Abstract
Patients with peripheral artery disease (PAD) face a range of treatment options to improve survival and quality of life. An evidence-based shared decision-making tool (brochure, website, and recorded patient vignettes) for patients with new or worsening claudication symptoms was created using mixed methods and following the International Patient Decision Aids Standards (IPDAS) criteria. We reviewed literature and collected qualitative input from patients (n = 28) and clinicians (n = 34) to identify decisional needs, barriers, outcomes, knowledge, and preferences related to claudication treatment, along with input on implementation logistics from 59 patients and 27 clinicians. A prototype decision aid was developed and tested through a survey administered to 20 patients with PAD and 23 clinicians. Patients identified invasive treatment options (endovascular or surgical revascularization), non-invasive treatments (supervised exercise therapy, claudication medications), and combinations of these as key decisions. A total of 65% of clinicians thought the brochure would be useful for medical decision-making, an additional 30% with suggested improvements. For patients, those percentages were 75% and 25%, respectively. For the website, 76.5% of clinicians and 85.7% of patients thought it would be useful; an additional 17.6% of clinicians and 14.3% of patients thought it would be useful, with improvements. Suggestions were incorporated in the final version. The first prototype was well-received among patients and clinicians. The next step is to implement the tool in a PAD specialty care setting to evaluate its impact on patient knowledge, engagement, and decisional quality.
Keywords
Introduction
Peripheral artery disease (PAD) is a burdensome condition that impacts an estimated 8.5 million Americans. 1 It is an indicator of systemic atherosclerosis that results in lower extremity arterial blockages leading to impaired blood flow. Symptoms can present as exertional calf (or buttock or thigh) pain while walking that is relieved with rest (‘intermittent claudication’). PAD leg symptoms can severely affect a patients’ health status.2–4 In addition, 1-year cardiovascular event rates are estimated to be over 20% in patients with PAD, and mortality rates of 15–30% within 5 years of diagnosis have been noted.5–7
While cardiovascular risk management is recommended for all patients with PAD, multiple effective treatment options are available to relieve claudication symptoms.8,9 These options range from non-invasive strategies, such as pharmacologic treatment and supervised and home-based exercise therapy, to endovascular or surgical revascularization procedures. While there is no ‘gold-standard’ treatment for PAD symptom relief, less invasive options are recommended as a first-choice treatment and decisions to proceed with revascularization depend on the severity of symptoms, comorbidities, and response to previously tried non-invasive options. 8 Importantly, the preferences and treatment goals of the patient are major factors when considering PAD treatment options. 8
Ideally, patients with PAD receive education on available treatments, including the risks and benefits of each, to empower them to decide which treatment strategy most aligns with their personal goals and values. Achieving this vision requires developing tools for patients with new or worsening symptoms of claudication. After reviewing publicly available decision-making tools for PAD, we created an evidence-based PAD treatment decision aid for patients with mild to severe claudication symptoms (Rutherford Grade I) 10 and evaluated its acceptability as a foundation for the broader integration of shared decision-making into the clinical care processes for patients with PAD.
Methods
To develop the decision aid, we adhered to the International Patient Decision Aids Standards (IPDAS). 11 Qualitative input was collected from patient and clinician stakeholders in three waves (Figure 1).
