Abstract
Background:
Prior research has demonstrated that individuals with peripheral artery disease (PAD) often have comorbid opioid use disorder (OUD) and major depressive disorder (MDD), with limited data regarding their impact on readmission outcomes, length of stay, and cost. This study aimed to investigate these healthcare utilization outcomes in patients with PAD who have comorbid OUD and MDD.
Methods:
Data were obtained from the National Readmission Database from 2011 through 2018. The study population included all hospitalizations with PAD as the primary or secondary diagnosis, from which hospitalizations with OUD and MDD were extracted using appropriate ICD-9/10 diagnosis codes. Primary outcomes were 30-day and 90-day readmission, total cost, and total length of stay within the calendar year. We created hierarchical multivariable logistic regression models examining OUD with and without MDD, with a random effect for healthcare facility location.
Results:
From 2011 to 2018, 13,265,817 weighted admissions with PAD were identified. These admissions were segmented into four categories: No OUD/No MDD (12,056,466), OUD/No MDD (323,762), No OUD/MDD (867,641), and OUD/MDD (17,948). The group with No OUD/No MDD was used as the reference group for all subsequent comparisons. Regarding 30-day and 90-day readmissions, patients with OUD/MDD had odds of 1.14 (95% CI 1.10, 1.18) and 1.09 (95% CI 1.06, 1.13), respectively. Patients with OUD/No MDD bore the highest median cost of $64,354 (IQR $30,797–137,074), and patients with OUD/MDD marked the lengthiest median stay of 6.01 days (IQR 2.01–13.30).
Conclusion:
This study found a significant association between these comorbidities and outcomes and therefore calls for targeted interventions and pain management strategies.
Background
Peripheral artery disease (PAD) is a significant public health concern affecting approximately 230 million individuals globally. 1 This includes nearly 21.3 million people in the United States at an estimated economic cost of $283–539 billion annually in 2022.2,3 High opioid use is defined as receiving two or more opioid prescriptions within 1 year, and about 25% of individuals with PAD meet this criteria. 4 Given the national increase in opioid use and related deaths,4–6 as well as the susceptibility of vulnerable populations such as those with chronic pain conditions, chronic limb-threatening ischemia, postrevascularization, and/or amputation, there is a pressing need to understand the frequency of opioid use disorder (OUD) with or without major depressive disorder (MDD) in patients admitted with PAD.7–9
Individuals with PAD are more prone to experience depressive symptoms, which are linked with adverse prognostic outcomes such as increased mortality, 10 recurrence of symptomatic PAD, and failure of lower-extremity revascularization.11,12 Opioid use is also associated with the development of MDD, and individuals experiencing MDD are at a higher risk of extended opioid use. 13 A Mendelian randomization study suggests the possibility of a causative relationship between the risk for MDD and the genetic propensity for greater prescription opioid use, although it is unclear whether one condition predisposes an individual to the other, or if a shared genetic component increases susceptibility to both. 14 These intertwined relationships underscore the complexity of managing PAD in individuals experiencing these comorbidities, emphasizing the need for a better understanding of their impacts on healthcare utilization and outcomes.
The frequency of OUD with or without MDD in patients admitted with PAD, the patient risk profile associated with these comorbidities, and their impact on outcomes, are currently unclear. Therefore, this study had two primary aims. First, we sought to examine the prevalence of these factors and second, examine their association with 30-day and 90-day readmission outcomes, costs, and lengths of stay. The goal is to improve overall health outcomes and reduce healthcare utilization among patients with PAD with these comorbidities.
