Abstract
Keywords
Background
Over 230 million persons are estimated to have peripheral artery disease (PAD), and this population has increased risk of adverse atherothrombotic outcomes.1,2 Although clinical trial subgroups have observed heterogeneity in the risk of limb events based on clinical presentation, particularly lower-extremity revascularization (LER), there are few additional risk stratification tools that incorporate anatomy into prediction of outcomes.3 –7 Certain risk stratification systems have been developed outside of randomized trials and focus on patients with chronic limb-threatening ischemia (CLTI), combining hemodynamics and wound characteristics but not integrating patient or anatomic variables. 8 Other anatomic classifications have been proposed and have been used in device trials but are largely based on expert opinion and not derived from a dataset with independently adjudicated limb outcomes.9 –16
In contrast, for coronary artery disease (CAD), anatomy has informed risk stratification, diagnostic modalities of choice, and revascularization strategies. For example, in the SYNergy between percutaneous coronary intervention with TAXus and cardiac surgery (SYNTAX) trial of patients with CAD randomized to surgical versus percutaneous revascularization, anatomic complexity predicted which revascularization modality was more beneficial. 17 In the Providing Regional Observations to Study Predictors of Events in the Coronary Tree (PROSPECT) study, in 697 patients with acute coronary syndromes, particular angiographic and intravascular ultrasound characteristics of atherosclerotic coronary plaque predicted subsequent cardiovascular events. 18 Furthermore, international guidelines acknowledge certain anatomic features that benefit from intravascular imaging and physiologic evaluation.19,20 As with CAD, it is plausible that better understanding of the anatomy of patients with PAD could yield similar insights for optimal management in improving limb and perhaps overall cardiovascular outcomes.
The Vascular Outcomes Study of Aspirin Along with Rivaroxaban in Endovascular or Surgical Limb Revascularization for Peripheral Artery Disease (VOYAGER-PAD) trial was an international, multicenter, double-blind, placebo-controlled randomized trial comparing low-dose rivaroxaban versus placebo on a background of aspirin in patients with PAD who had undergone LER (surgical or endovascular) up to 10 days prior to enrollment. 2 The trial results showed a benefit in the treatment arm for the primary composite outcome of acute limb ischemia, major amputation from vascular causes, myocardial infarction, ischemic stroke, or death from cardiovascular causes and more bleeding but overall a net clinical benefit. 3 Whether the knowledge of PAD anatomy and its relationship with outcomes can inform the decision to implement certain PAD therapies, such as low-dose Factor Xa inhibition, is unknown. Therefore, VOYAGER-PAD provides an opportunity to characterize anatomic disease patterns in PAD, correlate these findings with clinical characteristics and outcomes, and yield insights into optimal management. 21 Herein, we describe the design and methods of a catheter-based angiographic core lab from VOYAGER-PAD, its study population, and baseline anatomic data.
Methods
Design
The overall schema of the VOYAGER-PAD (
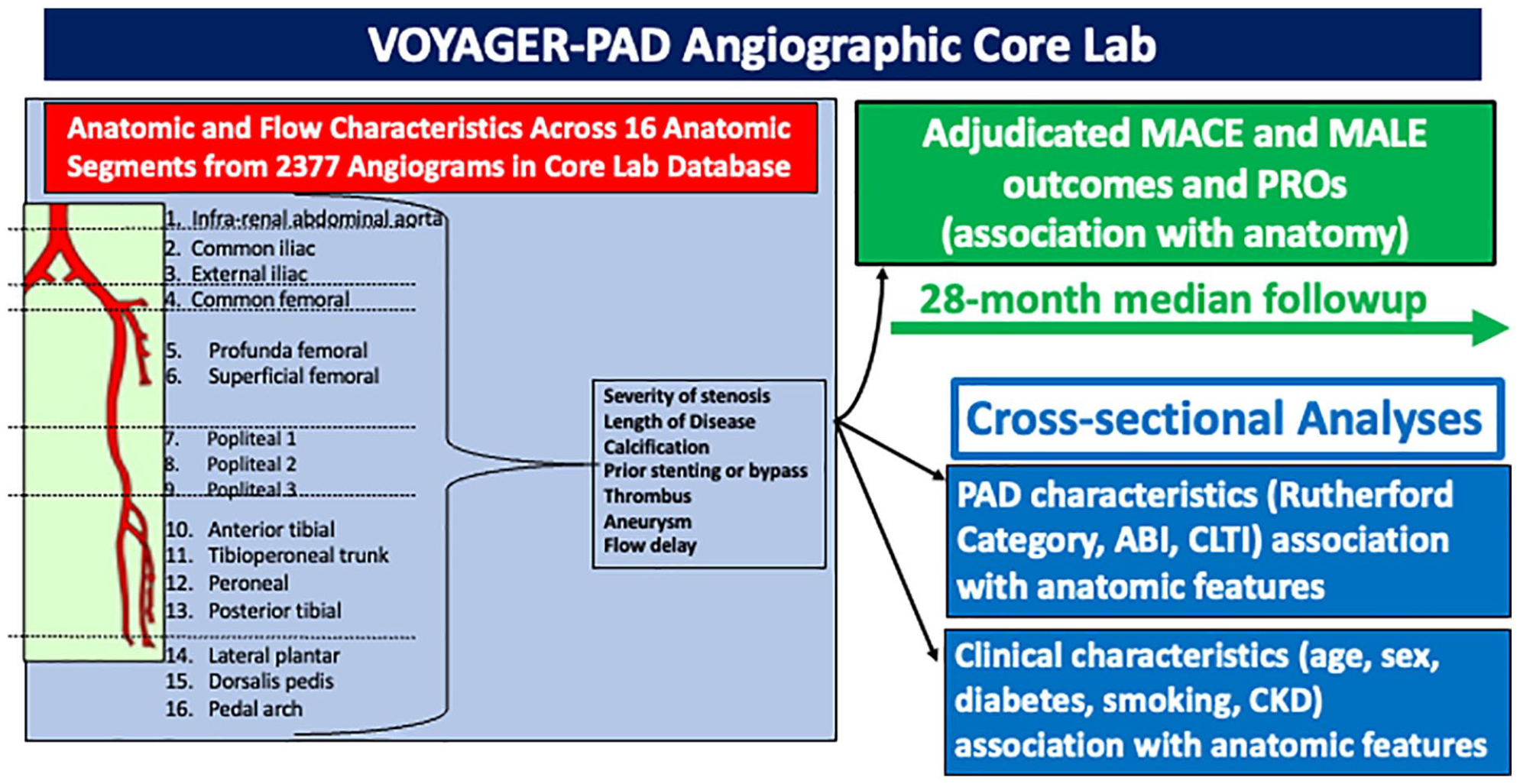
Overall schema of the VOYAGER-PAD angiographic core lab.
