Abstract
Background:
In 2019, the US Food and Drug Administration issued a warning that symptomatic relief from claudication using paclitaxel-coated devices might be associated with an increase in mortality over 5 years. We designed a discrete-choice experiment (DCE) to quantify tradeoffs that patients would accept between a decreased risk of clinically driven target-vessel revascularization (CDTVR) and increased mortality risk.
Methods:
Patients with claudication symptoms were recruited from seven medical centers to complete a web-based survey including eight DCE questions that presented pairs of hypothetical device profiles defined by varying risks of CDTVR and overall mortality at 2 and 5 years. Random-parameters logit models were used to estimate relative preference weights, from which the maximum-acceptable increase in 5-year mortality risk was derived.
Results:
A total of 272 patients completed the survey. On average, patients would accept a device offering reductions in CDTVR risks from 30% to 10% at 2 years and from 40% to 30% at 5 years if the 5-year mortality risk was less than 12.6% (95% CI: 11.8–13.4%), representing a cut-point of 4.6 percentage points above a baseline risk of 8%. However, approximately 40% chose the device alternative with the lower 5-year mortality risk in seven (20.6%) or eight (18.0%) of the eight DCE questions regardless of the benefit offered.
Conclusions:
Most patients in the study would accept some incremental increase in 5-year mortality risk to reduce the 2-year and 5-year risks of CDTVR by 20 and 10 percentage points, respectively. However, significant patient-level variability in risk tolerance underscores the need for systematic approaches to support benefit-risk decision making.
Keywords
Background
Paclitaxel-coated devices increase arterial patency and reduce restenosis rates resulting in more sustained relief of symptoms compared with uncoated devices in patients with peripheral artery disease (PAD) and have market approval by the US Food and Drug Administration (FDA) and the European Medicines Agency. Clinical trials investigating paclitaxel-coated devices have demonstrated significant reductions in clinically driven target vessel revascularization (CDTVR, generally defined as reintervention at or near the target lesion with > 50% stenosis and recurrent clinical symptoms) in the first 2 years through 5 years postprocedure compared to uncoated devices.1–3 In 2018 and 2020, a trial-level meta-analysis by Katsanos et al. 4 and a patient-level meta-analysis by Rocha-Singh et al. 5 reported that paclitaxel-coated devices increase the risk of late mortality compared to standard devices, reporting 5-year relative risk ratios of 1.93 (95% CI: 1.27–2.93) and 1.38 (95% CI: 1.06–1.80), respectively. A subsequent meta-analysis by the FDA reported a 5-year mortality hazard ratio of 1.57 (95% CI: 1.16–2.13). 6 Summary estimates representing increases in the absolute risk of 5-year mortality ranged from 4.6% to 7.2% across the three independent studies.4–6
Beginning in 2019, the FDA issued several communications to healthcare providers recommending risk-benefit discussions with patients regarding paclitaxel-coated devices, including consideration of patient preferences, acknowledging that the balance of benefits and risks may vary across patients. 7 To better understand how patients weighed the potential benefits and risks associated with paclitaxel devices, a quantitative patient-preference study was initiated in October 2020 through a collaboration convened by the MDEpiNet PASSION program, 8 including clinical researchers, patient representatives, regulators, and experts in stated-preference research. The project was designed to align with guidance from the FDA’s Center for Devices and Radiological Health (CDRH), which stresses the importance of incorporating patients’ perspectives to inform judgments about the acceptability of device-associated risks. 9
During the execution of the project (through September 2022), additional studies using randomized and nonrandomized designs did not observe an increased mortality risk with paclitaxel-coated devices, contributing uncertainty to the associations between paclitaxel-coated devices and late mortality risk.10–18 Based on a review of these studies and internal analyses using data provided by device manufacturers, the FDA issued an update in July 2023 informing providers that the data do ‘not support an excess mortality risk for paclitaxel-coated devices.’ This conclusion was supported by a meta-analysis that pooled patient-level data across 10 randomized clinical trials. 19
Despite changes in the clinical evidence base, findings from the patient preference study can provide useful insights on how patients viewed the previously presumed increased mortality risks. The study aimed to quantify the relative importance of improvements in health outcomes and trial endpoints that are frequently used to compare alternative treatments by quantifying benefit-risk tradeoffs that would be acceptable to patients with claudication, including whether and to what extent they would accept higher, longer-term mortality risk for more durable revascularization outcomes.