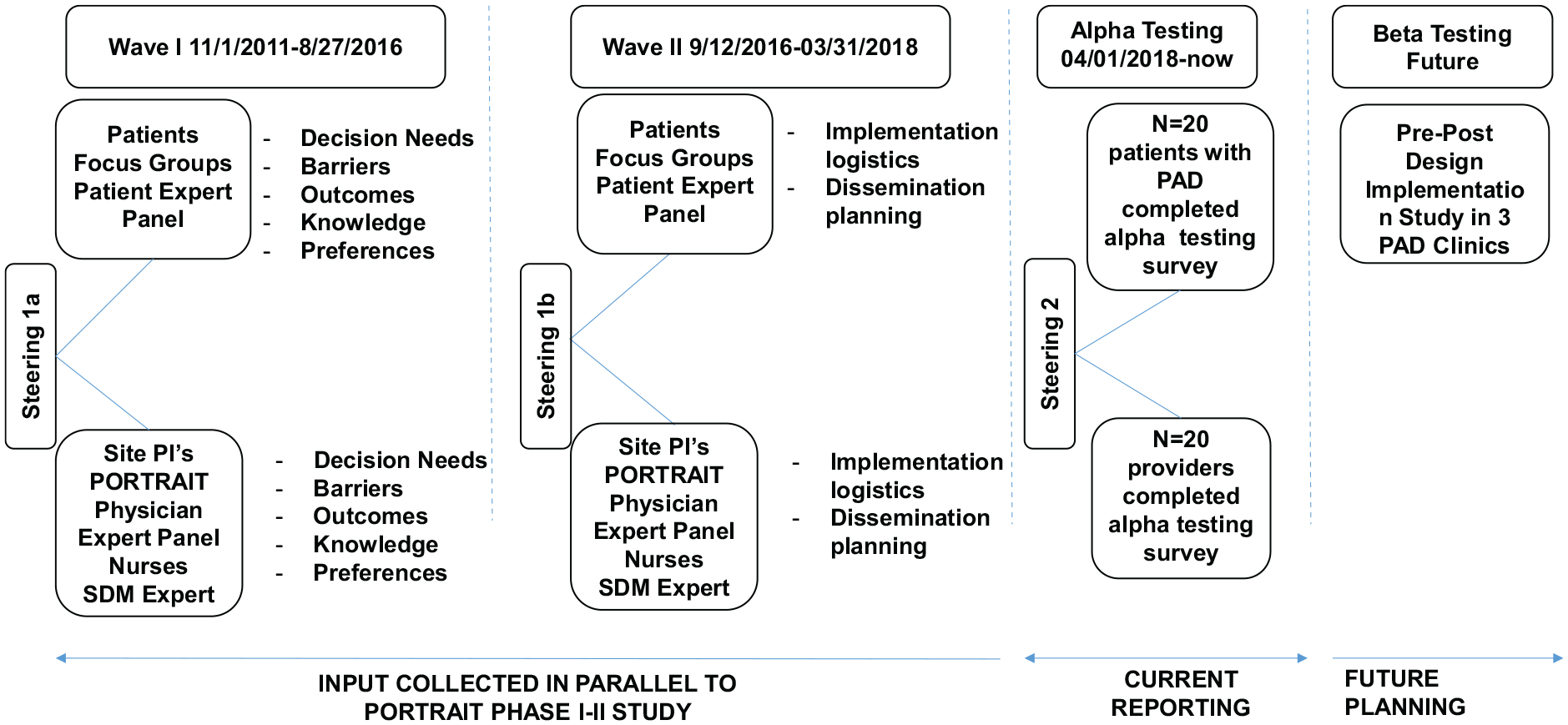
Timeline of the development process of the SHOW-ME PAD decision aid.
In
The latest clinical guidelines (only Class I recommendations) from the American Heart Association and the American College of Cardiology 13 provided the evidence base used to describe PAD, treatments, and outcomes. 13
For the
The research was approved by the Institutional Review Board of the Saint Luke’s Health System, Kansas City, MO, and Yale University, New Haven, CT. All enrolled patients provided informed consent. Focus group discussions were recorded, transcribed, and coded using descriptive content analysis until thematic saturation occurred. The transcripts were coded by a multidisciplinary team of nurses, a psychologist, an interventional cardiologist, and community-based participatory health researchers (KS, CD, NS, CF, CP, DS) using the methodology by Hahn. 17 For each wave of focus groups, we kept interviewing stakeholders until we reached content saturation, meaning that no new themes emerged. The descriptive data obtained from the Alpha testing survey was generated through the reporting functionality of REDCap.
Finally, the SHOW-ME PAD decision tool was registered and listed in the Ottawa Hospital Research Institute shared decision aid database. 18
Results
For
The following PAD treatment outcomes were identified as important: (1) rapid pain relief, (2) living longer, (3) improving quality of life, (4) cost of treatment, (5) avoiding loss of toes or legs, (6) timeline to return to normal activities, (7) decreasing risk of heart attack and stroke, (8) long-lasting symptom relief, and (9) avoiding a procedure or surgery. In steering meeting 1a, it was decided that the treatment decision support should focus on highlighting the evidence 13 of two treatment pathways (or combinations thereof) for PAD symptom relief: a non-invasive pathway that included medications and supervised exercise therapy (SET) and an invasive treatment pathway that included endovascular and surgical options. The option of ‘do nothing’ was also included. Per the guidelines, lifestyle management for PAD is recommended for everyone, regardless of the treatment path(s) chosen to relieve the PAD symptoms (Figure 2, left panel). The following consolidated treatment outcomes were highlighted in the decision aid concept: (1) symptom relief and quality of life; (2) cost; (3) decreased risk of heart attack and stroke; (4) avoid loss of toes or legs; and (5) timeline of return to normal activities (Figure 2, middle panel).