Methods
Data source
The data were obtained from the Nationwide Readmissions Database (NRD) from 2011 through 2018. The NRD is a subset of the Healthcare Cost and Utilization Project (HCUP) databases developed by the Agency for Healthcare Research and Quality, which focuses specifically on hospital readmissions.15,16 The NRD captures information on all US hospitalizations that led to unplanned readmissions within 30 days during each year. The database encompasses data from over 20 states, representing over 14 million hospitalizations each year. The stratified sampling of hospitals is based on criteria such as location, teaching status, and hospital size. Each hospitalization is deidentified and incorporated into the NRD as a unique observation, containing information about individual demographics, existing health conditions, 30-day readmission, 90-day readmission, associated costs, and length of stay. This study was exempt from Institutional Review Board approval, as the NRD is a publicly available, deidentified national database. This database has established its reliability and validity in depicting an accurate representation of hospital readmissions, confirmed by numerous studies, enhancing its credibility as a rich source for healthcare research.17–20
Study population
We initially identified all hospitalizations with PAD as the primary or secondary diagnosis using the International Classification of Diseases, Ninth Edition, Clinical Modification (ICD-9-CM) codes from 2011 to September 2015, and the International Classification of Diseases, Tenth Edition, Clinical Modification (ICD-10-CM) codes from October 2015 to 2018, as described in Table S1 in the online supplementary material. From these PAD admissions, we further categorized hospitalizations based on the presence or absence of OUD or MDD (exposures of interest) as a primary or secondary diagnosis, utilizing the appropriate ICD-9 and ICD-10 diagnosis codes. This delineation enabled us to form four distinct groups: patients diagnosed with PAD only (Group 1), individuals with PAD and OUD (Group 2), individuals with PAD and MDD (Group 3), and individuals with PAD who have both OUD and MDD concurrently (Group 4). Our use of these ICD codes is supported by the findings of Anantha-Narayanan et al., which have demonstrated the reliability of these codes in identifying PAD and related conditions in large datasets. 21
Exposure variables
We defined the exposure variable OUD using ICD-9 and ICD-10 codes. Specifically, OUD is defined as opioid use leading to significant impairment or distress, manifested by at least two of the symptoms outlined in (Table S2), occurring within a 12-month period. 22 These codes were also used to define the variable MDD, characterized by two core symptoms persisting for a minimum of 2 weeks, accompanied by at least three additional symptoms. 22