Methods
Recruitment
The target sample for the survey designed for this study included patients with claudication who: (1) were currently symptomatic; (2) were previously symptomatic with a history of peripheral revascularization; and (3) could read English. Screening questions in the survey limited participation to individuals who reported that they currently were experiencing or ever had experienced pain in the lower leg or buttock when walking but did not report symptoms or signs of limb-threatening ischemia, including leg pain at rest or active tissue loss. Patients also reported prior endovascular or surgical procedures.
Patients were recruited from seven US medical institutions in person, by phone, email, and mailed letters. A $45 (USD) electronic gift card was offered as compensation for time completing the survey. The study protocol was approved by institutional review boards at all participating centers, and all participants provided informed consent.
Discrete-choice experiments
Choice experiments are designed to quantify the relative importance of different features that, when considered together, represent a product, treatment, health policy or other relevant construct. Discrete-choice experiments (DCEs) are increasingly used to quantify acceptable benefit-risk tradeoffs associated with medical interventions to aid in regulatory and clinical decision making.20,21 In a DCE designed for a medical decision context, participants answer a series of questions in which they choose between two or more profiles defined by treatment-related attributes (e.g., benefits, risks, outcomes, process factors). Attribute levels can represent varying probabilities, degrees of severity, and/or type and are experimentally combined to construct treatment profiles. Each set of profiles requires participants to forgo one or more desirable features when selecting the preferred profile as if the profiles presented are the only options available. Statistical analysis of choices across experimentally controlled profiles reveals the implicit relative importance of each attribute level.
Attributes
The attributes selected for the DCE were limited to risks of CDTVR and all-cause mortality at 2 and 5 years to maintain focus on the primary study objective. Attribute levels were chosen to correspond with incidence rates and between-group differences in rates reported across randomized clinical trials investigating paclitaxel-coated and standard devices and meta-analyses of randomized trials (Table S1).1–6,22–26
