Abstract
The role of cilostazol after endovascular therapy (EVT) of peripheral artery disease (PAD) remains unclear. We conducted a meta-analysis for all studies reporting the outcomes of cilostazol after EVT of PAD from January 2000 through November 2018 with the outcomes of interest including primary patency, major adverse limb events (MALE), target lesion revascularization (TLR), and major amputation. We included eight studies (three randomized controlled trials (RCTs) and five observational studies) with a total of 3846 patients (4713 lesions). During a mean follow-up duration of 12.5 ± 5 months, the use of cilostazol was associated with higher primary patency (OR 2.28, 95% CI (1.77, 2.94), p < 0.001, I2 = 24%), lower risk of TLR (OR 0.37, 95% CI (0.26, 0.52), p < 0.001, I2 = 0%), and lower risk of major amputation (OR 0.15, 95% CI (0.04, 0.62), p = 0.008, I2 = 0%). The use of cilostazol in RCTs was associated with significantly higher odds of primary patency compared with observational studies (OR 3.37 vs 2.28, p-interaction = 0.03). After further subgroup analysis, cilostazol remained associated with higher primary patency regardless of the use of anticoagulants (warfarin) (p-interaction = 0.49). We conclude that the use of cilostazol after EVT of femoropopliteal and iliac lesions is associated with improved primary patency and lower risk of major amputation and TLR. The favorable impact of cilostazol is independent of the use of warfarin. PROSPERO identifier: CRD42018092715.
Keywords
Introduction
Peripheral artery disease (PAD) is considered a significant cause of functional impairment and limb loss.1,2 Revascularization is recommended for severe stenosis; it helps relieve claudication symptoms and prevent amputation.3,4 However, the rates of restenosis after endovascular therapy (EVT) remain high. The primary patency for balloon angioplasty has been reported to be as low as 40% in the first year.5–8 While the rates of primary patency have improved after the introduction of nitinol stents,9,10 drug-eluting stents,11–13 and drug-coated balloons, 14 restenosis after EVT remains associated with worse clinical outcomes including critical limb ischemia (CLI), limb loss, and mortality.2,6,15–17
Cilostazol is a phosphodiesterase inhibitor with antiplatelet, vasodilatory, and antiproliferative effects.18–21 Cilostazol is reported to prevent restenosis after coronary22–25 and carotid stenting. 26 The use of cilostazol after EVT for PAD has been sporadically studied.27–35 However, the role of cilostazol in preventing restenosis is still debatable. We conducted a comprehensive meta-analysis to examine the outcomes of cilostazol after peripheral endovascular interventions.
Methods
Literature search
The current study was conducted and reported according to the proposal for conducting and reporting Meta-analyses of observational studies (Moose) 36 and Preferred Reporting Items for Systematic Reviews and Meta-Analyses guidelines (PRISMA) 37 ; and is registered with the International Prospective Register for Systematic Reviews (PROSPERO: CRD42018092715). We performed a systematic computerized search through MEDLINE, EMBASE, and Cochrane databases from January 2000 to November 2018 using the following search terms separately and in combination; ‘cilostazol’, ‘Pletal’, ‘peripheral endovascular interventions’, ‘endovascular therapy’, and ‘peripheral vascular disease’. A similar search strategy was performed for abstracts of the major scientific sessions (American College of Cardiology (ACC), American Heart Association (AHA), European Society of Cardiology (ESC), etc.) through November 2018. We screened the references list of the retrieved articles for relevant studies not found through the initial search. Our search was limited to the English language.
Study selection
We included the published studies (randomized controlled trials (RCTs) or observational studies) that compared the outcomes with cilostazol versus no cilostazol after peripheral endovascular interventions. If more than one study reported outcomes in the same cohort of patients, we only included the most recent or most comprehensive publication. We excluded the studies that primarily compared cilostazol to ticlopidine.
Data extraction and quality assessment
The data were extracted by two independent investigators (AM, BA) and confirmed by a third investigator (MS). Data included the baseline study characteristics, clinical and angiographic features of the included patients and lesions, and the outcomes of interest. Discrepancies were settled by consensus. The risk of bias of the included studies was assessed using the Newcastle-Ottawa Scale for observational studies 38 and the Cochrane risk assessment tool for RCTs. 39
Study outcomes
The clinical outcomes of the current study included primary patency defined as freedom from restenosis or revascularization at follow-up. The imaging method used was duplex ultrasound in two studies,29,35 duplex ultrasound or angiography in five studies,28,31–34 and angiography in one study. 27 Secondary outcomes included major adverse limb events (MALE), target lesion revascularization (TLR), and major amputation defined as surgical amputation above the ankle. The definitions of the outcomes according to each included study are illustrated in online Supplementary Table 1. Outcomes were reported at the longest follow-up time. The total number of lesions was used when primary patency and TLR were assessed.
Data synthesis and statistical analysis
Statistical analysis was conducted using Review Manager (RevMan) software (Version 5.3.5. Copenhagen: The Nordic Cochrane Centre, The Cochrane Collaboration, 2014). Descriptive analyses were conducted using frequencies for categorical variables and means with SD for continuous variables. Categorical variables were compared using Fisher’s exact test or chi-squared test, while continuous variables were compared using the two-sample t-test. Tests were two-tailed, and a p-value ⩽ 0.05 was considered statistically significant.
