Abstract
Background:
Internal carotid artery (ICA) occlusion is the major cause of ischemic stroke. The effect of collateral vessels on cerebral hemodynamics in ICA occlusion remains unclear. This study investigated the correlation between collateral vessels and the peak systolic velocity of the middle cerebral artery (MCA) in patients with ICA occlusion.
Methods:
The relevant collateral vessels included the anterior communicating (ACoA), posterior communicating (PCoA), and internal-external carotid (IECCA) arteries, respectively. Patients with unilateral ICA occlusion (n = 251) underwent transcranial Doppler imaging to detect the peak systolic velocity (PSV) of the MCA and other intracranial arteries. The clinical symptoms were assessed using the National Institutes of Health Stroke Scale (NIHSS).
Results:
Patients with ACoA collaterals had significantly higher PSVMCA scores and significantly lower NIHSS scores than those without ACoA collaterals (p < 0.001). Patients without any notable collaterals and those with only IECCA had the lowest PSVMCA and highest NIHSS scores. The PSVMCA and NIHSS scores were negatively correlated (r = −0.566, p < 0.001).
Conclusion:
Collateral circulation patency in unilateral ICA occlusion was closely associated with clinical symptoms, and patients with ACoA collaterals may have favorable outcomes.
Keywords
Background
The annual risk of transient ischemic attack (TIA) or ischemic stroke of asymptomatic internal carotid artery (ICA) occlusion is estimated to be between 2% and 8%. 1 The risk of stroke recurrence in symptomatic ICA occlusion is 8%, 10%, and 14% at 30 days, 1 year, and 5 years, respectively.2,3 The absence of collateral circulation in patients with ICA occlusion is usually associated with a poor prognosis. 4 According to well-known and accepted definitions, four main collateral pathways, namely anterior communicating arteries (ACoA), posterior communicating arteries (PCoA), internal-external carotid artery collaterals (IECCA), and leptomeningeal collaterals, can be assessed. 5 According to previous studies, the various types and degrees of collateral circulation patency may be associated with different compensatory capacities, leading to variable clinical manifestations and outcomes.6,7 The National Institutes of Health Stroke Scale (NIHSS) is a systematic assessment tool for stroke with high reliability and validity, which has been widely used in stroke patients for clinical evaluation, therapeutic selection, and prognosis prediction. 8
However, few studies have used transcranial color code sonography and NIHSS to evaluate the effects of intracranial collateral circulation on clinical manifestations in patients with ICA occlusion. The present study used ultrasonography to investigate the collateral circulation and peak systolic velocity of the middle cerebral artery (PSVMCA) of patients with ICA occlusion and qualitatively assessed the clinical symptoms using NIHSS. Additionally, the correlation between PSVMCA and NIHSS scores in patients with ICA occlusion was analyzed.
Methods
This study was approved by the Xuanwu Hospital Ethics Committee (ClinicalTrials.gov Identifier: NCT02397655).
Participants
This prospective multicenter registered study was initiated by the China National Health and Family Planning Commission. We investigated 10,711 consecutive patients with ischemic cerebrovascular disease from 20 hospitals in China between June 2015 and April 2016. The 20 hospitals represented North, South, East, Central, Southwest, Northeast, and Northwest China. The inclusion criteria were as follows: (1) patients were diagnosed with ischemic cerebrovascular disease by neurologists upon admission and (2) ICA occlusion should be detected using ultrasonography and confirmed using digital subtraction angiography (DSA) or computed tomography angiography (CTA). Patients meeting any of the following were excluded: (1) concomitant ipsilateral MCA stenosis or occlusion; (2) severe stenosis or occlusion of contralateral ICA stenosis; (3) severe stenosis or occlusion of the vertebrobasilar artery; (4) localized mismatching of infarction and ICA occlusion; and (5) insufficient temporal windows. TIA was defined as a brief episode of neurologic dysfunction resulting from focal cerebral or retinal ischemia, with clinical symptoms typically lasting less than 1 h without evidence of acute infarction on imaging. Stroke was defined as a focal neurologic deficit lasting over 1 h with acute infarction on imaging such as computed tomography (CT) and magnetic resonance imaging (MRI), according to the guidelines of the American Stroke Association. 9
Ultrasonographic examinations
Ultrasonographic examinations, including carotid artery ultrasound, transcranial Doppler (TCD), and transcranial color-coded duplex sonography (TCCD), were performed by qualified practitioners at local hospitals. Ultrasound was conducted with 3.0–12.0-MHz linear array probes or 4.0–8.0-MHz micro-convex array probes, and examinations were conducted in the supine position. The depth was set at 5–10 cm, and the focus points were adjusted at the carotid level. The color scale was set at 6.0–12.0 cm/s to verify the weak blood flow signal, and gray and color gains were adjusted for the patients. ICA occlusion was diagnosed when the ICA was filled with homogenous or heterogeneous echoes and color Doppler flow imaging showed noncontinuous blood flow. 4
Patients were examined with a lateral supine position and underwent TCCD examinations with 1.0–5.0-MHz phased array probes. The depth was set at 10–15 cm, and the focus points were adjusted at the midbrain section. Through the temporal acoustic window, the hemodynamics of bilateral MCA, anterior cerebral arteries (ACA), posterior cerebral arteries (PCA), and terminal internal carotid arteries were detected through the occiput acoustic window to detect the hemodynamics of bilateral vertebral arteries and basilar artery. The hemodynamics of ophthalmic arteries were detected through the optic acoustic window. TCD examinations were conducted in the supine position. Over the bilateral temporal acoustic window, occiput acoustic window, and optic acoustic window, a 1.6-MHz probe was used to detect different arteries. The documented parameters included the PSV, end-diastolic velocity (EDV), pulsatility index, and blood flow direction. PSV and EDV were selected as the highest velocities in multiple beats.