Survey-instrument design
Portraying time-varying risks of CDTVR and mortality in the survey instrument presented a risk-communication challenge. Informed by a seven-arm randomized risk-communication study of 2242 participants, a vertical layout was chosen in which device alternatives are shown in columns. 27 In the current study, to test whether inclusion of icon arrays to portray risk levels affects benefit-risk preferences, participants were randomized to a version of the survey using icon arrays (Figure 1) or a version without icon arrays (Figure S1). We iteratively pretested and revised both versions in one-on-one virtual interviews with 15 patients with claudication. During the interviews, the study team evaluated the clarity and completeness of the survey material as well as patients’ ability to interpret the tradeoff questions accurately. The surveys included educational content about the attributes and five comprehension questions to evaluate participants’ ability to identify, compare, and calculate differences between device profiles. 28 When participants incorrectly answered a comprehension question, they were provided with an explanation about the correct answer before proceeding to the next part of the survey. The surveys also included questions on sociodemographic and health characteristics, the Vascular Quality of Life instrument (VascuQoL-6), 29 the three-item Subjective Numeracy Scale (SNS), 30 the Life Orientation Test–Revised (LOT-R; a measure of dispositional optimism), 31 and a single-item measure of an individual’s propensity to use medical care. 32
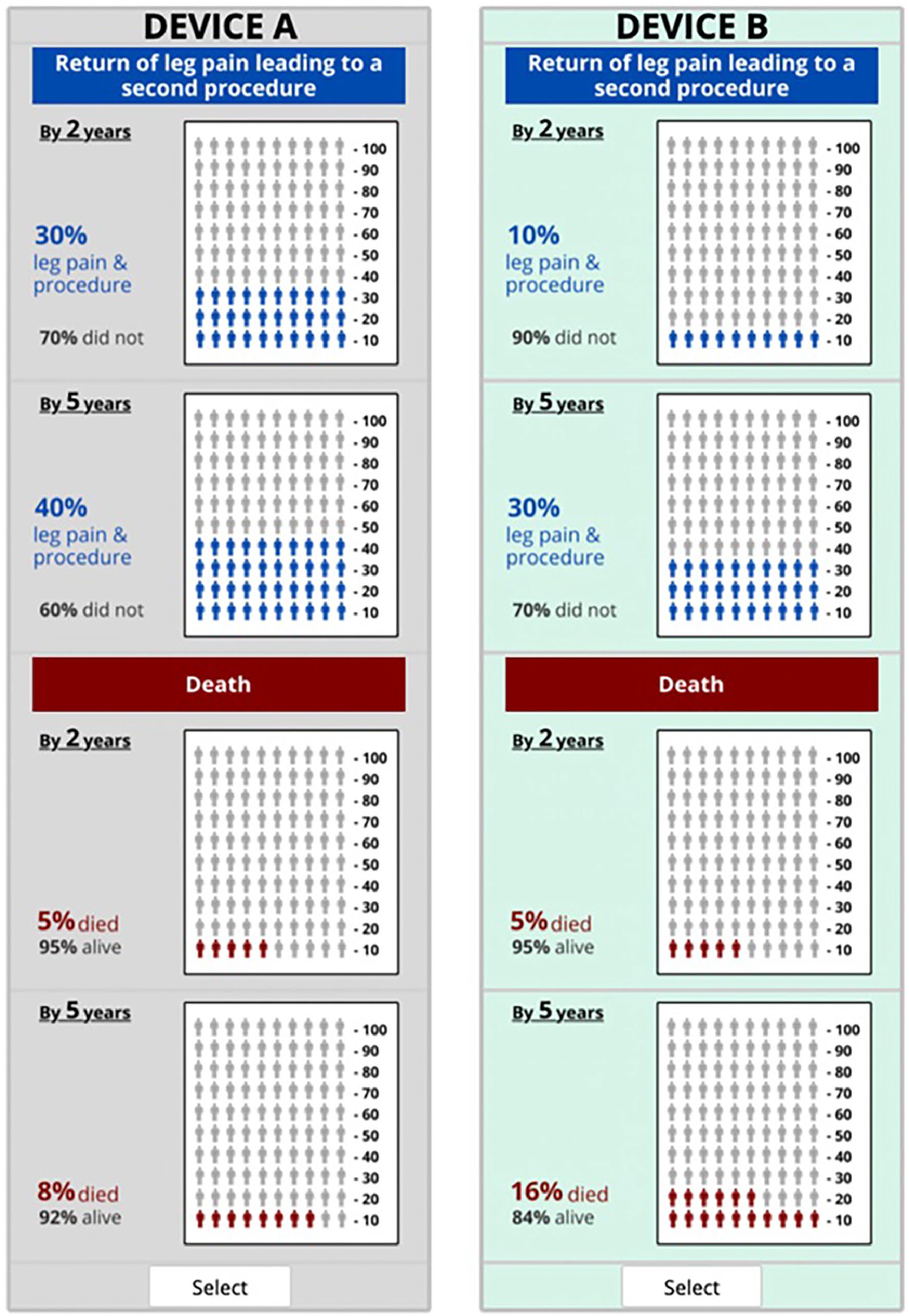
Example DCE question with icon arrays.
Decision context
Participants were asked to suppose that over the past several months their leg pain due to PAD had worsened, and they were unable to do usual activities of daily living (such as light housework, grocery shopping, or climbing stairs) without stopping to rest. They also were asked to suppose that their doctor informed them that they needed a procedure with a medical device that would allow them to resume their usual activities without being limited by pain. A no-device option was not included given the clinical acuity represented in the decision context.