Study outcomes and definition
Primary outcomes were readmission at 30 and 90 days, total associated costs, and the overall length of hospital stay. Owing to the nature of the NRD, which only links patient records within the same calendar year, the readmission rates are only recorded if they were within the same year. As such, for the 30-day readmission analysis, we included patients admitted from January through November. Similarly, for the 90-day readmission analysis, only patients admitted from January through September were considered to allow for the necessary follow-up within the calendar year. The total cost and length of stay were calculated over all linked records within the respective same year.
Other reported outcomes were amputations, revascularizations, in-hospital mortality, and major adverse cardiac events (MACE), which included ST-elevation myocardial infarctions (STEMI), non-ST-elevation myocardial infarctions (NSTEMI), and ischemic strokes.
Other variables
Patient demographics including age, sex, income, payer, rural/urban, elective admission, and disposition were reported. Additionally, comorbidities such as cardiac conditions, obesity, hypertension, coronary artery disease, heart failure, diabetes, chronic kidney disease, smoking, chronic limb-threatening ischemia, valve disease, and prior amputation were recorded (Table 1). Given the high prevalence of other mental health conditions in patients with PAD, we also examined several other mental health comorbidities, including schizophrenia, bipolar disorder, anxiety, and stress (Table 1). These other mental health disorders are associated with increased hospital readmissions in other populations, and chronic pain may also influence health outcomes and healthcare utilization.23,24
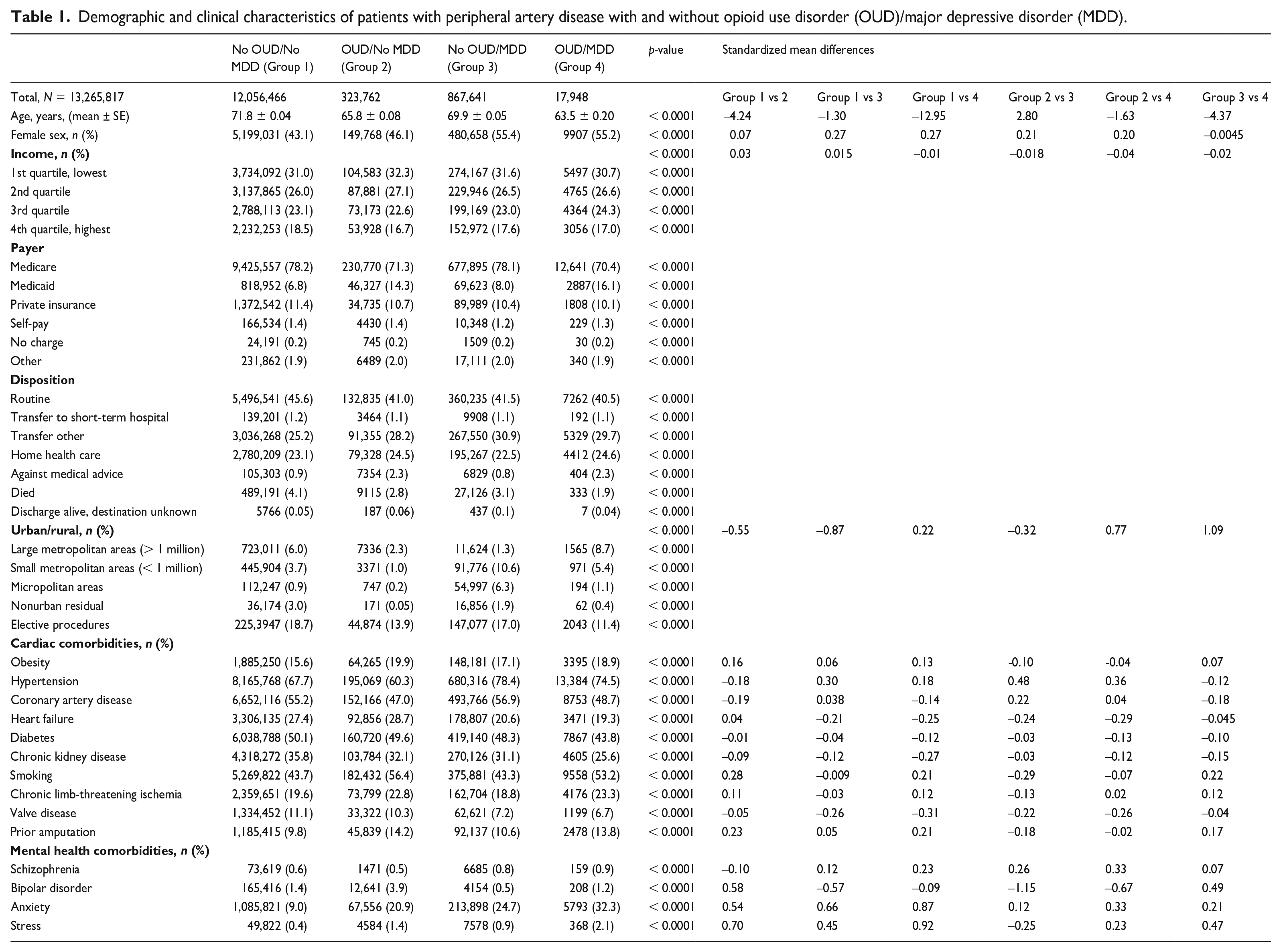
Demographic and clinical characteristics of patients with peripheral artery disease with and without opioid use disorder (OUD)/major depressive disorder (MDD).
Importantly, we also focused on pain-related disorders, including chronic limb-threatening ischemia, osteoarthritis, and nonspecific back and joint pain, as these factors have been associated with increased hospital readmissions and may significantly influence health outcomes and healthcare utilization in individuals with PAD. Individuals with these conditions were identified based on ICD-9-CM and ICD-10-CM codes. Given their high prevalence in individuals with PAD and their potential to influence readmission rates, we included these comorbidities as potential confounders or effect modifiers. It is important to note that although these comorbidities were considered in the analyses, our primary focus remained on the associations of OUD and MDD with readmissions, cost, and length of stay in patients with PAD.
