Abstract
The present study examines the association between depressive symptoms and inflammatory markers in peripheral artery disease (PAD) to better understand the mechanistic relationship between depression and PAD. A cross-sectional sample of 117 patients with PAD (97% male, 76% Caucasian) was recruited from the San Francisco Veterans Affairs Medical Center. Patients were categorized into three subgroups based upon current depressive symptom severity, as defined by Patient Health Questionnaire-8 scores: no symptoms (score of 0–4, n = 62), mild symptoms (score of 5–9, n = 33), and moderate/severe symptoms (score ≥ 10, n = 22). Serum levels of high-sensitivity C-reactive protein (hs-CRP), interleukin-6 (IL-6), soluble intercellular adhesion molecule-1 (ICAM-1), and tumor necrosis factor-alpha (TNF-α) were assayed and log-transformed for multivariable analysis. To decrease the possibility of Type 1 errors, inflammatory markers were standardized and summed to create a total inflammatory score. In a multivariable analysis controlling for demographics, PAD severity, and atherosclerotic risk factors, mild and moderate/severe depressive symptoms were predictive of a higher total inflammatory score when compared to the group with no symptoms (mild symptoms p = 0.04, moderate/severe symptoms p = 0.007). Exploratory multivariable analyses of individual inflammatory markers found IL-6 levels were significantly higher in the moderate/severe symptoms group (p = 0.006) than in the no symptoms group. Moreover, hs-CRP and ICAM-1 trended upwards with increasing depression severity. TNF-α was not associated with depression severity. We conclude that depressive symptom severity was independently associated with greater inflammation in PAD. Future research should examine the strength and directionality of this association through larger prospective cohort studies, as well as investigate the pathophysiological mechanisms responsible.
Introduction
Peripheral artery disease (PAD) has become a major health concern, affecting one in five men and women in the US by the age of 80. 1 Patients with PAD may suffer a lower quality of life from claudication, impaired mobility, limb loss, a dramatically higher risk of cardiovascular events, and higher total mortality.2,3 Despite widespread public campaigns aimed at improving cardiovascular health, such as Healthy for Good 4 and Stay in Circulation, 5 the prevalence of PAD is expected to increase with the aging of the world population. 6
In addition to traditional risk factors such as smoking, diabetes, hypertension, and hypercholesterolemia, recent research has brought attention to the role of mental health in PAD.7–11 Depression, the leading cause of disability worldwide, 12 afflicts patients with cardiovascular disease (CVD) more than the general population. 13 Patients with depression also face worsened coronary artery disease (CAD) outcomes,14,15 increased risk of cardiovascular events, 16 and worse outcomes after any surgery. 17 The INTERHEART study, an international case–control study, found that globally, psychosocial factors had a greater impact on risk of myocardial infarction than many traditional risk factors. 18
Given the overlapping pathogenesis between CAD and PAD, researchers have begun to explore the effects of depression on the PAD population specifically. Recent studies have found that patients with comorbid depression have more impaired lower limb function, 19 worse post-procedural outcomes, 20 and an increased risk of cardiovascular events. 10 Despite evidence of an association between depression and cardiovascular health independent of behavioral mechanisms, efforts aimed at understanding the pathophysiological processes linking depression and PAD remain limited.15,21
One proposed mechanism involves modification of the immune response. PAD is a disease of atherosclerosis, with inflammation driving critical pathologic processes from plaque formation to thrombus rupture.22,23 Markers of inflammation include high-sensitivity C-reactive protein (hs-CRP), interleukin-6 (IL-6), soluble intracellular adhesion molecule-1 (ICAM-1), and tumor necrosis factor-alpha (TNF-α), which are involved in both systemic inflammation and local plaque formation.24–27 These proteins have been identified as predictors of PAD development and progression. 28 McDermott et al. provide a comprehensive review of literature establishing the role of these inflammatory cytokines in PAD, 29 which has been confirmed by a recent study. 30 Inflammatory markers are also found at elevated levels in depressed individuals.31,32 A few studies have also explored the impact of depression on inflammation in patients with cardiovascular disease. For instance, levels of ICAM-1 were found to be significantly higher in depressed patients after an acute coronary event. 33 However, sufficient research examining inflammation in patients with PAD and depression is lacking.
The objective of the present study was to test the hypothesis that depression symptom severity is independently associated with increased inflammation in patients with PAD. Shedding light on this poorly understood relationship would provide a better understanding of the disease processes linking mental and vascular health.