The study population of VOYAGER-PAD has been described previously and consisted of those over 50 years old who had symptomatic PAD and underwent successful LER (surgical or endovascular) for infra-inguinal disease 10 days or less before enrollment. 3 The protocol was approved by the local ethics committees, and informed consent was obtained from all patients. To be eligible for VOYAGER-PAD, subjects were required to have documented imaging of peripheral arteries in the form of catheter-based angiography, computed tomography angiography, and/or magnetic resonance imaging. Study sites were asked to submit these clinically performed imaging studies in a de-identified format to a secure CPC Clinical Research database .
Because the peripheral artery imaging required for eligibility in VOYAGER-PAD was dictated by standard clinical practice of the site investigators, CPC accepted imaging studies in various formats, as were available from sites. Accepted formats included DICOM imaging files, digital movie files of cineangiography, and static photographs of angiographic images. Available angiographic studies varied in the number of anatomic segments that were available for analysis. For example, some angiographic studies included the infra-renal aorta and complete imaging of both lower extremities, whereas other studies focused on specific anatomic segments of a single lower extremity. Angiograms were rated as ‘adequate’ or ‘poor’ in quality.
A group of vascular experts convened to discuss prior proposed scoring systems and standard anatomic conventions and, by consensus, identified anatomic and flow variables for ascertainment. The information collected included assessment of 16 distinct anatomic segments of the lower-extremity arterial tree (online supplementary Table 1) for degree of stenosis, length of disease, presence of prior stents or bypass grafts, presence of calcification, aneurysms, and thrombus, the results of revascularization, and anatomic scoring according to the Trans-Atlantic Inter-Society Consensus (TASC) and the Global Limb Anatomic Staging System (GLASS) classifications15,22 (Figure 2; online supplementary Table 2).
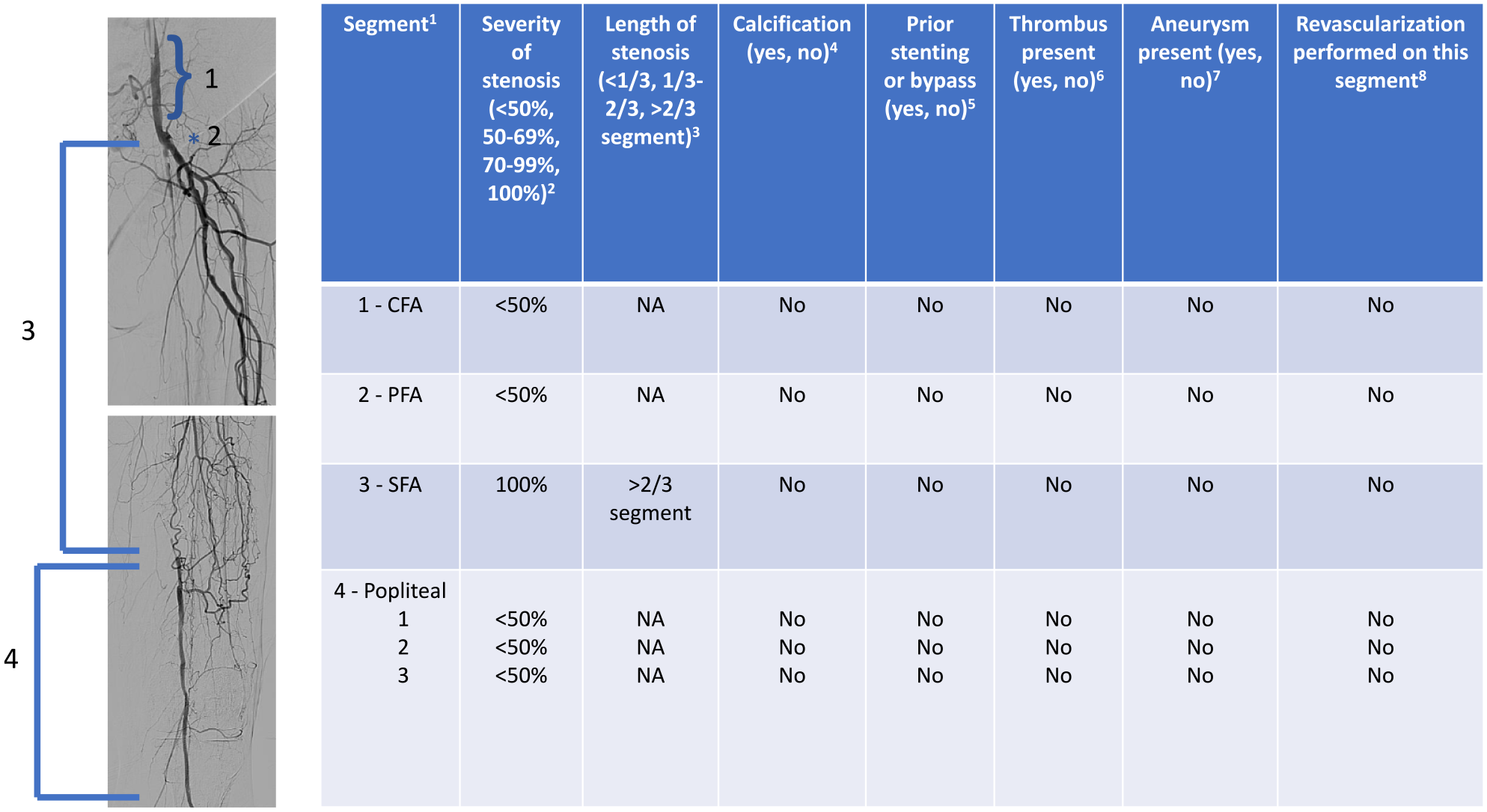
Example of angiogram and scoring for VOYAGER-PAD angiographic core lab for the segments: common femoral, profunda femoral, superficial femoral, and popliteal.