Statistical analysis
Data on patient demographics and comorbidities were presented as means with SD for continuous variables and frequencies with proportions for categorical variables. For continuous variables, we employed the Kruskal–Wallis test, and for categorical variables, we utilized the Rao–Scott chi-squared test. Standardized differences were calculated and used to compare baseline characteristics, with a standardized difference of 0.2 or less considered a small effect size. 25 For analysis, we distinctly categorized patients into four mutually exclusive groups based on OUD and MDD status: (1) No OUD, No MDD; (2) No OUD, MDD; (3) OUD, No MDD; and (4) OUD, MDD. This approach ensured that patients in one group were not included in another. A multilevel logistic regression, incorporating a random effect for each specific healthcare facility (defined by unique ID numbers in the database to represent distinct locations where care was delivered), was employed to analyze the association between OUD and MDD with readmissions whereas a quantile regression analysis was employed to assess the distribution of cost and length of stay. Covariates were selected based on a thorough review of existing PAD readmissions literature. Based on this literature, we selected covariates that have been consistently identified as risk factors for readmissions, including age, sex, history of chronic limb-threatening ischemia, hypertension, diabetes, coronary artery disease, heart failure, renal failure, obesity, smoking, and prior amputation. 26 To examine how the four groups based on their OUD and MDD status changed over a year, and their impact on readmissions over time, we conducted a multilevel ordinal logistic regression analysis among patients readmitted within 30 days with a random effect for healthcare facility location and adjusted for the same covariates identified above. Cost and length of stay were described as median (med) with IQR for each of the four groups based on their OUD and MDD status. Analysis was done using SAS 9.4 (SAS Institute Inc., Cary, NC, USA). The rates of missingness for selected covariates ranged from 0% to 3%, with complete case analysis performed to ensure the robustness of our findings.27,28
Results
During the years 2011–2018, a total of 13,265,817 weighted hospitalizations were diagnosed with PAD. These hospitalizations were segmented into four categories: No OUD/No MDD (12,056,466), OUD/No MDD (323,762), No OUD/MDD (867,641), and OUD/MDD (17,948).
Demographics and socioeconomic attributes
A significant age variation was evident among the groups. Patients with OUD/MDD recorded the youngest mean age of 63.5 ± 0.2 years, and patients with No OUD/No MDD were the oldest at 71.8 ± 0.04 years (p < 0.0001) (Table 1). A total of 55.4% of patients with No OUD/MDD were women; similarly, most patients with OUD/MDD (55.2%) were women (Table 1). For income, most of the groups were in the 1st quartile (p < 0.0001) (Table 1). Medicare was the prevailing payer across all classifications, with the highest being among patients with No OUD/No MDD, at 78.2% (p < 0.0001) (Table 1).
Comorbidities
Comorbidities like diabetes and hypertension bore notable prevalence. Patients with No OUD/No MDD manifested the peak prevalence of diabetes at 50.1%; however, there was no significant difference when compared with the other comorbid groups (d < 0.20). Patients with No OUD/MDD manifested the highest prevalence of hypertension at 78.4%, with a significant difference when compared to the No OUD/No MDD and OUD/No MDD group (d = 0.30, d = 0.48, respectively) (Table 1). Conversely, patients with OUD/No MDD displayed a considerable prevalence of smoking, reaching 56.4% (p < 0.0001) (Table 1). Cardiac comorbidities, such as coronary artery disease, were significantly prevalent. Patients with No OUD/MDD had the highest prevalence at 56.9% of cases with coronary artery disease, which showed a significant difference when compared with the No OUD/No MDD group (d = 0.22), whereas the patients with OUD/No MDD had the least at 47.0% (p < 0.0001) (Table 1). Patients with a history of chronic limb-threatening ischemia (CLTI) and prior amputation varied among the patients, with the OUD/MDD group showing the highest prevalence of 23.3% for CLTI (d < 0.20) and OUD/No MDD showing the highest at 14.2% for prior amputations, which showed a significant difference when compared with the No OUD/No MDD group (d = 0.23) (p < 0.0001) (Table 1). Mental health comorbidities presented distinctive variations. Anxiety, for instance, was substantially more pronounced in patients with OUD/MDD at 32.3% (d > 0.20), compared to the other groups with patients with No OUD/No MDD exhibiting the lowest anxiety rate at 9.0% (p < 0.0001) (Table 1).