Methods
Study population
A cross-sectional sample of 117 patients with PAD (97% male, 76% Caucasian) was selected from the OMEGA-PAD studies conducted by Grenon et al. (NCT01310270 and NCT01979874). 34 Participants were recruited between May 2011 and July 2016 from the vascular surgery outpatient clinic at the San Francisco Veterans Affairs Medical Center (SFVAMC). The goal of these studies was to determine the impact of omega-3 polyunsaturated fatty acid supplements on inflammation and endothelial function in patients with PAD. Baseline data were utilized for the purposes of the present study. PAD was defined as the presence of claudication symptoms and an ankle–brachial index (ABI) of less than 0.9, or a history of peripheral revascularization (bypass or angioplasty) for PAD. Patients had to be at least 35 years of age to be included in the study, and were required to have a reliable means of attending follow-up appointments as well as a current mailing address in order to participate. To eliminate confounding conditions that may intrinsically impact inflammation and to reduce overall health status variability, subjects were excluded if they were taking immunosuppressive medications, had a severe acute illness (infection, surgery, critical limb ischemia, or other illness) within the last 30 days, or had severe hepatic disease (Child–Pugh ≥ B), renal disease (creatinine ≥ 2 mg/dL), or a non-vascular inflammatory disease. The study was approved by the University of California San Francisco Committee on Human Research and the SFVAMC Research and Development Office. All participants provided written informed consent.
Measurements
Patient characteristics, demographics, medical history, and anthropometrics
Information on demographics and past medical history was obtained through an intake questionnaire and the SFVAMC electronic medical record. Significant comorbidities such as CAD, hypertension, hyperlipidemia, and diabetes mellitus were recorded. Participants also provided information regarding current use of aspirin, angiotensin-converting-enzyme inhibitors, beta-blockers, and statins. Selective serotonin reuptake inhibitor (SSRI) use up to 6 weeks prior to the baseline visit was determined through a chart review to denote antidepressant use. Smoking history was represented as pack years, calculated by multiplying the number of packs of cigarettes consumed per day by the number of years the person had smoked. Body mass index (BMI) and blood pressure were also recorded. ABIs were measured in each lower extremity using well-established techniques. 35
Mental health
The self-administered Patient Health Questionnaire-8 (PHQ-8) diagnostic instrument was used to determine depressive symptom severity. The PHQ-8 is a validated eight-item questionnaire scored on a scale from 0 to 24, with a score of ≥ 10 being a strong indicator of major depression. 36 For the present study, patients were categorized into three groups based upon worsening depressive symptoms defined by PHQ-8 scores: no symptoms (score of 0–4, n = 62), mild symptoms (score of 5–9, n = 33), and moderate/severe symptoms (score ≥ 10, n = 22).
Laboratory tests
Blood samples were collected from subjects in a fasting state, following standard guidelines to provide an accurate lipid profile. The SFVAMC clinical laboratory analyzed plasma samples to determine hs-CRP, creatinine, estimated glomerular filtration rate, glycated hemoglobin, and lipid levels (total cholesterol, low-density lipoprotein, high-density lipoprotein, and triglycerides) the same day as collection. These assays were performed according to standard kit protocol (Beckman Coulter Analyzer, Miami, FL, USA). Hs-CRP levels of < 1 mg/L are considered normal and have been associated with a low risk for future cardiovascular events. 37
Serum samples were isolated and frozen at −80°C for storage. Assays for IL-6, ICAM-1, and TNF-α were conducted according to the manufacturer’s instructions (R&D Systems Inc., Minneapolis, MN, USA). All samples were run twice and the average value recorded for results with a coefficient of variability less than 20%.
Statistical analyses
Data analyses were performed using Stata Statistical Software, Release 14 (StataCorp LP, College Station, TX, USA).
To minimize the risk of Type I statistical error due to multiple cytokine comparisons, a single total inflammatory score was created by adding the standardized z-scores of all inflammatory markers for each participant.38,39 The inflammatory markers were equally weighted in this calculation. The total inflammatory score was utilized for the primary analysis, and individual cytokines (hs-CRP, IL-6, ICAM-1, and TNF-α) were examined in exploratory analyses.
Unadjusted differences between groups were calculated for all variables of interest. Statistical analysis was performed using one-way analysis of variance (ANOVA) with Bonferroni-corrected post hoc pairwise comparisons for continuous variables, and Fisher’s exact test for categorical variables. For normally distributed continuous variables, summary statistics were reported using mean and standard deviations. For continuous skewed variables, such as unlogged inflammatory marker levels, comparisons were made using Kruskal–Wallis tests, and data were summarized with medians and interquartile ranges (IQRs). Categorical variables were reported in terms of frequency and percentage.