Experimental design
An experimental design consists of pairs of device profiles required to estimate all the preference weights in a study design. A design with known statistical properties was generated for this study using algorithms by Kuhfeld in SAS version 9.4 (SAS Institute, Inc., Cary, NC, USA). 33 See supplemental materials and Table S2 for more details.
Statistical analysis
Responses to non-DCE survey items (e.g., sociodemographic characteristics) were summarized using descriptive statistics. For DCE responses, a random-parameters logit (RPL) regression model was used to estimate aggregate-level preference weights, accounting for correlation at the participant level. An advantage with using an RPL model is that in addition to estimating mean preference weights, it also generates corresponding standard deviations which represent participant-level variations in preferences. Scale-controlled RPL models with interaction terms were used to investigate whether model fit significantly differed by sex, age, race, survey version (i.e., with or without icon arrays), and a history of a previous revascularization procedure. As an adjunctive approach to evaluate preference heterogeneity, latent-class logit models also were fit to the DCE data.
Results from the RPL model were then used to calculate the maximum-acceptable risk (MAR) for 5-year mortality that participants would accept for a device with lower risks of CDTVR. Although the magnitude of benefits varies across clinical trials comparing drug-coated versus standard devices, to estimate the 5-year mortality MAR, benefit was defined as a reduction in the risk of CDTVR from 30% to 10% at 2 years and from 40% to 30% at 5 years. When computing the 5-year mortality MAR using the estimated preference weights, 2-year mortality risks were set at 5% for both devices (i.e., Device A and Device B). As the lowest 5-year mortality shown in the DCE questions was 8%, MAR estimates represent the incremental increase in risk above a baseline risk of 8%.
Results
In total, 4968 patients were screened, and 1541 patients were invited to complete the survey. A total of 272 completed surveys were collected. The mean age of participants was 69.8 ± 10.1 years; 67.6% were men, and 5.1% were Black (Table 1). Education level varied, with nearly a quarter (23.1%) of the sample having a high-school education or less, and 39.3% had some college, technical school, or a 2-year college degree. About half of the sample (52.9%) reported prior endovascular lower-extremity intervention, whereas 20.2% reported prior surgical lower-extremity revascularization. The median time to complete the survey was 29 minutes.
Participant characteristics.

Participants could select more than one option; percentages may sum to more than 100.
VascuQoL-6 scores can range from 4 to 24; higher scores represent better health status.
LOT-R scores can range from 0 to 24; higher scores represent more optimism.
Represents the sum of three questions; scores can range from 3 to 18; higher scores represent greater subjective numeracy.
MM scale ranges from 1 (I strongly lean toward waiting and seeing) to 6 (I strongly lean toward taking action).
On average, participants correctly answered 3.1 ± 1.5 of five comprehension questions with both versions of the survey (i.e., with or without icon arrays to portray risk levels; p = 0.97). Twenty-one participants (7.7%) gave incorrect responses to all five comprehension questions, and 72 (26.4%) answered ‘don’t know or not sure’ to one or more questions.
Preferences
Figure 2 displays the parameter estimates from two RPL models: one in which DCE attribute levels were fit as linear functions, and one in which attribute levels were fit as categorical variables (preference weights reported in Table 2 for linear functions and Table S3 for categorical). The downward slopes across risk levels for each attribute represent decreased patient preference for devices with higher levels of risk. The slopes for mortality risks at 2 and 5 years were steeper than the corresponding slopes for CDTVR risks (p < 0.0001 for both), indicating that increases in mortality risks were more important than equivalent increases in CDTVR risks. No significant interactions between attribute levels and sex, age, race, survey version, or previous revascularization procedure were identified (Table S4).