Odds ratios (ORs) with 95% CIs are presented as summary statistics. CIs were calculated at a 95% level for an overall estimates effect. The choice between a fixed or random-effects model was determined by evaluating the clinical, methodological, and statistical heterogeneity between the included studies as recommended by a scientific statement of the AHA. 40 Statistical heterogeneity across trials was assessed by I2 statistics, with I2 statistic values < 25%, 25–50%, and > 50% considered as low, moderate, and a high degree of heterogeneity, respectively. 41 We anticipated a high degree of clinical and methodological heterogeneity between the included studies; therefore, we utilized the DerSimonian and Laird random-effects method to calculate odds ratios (ORs). Follow-up periods, baseline characteristics, and event rates presented are weighted; these were calculated with the sample size being the weight. Potential publication bias was assessed using Egger’s test through funnel plot visual examination. 42 When applicable, we calculated the number needed to treat (NNT) using the OR according to the formula: NNT = [1–(PEER*(1–OR))] / [(1–PEER)*(PEER)*(1–OR)], where ‘PEER’ is the patient expected event rate. 43
A sensitivity analysis was performed using the following exclusion criteria: (1) excluding the study by Nanto et al. 32 as it constitutes the most significant weight in the analysis; (2) excluding the study by Miura et al. 35 that indirectly compared cilostazol to clopidogrel; and (3) excluding studies that used ticlopidine. The following subgroup analyses were performed: (1) observational studies versus RCTs; and (2) studies that reported the use of oral anticoagulants versus those that did not.
Results
Characteristics of the included studies and quality assessment
The study selection process is described in Figure 1. Eight studies (three RCTs29,31,35) and five observational studies27,28,32–34) with a total of 3846 patients (4713 lesions) met our inclusion criteria. The characteristics of the included studies are described in Table 1. Oral anticoagulant use (warfarin) was reported in three studies.27,28,33 Patients’ symptoms ranged from claudication to chronic limb ischemia, as shown in Table 1.
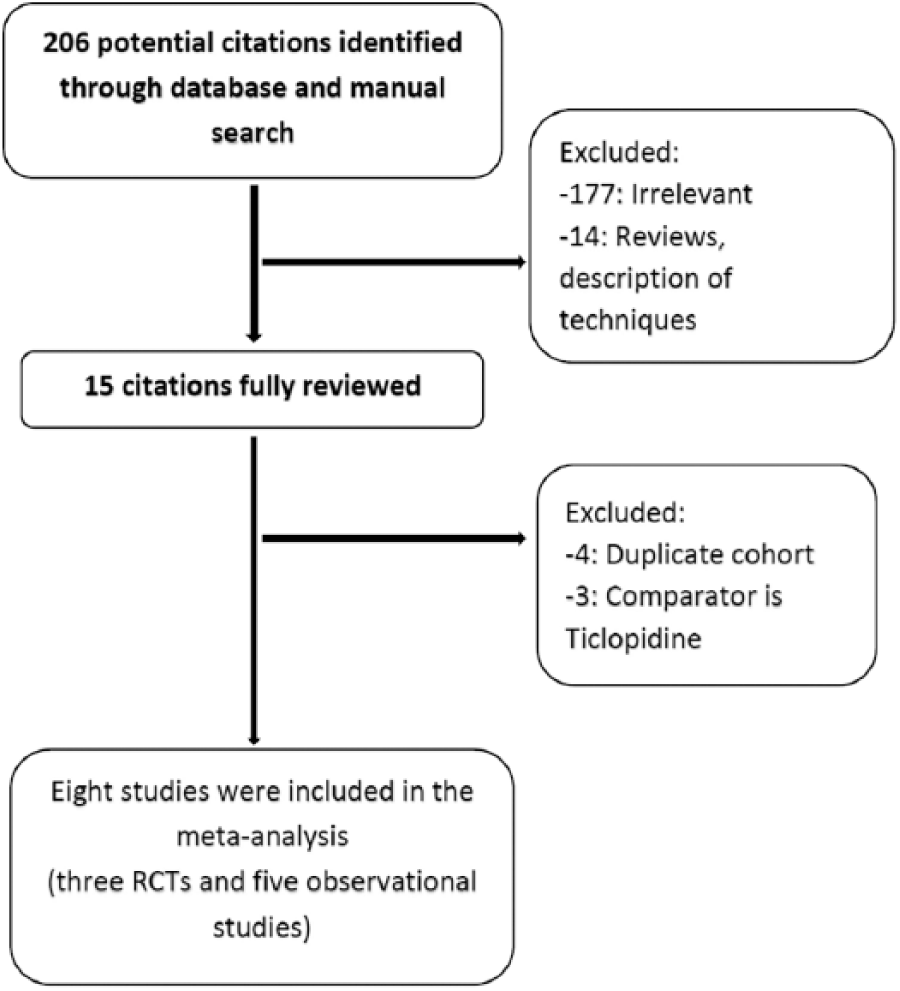
Flow diagram of the study selection process.
Baseline characteristics of the included studies.