A multivariable model was built using PHQ-8 groups (categorized as no symptoms, mild symptoms, and moderate/severe symptoms) as the primary predictor of the total inflammatory score. Covariates were selected a priori and included well-established atherosclerotic risk factors such as age, race (defined as Caucasian or not), hypertension, hyperlipidemia, diabetes mellitus, pack years, and eGFR. Additionally, ABI was included to control for severity of PAD, and coronary artery disease was included to control for overall severity of systemic atherosclerosis. Sex was excluded from the model as 97% of the sample was male. All results were audited for normality of residuals, constant variance, and influential values. As continuous variables, hs-CRP, IL-6, ICAM-1, and TNF-α levels were graphed and inspected for normality. All had right skewed distributions, and were thus log transformed for the multivariable linear regression analysis.
Results
Comparisons of patient characteristics and comorbidities between groups with no depressive symptoms (n = 62), mild symptoms (n = 33), and moderate/severe symptoms (n = 22) are depicted in Table 1. Age, sex, and ethnic make-up largely reflect the patient population at SFVAMC. Patients with moderate/severe depressive symptoms were more likely to be Caucasian compared to patients with mild symptoms or no symptoms (95% vs 85% and 65%, respectively, p = 0.005). Otherwise, there were no statistically significant differences between groups in BMI, rates of comorbidities, medication use, and lipid levels. Additionally, there was no significant difference in PAD symptom severity (represented by Rutherford categories 40 ) between depressive symptom groups. The median PHQ-9 score in the moderate/severe symptoms group was 14.
Characteristics of PAD patients by depressive symptom groups (n = 117).
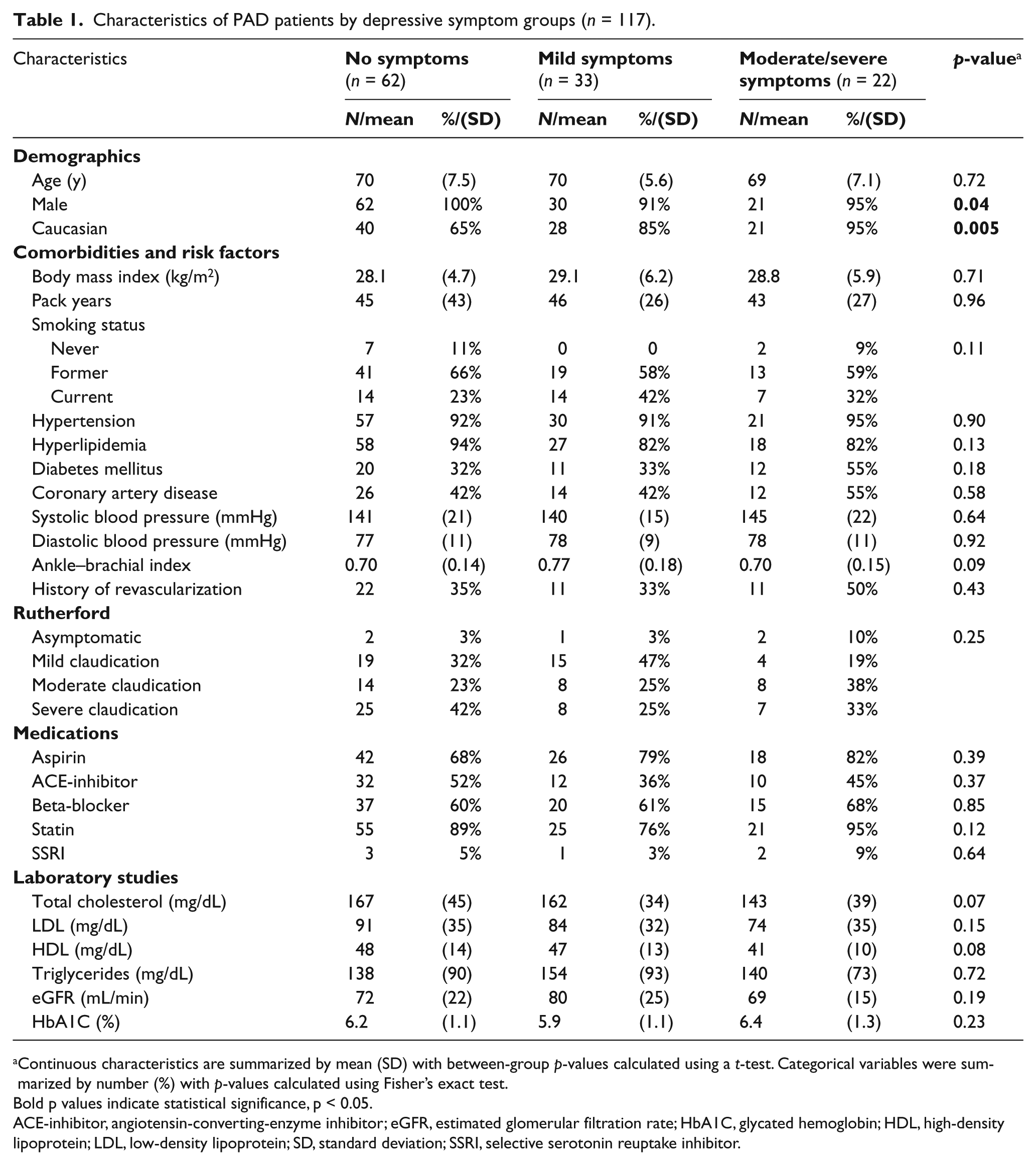
Continuous characteristics are summarized by mean (SD) with between-group p-values calculated using a t-test. Categorical variables were summarized by number (%) with p-values calculated using Fisher’s exact test.