Reviewers were required to have training in vascular surgery, vascular medicine, interventional cardiology, or radiology and clinical experience in interpreting vascular angiography. Reviewers were provided with a manual of definitions and guidance, and underwent standardization training in which they interpreted a common set of angiographic studies. Results from these standardization reviews were compared among reviewers, and concordance and discrepancies were shared with reviewers to promote inter-rater agreement. Those who completed the standardization training proceeded to review angiographic images and enter data into forms on a web-based platform (Datatrak International, Inc., Mayfield Heights, OH, USA). Reviewers had access only to studies to which they were assigned and were blinded to outcomes and treatment allocation.
Clinical outcomes
The primary efficacy outcome for the overall trial was a composite of acute limb ischemia, major amputation for vascular causes, myocardial infarction, ischemic stroke, or death from cardiovascular causes. Additional limb-specific outcomes included major amputation of any cause, all minor amputations, and unplanned index limb revascularization. The principal safety outcome for the overall trial was thrombolysis in myocardial infarction (TIMI) major bleeding. Additional safety outcomes included TIMI nonmajor bleeding and bleeding according to the International Society on Thrombosis and Hemostasis (ISTH) and the Bleeding Academic Research Consortium bleeding scales. These efficacy and safety events were adjudicated by an Independent Central Adjudicating Committee. 2
Analyses
Baseline characteristics for the VOYAGER-PAD participants included in the angiographic subgroup were compared to those not in the angiographic core lab cohort (Figure 3). For discrete variables, summary statistics are presented as frequencies and percentages. Continuous variables were summarized using mean values with SDs or medians with IQR ranges. Dichotomous variables between groups were compared using Fisher’s test. If discrete variables assumed multiple values, they were compared by the chi-squared test. Continuous variables were compared using a t-test if they were normally distributed and a nonparametric test if their distribution was not normal.
