Abstract

Keywords
Among patients hospitalized with an acute medical illness, the risk of venous thromboembolism (VTE) may persist for weeks to months after hospital discharge. 1 It is estimated that 75% of VTE events occurred after index hospitalization, with a median time-to-event of 19.5 days. 2 However, the benefit of VTE reduction associated with extended anticoagulation may be offset by the harm of hemorrhagic complications. In view of limited data and unclear net benefit, current guidelines up to 2012 recommend against extended-duration thromboprophylaxis beyond the period of immobilization or hospitalization.3,4
We systematically searched the literature on PubMed and extracted published data from randomized controlled trials comparing the efficacy and safety of extended- versus shorter-duration anticoagulation for VTE prevention in acutely ill hospitalized patients. A total of five studies were identified – three were released after the publication of current guidelines. While the exact eligibility criteria were different, these studies generally enrolled adult patients who were hospitalized for an acute medical illness (such as acute heart failure, acute respiratory failure, acute infectious or inflammatory disease, or acute ischemic stroke) and had additional risk factors for VTE (such as advanced age, reduced mobility, history of VTE, or history of cancer). Compared with a shorter-duration of enoxaparin for 6–14 days, extended-duration treatments include: (1) EXCLAIM (ClinicalTrials.gov identifier: NCT00077753): enoxaparin 40 mg daily for 6–14 days plus 24–32 days; 5 (2) ADOPT (NCT00457002): apixaban 2.5 mg twice daily for 30 days; 6 (3) MAGELLAN (NCT00571649): rivaroxaban 10 mg daily for 31–39 days; 7 (4) APEX (NCT01583218): betrixaban 80 mg daily for 35–42 days; 8 and (5) MARINER (NCT02111564): rivaroxaban 10 mg daily for 45 days after hospital discharge.9,10 The efficacy (symptomatic VTE events including symptomatic deep vein thrombosis (DVT), non-fatal pulmonary embolism (PE), and VTE-related mortality) and safety (major bleeding and clinically relevant non-major bleeding) were analyzed at the end of extended therapy. Risk difference was computed with the inverse variance-weighted method by fitting a fixed-effect model or random-effect model in the presence of heterogeneity.
Pooled results are illustrated in Figure 1. With respect to the symptomatic VTE, extended therapy reduced the risk by 0.46% (95% CI: 0.27% to 0.65%; p < 0.00001). Significant risk reduction was observed in an extended course of enoxaparin (0.75% [0.34% to 1.17%]; p = 0.0004), apixaban (0.39% [0.01% to 0.78%]; p = 0.0432), or betrixaban (0.51% [0.02% to 1.00%]; p = 0.0426). With respect to VTE components, extended therapy reduced the risk of symptomatic DVT by 0.21% (95% CI: 0.10% to 0.31%; p < 0.0001) or non-fatal PE by 0.13% (95% CI: 0.04% to 0.23%; p = 0.005) but not VTE-related death (p = 0.20). With respect to safety, extended therapy increased the risk of major bleeding by 0.31% (95% CI: 0.11% to 0.52%; p = 0.003), with moderate-to-high heterogeneity (Higgins’s I2 statistic = 59%; Cochran’s Q test p = 0.04). A greater risk of major bleeding was observed in an extended course of enoxaparin (0.51% [0.12% to 0.89%]; p = 0.0106), apixaban (0.28% [0.00% to 0.57%]; p = 0.0469), and rivaroxaban (0.70% [0.33% to 1.07%]; p = 0.0002) but not in betrixaban (p = 0.55) or post-discharge rivaroxaban (p = 0.12). Extended therapy was also associated with a higher rate of clinically relevant non-major bleeding (1.35% [0.62% to 2.08%]; p = 0.0003), with high heterogeneity (Higgins’s I2 statistic = 84%; Cochran’s Q test p < 0.0001).