Bold p values indicate statistical significance, p < 0.05.
ACE-inhibitor, angiotensin-converting-enzyme inhibitor; eGFR, estimated glomerular filtration rate; HbA1C, glycated hemoglobin; HDL, high-density lipoprotein; LDL, low-density lipoprotein; SD, standard deviation; SSRI, selective serotonin reuptake inhibitor.
An unadjusted analysis examining total inflammatory scores in the depressive symptom groups revealed an increasing trend with worsening symptom severity (median, IQR): no symptoms (–1.1, –1.9 to 0.8); mild symptoms (–0.3, –1.6 to 0.9); moderate/severe symptoms (1.3, –1.1 to 3.2); p = 0.06 (Figure 1). In multivariable analyses, mild symptoms and moderate/severe symptoms were independently associated with a higher total inflammatory score compared to the no symptoms group: mild symptoms versus no symptoms (p = 0.04) and moderate/severe symptoms versus no symptoms (p = 0.007) (Table 2). Additional sensitivity analyses in which the model was adjusted for aspirin, statin, or SSRI use did not affect the results. A sensitivity analysis adjusting for smoking status rather than pack year history attenuated the association, with the association between the moderate/severe symptoms group remaining statistically significant (mild symptoms: p = 0.17; moderate/severe symptoms: p = 0.02).
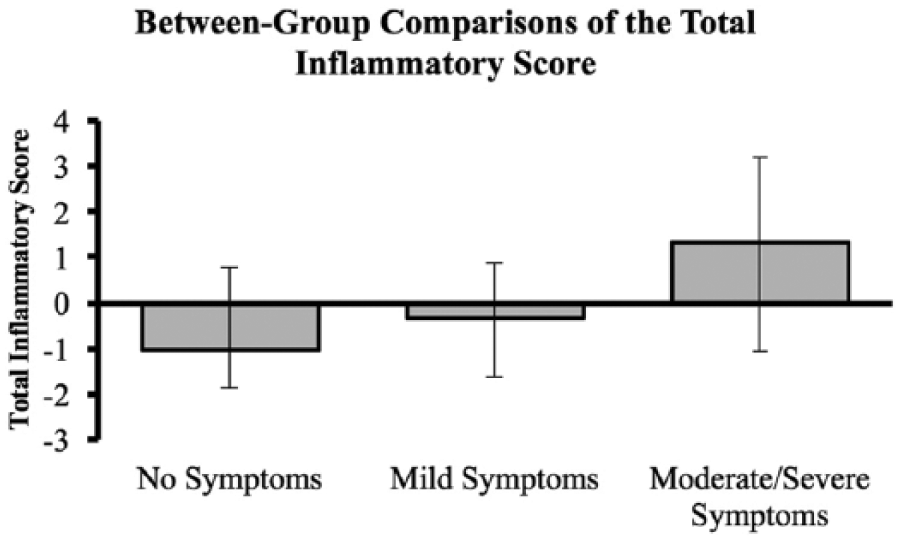
Total inflammatory scores trend higher in patients with worsening depressive symptoms (p = 0.06) in unadjusted analysis. The medians and IQRs of the total inflammatory score are depicted.
Multivariable associations of total inflammatory score with depressive symptom groups (n = 79).