Trends in 30-day and 90-day readmissions
In terms of likelihood of readmissions (0, 1, or 2+), patients with MDD/no OUD had the least increase in odds of readmission: odds ratio (OR) for one readmission: 1.05 (95% CI 1.04–1.06); OR for 2+ readmissions: 1.09 (95% CI 1.06–1.13). The highest risk was observed in patients with both OUD and MDD: OR for 1 readmission: 1.15 (95% CI 1.11–1.19); OR for 2+ readmissions: 1.48 (95% CI 1.35–1.63) (Table S3).
The overall 30-day readmission rate was 72.2% with the odds of 30-day readmission being highest for patients with OUD and MDD (odds ratio [OR] 1.14, 95% CI 1.10–1.18), followed by OUD without MDD (OR 1.11, 95% CI 1.09–1.13), compared with patients with no OUD and no depression. For 90-day readmissions, patients with OUD and MDD had the highest odds of readmission (OR 1.09, 95% CI 1.06–1.13), followed by OUD without MDD (OR 1.08, 95% CI 1.07–1.10) and no OUD with MDD (OR 1.05, 95% CI 1.04–1.06) when compared with no OUD and no MDD (Table S4). The study found that readmission odds increased annually from 2011 to 2018, with the highest odds of 30-day and 90-day readmissions observed in patients with OUD and MDD. Patients with OUD and MDD in 2018 had 6.81 (95% CI 5.56–8.35) times higher odds of readmission than those with no OUD and no MDD in 2011 (Table S5).
Cost and length of stay
The median cost was highest for those with OUD and no MDD (med $64,354, IQR $30,797–137,074), followed by OUD and MDD (med $61,932, IQR $29,831–132,059), no OUD and no MDD (med $52,477, IQR $25,538–111,125), and no OUD with MDD (med $51,122, IQR $25,070–$108,364) (Table S4). Length of stay was longest for patients with OUD and MDD (med 6.01, IQR 2.01–13.30), followed by OUD with no MDD (med 5.82, IQR 2.71–12.50) and no OUD with MDD (med 5.25, IQR 2.50–11.36) with length of stay of no OUD and no MDD being (med 4.69, IQR 2.17–10.00) (Table S4).
Hospitalization outcomes
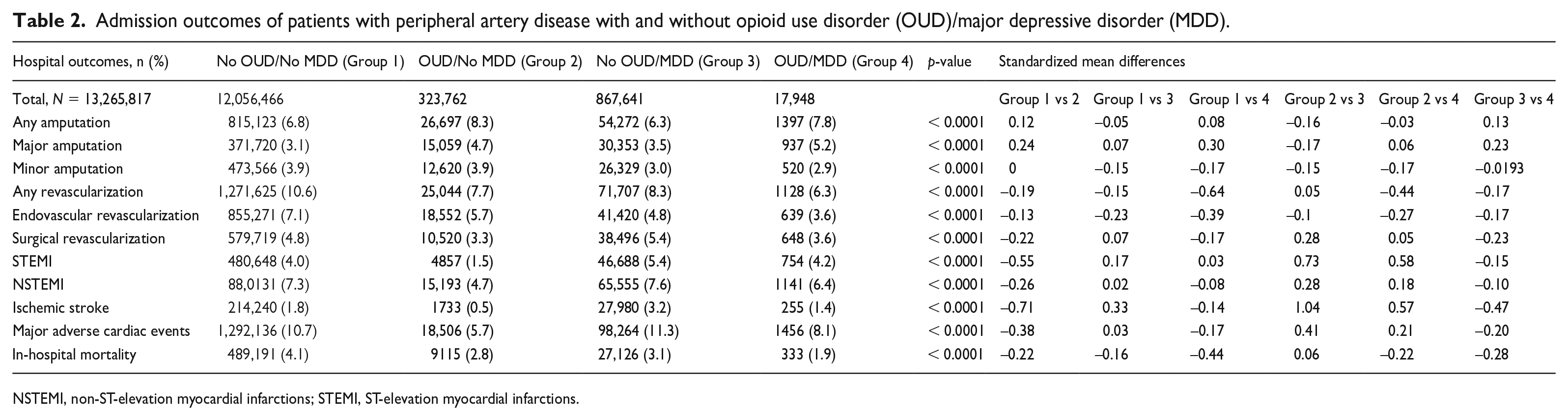
For revascularization procedures, patients without either condition had the highest rate at 10.6% but indicated a significant difference when compared with the OUD/MDD group (d = 0.64); those with OUD/MDD had the lowest rate of 6.3% (p < 0.0001 for all) (Table 2). In MACE outcomes, the No OUD/MDD group had the highest prevalence of 11.3% and was significant when compared with the OUD/No MDD and OUD/MDD groups (d > 0.20), yet patients with OUD/MDD registered the lowest at 8.1% (p < 0.0001 for all) (Table 2). Regarding in-hospital mortality, patients without either condition reported a rate of 4.1%, whereas those with OUD/MDD showed the lowest rate of 1.9% (p < 0.0001 for all) (Table 2). For major amputation, patients with OUD/MDD experienced the highest rate at 5.2%, which was significant when compared with the No OUD/No MDD and No OUD/MDD groups (d = 0.30, d = 0.23, respectively). This was followed by patients with OUD/No MDD at 4.7%, and patients with No OUD/No MDD recorded the lowest at 3.1% (p < 0.0001 for all) (Table 2).
Admission outcomes of patients with peripheral artery disease with and without opioid use disorder (OUD)/major depressive disorder (MDD).