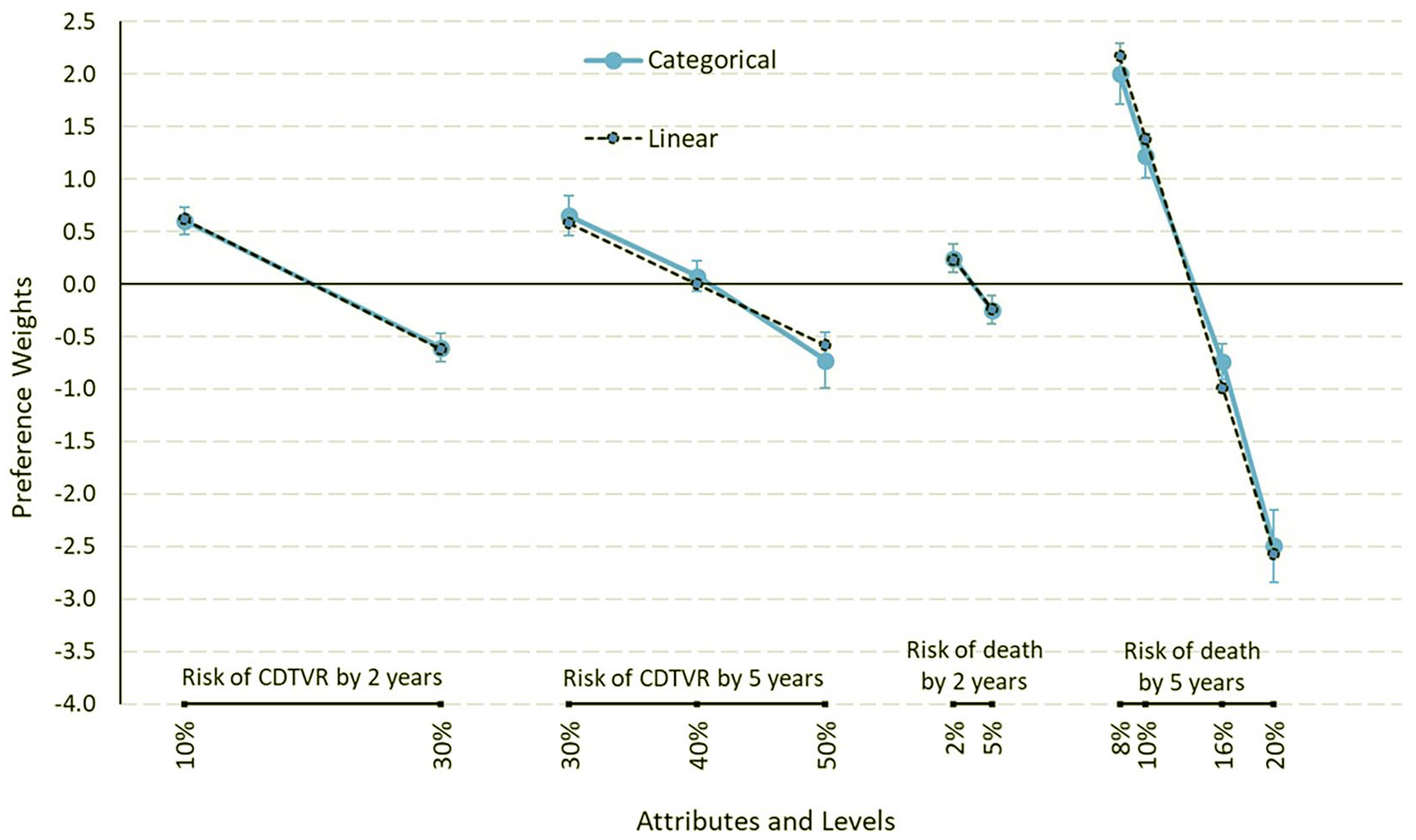
Preference weights for categorical and linear specifications for risk levels.
Preference weights from a random-parameters logit model with linear-fit attributes.