Evaluation of collateral circulation
Intracranial collateral circulation was evaluated according to previous criteria. 10 The establishment of ACoA collaterals was identified when the blood flow direction of the A1 segment of ipsilateral ACA was reversed or when the blood flow velocity of ipsilateral MCA decreased after compressing the contralateral common carotid artery (Figures 1A and 1B). The establishment of PCoA collaterals was verified when TCCD showed PCoAs connecting the terminal ICA and PCA or when TCD revealed increased blood flow velocity of the ipsilateral P1 segment of the PCA (> 50% of the contralateral blood flow velocity) with increased blood flow velocity in the basilar artery (Figure 1C). The establishment of IECCA was confirmed when the blood flow direction of the ipsilateral ophthalmic artery was reversed with a low-resistance Doppler sonography waveform. The establishment of collateral circulation was confirmed using DSA or CTA (Figure 2).
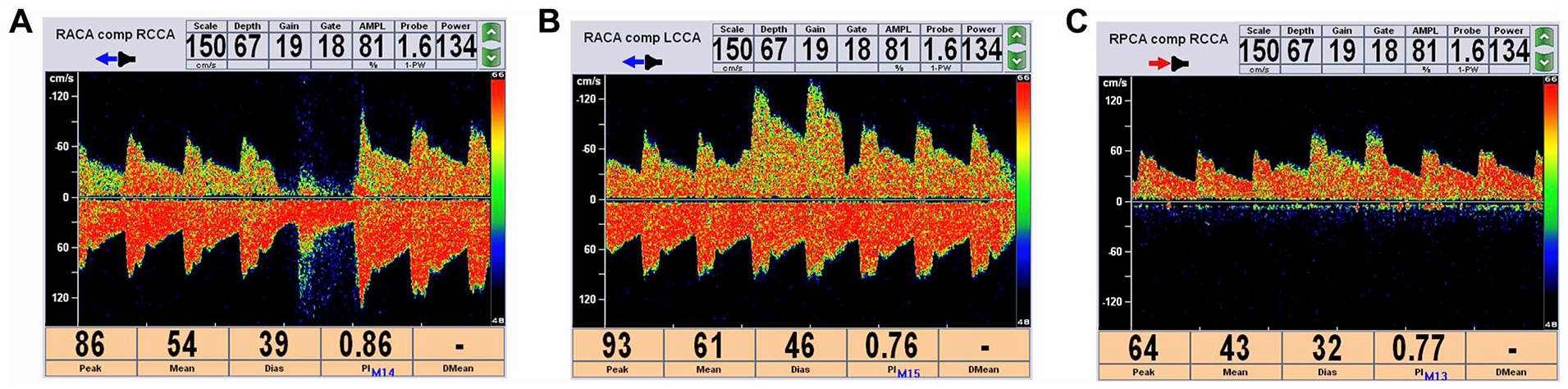
ACoA and PCoA in ICA occlusion performed by TCD.
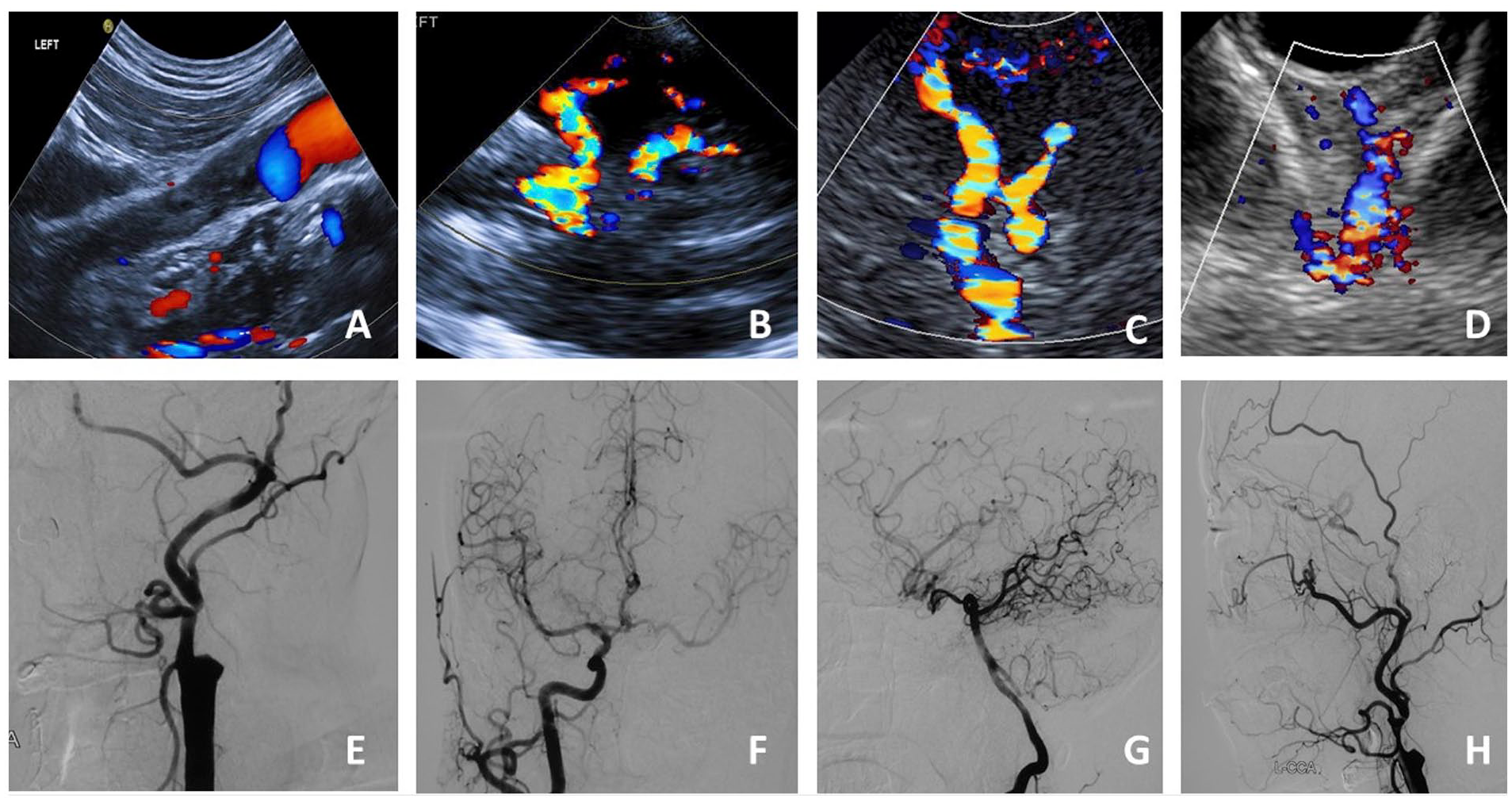
Collateral circulation in ICA occlusion performed using ultrasonography and DSA.