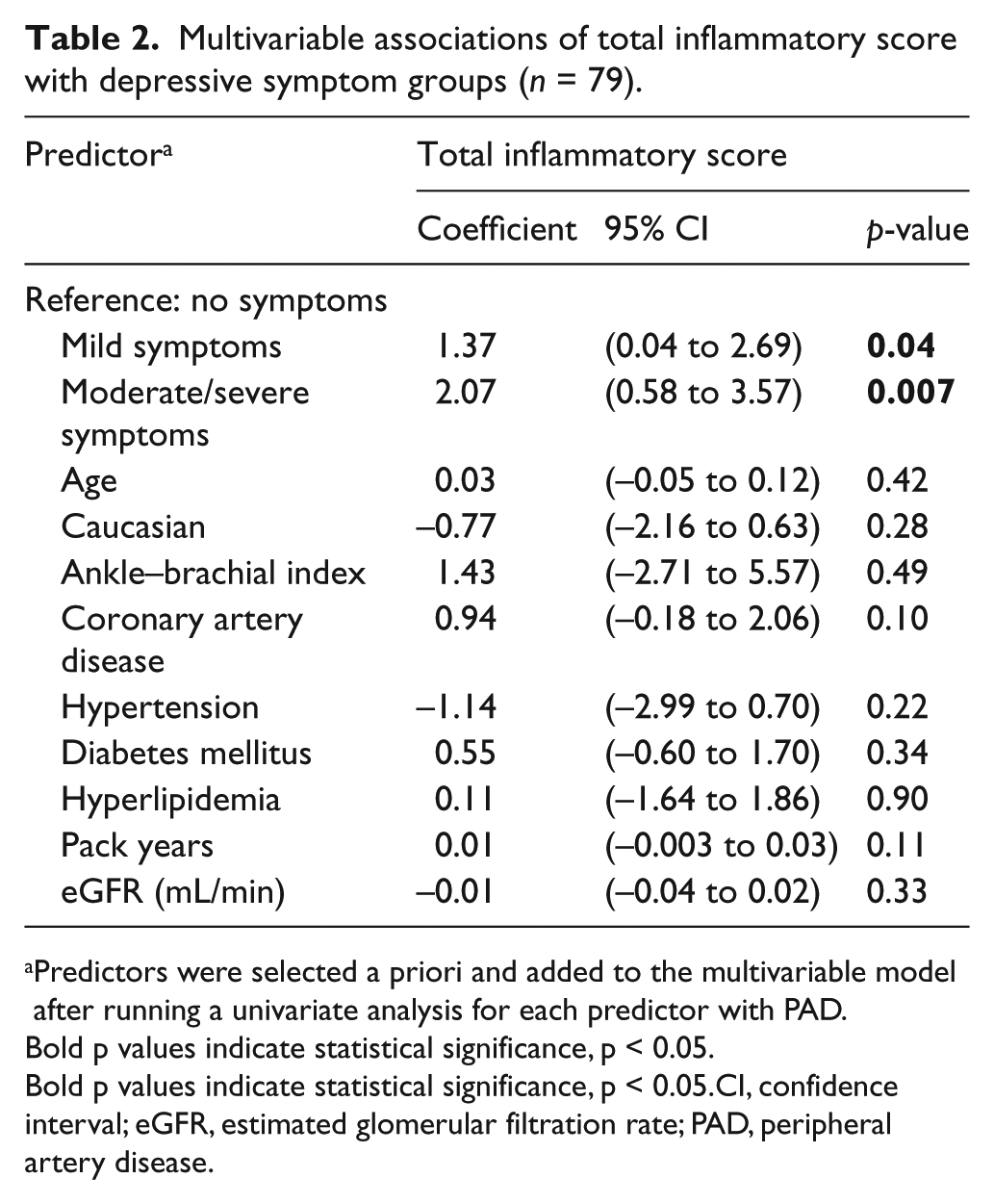
Predictors were selected a priori and added to the multivariable model after running a univariate analysis for each predictor with PAD.
Bold p values indicate statistical significance, p < 0.05.
Bold p values indicate statistical significance, p < 0.05.CI, confidence interval; eGFR, estimated glomerular filtration rate; PAD, peripheral artery disease.