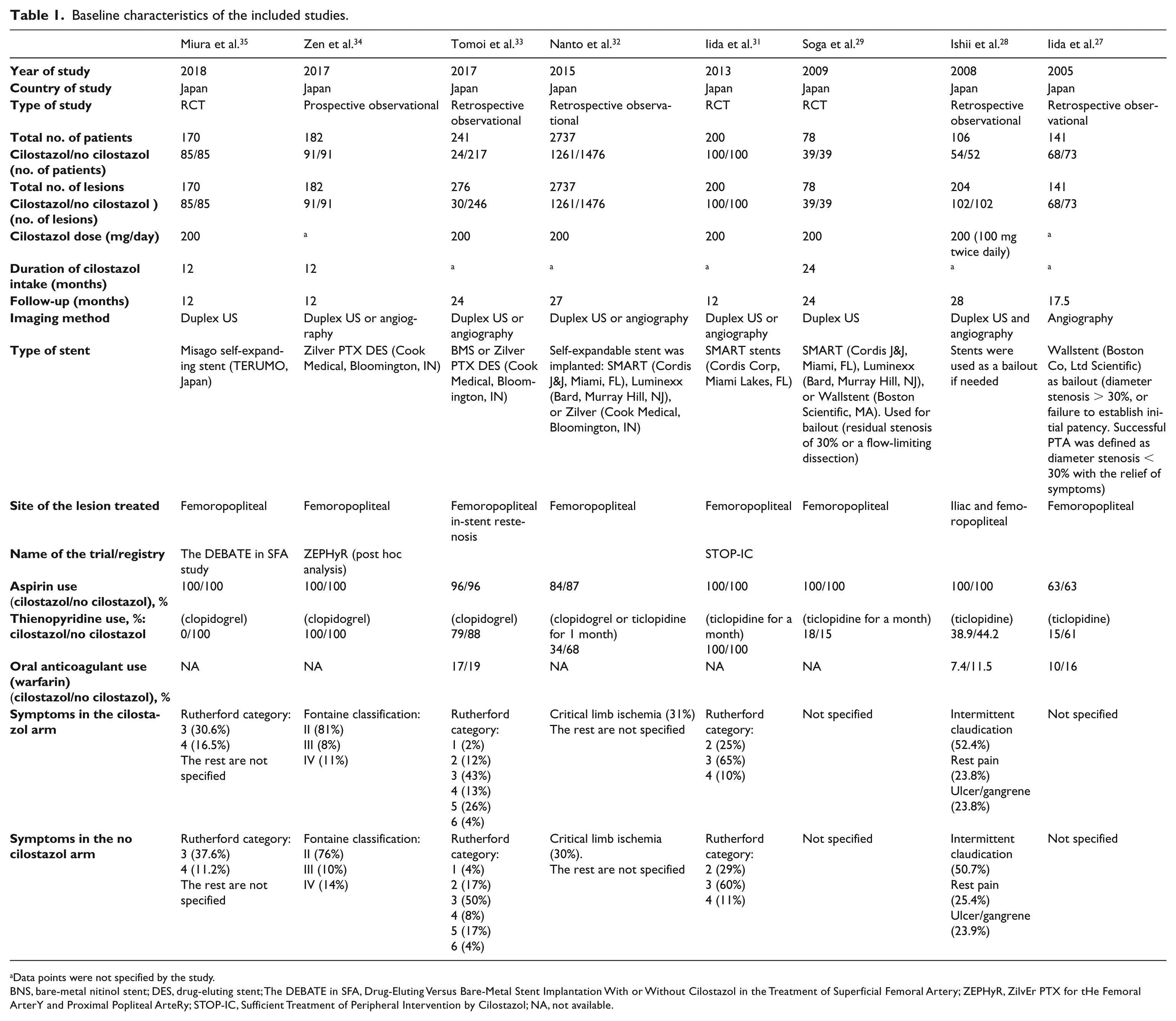
Data points were not specified by the study.
BNS, bare-metal nitinol stent; DES, drug-eluting stent; The DEBATE in SFA, Drug-Eluting Versus Bare-Metal Stent Implantation With or Without Cilostazol in the Treatment of Superficial Femoral Artery; ZEPHyR, ZilvEr PTX for tHe Femoral ArterY and Proximal Popliteal ArteRy; STOP-IC, Sufficient Treatment of Peripheral Intervention by Cilostazol; NA, not available.
The cilostazol arm included 1715 patients (2098 lesions), while the control arm included 2131 patients (2615 lesions). Antiplatelet therapy was used at the physician’s discretion in most of the observational studies. Follow-up for primary patency was completed in 63.7% of lesions and clinical follow-up was completed in 97% of patients. The weighted mean follow-up duration was 12.5 ± 5 months.
All studies met the inclusion criteria with no evidence of publication bias (online Supplementary Figure 1). Bias assessment as per the Newcastle-Ottawa Scale for observational studies and the Cochrane assessment tool for RCTs are illustrated in online Supplementary Tables 2 and 3.
Baseline characteristics of the included cohort
The baseline clinical and angiographic characteristics for cilostazol versus no cilostazol groups are summarized in Tables 2 and 3, respectively. The cilostazol group had a higher percentage of males (71.8% vs 68.2%, p = 0.02), and lower prevalence of chronic kidney disease (33.6% vs 62.1%, p < 0.001), patients on hemodialysis (25.3% vs 30.2%, p < 0.001), and coronary artery disease (43.1% vs 55.1%, p < 0.001) compared with the no cilostazol group. Patients who took cilostazol were prescribed less thienopyridines (37.5% vs 72.4%, p < 0.001) compared with patients who were not on cilostazol. Patients in the cilostazol group had a higher percentage of de novo lesions rather than in-stent restenosis (82.5% vs 50.6%, p < 0.001), as well as a lower percentage of poor distal below-knee run-off (25.1% vs 44.3%, p < 0.001). Patients who were prescribed cilostazol had more complex lesions (TASC C/D) (45.6% vs 37%, p < 0.001); however, they had a higher preprocedure ankle–brachial index (ABI) (0.64 ± 0.22 vs 0.59 ± 0.25, p < 0.001) compared with patients who were not prescribed cilostazol.
Baseline characteristics of the study patients.