Patients were classified into eight groups based on the collateral circulation type: ACoA, PCoA, IECCA, ACoA + PCoA, ACoA + IECCA, PCoA + IECCA, ACoA + PCoA + IECCA, and a noncollateral group.
Data collection
At the time of hospital admission, information about the patients’ demographics, medical history, AIS risk factors, previous stroke medications, and clinical laboratory findings were gathered. All patients underwent MRI or CT to evaluate infarction. A DSA or CTA was performed to evaluate occlusion of the carotid artery and to verify the intracranial collateral circulation assessed by TCD/TCCD. Neurologists evaluated the NIHSS score at admission, and the researchers referred to the hospital management system to obtain the NIHSS score and clinical symptoms. 8 The quality control protocols have been described in our previous study. 11
Statistical analysis
SPSS software (version 24.0; IBM Corp., Armonk, NY, USA) was used for statistical analyses. The one-sample Kolmogorov–Smirnov test was used to identify the distribution of continuous variables. Continuous variables with normal distribution are presented as mean ± SD and nonnormally distributed variables are presented as median (IQR); frequencies (percentages) were measured for categorical variables. The two-sided Student’s t-test (normally distributed), Wilcoxon test (not normally distributed), and chi-squared test were performed to detect differences between groups. Linear regression analysis was performed to identify the correlations between individual PSVMCA and NIHSS scores. Furthermore, p ⩽ 0.05 was considered statistically significant.
Results
Patient characteristics
A total of 315 patients had ICA occlusion. Of these, 64 were excluded based on the following conditions: 15 had severe stenosis or occlusion of the contralateral ICA; 12 had stenosis or occlusion of the ipsilateral MCA; 15 had severe stenosis or occlusion of the vertebrobasilar artery, localizing mismatch of the infarction and ICA occlusion, or overlapping factors; and 22 had insufficient temporal windows from which cerebral vascular hemodynamics could not be obtained. Ultimately, 251 patients with unilateral ICA occlusion were enrolled in the study (Figure 3).
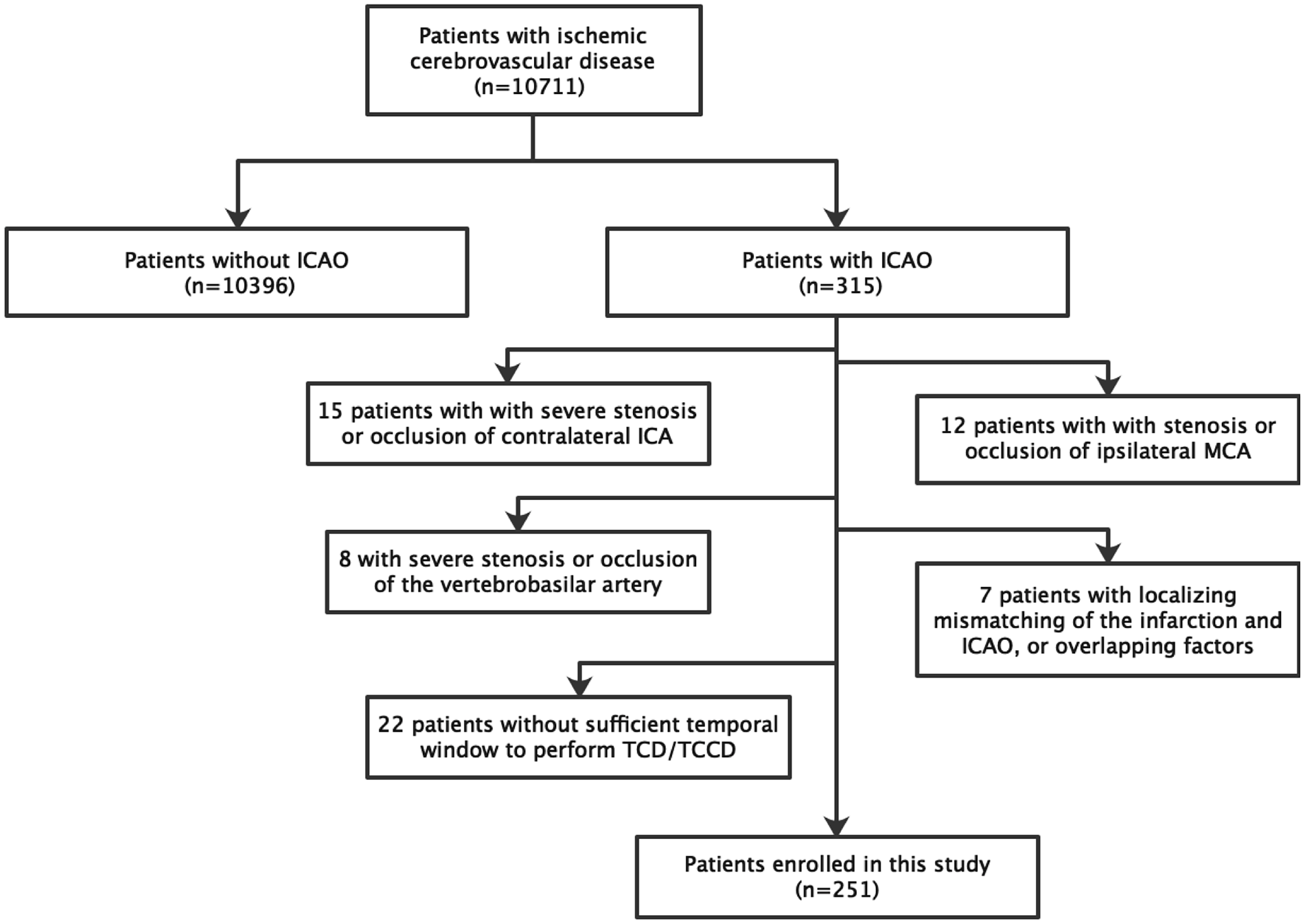
Flow chart of the study.