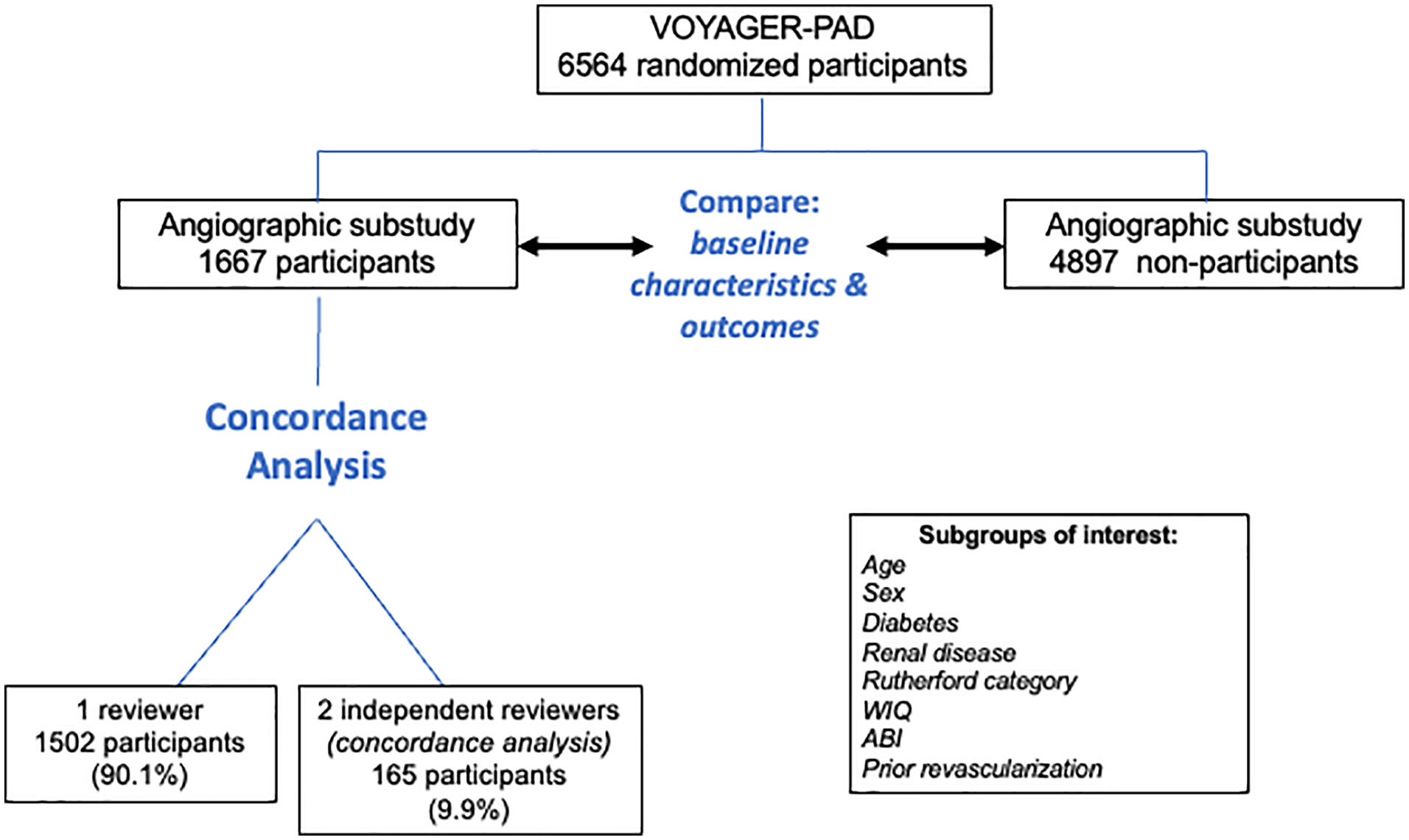
Consort diagram of the VOYAGER-PAD study population, showing subset with available catheter-based angiographic images, subgroups, and planned analyses.
Event rates for the efficacy and safety outcomes were determined in the VOYAGER-PAD core lab population. Event probabilities were expressed as Kaplan–Meier estimates of the cumulative incidence at 3 years after randomization. Hazard ratios were based on the Cox proportional hazards model stratified by type of revascularization and clopidogrel use with treatment as the only covariate. The p-values were based on the log rank test stratified by type of procedure and clopidogrel use.
Subgroups of interest were prespecified (online supplementary Table 3). Ten percent of the total angiographic reviews were independently adjudicated by two reviewers to allow for assessment of inter-rater agreement (Figure 3). Concordance between two reviewers was calculated as: (reviewer 1 and reviewer 2 agreement for anatomic or flow variable per segment) / (all anatomic or flow variables per segment).
Results
Of the 6564 patients enrolled in the VOYAGER-PAD trial, evaluable angiograms from 1666 patients (2377 limbs) were obtained and included in the VOYAGER-PAD angiographic core lab substudy. Fifteen reviewers underwent standardization training and scored these studies. Of all images, 165 (9.9%) were adjudicated independently by two reviewers, and 90.0% were scored as adequate quality (Figure 3). Overall concordance between reviewers for all anatomic and flow variables across each anatomic segment was 90.5% (24,417/26,968). For stenosis severity, concordance was ⩾ 80% in the aorta, common and external iliac, common femoral, and popliteal 3 segments; ⩾ 75–80% in the profunda femoral, superficial femoral artery (SFA), tibioperoneal trunk, and dorsalis pedis segments; 70–75% in the popliteal 1/2, peroneal, and posterior tibial segments; and 65–70% for the pedal arch and anterior tibial segments. For length severity, concordance was > 90% for the aorta, common and external iliac, common and profunda femoral, and popliteal 3 segments; 80–90% for the tibioperoneal segments; 70–80% for the popliteal 1/2 and anterior tibial segments; and 65% for the SFA.
The concordance for calcification was 80–100% in all segments except the SFA (69%) and pedal arteries (75%). Concordance for the presence of preexisting stents, aneurysm, flow delay, and thrombus was greater than 80% for all segments.
Clinical characteristics and outcomes of the VOYAGER-PAD core lab cohort
Subjects in the VOYAGER-PAD trial were followed for a median of 28 months (IQR 22–34 months) for clinical outcomes. 3 The mean age was 67 years in both groups; 463 (27.8%) participants in the angiographic core lab cohort were women, and 1241 (25.3%) subjects not in the core lab were women (p = 0.05). The prevalence of comorbidities such as a history of CAD and carotid disease were similar (Table 1). Participants in the angiographic core lab were more likely to be from North America and Western Europe and less likely to have had their index procedure in Eastern Europe, Asia Pacific, and South America. There were more White (86.9%) and Black (4%) individuals in the core lab cohort versus 78.7% and 1.8% in the noncore lab cohort, respectively, based on patient-reported race (p < 0.001). An endovascular index LER was more common in the angiographic core lab cohort: 1320 (79.2%) LER procedures versus 3059 (62.5%) in those not in the core lab group (p < 0.01) (Table 1).