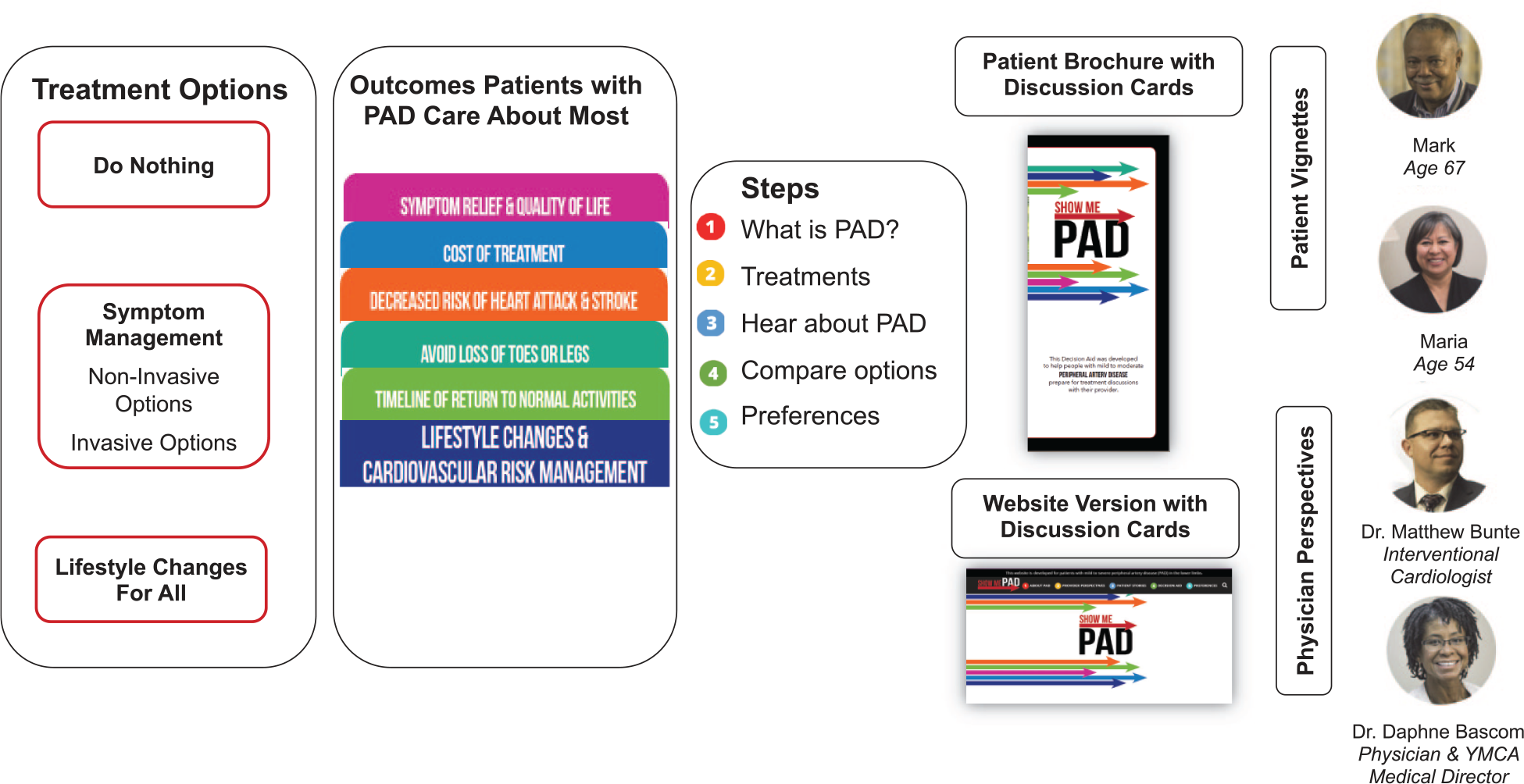
Original conceptual overview of the SHOW-ME PAD decision aid.