Among the 251 patients, 216 were men (86.1%) and 35 were women (13.9%), with ages ranging from 40 to 87 years (mean 64.2 ± 10.3 years) (Table 1). Based on radiological profiles, 114 and 137 patients were diagnosed with TIA and new-onset cerebral infarction, respectively. The rates of ACoA collateral, IECCA, PCoA, and noncollateral were 55.8%, 58.6%, 49.8%, and 9.6%, respectively.
Demographic and clinical characteristics of patients.
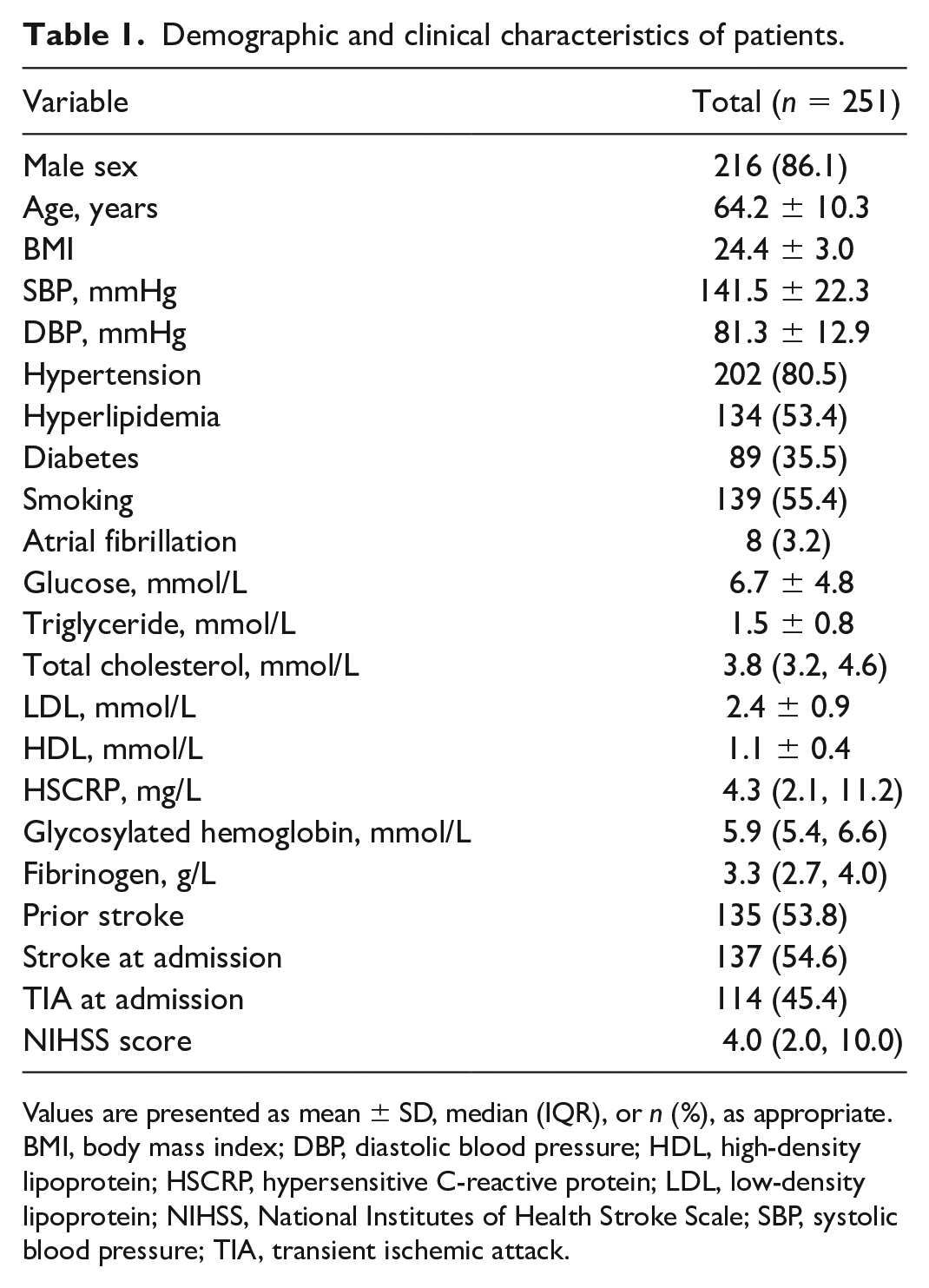
Values are presented as mean ± SD, median (IQR), or n (%), as appropriate.