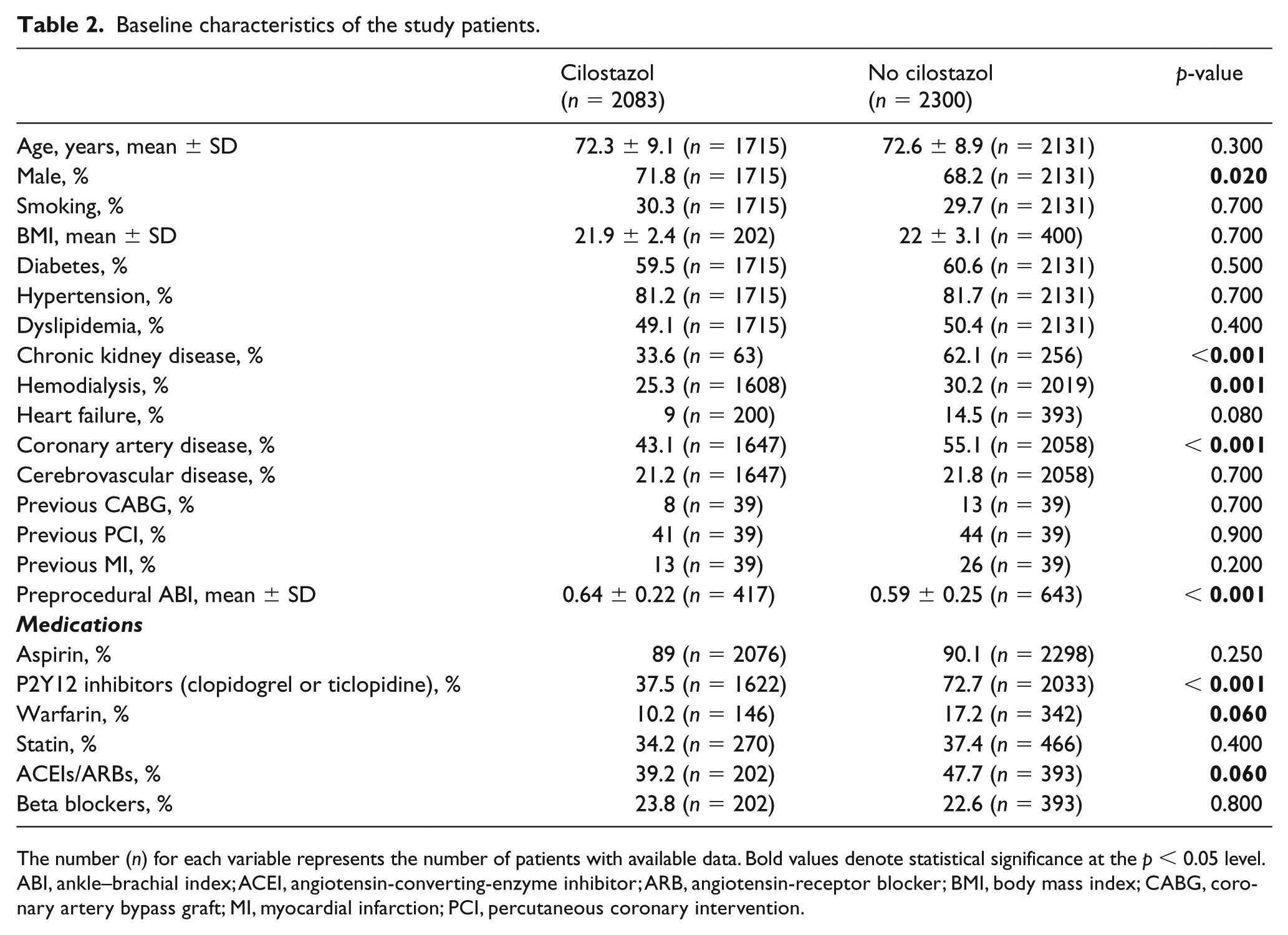
The number (n) for each variable represents the number of patients with available data. Bold values denote statistical significance at the p < 0.05 level.
ABI, ankle–brachial index; ACEI, angiotensin-converting-enzyme inhibitor; ARB, angiotensin-receptor blocker; BMI, body mass index; CABG, coronary artery bypass graft; MI, myocardial infarction; PCI, percutaneous coronary intervention.
Characteristics of the study lesions and procedural characteristics.
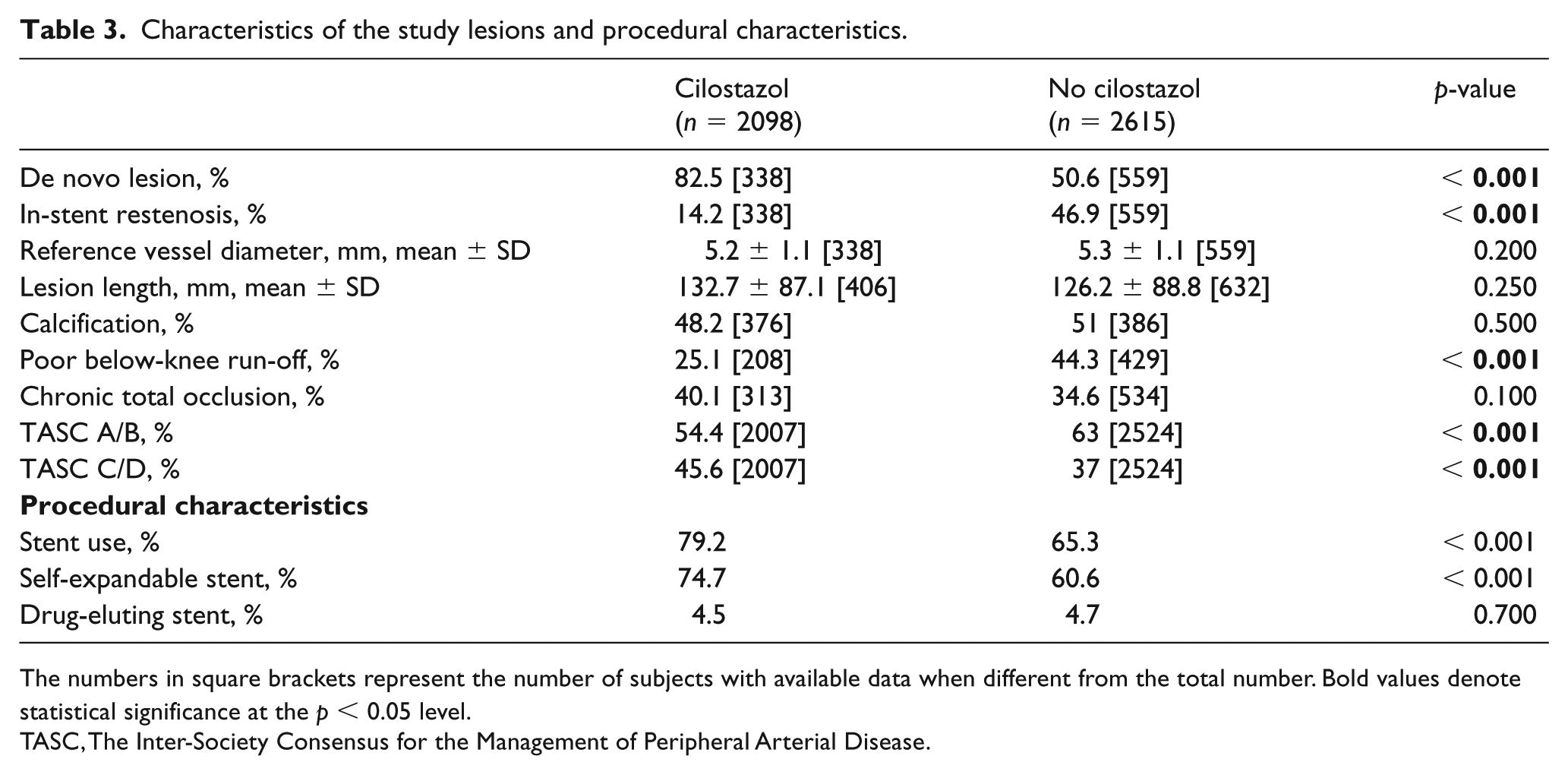
The numbers in square brackets represent the number of subjects with available data when different from the total number. Bold values denote statistical significance at the p < 0.05 level.
