Abstract

Keywords
Claudication is present in up to 10% of patients with lower-extremity peripheral artery disease (PAD) and is associated with significant impairment in patient function and quality of life. 1 Endovascular revascularization is a recommended treatment for patients with claudication refractory to optimal medical therapy or based on patient preference. 2 However, in some patients, improvements after treatment may be short-lived, yet others may experience sustained improvement in symptoms over time. The baseline characteristics of patients who can maintain an improved course over time remain unclear. We hypothesized that low baseline health status will be associated with sustained improvement in reported walking impairment after intervention.
We analyzed data of patients from three Stellarex (Phillips, Maple Grove, MN, USA) clinical studies who underwent initial endovascular revascularization between January 1, 2013 and December 31, 2019. The ILLUMENATE Pivotal trial (Pivotal Trial of a Novel Paclitaxel-coated Percutaneous Angioplasty Balloon) was a prospective, multicenter, randomized, single-blinded trial that included patients treated with uncoated percutaneous transluminal angioplasty (PTA) (ClinicalTrials.gov Identifier: NCT01858428). 3 The ILLUMENATE Global (Global Study of a Drug-coated Balloon to Treat Obstructive SFA and/or Popliteal Lesions) and the in-stent restenosis (ISR) cohort were international, longitudinal, multicenter, single-arm studies designed to assess the safety and performance of Stellarex in patients with de novo or restenotic lesions in the superficial femoral (SFA) and/or popliteal arteries (ClinicalTrials.gov Identifier: NCT01927068). 4 Detailed inclusion and exclusion criteria for these studies have been previously reported.3,4 Briefly, adults with Rutherford classification < 4 who had severe de novo or restenotic femoral and/or popliteal stenosis were included. Exclusion criteria included patients with a contraindication to dual-antiplatelet therapy, prior bypass surgery in the target limb, and intolerance to the study drug. All studies were approved by independent institutional review boards or ethics committees and all patients provided written informed consent.
The main outcome was sustained improvement in the Walking Impairment Questionnaire (WIQ) overall score from baseline to 6 months and from baseline to 1 year. The WIQ is a PAD-specific measure of self-reported walking limitation (score range: 0–100; worst–best) that includes a speed, distance, and stair-climbing subscale. Responses are weighted and summarized into an overall score. 5 A clinically meaningful improvement was defined as a change in score from baseline of > 11 at 6 months and at 1 year. 6 Only those with complete WIQ assessments were included. To assess baseline characteristics associated with sustained improvement, we performed complete case, hierarchical logistic regression (site as random variable) with stepwise backward elimination to minimize the Akaike information criterion (AIC). 7 Candidate variables were selected based on clinical judgment and prior associations with health status and limb outcomes. These included age, sex, race, country (US vs Non-US), baseline WIQ, ankle–brachial index (ABI), body mass index, hypertension, diabetes, hyperlipidemia, heart failure, lung disease, liver disease, coronary artery disease, stroke, smoking status, antiplatelet, anticoagulant, and statin use, Rutherford classification, lesion characteristics (Trans-Atlantic Inter-Society Consensus [TASC] II category, eccentric lesion, lesion length, lesion location), and prior peripheral intervention.
Among 425 patients, 307 (72%) demonstrated improvement at 6 months and 256 (60%) had sustained improvement in WIQ at 1 year. Patients with sustained improvement were younger, had lower WIQ and ABI, were more likely Non-US, had fewer comorbidities, and had fewer SFA lesions than those in other categories (Supplemental Table 1). In the multivariable model, statin use (odds ratio, 95% CI) (3.81, 1.17–12.4, p = 0.026) was associated with higher odds of sustained improvement at 1 year (Table 1). Higher baseline WIQ (0.98, 097–0.99, p < 0.001), undergoing treatment in the US (0.12, 0.04–0.39, p < 0.001), history of hypertension (0.40, 0.18–0.87, p = 0.022), and SFA location (0.29, 0.10–0.81, p = 0.018) were associated with lower odds of sustained improvement at 1 year.
Hierarchical multivariable logistic regression (site as random variable): Baseline factors associated with sustained improvement (WIQ change from baseline > 11 at 6 months and > 11 at 1 year) after endovascular intervention.