Unadjusted analyses examining individual inflammatory markers found that hs-CRP and IL-6 increased with depressive symptom severity, but this association did not reach statistical significance. Levels of ICAM-1 and TNF-α increased from the no symptoms groups to the mild symptoms group but did not continue to rise in the moderate/severe symptoms group (Figure 2). In a multivariable analysis, IL-6 was marginally higher in the mild symptoms group (p = 0.06) and significantly higher in the moderate/severe symptoms group (p = 0.006), compared to the no symptoms group. Five patients had IL-6 values above the upper limit of reliably detectable levels for the assay (> 10 pg/mL), so their values were dropped from the main analysis. However, a sensitivity analysis was performed with these five values reported as 9.99 pg/mL and the results were unchanged. Additionally, hs-CRP levels were marginally higher in both the mild symptoms group (p = 0.11) and the moderate/severe symptoms group (p = 0.07) compared to the no symptoms group, although the results did not reach statistical significance. ICAM-1 was elevated in the mild symptoms group (p = 0.05), but not in the moderate/severe symptoms group (p = 0.17). TNF-α levels were not associated with depressive symptoms in this study. Please see the supplementary material for full results from the multivariable analysis of individual inflammatory markers.
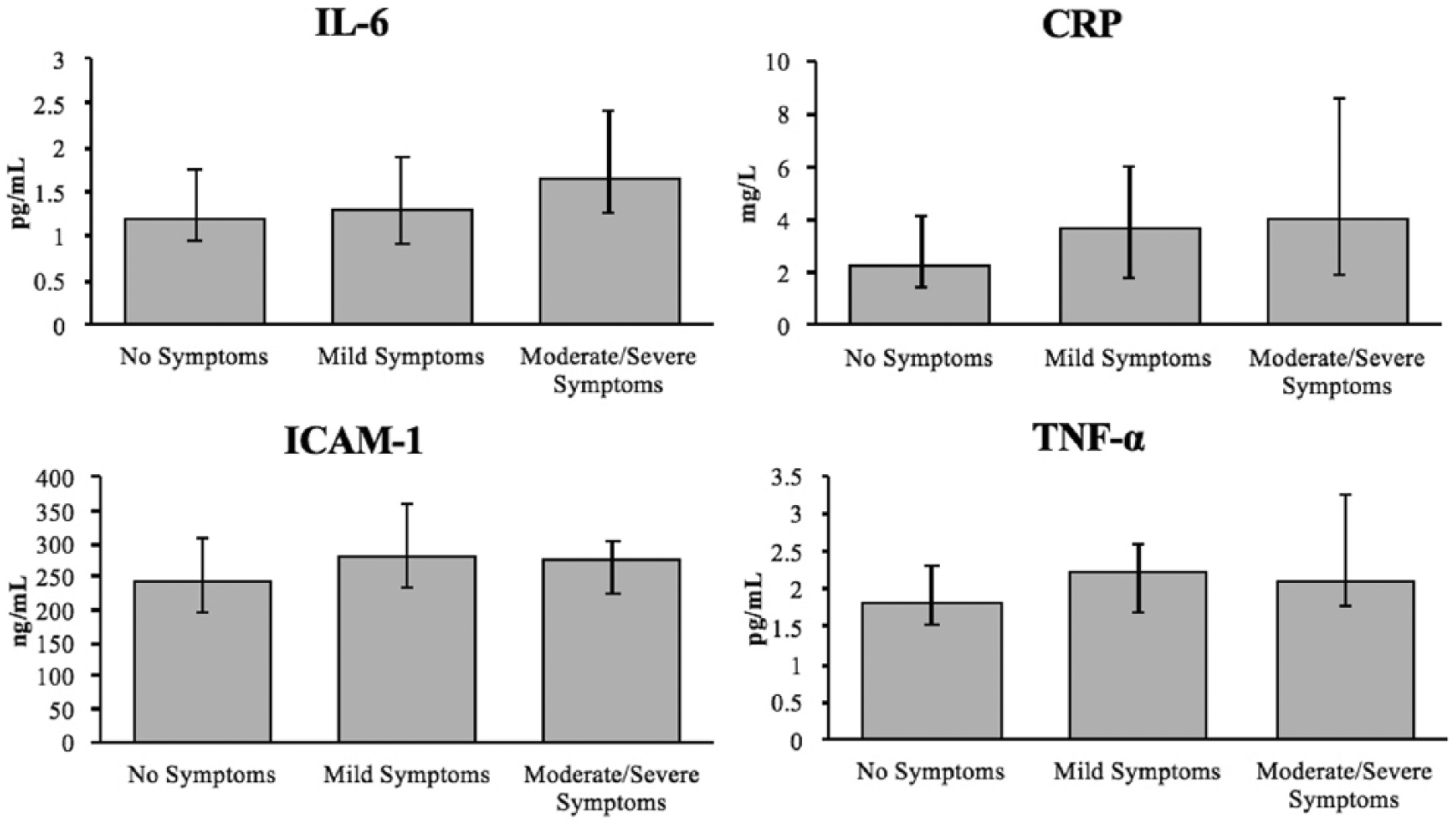
Unadjusted comparisons of individual inflammatory markers with depressive symptom groups show trends of increase with worsening symptoms: CRP (p = 0.06), IL-6 (p = 0.10), ICAM-1 (p = 0.15), and TNF-α (p = 0.27). The medians and IQRs of the inflammatory markers are depicted.