TASC, The Inter-Society Consensus for the Management of Peripheral Arterial Disease.
Outcomes
Primary patency
The summary of clinical and angiographic outcomes is illustrated in Figure 2. During a mean follow-up duration of 12.5 ± 5 months, the use of cilostazol was associated with higher primary patency (80.6% vs 66.6%, NNT = 6), (OR 2.28, 95% CI (1.77, 2.94), p < 0.001, I2 = 24%) (Figure 3). Sensitivity analyses demonstrated that the use of cilostazol remained associated with higher primary patency after excluding the study by Nanto et al. (OR 2.65, 95% CI (1.98, 3.54), p < 0.001, I2 = 0%), the study by Miura et al. (OR 2.25, 95% CI (1.71, 2.96), p < 0.001, I2 = 28%), and the studies that used ticlopidine (OR 1.95, 95% CI (1.21, 3.14), p = 0.006, I2 = 0%) (online Supplementary Figure 2). Subgroup analysis demonstrated that cilostazol remained associated with higher primary patency when comparing studies that used oral anticoagulants versus not (OR 2.72 vs 2.23, p-interaction = 0.49) (online Supplementary Figure 3). The use of cilostazol in RCTs was associated with significantly higher odds of primary patency compared with observational studies (OR 3.37 vs 1.93, p-interaction = 0.03) (online Supplementary Figure 4).
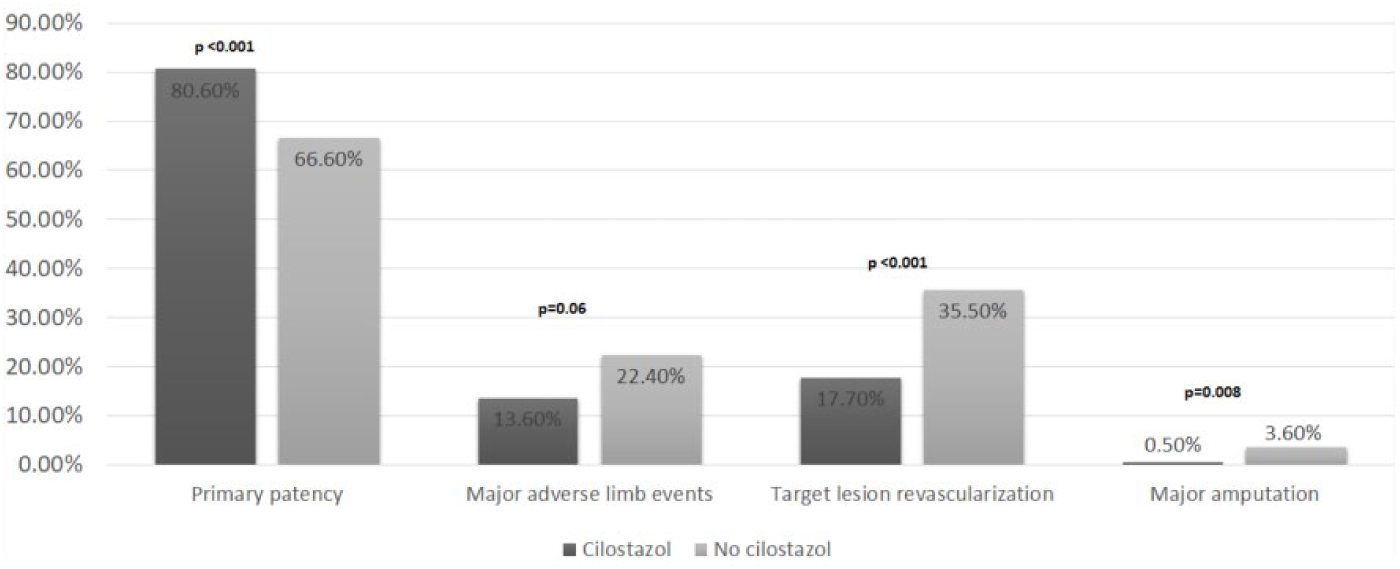
Outcomes with cilostazol after EVT of PAD.