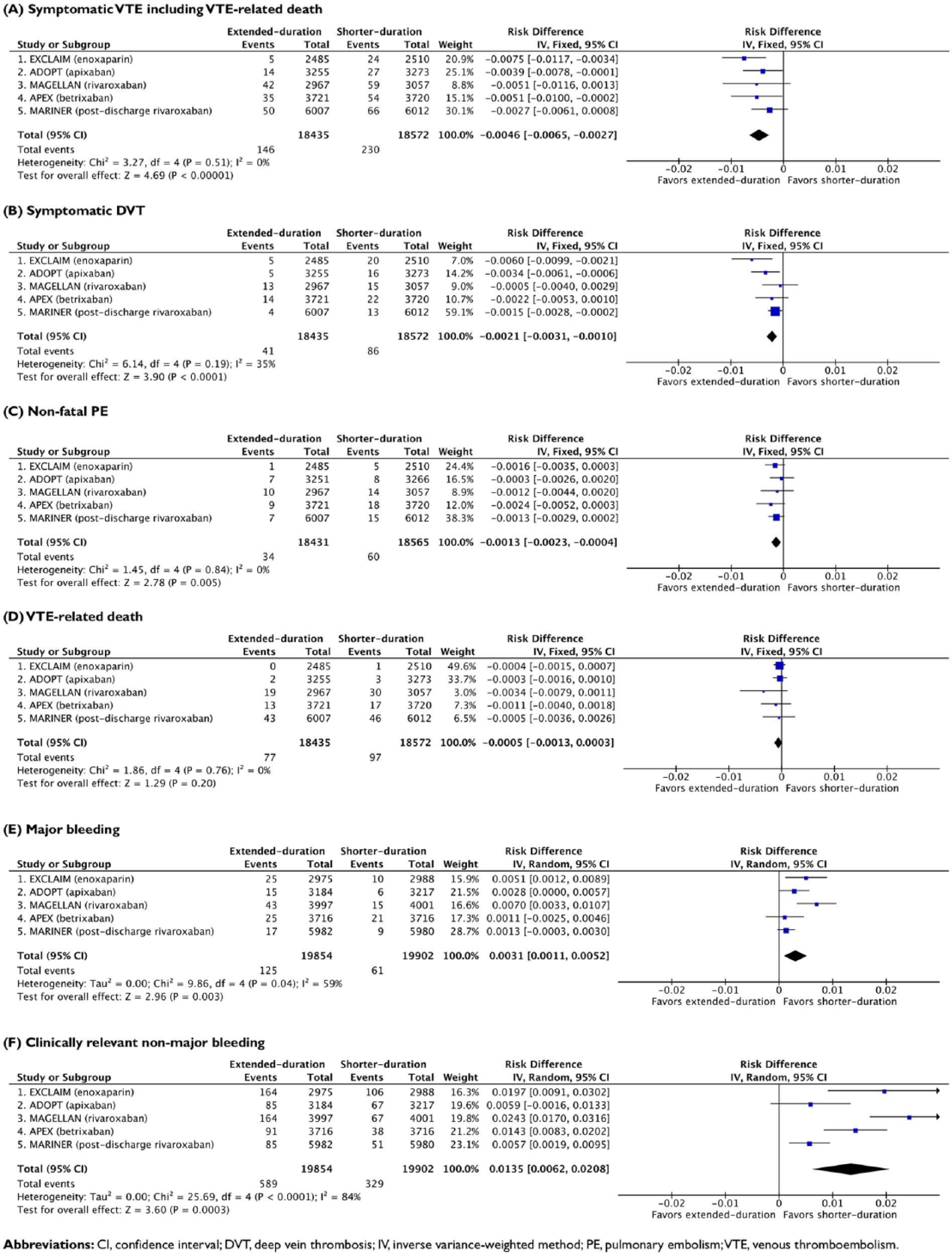
Efficacy and safety of extended- versus shorter-duration prophylaxis.
Taken together, extended anticoagulation decreased the risk of symptomatic VTE at a cost of increased major bleeding when compared with shorter-duration enoxaparin. The effect size of thromboembolic prevention (0.46%; number-needed-to-treat = 218) was comparable to excess bleeding (0.31%; number-needed-to-harm = 323), yielding a modest net clinical benefit of 0.15%. Considering the additional burden and cost of extended anticoagulation, results from this updated meta-analysis support in-hospital thromboprophylaxis when symptomatic VTE is weighed against major bleeding. Although the study participants were at increased risk of VTE according to inclusion criteria, none of the studies utilized a standardized approach for stratifying bleeding risk at baseline. Substantial heterogeneity across studies was observed in safety end points but not efficacy end points. This analysis points to the unmet demand for a risk assessment model that collectively evaluates the benefit–harm of thromboprophylaxis. A joint VTE–bleeding score comprised of predictors with discriminatory ability for net clinical benefit will assist decisions on thromboprophylaxis.
Footnotes
Declaration of conflicting interests
Dr. Chi is supported by a grant from Portola Pharmaceuticals, Bayer, and Janssen Scientific Affairs, paid to Beth Israel Deaconess Medical Center, Harvard Medical School. All other authors declare no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