Demographics and baseline characteristics.
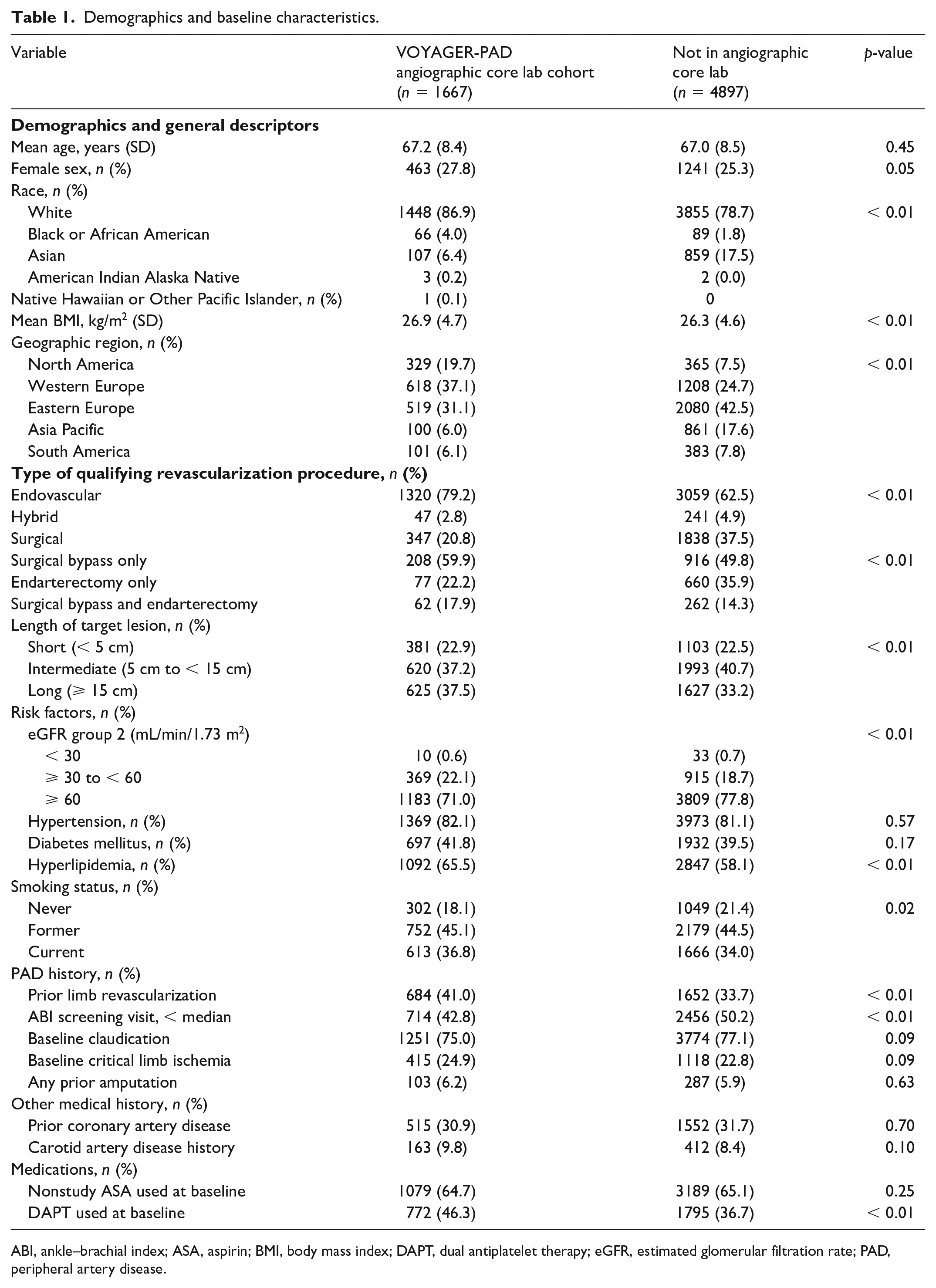
ABI, ankle–brachial index; ASA, aspirin; BMI, body mass index; DAPT, dual antiplatelet therapy; eGFR, estimated glomerular filtration rate; PAD, peripheral artery disease.
The effect of rivaroxaban on the primary efficacy outcome, and the components of the primary efficacy outcome, was consistent in patients included and not included in the angiographic core lab substudy (Figure 4A). The effect of rivaroxaban on the primary safety outcome was also consistent in the participants included in the VOYAGER-PAD angiographic core lab, as compared to those in the overall trial not in the core lab (Figure 4B).