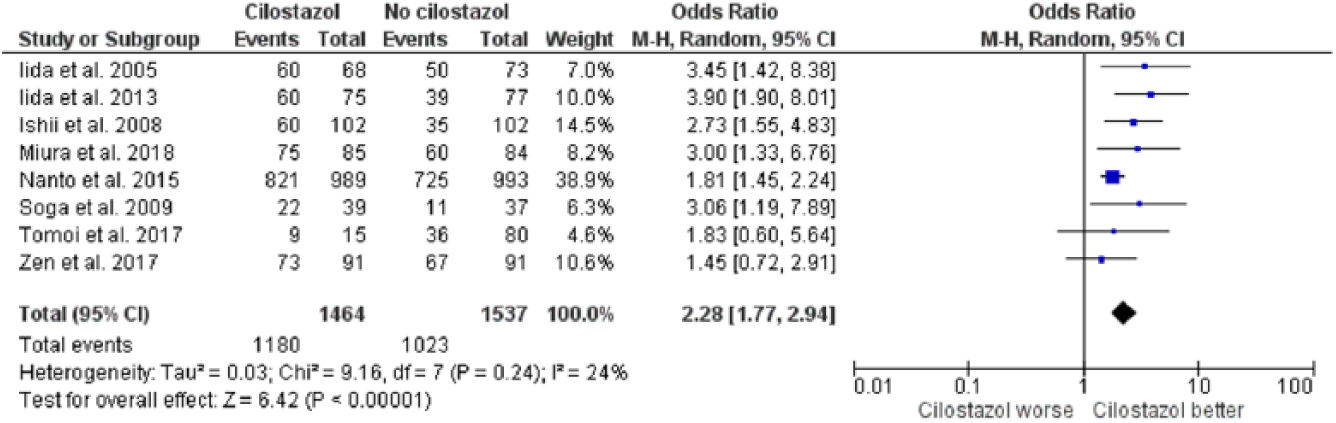
Pooled analysis of the odds of primary patency with cilostazol versus no cilostazol after EVT of PAD.
Major adverse limb events
Major adverse limb events were reported in four studies29,31,34,35 including 620 patients. During a mean follow-up duration of 13.5 ± 5 months, the use of cilostazol had no impact on the incidence of MALE (OR 0.51, 95% CI (0.26, 1.02), p = 0.06, I2 = 57%) (Figure 4).
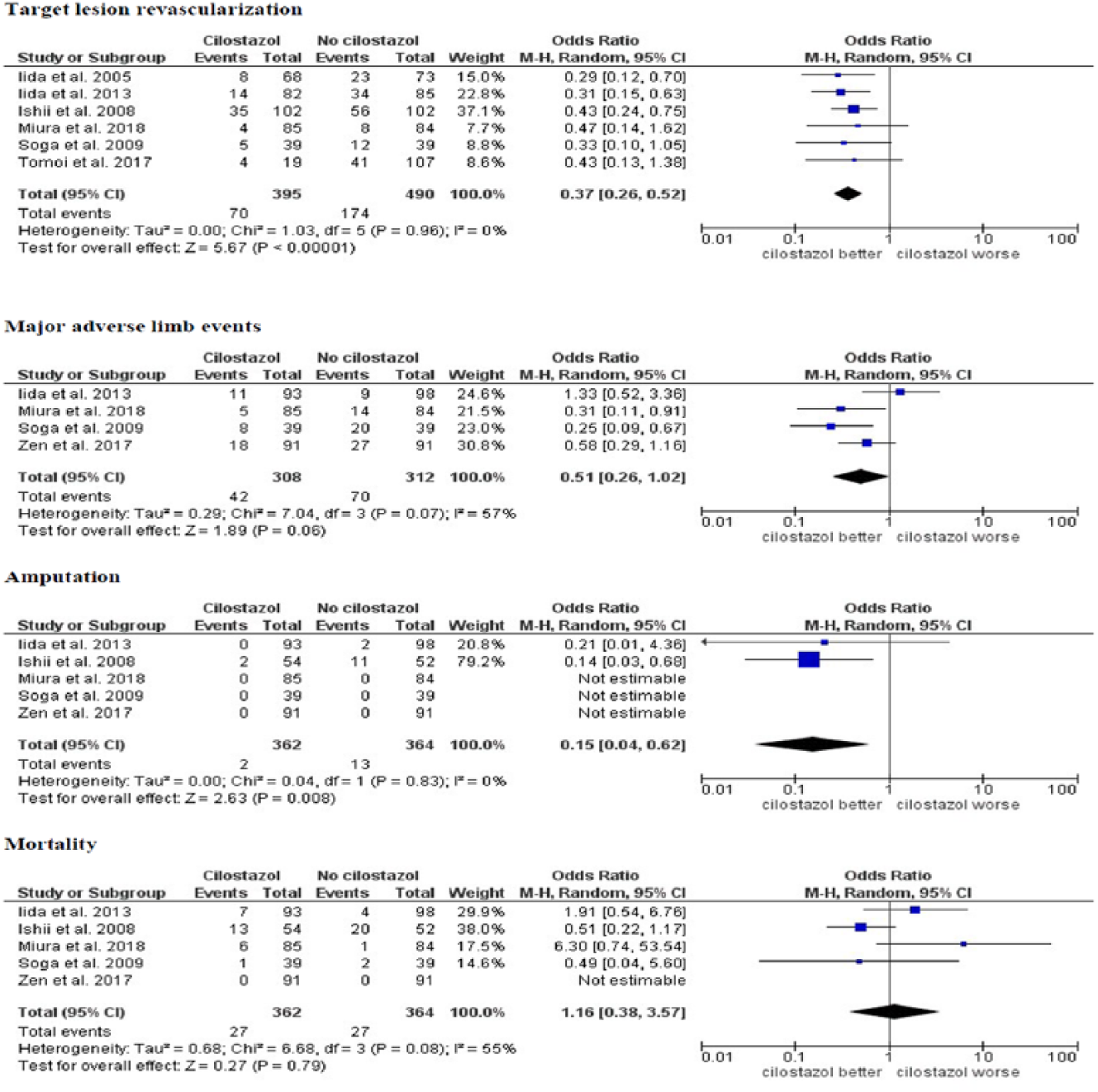
Pooled analysis of the odds of MALE, TLR, major amputation, and mortality with cilostazol versus no cilostazol after EVT of PAD.