Discussion
The results of the present study demonstrate a significant association between depressive symptom severity and systemic inflammation in patients with PAD, indicating immune activation may be a common underlying factor for PAD and comorbid depressive symptoms. These findings in PAD complement and support those previously reported in depressed patients with coronary heart disease (CHD). 41 While cardiovascular morbidity and mortality in depressed patients can be partially explained by behavioral determinants such as decreased exercise, higher rates of smoking, and worse adherence to medications,42,43 there remains an unexplained, independent link between depression and vascular health suggestive of additional pathophysiological mechanisms.10,15,43 By controlling for ABI (a measure of PAD severity) and several cardiovascular risk factors, the results of the present study suggest an independent relationship between depression severity and heightened inflammation.
Previous evidence from prospective cohort studies have found that the relationship between depression and inflammation may be bidirectional, with data demonstrating both that depression scores predict IL-6 levels 44 and that inflammation predicts the development of depressive symptoms. 45 Recent literature outlines the complex manner by which these processes may modify one another.31,46 One such example is that during a biologic stressor, activation of the immune system influences the brain to produce psychosocial changes called ‘sickness behaviors’: fatigue, anhedonia, and social isolation. This evolutionary reaction, likely meant to conserve energy for the host to fight off infections, also reflects behaviors seen in depression. 47 Additionally, inflammatory cytokines have been found to cause direct damage to neurons. 48 Indeed, increasing evidence suggests a role of inflammation in the pathogenesis of depression, at least in a subset of depressed patients. 49 In exploring the means by which depression may lead to heightened inflammation, research has found lower levels of serum omega-3 fatty acids (which have anti-inflammatory properties) in depressed individuals. 50 The psychosocial stress of depression may also directly trigger an inflammatory response through a complex biomolecular chain reaction. Miller and Raison describe a pathway involving the activation of inflammasomes and nuclear factor-κB by stress-induced catecholamines, thus stimulating the release of inflammatory cytokines, and further exacerbating depression pathology. 31 Additionally, hypercortisolemia, seen in a subset of depressed individuals, can lead to insufficient glucocorticoid signaling through down-regulation of leukocyte glucocorticoid receptors and, hence, disinhibited inflammatory cytokine release. 41
The role of inflammation in PAD is comparatively more well-established51,52 – both as a risk factor 27 as well as a predictor of progressive atherosclerosis, 53 than the connection between depression and PAD. In fact, higher levels of hs-CRP have been associated with an increased incidence of adverse cardiovascular outcomes.54,55 Elevated levels of hs-CRP, IL-6, ICAM-1, and TNF-α were found to be predictive of symptomatic PAD events in the Women’s Health Study and the Heart and Soul Study.56,57 This may be due to cytokines directly exacerbating atherosclerosis and plaque destabilization.58,59 Patients with worse inflammation also experience greater mortality60,61 from predominantly cardiovascular causes. 62
Aside from inflammation, other potential mediators of the depression–vascular health relationship include dysregulation of the hypothalamic-pituitary-adrenal (HPA) axis63,64 and autonomic nervous system, 65 reduced heart rate variability, 66 altered platelet activity, 67 and endothelial dysfunction. 67 Recent review articles on depression and CVD acknowledge the potential function of immune markers as mediators due to their role in both disease processes. 68 Investigations examining this relationship in PAD are lacking; however, there are a few studies that explore the role of the immune system in depressed individuals with CVD. One nested case study within the Prospective Epidemiological Study of Myocardial Infarction (PRIME) found that depressed participants had a significantly higher incidence of CHD events, and there was a significant association between depressive symptoms and elevated levels of hs-CRP, IL-6, and ICAM-1 after adjustment for behavioral and social factors. 69 Another study within the Women’s Ischemia Syndrome Evaluation (WISE) found that depression and inflammation were associated with each other and with CVD. However, inflammation only modestly contributed to the increased risk of CVD events in depressed individuals. 70 Given the paucity of research investigating PAD and mental health, the current study serves to further expand understanding of the relationship between the two, with the ultimate goal of preventing PAD progression and mortality in patients suffering from depression.