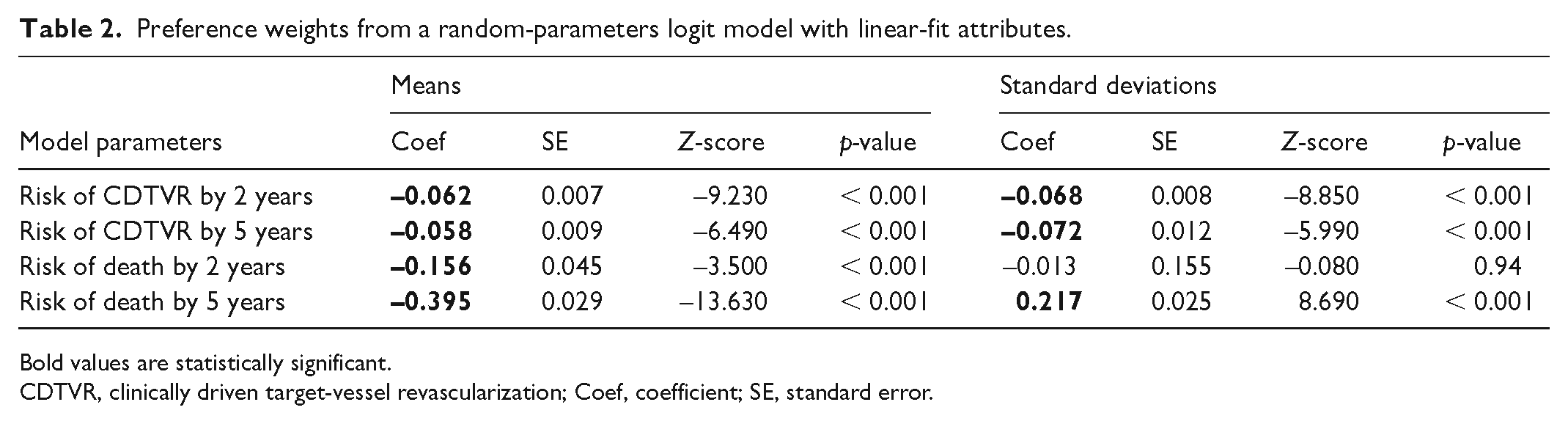
Bold values are statistically significant.
CDTVR, clinically driven target-vessel revascularization; Coef, coefficient; SE, standard error.
The preference estimates were then used to calculate the maximum level of 5-year mortality risk that would fully offset the perceived value of the incremental benefit with a device offering a 20 percentage-point reduction in the 2-year CDTVR risk (from 30% to 10%) and a 10 percentage-point reduction in the 5-year CDTVR risk (from 40% to 30%). For the full sample, the mean MAR corresponds to a 4.6 percentage-point (95% CI: 3.9–5.4) increase in the risk of 5-year mortality. Accounting for the baseline 5-year mortality risk of 8% in the DCE, the finding indicates that, on average, patients perceive that the defined CDTVR benefits outweigh mortality risks as long as the total 5-year mortality risk is less than 12.6% (i.e., 8% + 4.6%), all else being equal. Incremental MAR estimates from separate RPL models are similar across subgroups based on age, sex, race, previous procedure, and use or nonuse of icon arrays (Figure 3).

Incremental MAR forest plot for subgroups by age, sex, race, previous procedure, and use of icon arrays.
Among the pairs of hypothetical device profiles shown across the eight DCE questions, 49 (18.0%) participants chose the device option with the lower 5-year mortality risks every time. Another 56 (20.6%) chose the device option with the lower 5-year mortality risks in seven of the eight DCE questions. When excluding these patients, the incremental MAR for 5-year mortality risk increases from 4.6 percentage points (in the full sample) to 9.9 percentage points (95% CI: 8.4–11.4). Limiting the sample to 46% of patients who correctly answered four or all five comprehension questions, the incremental MAR is 5.2 percentage points (95% CI: 4.3–6.2). Results of latent-class models are reported in Figures S2 and S3.