For
For the
Overview of patient characteristics.

Data are presented as numbers (percentages), unless otherwise stated.
PAD, peripheral artery disease.
Overview of PAD clinician characteristics.
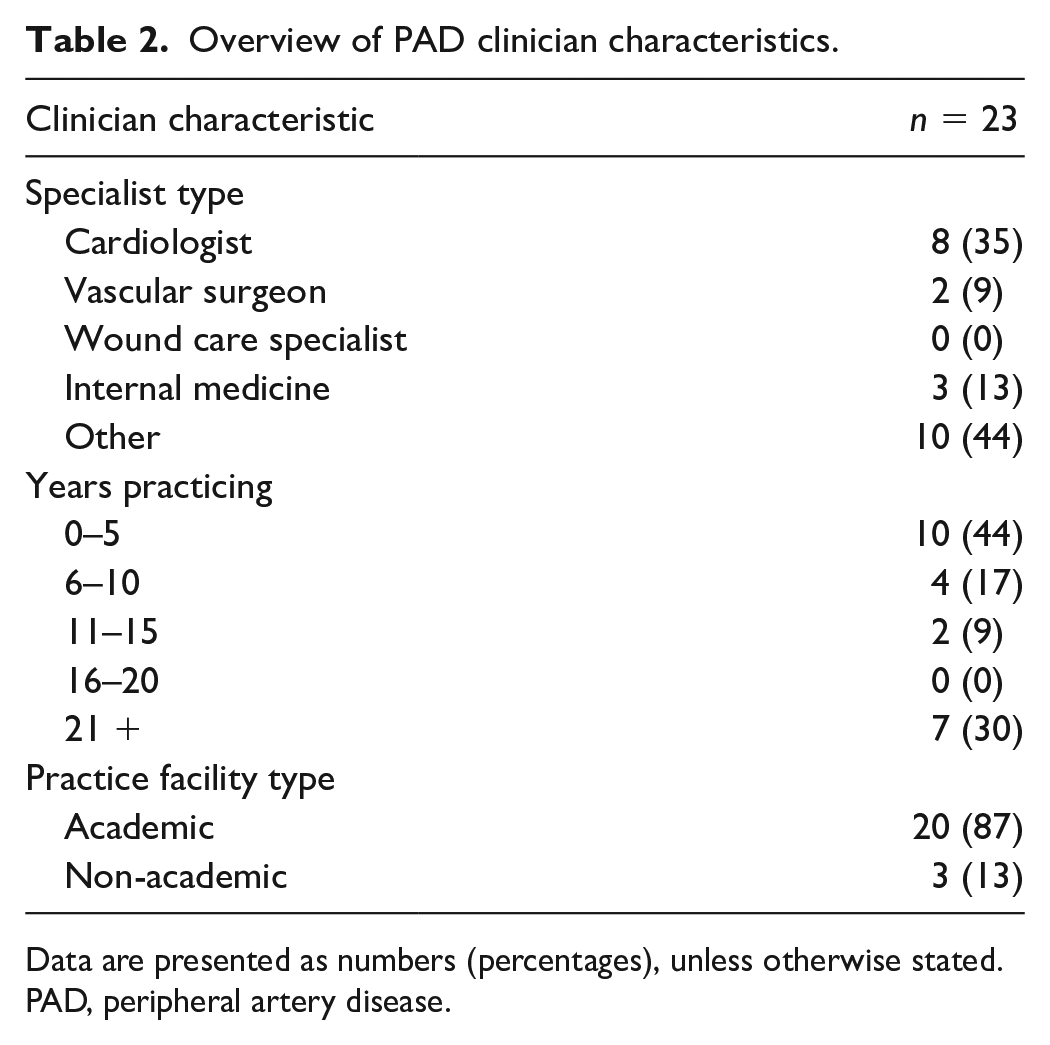
Data are presented as numbers (percentages), unless otherwise stated.
PAD, peripheral artery disease.
Alpha testing results.

Data are presented as numbers (percentages).
PAD, peripheral artery disease.