NSTEMI, non-ST-elevation myocardial infarctions; STEMI, ST-elevation myocardial infarctions.
Discussion
In this study, we aimed to investigate the association between OUD, MDD, and readmission outcomes in patients with PAD using a large national database. A closer look at the patient profiles reveals distinct patterns among those diagnosed with PAD. Age-wise, patients with PAD who have concurrent OUD and MDD were generally younger, averaging about 63.5 years, as compared to those without either condition who averaged about 72 years. We observed variation in sex distribution across the groups. In the OUD/MDD group, approximately 55% of patients were women. Similarly, the No OUD/MDD group also had a female majority, comprising around 55% of the group. Conversely, the group with OUD/No MDD had a lower proportion of female patients, accounting for 46% of this group followed by the No OUD/No MDD group, which had a 43% female population. Socioeconomic disparities were evident, with a majority across all groups falling into the lowest income quartile. Our key findings suggest that patients with PAD who have OUD and MDD have more than 14% higher odds of readmission within 30 days and a 9% increase in the odds for a 90-day readmission compared to those without these comorbidities. Furthermore, we found that the odds of readmissions increased each year from 2011 to 2018 for all categories of opioid use and MDD, especially for patients with both comorbidities, who had 6.81 times the odds of 30-day readmission in 2018. Patients with OUD and MDD also had a longer stay, averaging about a median 2-day difference compared to patients with no comorbidities. Additionally, patients with both OUD and MDD had a median cost difference of more than $9455 compared to those without these comorbidities, highlighting the significant burden that OUD and MDD can place on healthcare utilization among patients with PAD.
OUD poses a significant hurdle for patients with PAD, as evidenced by previous studies highlighting elevated rates of opioid use and associated complications. Among these complications are in-hospital issues such as acute renal failure and respiratory complications.7,29 Additionally, other studies have investigated the association between MDD and PAD and poor outcomes, including higher odds of major amputation among patients undergoing endovascular revascularization, longer median length of stay, and higher median cost.30–32 The previous studies have independently investigated the relationships between OUD and PAD, MDD and PAD, as well as OUD and MDD. If we visualize these relationships as a triangle, with PAD, OUD, and MDD forming the vertices, existing research has delved into the connections between each pair of vertices, but the interplay of all three conditions – the entire triangle – has not been the central focus. Our study seeks to fill this gap in the current literature by exploring the impact of the co-existence of OUD and MDD on patients with PAD. In essence, our study considers the entire triangle, thereby expanding upon previous research and emphasizing the importance of taking into account both comorbidities when treating patients with PAD.