BMI, body mass index; DBP, diastolic blood pressure; HDL, high-density lipoprotein; HSCRP, hypersensitive C-reactive protein; LDL, low-density lipoprotein; NIHSS, National Institutes of Health Stroke Scale; SBP, systolic blood pressure; TIA, transient ischemic attack.
PSVMCA in patients with different collateral circulation types
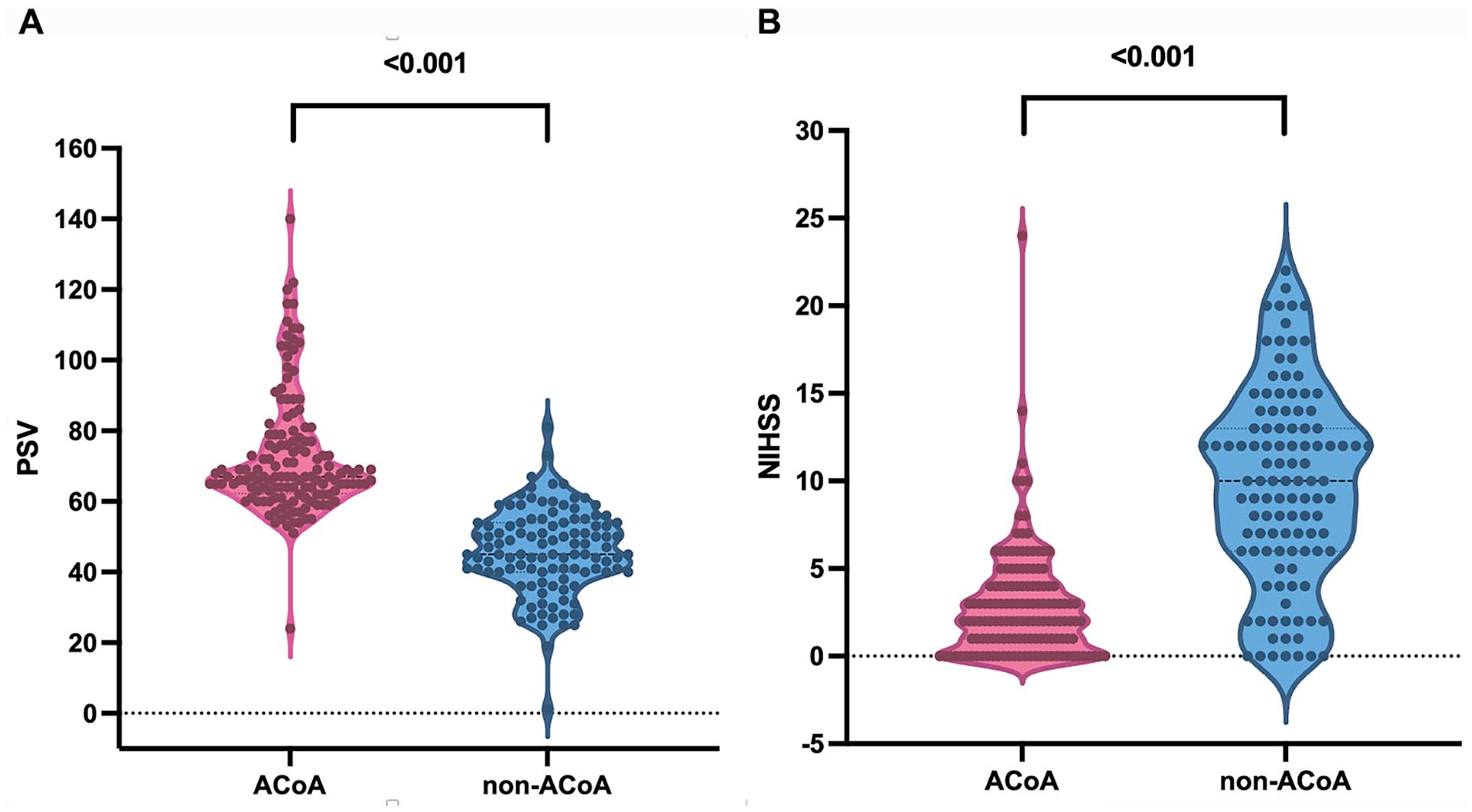
Intergroup one-way analysis of variance revealed significant differences in PSVMCA among the different collateral circulation types (F = 34.954, p < 0.001; Table 2, Figure 4A). The four groups with an ACoA collateral pathway had significantly higher PSVMCA levels than those without ACoA collaterals (p < 0.001). The noncollateral and IECCA groups had significantly lower PSVMCA values than the other groups (p < 0.001); however, these groups were statistically similar (p > 0.05). The PSVMCA values of PCoA and PCoA + IECCA groups were comparable (p > 0.05). The PSVMCA values of these two groups were significantly lower than those of the four groups with open ACoA yet were significantly higher than those of the noncollateral and IECCA groups (p < 0.001).
PSVMCA and NIHSS scores in patient groups with different types of collateral circulation.