Major amputation
Major amputation was reported in five studies28,29,31,34,35 including 726 patients. Although several studies reported major amputation, due to the lack of events in these studies, they were not used in the statistical calculations. During a mean follow-up duration of 16 ± 7 months, the use of cilostazol was associated with a significantly lower risk of major amputation (0.5% vs 3.6%, NNT = 33), (OR 0.15, 95% CI (0.04, 0.62), p = 0.008, I2 = 0%) (Figure 4).
Target lesion revascularization
TLR was reported in six studies27–29,31,33,35 including 885 lesions. During a mean follow-up duration of 17.5 ± 6 months, cilostazol use was associated with a significantly lower risk of TLR (17.7% vs 35.5%, NNT = 5), (OR 0.37, 95% CI (0.26, 0.52), p < 0.001, I2 = 0%) (Figure 4).
Mortality
All-cause mortality was reported in five studies28,29,31,34,35 including 726 patients. Although several studies reported mortality, due to the lack of events in these studies, they were not used in the statistical calculations During a mean follow-up duration of 16 ± 7 months, cilostazol use had no impact on mortality (OR 1.16, 95% CI (0.38, 3.57), p = 0.79, I2 = 55%) (Figure 4). A summary of the study results is illustrated in Figure 5.
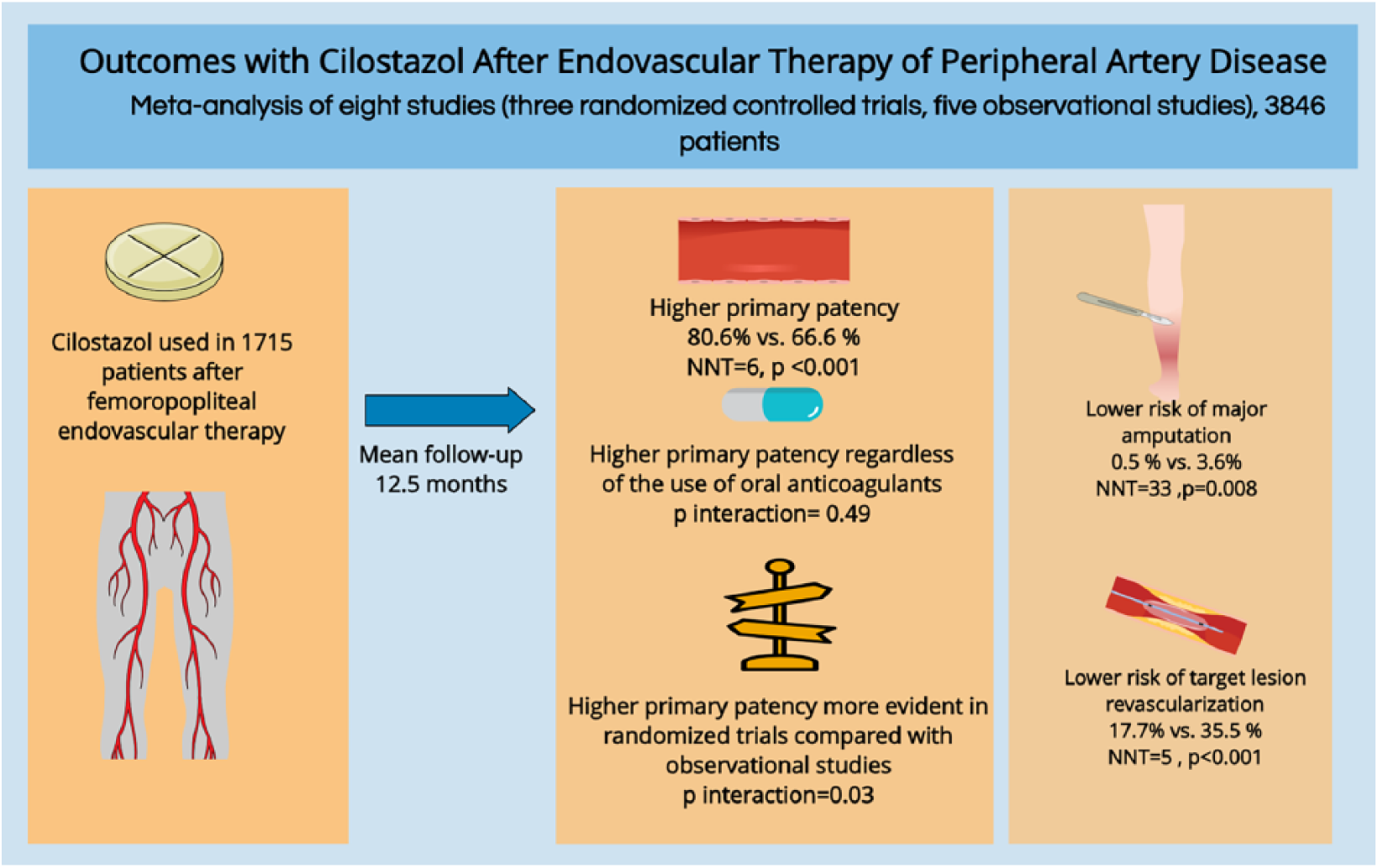
Summary of the study results.
