexin-1 receptor (a mechanoreceptor known to respond to shear stress) as well as TUS-mediated permeabilization and release of adenosine triphosphate (ATP) from within endothelial and red blood cells into the extracellular vascular space. 16 Both Pannexin-1 and ATP are vasodilators known to act via nitric oxide pathways. The observed acute digital blood flow and tissue oxygenation increases were likely due to the vasodilatory effect of TUS as mentioned above.
After completion of all sessions of the TUS therapy, the improvements in laser Doppler and TcPO2 measurements were not as robust as tissue oxygenation increase by spatial frequency domain. This may be due to the small number of subjects who had laser Doppler and TcPO2 (n = 5). The modest changes in TcPO2 may also be due to the subjects’ surprisingly high values at baseline (56.2 ± 15.1%). Prior studies focusing on TcPO2 have shown relative stability of values in this higher range even despite successful revascularization. 17
TBI did not change significantly in this study despite improvements in blood flow. One explanation for lack of change in TBI may be the diffuse and down-stream TUS-mediated vasodilation. Preclinical TUS studies have shown that the adenosine, pannexin, and nitric oxide-mediated effects cause vasodilation distally in the treatment limb beyond the acoustic field, as well as in the contralateral limb. 18 Thus, TUS applied at the level of the calf may dilate the pedal blood vessels distally, leading to lower toe pressures despite the increased perfusion.
The acute vasodilatory effects of TUS were most prominent in Rutherford class 3 patients. This may be due to a relatively better-preserved endothelial function and reactivity compared with the more advanced disease in Rutherford 4 and 5 patients. Conversely, the chronic tissue oxygenation increases were consistent across all Rutherford classes studied. The reason for this observation is unclear but a potential explanation may be the lower baseline values for tissue oxygenation in more advanced disease states and having more oxygen-avid distal tissue and thus greater room for improvement.
This was a pilot study designed to evaluate the feasibility and tolerability of the TUS in this patient population. As such, it was conducted in a small number of patients without a control group (patients served as their own internal controls). Hence, results must be interpreted with caution. Further statistically powered investigations are needed as this was a small study where most improvements observed were statistically nonsignificant. Although the focus was not wound healing, it is encouraging that both patients with active ischemic ulcers experienced complete healing over the course of the study and there was a measurable improvement in quality of life of the participants. Though the 90-minute therapy is time-consuming, duration or frequency of therapy was not cited as a reason for intolerance or study withdrawal, and many patients with advanced PAD attend hyperbaric oxygen therapy (not in their own home) for 90-minute sessions several times a week.
A major limitation in assessing efficacy of any new technology in patients with CLTI is the lack of validated and universally accepted instruments to assess outcome. Advances in patients’ functional status or clinical endpoints such as amputation, amputation-free survival, and/or wound healing are multifactorial and therefore insensitive to changes in any one variable such as improved foot perfusion or blood flow to the toes. Furthermore, measurement of such hemodynamic parameters as perfusion or blood flow and their correlation to physiologic changes have been problematic in patients with CLTI. ABI/TBI, TcPO2, and laser Doppler all have well-established limitations and neither laser speckle nor spatial frequency domain imaging are validated in large studies. This created a challenge in selecting an instrument or technique that could detect directional changes in limb perfusion or blood flow in this study. Hence, multiple techniques and devices were used to assess outcome, including quality of life instruments.
This was the first assessment of TUS in patients with advanced PAD showing that the approach is feasible and well tolerated. Observations of improved acute blood flow and increased tissue oxygenation lasting for several weeks are encouraging, although efficacy in our broad array of endpoints was mixed, and quantitative improvements in our PROMs did not reach statistical significance in this small study. Our outcomes support TUS’s acute vasodilatory effects to a greater degree than its chronic angiogenesis/collateralogenesis effects, and the more robust acute perfusion increases in Rutherford class 3 patients suggest patients with better endothelial function may be more acutely responsive. To better determine the role of this technology in the spectrum of treatments for patients with CLTI, we plan larger studies that include a sham control. Differences in its role as stand-alone therapy versus adjunctive (to surgical or percutaneous revascularization) may also be studied. Finally, though the device has a form factor and automated compliance and transducer–skin coupling detection features designed for patient self-administration, TUS treatments in this study were supervised by study staff, and the efficacy of unsupervised patient self-administration still needs to be studied.
Footnotes
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Drs. Nazer and Razavi are consultants for and have equity interests in Vibrato Medical. The remaining authors have no conflicts of interest to declare.
Funding
Financial support for this study was provided by Vibrato Medical, Inc.