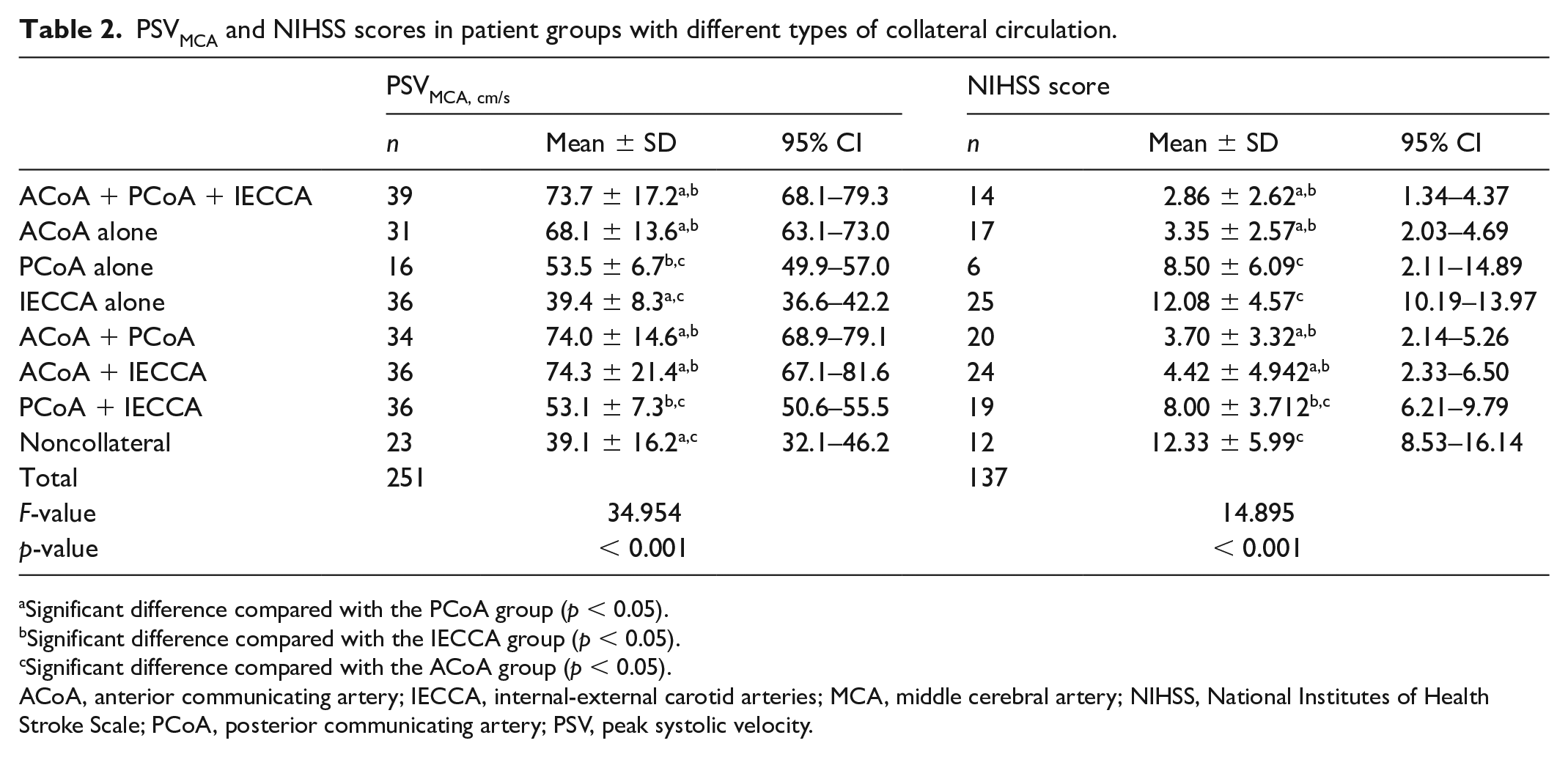
Significant difference compared with the PCoA group (p < 0.05).
Significant difference compared with the IECCA group (p < 0.05).
Significant difference compared with the ACoA group (p < 0.05).
ACoA, anterior communicating artery; IECCA, internal-external carotid arteries; MCA, middle cerebral artery; NIHSS, National Institutes of Health Stroke Scale; PCoA, posterior communicating artery; PSV, peak systolic velocity.
Distribution between
NIHSS scores in patients with different collateral circulation types
Intergroup one-way analysis of variance revealed significant differences in the NIHSS scores among the groups (F = 14.895, p < 0.001; Table 2, Figure 4B). Comparisons between paired groups showed that the four groups with an ACoA collateral pathway had significantly lower NIHSS scores than those without ACoA collaterals (p < 0.05). The noncollateral group had the highest NIHSS scores. The NIHSS scores of the noncollateral, PCoA, and IECCA groups were significantly higher than those of the other groups (p < 0.05), whereas the scores of the three groups were comparable (p > 0.05).
Correlation analysis
Linear regression analysis indicated a significant negative correlation between individual PSVMCA and NIHSS scores (r = −0.566, p < 0.001), yielding a regression equation of y = −0.156x + 15.388, where y is the NIHSS score and x is PSVMCA. After dividing the patients into two groups depending on whether ACoA existed, the correlation between PSVMCA and NIHSS score still showed a significant difference in the ACoA un-existing group (r = −0.504, p < 0.001); the regression equation was y = −0.231x + 20.321. Interestingly, there was no statistical difference between PSVMCA and NIHSS scores in the existing ACoA group (p = 0.240) (Figure 5). Furthermore, we analyzed the distribution of NIHSS scores between the ACoA and non-ACoA groups. The results showed that patients in the ACoA group had a concentrated distribution of NIHSS scores within 2 (1, 4) and patients without ACoA had a discrete distribution trend of NIHSS scores within 10 (6, 13). There was a significant difference in the NIHSS scores between ACoA and non-ACoA groups (p < 0.001).