Discussion
The anatomy of a medical decision, as described by Eddy in 1990, requires two steps. 34 The first step requires estimating outcomes of alternative management options. The second step requires comparing the desirability of the outcomes for each option. As evidenced by multiple meta-analyses of randomized clinical trials, numerous nonrandomized comparative effectiveness studies, and ongoing randomized trials, there has been a massive investment in generating empirical evidence about the benefits and risks associated with paclitaxel-coated devices versus standard devices for endovascular procedures in patients with PAD.4–6,10–19 The debate continues regarding the strengths and weaknesses of previous studies of risk associated with paclitaxel-coated devices, their estimated effect sizes, and associated uncertainty. Our study is the first to address the second step of medical decision making in providing evidence on patients’ preferences pertaining to the relative importance of incremental benefits and risks associated with medical devices used in lower-limb endovascular procedures.
In our DCE study, 272 patients with claudication evaluated alternative device profiles offering varying combinations of CDTVR and mortality risk at 2 and 5 years. Approximately 40% chose the device alternative with the lower risk of 5-year mortality in seven or all eight DCE choice questions. This finding highlights the importance to many patients of avoiding an increase in 5-year mortality risk for any of the offered benefit levels. An equally important finding is that most patients indicated they would accept higher mortality risks for more durable outcomes.
The average maximum-acceptable increase in 5-year mortality risk estimated in this study was 4–5 percentage points, which overlaps with the absolute mortality risk differences between paclitaxel-coated and noncoated devices reported across three meta-analyses (4.6% to 7.2%).4–6 Some clinicians might assume that patients would not accept a higher risk of long-term mortality for the reductions in CDTVR as specified in our representative scenario. Although patient acceptance of a higher long-term mortality risk for reductions in CDTVR might not be anticipated by many clinicians, revascularization procedures for claudication are performed to improve symptoms, rather than survival. Accordingly, survey revisions in the current study clarified that recurrent leg pain symptoms were the reason for a second procedure. This clarification may have aided participants in considering the impact of pain on their physical functioning and quality of life, and helped motivate their acceptance of mortality risk. Similar acceptance of mortality risk for improvements in physical functioning and reductions in pain has been reported in studies employing standard gamble and DCEs to estimate health-state utilities.35,36
A strength of the study is the recruitment of patients with claudication from seven medical centers. Participant demographics are consistent with the increasing prevalence of PAD with age and among men. 37 However, despite inclusion of study sites in cities with racially and ethnically diverse patient populations and a higher prevalence of PAD among Black Americans, 38 the recruited sample was predominantly non-Hispanic White (92%), in part because our survey was administered only in English. As such, though the tight confidence intervals on the preference weights indicate high levels of precision, our estimates may not reflect the preferences of a representative sample of US patients with PAD. The study sample reported levels of health status (VascuQoL-6D) similar to other cohorts with claudication,39,40 and had a moderate level of optimism (LOT-R). 41 Subjective numeracy in our sample (13.1; SD = 3.7) was slightly lower than reported for a diverse sample of nearly 16,000 adults across the mid-southern USA (14.3; SD = 3.6). 42 About half (53%) previously had a lower-extremity endovascular revascularization procedure which aligned with our recruitment goal.
Results of the current study should be interpreted with consideration of commonly cited concerns about stated-preference studies, including whether people would make the same choices in real life. Although we are uncertain as to the influences of real-world circumstances, advice from others, and external factors, patients participating in pretest interviews provided rational reasons for choices they made in ways that aligned with the study findings.
Although the scrutiny over potential mortality risks posed by paclitaxel-coated devices has subsided, our study results remain valuable to clinicians and regulators to support transparent consideration of patient perspectives across a range of benefit-risk scenarios relevant to interventions for claudication. These decisions can be better informed by understanding patients’ benefit-risk preferences through studies like ours. The study results underscore the importance of reducing CDTVR rates to patients and its relevance as a clinical trial endpoint.