Online supplemental Tables 4–6 provide an overview of all alpha testing stakeholder suggestions and the Steering 3 action items. The main change was the consolidation of the nine separate discussion cards (both for brochure and website) into a streamlined side-by-side overview of the pros and cons for each treatment modality. In direct response to the feedback that materials were containing too much information, we only focused on two patient-centered outcomes: symptoms and quality of life; and returning to usual activities. Generic cost information, tips, and resources for medical treatments were summarized underneath the decision aid. The decision aid was made central in the brochure as a fold out. The website was updated so that patients could choose to go straight to the decision aid, or they could review a 1.5-minute compilation video. Online supplemental Figure 1 provides an overview of the revised decision aid.
Discussion
Given the preference-sensitive nature of PAD treatments, having a reliable, objective, evidence-based decision-making tool is an important, currently unmet, need. To address this, we designed and tested a decision tool to assist patients and clinicians to make more informed, participatory decisions about PAD treatment. We worked intensely with patient and clinician stakeholders to design and alpha test a tool that would support patients with new or worsening symptoms of PAD in understanding and considering different treatment options for PAD symptom relief. Patients expressed interest in learning more about five different PAD treatment outcomes: (1) symptom relief and quality of life; (2) cost of treatment; (3) decreased risk of heart attack and stroke; (4) avoid loss of toes or legs; and (5) timeline of return to normal activities. These outcomes defined the framework for presenting levels of evidence for non-invasive and invasive treatment options for PAD symptom relief provided in a variety of modalities (website, videos, and printed materials) to facilitate the treatment discussion with the clinician, but also to be used as a resource to patients in preparation of their visit.
Overall, the current SHOW-ME PAD decision aid was well received and next steps for an update of the tool for implementation testing (beta testing) in routine clinical practice (PAD specialty setting) are being developed. To date, tools for shared decision-making in PAD have not been widely developed or studied. A decision aid has been developed by Healthwise, 19 but it does not highlight the patient-centered aspects of outcomes following PAD treatment options and does not highlight specific benefits of individual evidence-based strategies, such as SET, whereas the SHOW-ME PAD tool focuses on all potential treatment options as well as the patient experience. With patients and clinicians, we co-created a platform that takes into account the latest evidence about all available treatment options and their outcomes for PAD symptom relief, 13 as well as patients’ preferences with regards to treatment and potential outcomes that matter to them. 20
Decision aids that facilitate shared decision-making have demonstrated the potential to be cost saving because less invasive options are often preferred by patients when given a choice; higher treatment satisfaction and knowledge are provided; and there is less decision conflict after the decision has been made.20,21 While there is no ‘gold-standard’ treatment for PAD, less invasive options are generally recommended as a first-choice treatment; however, personal preferences, quality of life considerations, or anatomical factors should also weigh into a treatment decision. 8 Each of the treatment options has specific risks and benefits. Patients may not know the risks and benefits associated with each treatment and how these align with their treatment preferences and goals. Patient-centric tools like the SHOW-ME PAD decision aid could be used to better tailor decision-making with patients’ preferences and improve the quality of medical decision-making.
The next steps for the evaluation of the SHOW-ME PAD decision aid are designing and testing clinical workflows for implementation in a PAD specialty care setting. Testing of the tool is currently being conducted a pre-post design study 22 in patients with new or worsening PAD symptoms who consult with a PAD specialty care clinic in the US. The study goals are to understand if a decision aid reduces ‘decisional conflict’ (i.e. uncertainty and insufficient support and information to make a medical decision) and increases patient knowledge of treatment options. Also, as SHOW-ME PAD is currently designed as a static tool (i.e. no individualized risk prediction for certain outcomes), further development is needed to integrate validated risk prediction models for relevant outcomes (e.g. risk of bleeding or developing acute kidney injury associated with peripheral angioplasty, health status outcomes), and integrate the newest results of comparative effectiveness research as evidence becomes available. Further testing of the decision aid in a randomized design is also anticipated to compare in the most valid method the benefits of shared decision-making in PAD.