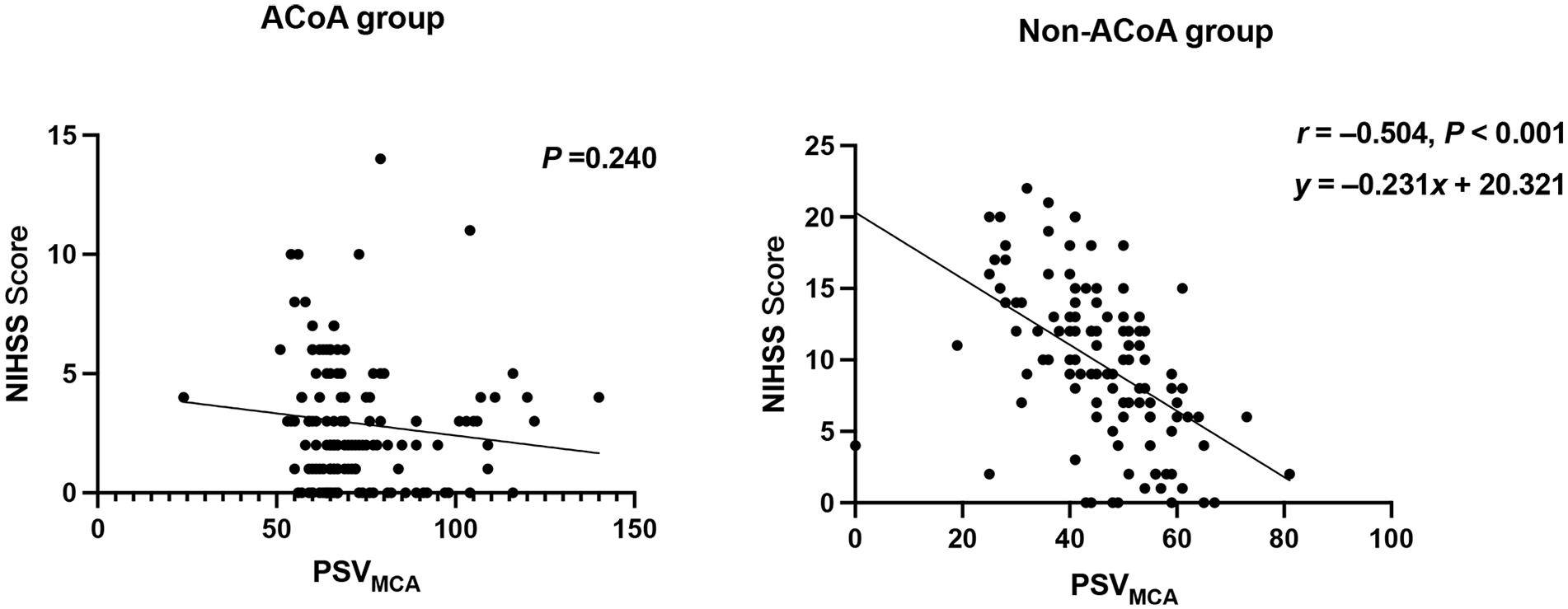
Correlation analysis between NIHSS score and PSVMCA in the ACoA and non-ACoA groups.
Discussion
To date, ischemic stroke is a leading cause of morbidity and mortality worldwide 12 and ICA occlusion is an important reason for ischemic stroke. The main causes of occlusion were atherosclerosis (55–65%), cardioembolic (19–22%), dissection (9–14%), and other uncommon causes (4%).13,14 The outcome of ICA occlusion is considerably influenced by the cerebral hemodynamic compensatory adaptation of intracranial collateral circulation. Multiple imaging modalities such as CTA7,15 MRA, DSA, and ultrasonography have been used for evaluating intracranial collateral circulation in ICA occlusion.16–18 The circle of Willis, which includes the ACoA and PCoA, respectively, are the primary collaterals that can rapidly compensate for decreased cerebral perfusion pressure. In cases with poor communicating artery collateral establishment, IECCA can also compensate for occluded cerebral blood flow. 19 Leptomeningeal collaterals supply blood to the cerebral cortex. 5
In the current study, we used ultrasonographic methods to evaluate the patency of intracranial collateral circulation and quantitatively assess the flow velocity of MCA. Patients with ACoA collaterals had significantly higher PSVMCA and significantly lower NIHSS scores than those without ACoA collaterals. Patients without any notable collaterals and those with only IECCA had the lowest PSVMCA and highest NIHSS scores. The PSVMCA and NIHSS scores were negatively correlated (r = −0.566, p < 0.001). Collateral circulation patency in unilateral ICA occlusion was closely associated with clinical symptoms. Patients with ACoA collaterals may have more favorable outcomes and significantly higher PSVMCA than those without ACoA collaterals. PSVMCA in patients with PCoA collaterals was significantly higher than that in the IECCA group. There was no significant difference in PSVMCA between IECCA and noncollateral groups. We speculated that in ICA occlusion, ACoA collaterals are best able to compensate followed by PCoA and IECCA collaterals. The IECCA collaterals supply the ipsilateral MCA via the ophthalmic artery, which is thin in diameter, long in route, and slow in flow velocity, which may explain the weak compensatory capacity of IECCA collaterals compared with the collateral vessels via the circle of Willis.