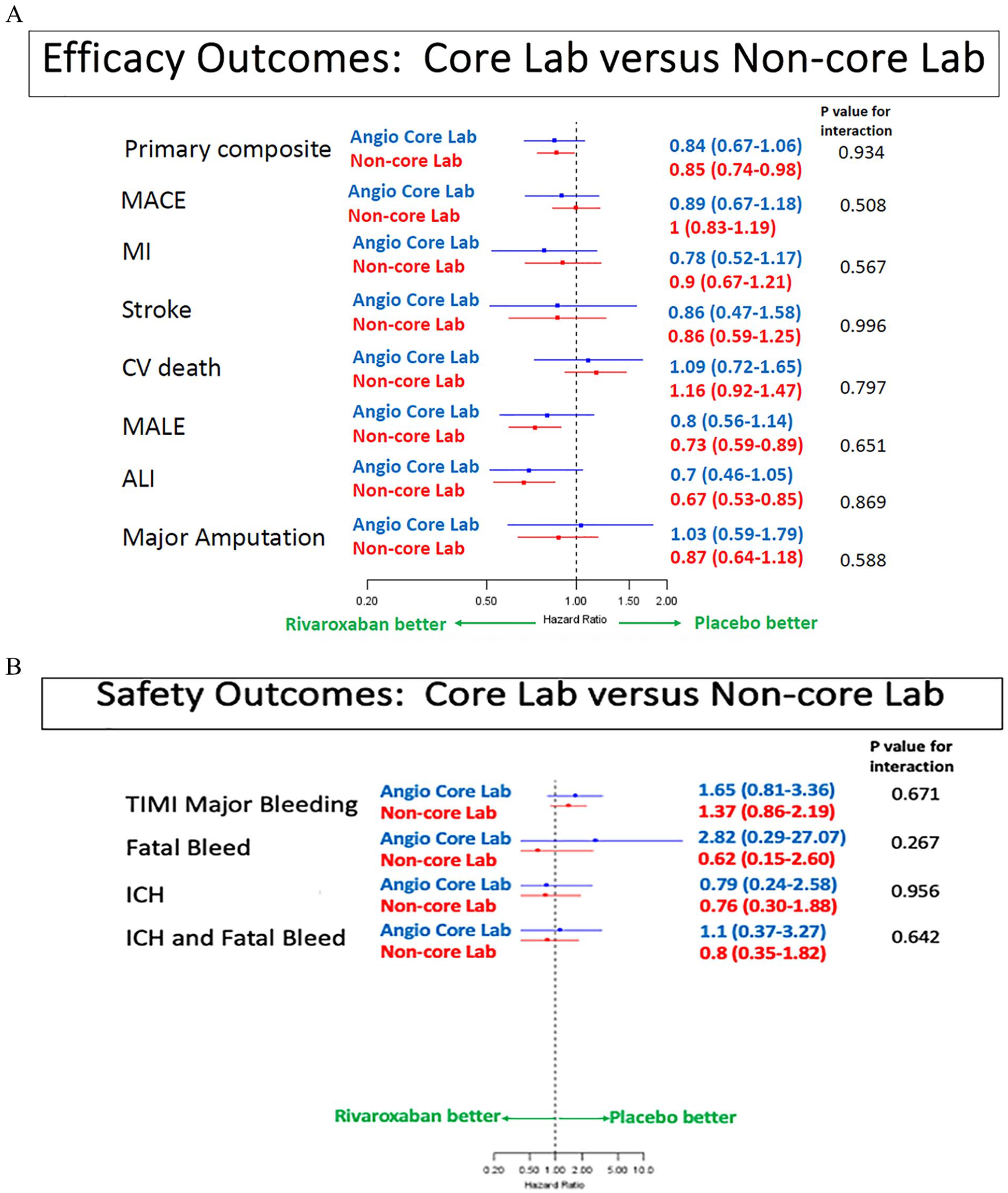
Forest plots of
Anatomic characteristics at baseline
Among the angiograms collected and analyzed, the SFA was the most frequently scored segment (1468 or 88% of 1666 index limbs), whereas the pedal arch was the least frequently scored (365 or 22% of index limbs). The SFA was also the most diseased segment, with the greatest proportion of complete occlusions (n = 742, 45%), was the most likely to be scored as calcified (n = 627, 43%), and had the highest prevalence of preexisting stents (n = 169, 11.5%) (Figures 5A-B; online supplemental Figure 1). The popliteal 3 segment, the anterior tibial, and the posterior tibial arteries were the most likely to have a length of disease > 2/3 of the segment (56%, 53%, and 56%, respectively) (Figure 5C). The presence of aneurysm (0.2% of all segments scored) and thrombus (1.7% of all segments scored) was uncommon. The variable for ‘postprocedure flow delay’ was evaluable for 9.6% of total segments scored. Of these scored segments, the prevalence of flow delay was 4.6%.