Our study primarily delved into the association between OUD, MDD, and readmission outcomes, uncovering significant demographic and hospitalization trends. We found that the OUD/MDD group was predominantly younger, this trend may reflect broader societal trends observed during this period, including increased awareness and diagnosis of opioid use and mental health issues. However, further research is needed to establish a direct causal link between these societal trends and the specific patterns observed in our study population.8,33 Surprisingly, the group without both OUD and MDD exhibited the highest in-hospital mortality, potentially due to older age, several co-existing conditions, and a higher number of admissions. Notably, the incidence of MACE was highest among patients without OUD but with MDD, suggesting a potential link between MDD and cardiac complications that merits deeper exploration. This is consistent with other studies that have posited MDD as a significant risk factor for adverse cardiovascular events.34–36 The elevated rates of STEMI and NSTEMI in this group further underscore the critical need for targeted cardiovascular monitoring in patients with MDD. Furthermore, data revealed that the highest major amputation rate was in patients with both OUD and MDD, followed by those having only OUD and No MDD. Though a significant number of studies have linked MDD to amputations,37–39 the association between OUD and amputations has been explored in a smaller number of studies and warrants further in-depth research. 40
PAD is a chronic disease, with one of its most prominent manifestations being significant pain, especially in the advanced stages of the disease, and associated functional limitations. Pain is a central experience in PAD, yet we know very little about the best management strategies and the burden of complex addiction patterns associated with opioid prescriptions for patients with PAD. Prior work found an average of 24.4% of patients with PAD met the high opioid use criteria in any given calendar year. 4 This was consistent with our work. The increase in odds ratios over the years, particularly for the OUD/No MDD group, signals a potential rise in the prevalence of opioid use among patients with PAD. By 2018, the odds for the OUD/No MDD group surged to 4.91, representing a nearly fivefold increase relative to the baseline year of 2011. This finding suggests that the population of patients with PAD may be at a greater risk for opioid use and use disorder. At this juncture, we must raise an interesting and crucial question: how do these rates of OUD in patients with PAD compare to those in other disease populations, particularly those with other kinds of chronic pain conditions? Answering this question can offer two key insights. First, by comparing OUD rates, we can identify whether patients with PAD are uniquely vulnerable to developing OUD, or if this high prevalence is a common feature among chronic pain conditions. Second, understanding this comparison may help us adapt and implement successful strategies that have worked in managing OUD in other disease populations. As such, further studies are needed to draw these comparisons, and consequently, devise effective treatment strategies for patients with PAD who have OUD and MDD.
Patients with PAD are also known to be at risk of experiencing MDD. Patients diagnosed with MDD and PAD have an increased risk of mortality, worse revascularization patency, and increased progression of PAD.10,12,37 Our study documents an additional 32% increase in the odds of 30-day readmission in 2018 and a greater length of stay compared to patients with no MDD and no OUD. OUD and MDD often co-occur. Studies have found an increased risk of depression in patients with opioid dependence and other studies have found an increased risk of opioid misuse in depressed patients.14,41 Depression and prescription opioid use have a bi-directional relationship. 42 There is, however, a paucity of information on this reciprocal association between patients with PAD and outcomes. In our study, we compared individuals without comorbidities to assess the impact of MDD and OUD on patient outcomes. We found that patients with both MDD and OUD had the highest risk of poor outcomes. Given the reciprocal relationship between MDD and OUD, it is vital for clinicians to recognize this association and remain vigilant when diagnosing one condition to monitor the presence of the other.