In addition, the study results have implications for clinical practice; patients have varied views on benefit-risk tradeoffs, but patient characteristics were not significantly associated with lower or higher levels of risk tolerance. Thus, practical approaches are needed to determine an individual patient’s acceptance of benefit-risk tradeoffs. Patients routinely discuss treatment options with a provider and make a ‘yes’ or ‘no’ decision, but quantitative findings from experiment-based studies like this one can provide the foundation for decision support tools. Such tools can be designed to elucidate an individual’s acceptance of adverse-event risks for defined treatment benefits which could be documented in patients’ electronic health records to confirm their understanding and assessment of benefit-risk tradeoffs or for use in risk adjustment algorithms.
Conclusion
Our study findings suggest that patients, on average, would accept incremental 5-year mortality risks of up to four to five percentage points with interventions delivering similar reductions in clinically driven target-vessel revascularization as provided by paclitaxel-coated devices, all else being equal. The study also demonstrates that patients have varying levels of risk tolerance that are not associated with demographic characteristics. This observation indicates potential value in developing systematic, efficient approaches to eliciting reliable patient-level measures of risk tolerance that could be used in shared decision making to ensure patients receive preference-concordant medical interventions.
Supplemental Material
sj-pdf-1-vmj-10.1177_1358863X241290233 – Supplemental material for Quantifying patients’ preferences on tradeoffs between mortality risk and reduced need for target vessel revascularization for claudication
Supplemental material, sj-pdf-1-vmj-10.1177_1358863X241290233 for Quantifying patients’ preferences on tradeoffs between mortality risk and reduced need for target vessel revascularization for claudication by Shelby D Reed, Jessie Sutphin, Matthew J Wallace, Juan Marcos Gonzalez, Jui-Chen Yang, F Reed Johnson, Jennifer Tsapatsaris, Michelle E Tarver, Anindita Saha, Allen L Chen, David J Gebben, Misti Malone, Andrew Farb, Olufemi Babalola, Eva M Rorer, Sahil A Parikh, Jessica P Simons, W Schuyler Jones, Mitchell W Krucoff, Eric A Secemsky and Matthew A Corriere in Vascular Medicine
Footnotes
Acknowledgements
The authors would like to acknowledge the contributions of study site personnel who were instrumental in recruiting patients to participate in the study: Ashley Duby (University of Michigan); Shylie Ati (Harvard University); Suzanne Edwards and Kate Dalton (Columbia University); Shauneen Valliere (University of Massachusetts); Dr Ehrin Armstrong, Cheyanne Johnson, and Jennifer DeBattista (St Helena Hospital); Dr Omar Hyder, Bailey Nevins, and Elizabeth Nowak (The Miriam Hospital); and Krista Camuglia (Duke University). The authors would also like to acknowledge the contributions of our patient advisors: Frank Dent and Rick Black.
See commentary by Hogan and Coyelwright. 43
Data availability statement
Study data are available from the corresponding author on reasonable request.
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Sahil Parikh reports institutional research support from Abbott Vascular, Acotec, Boston Scientific, Concept Medical, Shockwave Medical, Surmodics, Reflow Medical, TriReme Medical, and Veryan Medical; advisory board for: Abbott Vascular, Boston Scientific, Cordis, Medtronic, and Philips; and consulting for Canon, Inari, Penumbra, and Terumo. Eric A. Secemsky reports grants (to institution) from Abbott/CSI, BD, Boston Scientific, Cook Medical, Medtronic, and Philips; and consulting for Abbott/CSI, BD, BMS, Boston Scientific, Cagent, Conavi, Cook, Cordis, Endovascular Engineering, Gore, InfraRedx, Medtronic, Philips, RapidAI, Shockwave, Terumo, Thrombolex, VentureMed, and ZOLL. The remaining authors have no relevant conflicts of interest.
Funding
This work was supported by a contract with FDA (75F40120C00179). The content is solely the responsibility of the authors and does not necessarily represent the official views of the US FDA.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