Discussion
In the largest meta-analysis of cilostazol after EVT for PAD, we found the use of cilostazol is associated with significantly higher odds of primary patency that was independent of the use of oral anticoagulants (warfarin). Furthermore, the impact of cilostazol remained favorable even with the concomitant use of clopidogrel. Importantly, the use of cilostazol was associated with a significantly lower risk of major amputation and TLR. These findings strongly support the use of cilostazol as an adjunct to antiplatelet therapy for patients undergoing EVT for PAD and CLI. Our analysis mainly reports the outcomes of cilostazol use after EVT of femoropopliteal de novo and in-stent restenosis (ISR) lesions.
Smaller studies have suggested that cilostazol may have a favorable effect in preventing restenosis after EVT, which is mostly attributed to the inhibition of neointimal growth, the vasodilatory effect through inhibiting phosphodiesterase type 3 (PDE-3) with subsequent elevation of intracellular cAMP levels, and the improvement of vascular endothelial function.22,44–46 In our analysis, we also demonstrated the significantly higher odds of primary patency with cilostazol. Moreover, the impact of cilostazol remained favorable regardless of the use of oral anticoagulants (warfarin). It is important to note that no trials studied the effect of cilostazol in patients who are using new oral anticoagulants (NOAC).
The common side effects of cilostazol are minor and mainly include headache, tachycardia, and diarrhea. However, it is generally safe in terms of major cardiovascular side effects including myocardial infarction and stroke. 47 The use of triple antiplatelet therapy (aspirin, a thienopyridine, and cilostazol) has been previously reported to be safe without increased risk of bleeding compared with dual antiplatelet therapy (DAPT).23,24,48 Unfortunately, bleeding was not reported consistently in the included studies and could not be analyzed. More studies are needed to compare the outcomes of cilostazol versus DAPT that might be more tolerated by patients than cilostazol. The main concern that is likely preventing the widespread use of cilostazol for PAD is the fact that similar phosphodiesterase inhibitors (i.e. milrinone) have been associated with higher mortality rates in patients with congestive heart failure (CHF). 49 Subsequently, the US Food and Drug Administration (FDA) issued a black box warning concerning the safety of cilostazol in CHF patients. Despite multiple studies that have shown that cilostazol is not associated with increased risk in these patients,48,50 the black box warning was not reversed.
Cilostazol is not currently used after coronary stenting, mainly because of the more powerful antiproliferative effect of drug-eluting stents (DES). 51 However, cilostazol use in peripheral lesions may be reasonable even with the use of DES because of longer lesions and higher prevalence to total occlusions. The favorable impact of cilostazol has been demonstrated after EVT of PAD with DES in 182 patients in a subgroup analysis of the ‘ZilvEr PTX for tHe Femoral ArterY and Proximal Popliteal ArteRy’ (ZEPHYR) study. 34 Moreover, cilostazol had favorable outcomes when used with second-generation bare-metal nitinol stents (BNS) shown by the RCT by Miura et al. on 95 patients. 35 Given the low cost and significant benefit, consideration should be given for its use in patients undergoing EVT for PAD and CLI. However, due to the small number of studies and patients, further trials are needed to outline the additional benefit of cilostazol use with newer-generation stents (DES, second-generation BNS) that already provide superior outcomes and improved primary patency.35,52,53
Our results are consistent with the results of prior meta-analyses including improved primary patency and lower TLR with cilostazol.54–56 However, unlike previous analyses, we excluded studies that compared cilostazol with ticlopidine, which is no longer used. In our analysis, we included studies that compared cilostazol versus no cilostazol regardless of the use of clopidogrel or oral anticoagulants. Furthermore, our analysis is most updated with a higher number of studies and patients.
Limitations
Our study has several limitations. First, our study included observational studies which carry the risk of selection bias. Second, some of the included studies did not report the dose and the duration of cilostazol used, which hindered further analysis to identify the optimal period of use. Third, bleeding was not reported consistently in the included studies and could not be analyzed. Fourth, we could not run relevant subgroup analyses (e.g. cilostazol use with DES, cilostazol use with second-generation BNS, and cilostazol use in ISR) due to the lack of an adequate number of studies. Fifth, there were significant differences in the baseline characteristics of the patients and lesions. However, our results remained significant when analyzing only RCTs, which eliminates the risk of patient selection bias. Finally, we encountered a high degree of clinical and methodological heterogeneity; we utilized a random-effects model in an attempt to overcome this limitation.
Conclusion
The use of cilostazol after EVT of femoropopliteal lesions is associated with improved primary patency and lower risk of major amputation and TLR. The favorable impact of cilostazol is independent of the use of oral anticoagulants (warfarin). Further studies are needed to evaluate the role of cilostazol after EVT with newer-generation stents and drug-coated balloons.
Supplemental Material
10.1177_1358863X19838327_Supplementary_material – Supplemental material for Outcomes with cilostazol after endovascular therapy of peripheral artery disease
Supplemental material, 10.1177_1358863X19838327_Supplementary_material for Outcomes with cilostazol after endovascular therapy of peripheral artery disease by Michael Megaly, Bishoy Abraham, Marwan Saad, Andrew Mekaiel, Peter Soukas, Subhash Banerjee and Mehdi H Shishehbor in Vascular Medicine
Footnotes
Declaration of conflicting interests
The authors declared the potential conflicts of interest with respect to the research, authorship, and/or publication of this article. Peter Soukas receives research support from Bard, Cordis Endovascular, Mercator, Spectranetics, and WL Gore. Subash Banerjee receives research support from Boston Scientific and The Medicines Company and consultant/speaker honoraria from Gilead, St. Jude, Cordis, Boehringer Ingelheim, Sanofi, and Medtronic. Dr. Banerjee is a consultant to Covidien and has ownership in MDCARE GLOBAL (spouse) and intellectual property in HygeiaTel. Mehdi Shishehbor provides education and consulting without compensation for Abbot Vascular, Medtronic, Covidien, Spectranetics, Cook, Terumo, and Boston Scientific. The other authors have nothing to disclose.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Supplementary material
The supplementary material is available online with the article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