In a previous study by Connolly et al., 20 the activation of primary and secondary collaterals in 113 patients with symptomatic ICA occlusion was assessed using TCD, which showed that ACoA collaterals were most frequently activated (81%) followed by IECCA (63%) and PCoA (53%). From these results, we can see that the discrepancy between our study and Connolly’s was mostly focused on the ACoA collateral percentage (55.8% vs 81%, respectively). In their study, the examinations were performed using TCCD and the diagnostic criterion was retrograde flow in the ipsilateral A1 segment of ACA, which may be confused with the contralateral ACA flow. Furthermore, previous studies showed that the incidence rate of ACoA was 47.7–67.7%,5,7,21 which is similar to our findings. Researchers have found that approximately 70% of blood flow in the anterior circulation (ICA system) supplies to the MCA. 13 Ishikawa et al. 22 indicated that the flow velocity of MCA decreased in 86% of all symptomatic patients with ICA occlusion and the NIHSS scores significantly improved in patients with ICA occlusion and increased flow velocity in the MCA. 23 In the present study, we observed a significant negative correlation between the individual PSVMCA and NIHSS scores. This suggests that PSVMCA can reflect compensatory collateral circulation in ICA occlusion and clinical severity is closely associated with ipsilateral PSVMCA. In patients with unilateral ICA occlusion, collateral vessels via the circle of Willis (i.e., the ACoA and PCoA), are important for hemodynamic compensation by complementing the PSVMCA. 4
Cheng et al. 15 performed a perfusion CT study involving 38 patients with symptomatic unilateral ICA occlusion and found that secondary collaterals (IECCA) were associated with lower cerebral blood volume and delayed time-to-peak compared with primary collaterals (ACoAs and PCoAs). This suggests that the establishment and functional status of intracranial collateral circulation are closely correlated with the severity of cerebral tissue injury and occurrence of ischemic stroke. Blood oxygen level-dependent cerebrovascular reactivity (BOLD-CVR) MRI measures change in the T2 of blood and tissue water secondary to decreases in the amount of venous capillary and venule deoxyhemoglobin (dHb). Specifically, in cases of BOLD-CVR measurements, a vasodilatory challenge increases cerebral blood flow and cerebral blood volume in the brain parenchyma while inducing a much smaller change in metabolism. As such, information regarding CVR can be inferred from the dHb reductions in and around veins. 24 In a previous study using TCD and BOLD-CVR MRI, 5 ipsilateral leptomeningeal collateral activation was the sole collateral pathway associated with severely impaired BOLD-CVR in patients with symptomatic unilateral ICA occlusion. An interesting pattern has been demonstrated – patients with ICA occlusion and activation of only the primary collateral pathways presented with preserved BOLD-CVR. In the present study, we observed an interesting phenomenon: PSVMCA was correlated with the NIHSS score in patients without ACoA but had no significant correlation in patients with ACoA. This might be because in the ACoA group, the NIHSS score was mostly concentrated in the range of 2–4 regardless of the PSVMCA. This result demonstrates that collateral ACoA is significantly associated with favorable neurological function in patients with ICA occlusion.
A previous study on the correlation between collateral circulation and clinical severity in unilateral ICA occlusion reported that the presence of collateral flow via the circle of Willis is associated with a low prevalence of border zone infarcts. 25 In the current study, we found that the NIHSS scores of groups with ACoA collateral pathways were significantly lower than those of the groups without ACoA collaterals. This indicates that the compensatory capacity of ACoA collaterals may be better than that of PCoA collaterals. The ACoA collaterals may be the most important contributors to clinical improvement in symptomatic unilateral ICA occlusion. 15 Additionally, we noted no significant difference in NIHSS scores between the IECCA and noncollateral groups, suggesting that IECCA collaterals have a slight compensative capacity.
However, the compensatory effects of ophthalmic arterial collaterals remain controversial. Previous studies have reported that the ophthalmic artery collaterals may increase blood flow and protect against cerebral hemodynamic compromise. 10 Other studies have hypothesized that secondary collaterals are associated with more severe cerebral hemodynamic impairment and their presence may be a marker of inadequate primary pathways.26,27 Our study supports the idea that secondary collaterals such as ophthalmic artery collaterals may be established following blood redistribution in patients with unilateral ICA occlusion, which may limit the compensatory capacity. However, the definitive effects of secondary collaterals on the clinical prognosis of symptomatic unilateral ICA occlusion require a much longer observation period.
Recently, noninvasive optimal vessel analysis quantitative magnetic resonance angiography (NOVA-qMRA) has been increasingly used to measure blood flow in cerebral vessels in milliliters per minute, allowing reliable quantification of cerebral collateral pathways in cases of large-vessel occlusion. This quantification of the collateral vessel status can be used as a decision-making tool for revascularization. 28 The NOVA-facilitated blood flow measurements were similar to those obtained using an invasive sonographic flow probe (R2 = 0.9869) in a phantom study, and the maximum error between the two was 7.9%. 29 Since patients with secondary collaterals and no collaterals suffered from a low velocity of MCA, the severity of cerebral tissue injury as well as the occurrence of ischemic stroke may deteriorate, and it is necessary to accept surgical revascularization to improve cerebral flow. In contrast, patients with primary collaterals have a lower risk of intraoperative ischemia. Therefore, collaterals play an important role in surgical revascularization in patients with ICA occlusion.
This study has some limitations. First, as the P2 of PCA was not measured, leptomeningeal collaterals were not assessed in this study. In future studies, we plan to consider this aspect. Second, the clinical evaluations using NIHSS in the current study were performed within 24 h after admission due to acute cerebral ischemia whereas the compensatory effects of IECCA collaterals formed in the late stage still require dynamic observations. Third, in the present study, we did not recruit patients without ICA occlusion as a control group. Finally, the correlation between collateral vessel status, PSV values, and recurrent stroke was not observed in the present study, and further research is needed to explore the relationship between PSV values and recurrent stroke.
Conclusion
Collateral circulation patency in unilateral ICA occlusion is closely correlated with clinical severity, and evaluation of collateral circulation using ultrasonography can be an effective method for clinical assessment and prognostic prediction. Collaterals eliminate poor blood flow, which is a mediator of worse (higher) severity scores.
Footnotes
Acknowledgements
The authors thank the staff in the Department of Vascular Ultrasonography, Xuanwu Hospital for their valuable help and all the researchers at other hospitals in this multicenter study for having provided meaningful data.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This study was funded by the Beijing Tongzhou District Science and Technology Plan Project, (KJ2023SS021) and Natural Science Foundation of Hebei Province (H2024112005).
Data availability statement
The raw data to support this article may be shared upon reasonable request with the corresponding author.