Study limitations
While the study has adhered to the IPDAS evaluation criteria, and was conceptualized to be patient-centric, it does have limitations. The tool was designed for the PAD specialty care setting and primarily in academic settings. It is unclear whether it will be acceptable or effective in other settings. Given the use of convenience sampling for this alpha testing stage, validation in larger populations and geographies would be warranted to ensure wider generalizability. Patients with claudication and clinicians treating this condition were intensely involved throughout all phases of design and development of the decision aid. However, in the alpha testing phase, patients with more advanced expression of the disease (those with wounds or a history of amputation) also evaluated the decision aid and it is unclear whether their advanced disease process may have impacted the way they rated the materials. Furthermore, while we relied on the most recent guideline information, the body of comparative effectiveness research in PAD is still being built and, as evidence becomes available, shared decision-making platforms such as SHOW-ME PAD will need periodic updating.
Conclusion
The goal of SHOW-ME PAD is to make the existing evidence on treatment outcomes – focusing on health status outcomes that reflect the patients’ perspective – easily available to patients and clinicians, such that more informed, evidence-based, shared treatment decisions occur. This study serves as the foundation for future efforts to build shared decision-making platforms for patients with PAD facing important treatment decisions. We demonstrated the feasibility of this vision and gained important insights into the barriers and facilitators of implementing personalized shared decision-making tools in routine clinical care. The SHOW-ME PAD platform has the potential to reorganize care delivery to patients with PAD in a way that creates more value for the patient and society.
Supplemental Material
sj-docx-1-vmj-10.1177_1358863X20988780 – Supplemental material for Treatment decisions for patients with peripheral artery disease and symptoms of claudication: Development process and alpha testing of the SHOW-ME PAD decision aid
Supplemental material, sj-docx-1-vmj-10.1177_1358863X20988780 for Treatment decisions for patients with peripheral artery disease and symptoms of claudication: Development process and alpha testing of the SHOW-ME PAD decision aid by Kim G Smolderen, Christina Pacheco, Jeremy Provance, Nancy Stone, Christine Fuss, Carole Decker, Matthew Bunte, Qurat-ul-ain Jelani, David M Safley, Eric Secemsky, Karen R Sepucha, Erica S Spatz, Carlos Mena-Hurtado and John A Spertus in Vascular Medicine
Footnotes
Acknowledgements
We thank all the patient, physician, and allied health stakeholders, members of the graphic design team (Dustin Fann, Kristi Ernstig, Maggie Goldsborough, Brian Ellis), the patient and physician experts who helped with developing the patient and physician videos, and the patients who participated in our focus groups. We would like to thank the authors of the paper on ‘Development of a decision aid about fertility preservation for women with breast cancer in The Netherlands’ for allowing us to use their evaluation questions for the alpha testing of our study. 16
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Dr Mena-Hurtado is a consultant for Abbott, Cook, Cardinal Health, Bard, Boston Scientific, Gore, Optum Labs, and Medtronic. Dr Spertus owns the copyright for the KCCQ; has equity interest in Health Outcomes Sciences; has received consulting income from Novartis, Bayer, AstraZeneca, V-Wave, Corvia, and Janssen; has served on the Advisory Board for United Healthcare; and has served on the Board of Directors for Blue Cross Blue Shield of Kansas City. Dr Smolderen is a consultant for Optum Labs and reports support through unrestricted research grants from Cardiva, Abbott, and Johnson & Johnson (for work unrelated to this study). The remaining authors report no relevant disclosures.
Funding
Research reported in this manuscript was partially funded through a Patient-Centered Outcomes Research Institute (PCORI) Award (IP2 PI000753-01; CE-1304-6677), The Netherlands Organization for Scientific Research (VENI Grant No. 916.11.179), unrestricted grants from W. L. Gore & Associates, Inc. (Flagstaff, AZ) and Merck & Company, Inc. (Kenilworth, NJ). Dr Eric Secemsky is funded in part by NIH/NHLBI K23HL150290.
The funding organizations had no role in the design and conduct of the study, collection, management, analysis, and interpretation of the data and preparation, review, or approval of the manuscript, or the decision to submit the manuscript for publication. The statements in this manuscript are solely the responsibility of the authors and do not necessarily represent the views of the PCORI, its Board of Governors or Methodology Committee. All manuscripts for the PORTRAIT study are prepared by independent authors who are not governed by the funding sponsors and are reviewed by an academic publications committee before submission.
Supplementary material
The supplementary material is available online with the article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