Moving forward, prospective cohort studies are needed to establish causality and directionality between depression and inflammation in PAD. Additionally, further investigations should be conducted to deduce the precise pathophysiological mechanisms responsible. Future studies may seek to further characterize inflammatory processes by the biomarkers studied in this paper in addition to cytokines that have newly discovered involvement in atherosclerosis, such as ADAMTS-5 and NUS1. 71 A particularly important focus for future research is whether treatment of comorbid depression improves PAD outcomes, in tandem with decreasing inflammatory markers. A recent study using a large VA database found that the link between depression and increased limb amputation and death in patients with PAD was attenuated when depressed patients were taking antidepressants, but it is not yet known whether this was through decreased inflammation or other mechanisms. 72 By better defining the impact of mental health on PAD, guidelines can be modified to identify patients with PAD who would most benefit from psychiatric treatments. Aside from pharmacologic treatment, efforts could be made to encourage positive health behaviors that impact both PAD and depression outcomes, such as increased physical activity.7,42
The American Heart Association currently recommends routine screening of all CVD patients for depression. 73 In the Heart and Soul Study, CAD patients who scored a 10 or higher on the PHQ-9 had an adjusted 41% greater rate of cardiovascular events. 74 Women and African Americans are more likely to have depression and PAD, respectively.75,76 Therefore, it would be crucial to address the impact of depression on PAD in more diverse populations in order to not perpetuate further health disparities.
Limitations
The observations made serve to generate further hypotheses to advance our understanding of mechanisms underlying the association between depression and PAD. The lack of longitudinal data and cross-sectional design of the present study limits the ability to make causal or directional conclusions. As this study examined a cohort of patients originally recruited for the OMEGA-PAD studies, the researchers were limited in subject selection. Additionally, the homogeneity of the patient population, which reflects the VA population, may not be generalizable to the US population at large. Other limitations include the small sample size, which reduced power to detect differences in individual inflammatory markers, and the data were incomplete for some socioeconomic factors, such as homelessness, which may contribute to health and inflammation. 77
Although the present study seeks to elucidate the connection between depression and PAD, it must be noted that the depression–inflammation link is not specific to PAD and may be seen in a variety of conditions. Additionally, while there is evidence that treatment of depressive symptoms can reduce inflammation and vice versa, there are currently no studies examining the effects of such treatment in patients with PAD.78,79
Conclusions
In conclusion, a cross-sectional analysis of patients with PAD revealed an independent association between depression, as measured by the PHQ-8, and inflammation, as measured by peripheral levels of several inflammatory markers. Specifically, individuals with moderate/severe depressive symptoms demonstrated elevated total inflammatory scores and IL-6 levels compared to patients with mild or no depressive symptoms. The findings of this study provide further evidence that inflammation is associated with co-occurring depression and PAD, although this study design cannot address causality or mediation. Prospective research should examine the directionality between depression and inflammation in PAD in more diverse populations. Such findings would inform future studies investigating effective treatment strategies and screening guidelines for depressed individuals with PAD, and thus better serve patients at higher risk for adverse outcomes.
Supplemental Material
VMJ787640_Supplemental_Material – Supplemental material for Depression severity is associated with increased inflammation in veterans with peripheral artery disease
Supplemental material, VMJ787640_Supplemental_Material for Depression severity is associated with increased inflammation in veterans with peripheral artery disease by Natalie VM Hernandez, Joel L Ramirez, Sukaynah A Khetani, Kimberly A Spaulding, Warren J Gasper, Jade Hiramoto, Daniel Lindqvist, Owen M Wolkowitz, Nancy K Hills, S Marlene Grenon and Greg J Zahner in Vascular Medicine
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: this project was supported by the National Center for Advancing Translational Sciences, National Institutes of Health, through UCSF-CTSI Grant Number TL1 TR001871, with additional student research support from the Society for Vascular Surgery Student Research Fellowship Award and the American Heart Association Student Scholarship (GZ). Furthermore, this work was supported by start-up funds from the University of California, San Francisco and the Northern California Institute for Research and Education, by Award Number KL2RR024130 from the National Center for Research Resources, Award Number 1K23HL122446-01 from the National Institutes of Health/National Heart, Lung, and Blood Institute, and a Society for Vascular Surgery Seed Grant and Career Development Award (SMG). The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Center for Research Resources or the National Institutes of Health. DL was supported by the Swedish Research Council (registration number 2015-00387), Marie Sklodowska Curie Actions, Cofund (Project INCA 600398), the Swedish Society of Medicine, the Söderström König Foundation, the Sjöbring Foundation, the OM Persson Foundation, and the province of Scania (Sweden) state grants (ALF).
Supplementary material
The supplementary material is available online with the article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