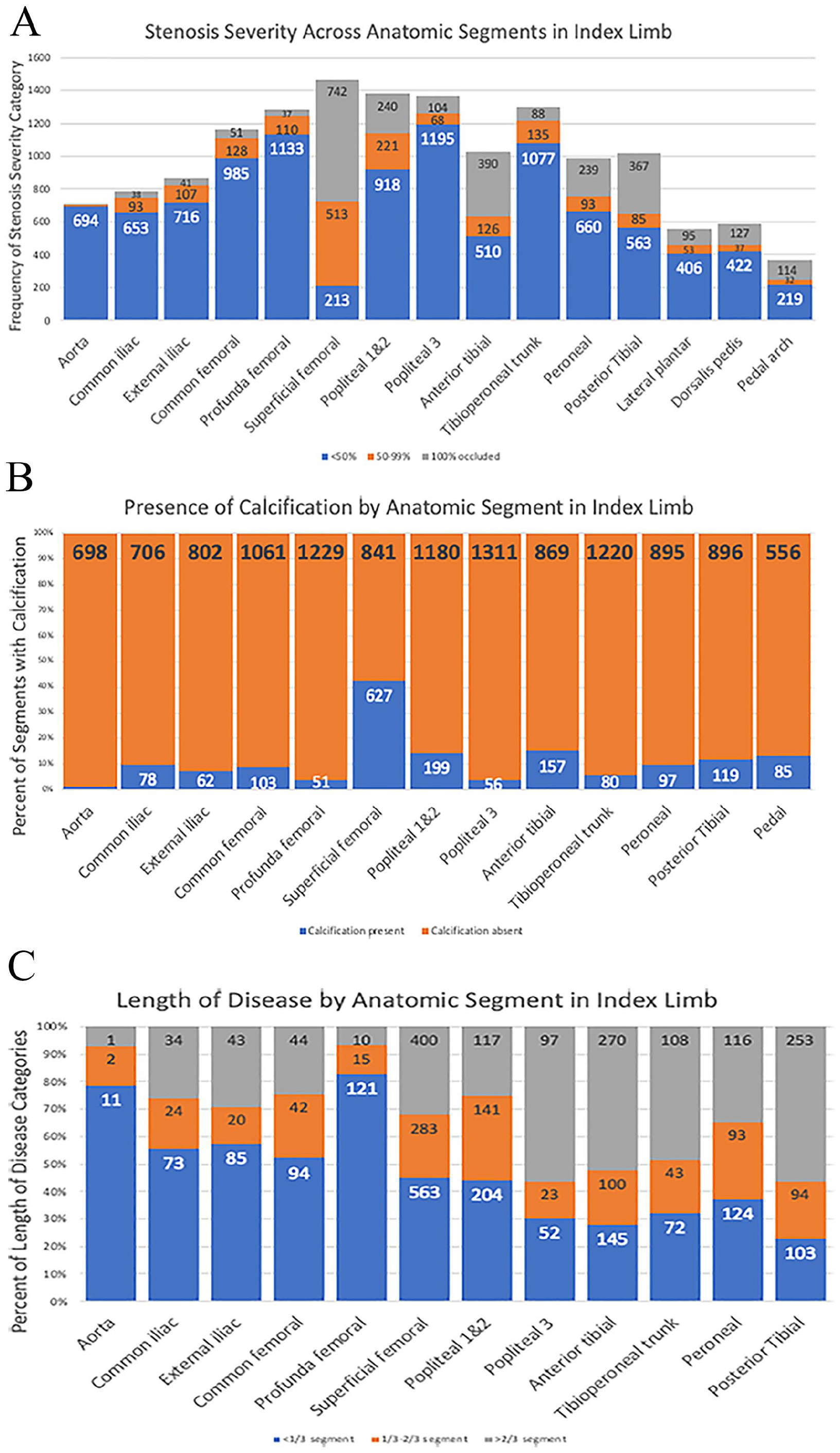
Across anatomic segments:
Discussion
Herein, we describe the design and baseline characteristics of an angiographic core lab from evaluable peripheral catheter-based angiograms in subjects from the VOYAGER-PAD trial. Strengths of this core lab include having: (1) a large dataset of angiographic studies (2377 limbs) in patients with PAD who are clinically similar to overall trial participants; (2) ascertainment of detailed anatomic information across 16 distinct anatomic segments, including severity of stenosis, length of disease, presence of calcification, presence of aneurysms, presence of thrombus, and flow characteristics; (3) independently adjudicated cardiovascular and limb events; (4) detailed characterization of clinical and PAD-related variables; (5) a high rate of acute limb ischemia events (98 events in 1667 patients, 5.9%) in this at-risk population following LER; and (6) nesting within a randomized trial of a therapy shown to reduce limb outcomes. These strengths facilitate the ability to examine the relationship between specific lower-extremity anatomic characteristics and clinical phenotypes, PAD subgroups, and cardiovascular outcomes – particularly limb outcomes.
Study limitations
Limitations include that despite analyzed angiograms in over 1600 patients, this subgroup represents only a subset of the overall trial. In addition, since images were obtained for clinical use and performed by provider discretion, not all anatomic segments in the lower extremities were imaged in a standardized fashion; however, we believe that anatomic segments not imaged in these catheter-based angiograms are less likely to be severely diseased based on the angiographers’ knowledge of patient presentation. Additional limitations include that the VOYAGER-PAD angiographic core lab cohort was disproportionately more likely to be White, men, and from North America or Europe, which affects the generalizability of this data set. Finally, inter-rater agreement, though greater than 90% overall for all variables across segments as a whole, was variable for some segments. For example, concordance was 68% for stenosis severity in the pedal arch and 65% for length severity in the SFA. We believe these limitations in concordance reflect the practical issues of performing and interpreting pedal images and defining length of disease in a segment such as the SFA from clinically performed angiograms.