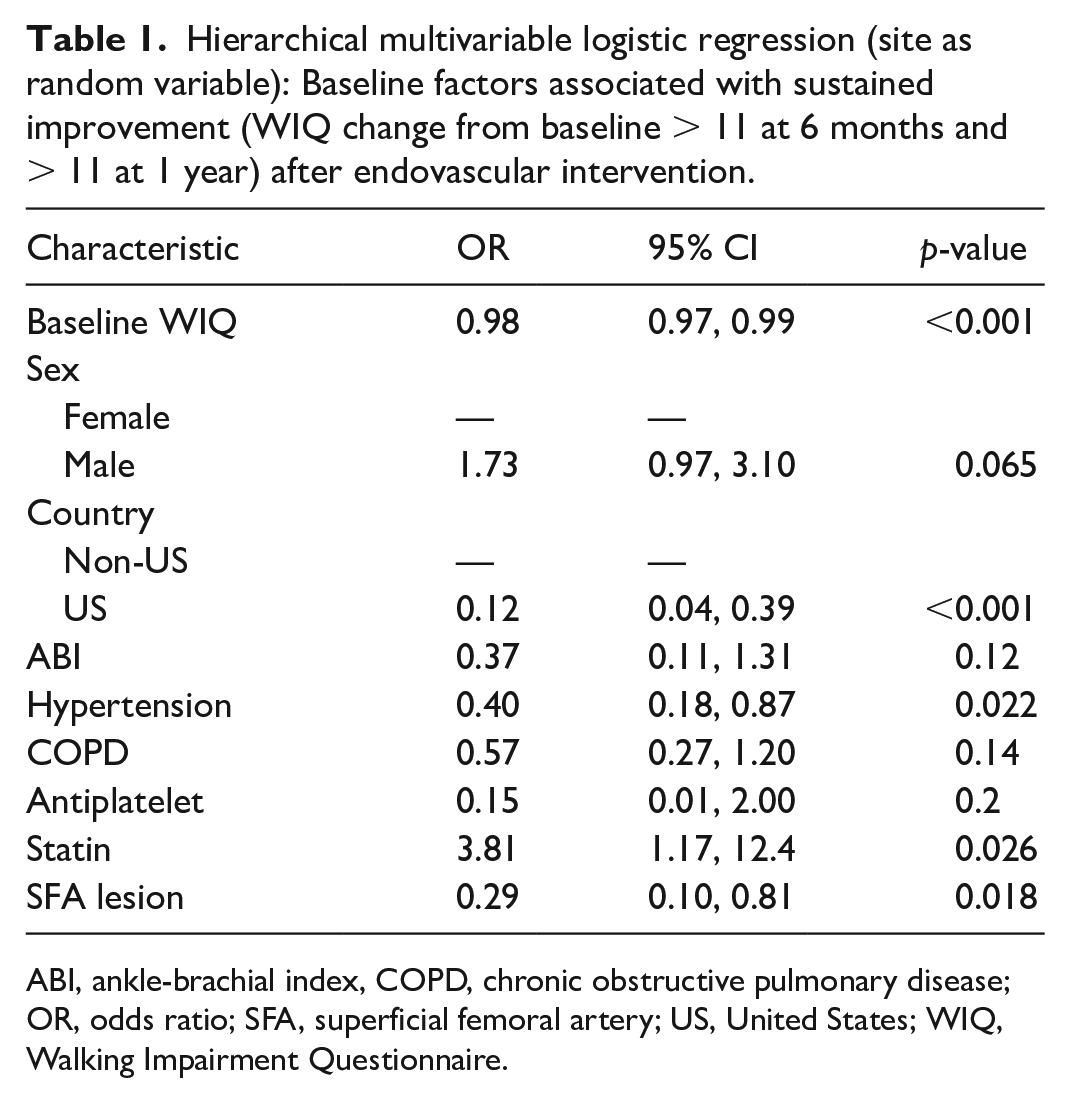
ABI, ankle-brachial index, COPD, chronic obstructive pulmonary disease; OR, odds ratio; SFA, superficial femoral artery; US, United States; WIQ, Walking Impairment Questionnaire.
We identified baseline characteristics associated with sustained improvement in WIQ after peripheral vascular intervention (PVI) for SFA/popliteal lesions in claudicants. We noted an association of statin use with a greater likelihood of sustained improvement. Statins are associated with lower risk of cardiovascular and limb events in patients with PAD, despite their low rates of use in clinical practice. 8 We add to the literature by demonstrating that statin use is also associated with sustained WIQ improvement over time. Hypertension is common in PAD and leads to adverse vascular remodeling, which may negatively impact the durability of endovascular intervention. 9 Our finding of a lower risk of sustained improvement in the US may reflect the higher comorbidity profile and overall lower quality of health in patients from the US in this cohort. SFA lesions were longer than popliteal (86 vs 54 mm, p < 0.01). Thus, the association of SFA lesions with lower odds of sustained improvement likely reflects the effect of lesion length on vessel patency after intervention. 10 Study limitations include use of one type of drug-coated technology, short follow up, and the occurrence of more recent advances in endovascular technology since data collection. Further analysis in larger patient populations is needed to further characterize patient trajectories after PVI in context of a larger societal backdrop and social determinants of health/health policy.
Supplemental Material
sj-pdf-1-vmj-10.1177_1358863X241261369 – Supplemental material for Factors associated with sustained improvement after peripheral vascular intervention in patients with claudication
Supplemental material, sj-pdf-1-vmj-10.1177_1358863X241261369 for Factors associated with sustained improvement after peripheral vascular intervention in patients with claudication by Poghni A Peri-Okonny, Gaëlle Romain, Vikash Rambhujun, Santiago Callegari, Aseem Vashist, Jacob Cleman, Kim G Smolderen and Carlos Mena-Hurtado in Vascular Medicine
Footnotes
Declaration of conflicting interests
Dr Mena-Hurtado reports unrestricted research grants from Philips, Shockwave, Abbott, and Merck; he is a consultant for Abbott Vascular and Cook. Dr Smolderen reports unrestricted research grants from Philips, Shockwave, Abbott, and Merck; she is a consultant for Cook and Happify. The other authors report no conflicts of interest.
Funding
The ILLUMENATE studies were sponsored by the Spectranetics Corp, Maple Grove, MN, USA, with no involvement in the study design or manuscript preparation.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