Our study highlights the need for targeted prevention and pain management strategies for patients with PAD who are at risk for OUD and MDD. These interventions may include screening for MDD and substance use disorders, with care linkage to integrated behavioral health services and multimodal pain management programs. The results of this study emphasize the importance of implementing nonopioid pain management strategies in patients with PAD. As OUD and MDD are highly prevalent in the PAD population, they are likely to contribute to an increased risk of complications and readmissions, which underscores the need to explore alternative pain management approaches that can reduce the reliance on opioids and, subsequently, minimize adverse outcomes. As we move forward, it becomes imperative to champion integrated care models. Such models, which cater to both physical and mental health dimensions, can holistically address the needs of patients with PAD. Integrating multidisciplinary care that involves collaboration between vascular specialists, pain management experts, mental health professionals, and other allied health providers can facilitate the implementation of comprehensive nonopioid pain management plans tailored to individual patients’ needs. Our research underscores that by embracing such comprehensive approaches, we can potentially mitigate the adverse outcomes tied to these comorbidities and significantly improve the quality of life for patients with PAD.
Study limitations
Our study has some limitations that should be considered. First, administrative data from which ICD codes are derived tend to be specific but not sensitive when used for PAD case ascertainment. This suggests a probable under-diagnosis of PAD in community settings, indicating that relying solely on administrative data might not robustly identify all forms of PAD. 43 Next, the NRD, which only records readmissions within the same year, could underestimate actual read-mission rates. It also only reflects outcomes for patients readmitted, excluding those not seeking care after discharge. The NRD does not cover the entire US, nor does it provide certain demographic details like race/ethnicity, but it represents a diverse geographical spread. The study also lacked details about PAD severity, specific opioid medications, dosages, and prescription changes over time, potentially under-representing opioid dependence in patients with PAD. Though our dataset offers insights into revascularization procedures, it does not allow us to conclusively determine the exact condition or disease for which the opioid was prescribed. Even though PAD was our study’s primary concern, we cannot definitively verify if opioids were always prescribed for PAD or another condition. Furthermore, concerning understanding prior vascular procedures on these patients, the NRD provides specific information about revascularization procedures, distinguishing between surgical and endovascular procedures. Yet, for vascular procedures beyond revascularization, our dataset does not provide a comprehensive historical overview. This creates a limitation in terms of acquiring a full picture of the vascular interventions a patient may have undergone. Addressing these in future research would yield a clearer understanding of opioid use disorder and MDD in patients with PAD.
Conclusion
Our study findings confirm that patients with PAD who have comorbid OUD and MDD face a significantly increased risk of readmission, longer length of stay, and higher costs compared to those without these comorbidities. This emphasizes the importance of addressing both pain and mood disorders in the management of PAD, as they are closely interconnected and have a profound impact on patient outcomes.
Supplemental Material
sj-pdf-1-vmj-10.1177_1358863X241228540 – Supplemental material for Impact of comorbid opioid use disorder and major depressive disorder on healthcare utilization outcomes in patients with peripheral artery disease: A National Readmission Database analysis
Supplemental material, sj-pdf-1-vmj-10.1177_1358863X241228540 for Impact of comorbid opioid use disorder and major depressive disorder on healthcare utilization outcomes in patients with peripheral artery disease: A National Readmission Database analysis by Kelvin Amenyedor, Megan Lee, Miguel Algara, Waleed Tariq Siddiqui, Madeleine Hardt, Gaëlle Romain, Carlos Mena-Hurtado and Kim G Smolderen in Vascular Medicine
Footnotes
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Dr. Mena-Hurtado receives unrestricted grant funding from Janssen, Philips, Abbott, Shockwave, and Merck and serves as a consultant for Abbott, Cook Medical, Terumo, and Penumbra. Dr. Smolderen receives unrestricted grant funding from Janssen, Philips, Abbott, Shockwave, and Merck and serves as a consultant for Happify, Cook Medical, and Terumo.
Funding
Research reported in this publication was supported by the National Center For Complementary & Integrative Health of the National Institutes of Health under Award Number R21AT012430.The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Institutes of Health.
Supplementary material
The supplementary material is available online with the article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