Certain anatomic classification systems for PAD do exist, such as the widely used TASC system, the more recently developed GLASS classification,15,22 and others. 14 The development of these anatomic classification schemes are important and address treatment gaps in patients with PAD. 23 However, these anatomic descriptions are largely based on expert opinion or chart review, rather than direct examination of peripheral angiograms. Additionally, these anatomic classifications have not been developed in datasets with independently adjudicated limb outcomes. In contrast, the VOYAGER-PAD angiographic core lab provides an opportunity to analyze the relationship of anatomic variables with adverse limb outcomes by establishing a database of patients with PAD enrolled in a randomized trial setting with independently adjudicated cardiovascular and limb outcomes on a background of well-defined medical therapy. Such a project not only facilitates the ability to understand better the association of peripheral anatomy with outcomes but also what therapy might reduce adverse outcomes.
Conclusion
In the field of CAD, coronary anatomy has informed risk stratification, diagnostic modalities of choice, and revascularization strategies.17 –20 Extrapolating, we believe that the VOYAGER-PAD angiographic core lab can yield insight into which anatomic factors place patients with PAD at risk for adverse limb outcomes and how interventions, such as revascularization and medical therapy, can modify these risks.
Supplemental Material
sj-pdf-1-vmj-10.1177_1358863X241228542 – Supplemental material for Methods, design, and initial results of an angiographic core lab from VOYAGER-PAD
Supplemental material, sj-pdf-1-vmj-10.1177_1358863X241228542 for Methods, design, and initial results of an angiographic core lab from VOYAGER-PAD by R Kevin Rogers, Joerg Herold, Nicholas Govsyeyev, Roberto Iezzi, Justin Morrison, Shea E Hogan, Mark Nehler, Rory Bricker, Brice Andring, Brian Bergmark, Matt Cavender, Emily Malgor, Donald Jacobs, Michael N Young, Warren Capell, Joseph W Yčas, Sonia S Anand, Scott D Berkowitz, E Sebastian Debus, Lloyd P Haskell, Eva Muehlhofer, Manesh R Patel, Connie N Hess, Rupert M Bauersachs, Victoria Anderson and Marc P Bonaca in Vascular Medicine
Footnotes
Acknowledgements
The authors would like to acknowledge the contributions of the late Dr. William R. Hiatt, a leader in vascular medicine, co-chair of VOYAGER PAD, and supporting investigator of the angiographic core lab.
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Drs Rogers, Hogan, Nehler, Capell, Berkowitz, Hess, Yčas, and Bonaca receive salary support from CPC, a nonprofit academic research organization affiliated with the University of Colorado that receives or has received research grant/consulting funding between February 2021 and June 2023 from the following organizations: Abbott Laboratories, Adamis Pharmaceuticals Corporation, Agios Pharmaceuticals, Inc., Alexion Pharma, Alnylam Pharmaceuticals, Inc., Amgen Inc., Angionetics, Inc., ARCA Biopharma, Inc., Array BioPharma, Inc., AstraZeneca and Affiliates, Atentiv LLC, Audentes Therapeutics, Inc., Bayer and Affiliates, Beth Israel Deaconess Medical Center, Better Therapeutics, Inc., BIDMC, Boston Clinical Research Institute, Bristol-Meyers Squibb Company, Cambrian Biopharma, Inc., Cardiol Therapeutics Inc., CellResearch Corp., Cook Medical Incorporated, Covance, CSL Behring LLC, Eidos Therapeutics, Inc., EP Trading Co. Ltd, EPG Communication Holdings Ltd, Epizon Pharma, Inc., Esperion Therapeutics, Inc., Everly Well, Inc., Exicon Consulting Pvt Ltd, Faraday Pharm-aceuticals, Inc., Foresee Pharmaceuticals Co. Ltd, Fortress Biotech, Inc., HDL Therapeutics Inc., HeartFlow Inc., Hummingbird Bioscience, Insmed Inc., Ionis Pharmaceuticals, IQVIA Inc., JanOne Biotech Holdings Inc., Janssen and Affiliates, Kaneka, Kowa Research Institute, Inc., Kyushu University, Lexicon Pharm-aceuticals, Inc., LSG Kyushu University, Medimmune Ltd, Medpace, Merck & Affiliates, Novartis Pharmaceuticals Corp., Novate Medical, Ltd, Novo Nordisk, Inc., Pan Industry Group, Pfizer Inc., PhaseBio Pharmaceuticals, Inc., PPD Development, LP, Prairie Education and Research Cooperative, Prothena Biosciences Limited, Regeneron Pharmaceuticals, Inc., Regio Biosciences, Inc., Rexgenero, Sanifit Therapeutics S.A., Sanofi-Aventis Groupe, Silence Therapeutics PLC, Smith & Nephew plc, Stealth Bio-Therapeutics Inc., State of Colorado CCPD Grant, The Brigham & Women’s Hospital, Inc., The Feinstein Institutes for Medical Research, Thrombosis Research Institute, University of Colorado, University of Pittsburgh, VarmX, Virta Health Corporation, WCT Atlas, Worldwide Clinical Trials Inc., WraSer, LLC, and Yale Cardiovascular Research Group. Dr Bonaca receives support from the AHA SFRN under award numbers 18SFRN3390085 (BWH-DH SFRN Center) and 18SFRN-33960262 (BWH-DH Clinical Project). Dr Bonaca also reports stock in Medtronic and Pfizer.
Funding
The VOYAGER PAD trial was funded through grants from Bayer and Janssen to CPC Clinical Research.
Supplementary material
The supplementary material is available online with the article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
