Abstract
Background:
Although the 1-year clinical outcomes of fluoropolymer-based drug-eluting stents (FP-DES) were favorable for the treatment of real-world femoropopliteal lesions in symptomatic peripheral artery disease (PAD), their performance beyond 1 year remained unknown. The current study determined the 3-year clinical course of FP-DES implantation for real-world femoropopliteal lesions.
Methods:
This multicenter, prospective, observational study evaluated 1204 limbs (chronic limb-threatening ischemia, 34.8%; mean lesion length, 18.6 ± 9.9 cm, chronic total occlusion: 53.2%) of 1097 patients with PAD (age, 75 ± 9 years; diabetes mellitus, 60.8%) undergoing FP-DES implantation for femoropopliteal lesions. The primary outcome measure was 3-year restenosis. The secondary outcome measures included 3-year occlusive restenosis, stent thrombosis, target lesion revascularization (TLR), and aneurysmal degeneration.
Results:
The 3-year cumulative occurrence of restenosis was 27.3%, whereas that of occlusive restenosis, stent thrombosis, and TLR was 16.1%, 7.3%, and 19.6%, respectively. The annual occurrence of restenosis decreased by 12.0%, 9.5%, and 5.8% in the first, second, and third year, respectively (p < 0.001). Similarly, the rates of occlusive restenosis and stent thrombosis decreased (p < 0.001 and p = 0.007, respectively), whereas the rate of TLR remained unchanged for 3 years (p = 0.15). The incidence of aneurysmal degeneration at 3 years (15.7%) did not significantly differ from that at 1 and 2 years (p = 0.69 and 0.20, respectively).
Conclusions:
This study highlights the favorable long-term clinical course of FP-DES in real-world practice, emphasizing the importance of monitoring for occlusive restenosis and stent thrombosis while considering the potential onset of aneurysmal degeneration.
Background
Recent clinical trials have revealed that drug-eluting endovascular therapies significantly reduce the rates of restenosis and target lesion revascularization (TLR) compared with conventional therapies for femoropopliteal lesions.1–3 Consequently, these therapies have been positioned as first-line strategies for endovascular femoropopliteal revascularization. 4 The femoropopliteal artery is subject to frequent flexion, bending, and compression. In such cases, nonstenting drug-coated balloon (DCB) treatment is preferred over drug-eluting stent (DES) treatment.5–7 However, standalone DCB treatment presents challenges such as recoil and dissection, especially for complex lesions. Thus, stent-based strategies are favored for treating such complex lesions.8–10
Clinical studies have reported that fluoropolymer-based drug-eluting stents (FP-DES) have a low 1-year restenosis rate, yet aneurysmal degeneration and stent thrombosis, on which clinical trials have not focused, have emerged as rising safety concerns.10,11 Notably, the long-term clinical outcomes after FP-DES implantation are yet to be revealed. Therefore, this study aimed to reveal the 3-year clinical outcomes, including restenosis, stent thrombosis, and aneurysmal degeneration, after FP-DES implantation for symptomatic atherosclerotic peripheral artery disease (PAD) in real-world practice.
Methods
We analyzed the 3-year database of the CAPSICUM (Con-temporary outcomes After Paclitaxel-eluting peripheral Stent implantation for symptomatic lower limb IsChemia with sUperficial feMoral or proximal popliteal lesion) study, a prospective multicenter observational study. Details of the CAPSICUM study have been reported elsewhere. 10 Briefly, the study enrolled adult patients (aged 20 years or older) primarily undergoing femoropopliteal implantation of an Eluvia DES (Boston Scientific, Marlborough, MA, USA) for symptomatic atherosclerotic femoropopliteal artery disease between February 2019 and June 2020. The study was conducted in accordance with the Declaration of Helsinki and approved by the ethics committees of the participating centers. Informed consent was obtained from the participants or their families if not possible.
Intervention
The details of the revascularization steps have been previously reported. 10 In brief, symptomatic PAD with femoropopliteal lesions presenting with ⩾ 50% stenosis, evaluated using imaging tests, is indicated for revascularization. The decision to perform Eluvia DES implantation was left to the discretion of the physicians, but all cases were treated with primary Eluvia DES implantation without any prior DCB intervention. A 6-Fr sheath was successfully inserted into the femoral artery using a contralateral approach. Generally, predilation and full-coverage strategies are used. The package insert of the Eluvia DES recommends dual antithrombotic therapy for at least 2 to 3 months after stent implantation. The use of intravascular ultrasound (IVUS) was recommended but was left to the discretion of the operators based on their usual clinical practice. Owing to their limited availability, atherectomy devices were not commonly utilized in Japan for the treatment of PAD.
Follow-up protocol
All participants were asked to visit their center annually after Eluvia DES implantation, with a time window of ± 2 months, for the evaluation of restenosis occurrence and the presence of aneurysmal degeneration, regardless of the presence of ischemic symptoms.
Definitions
As previously reported, restenosis was defined as a peak systolic velocity ratio > 2.4 based on duplex ultrasound (DUS) findings, whereas stenosis recurrence was defined as ⩾ 50% of the arterial diameter based on angiographic findings. 12 Restenosis was classified as nonocclusive or occlusive restenosis, and occlusive restenosis was further classified as nonthrombotic occlusive restenosis or stent thrombosis. Stent thrombosis was defined as occlusive restenosis that met the following criteria: (1) initial procedural success; (2) rapid symptom occurrence; (3) thrombus present during the procedure; and (4) lesion resolution with < 50% narrowing of the diameter by thrombolysis therapy. 13 TLR was clinically driven and defined as a reintervention performed for lesions with ⩾ 50% diameter stenosis identified by angiography within ± 5 mm of the target lesion after documentation of recurrent clinical symptoms of PAD. Aneurysmal degeneration was defined as the presence of a dark halo around the stent site on DUS. 10
Study outcomes
Restenosis was the primary outcome measure. Secondary outcome measures included occlusive restenosis, stent thrombosis, TLR, and aneurysmal degeneration.
Statistical analyses
Data are presented as means ± SD for continuous variables or as median (IQR) and percentage for categorical variables unless otherwise indicated. Statistical significance was set at p < 0.05, and 95% CIs were reported when appropriate. The occurrence of clinical outcomes is reported as a percentage. Of the cases in which a clinical outcome of interest occurred within 3 years after FP-DES implantation, those with an occurrence in the first year and those with an occurrence in the second year were compared using the chi-squared goodness-of-fit test. The proportions of occlusive restenosis, thrombosis, and TLR to any restenosis were compared between those in whom restenosis occurred in the first year and those in whom restenosis occurred in the second year using the chi-squared test of independence. The presence of aneurysmal degeneration at 3 years was compared with that at 1 year using McNemar’s test. Missing data were addressed using multiple imputations by the chained equations method, where we generated 10 imputed datasets and combined them with the analytic results. All statistical analyses were performed using R Statistical Software, v4.1.1 (R Foundation for Statistical Computing, Vienna, Austria).
Results
The 1-year outcomes of the CAPSICUM study have been published previously. 10 The baseline characteristics are shown in Table 1. Briefly, notable comorbidities included end-stage renal disease (n = 311, 28.4%) and chronic limb-threatening ischemia (n = 419, 34.8%). The average lesion length and reference vessel diameter were 18.6 ± 9.9 cm and 5.3 ± 0.9 mm, respectively. A history of revascularization, chronic total occlusion, bilateral calcification, and spot stenting was found in 200 (16.7%) 200, 640 (53.2%), 504 (41.9%), and 223 limbs (18.5%), respectively.
Baseline characteristics.
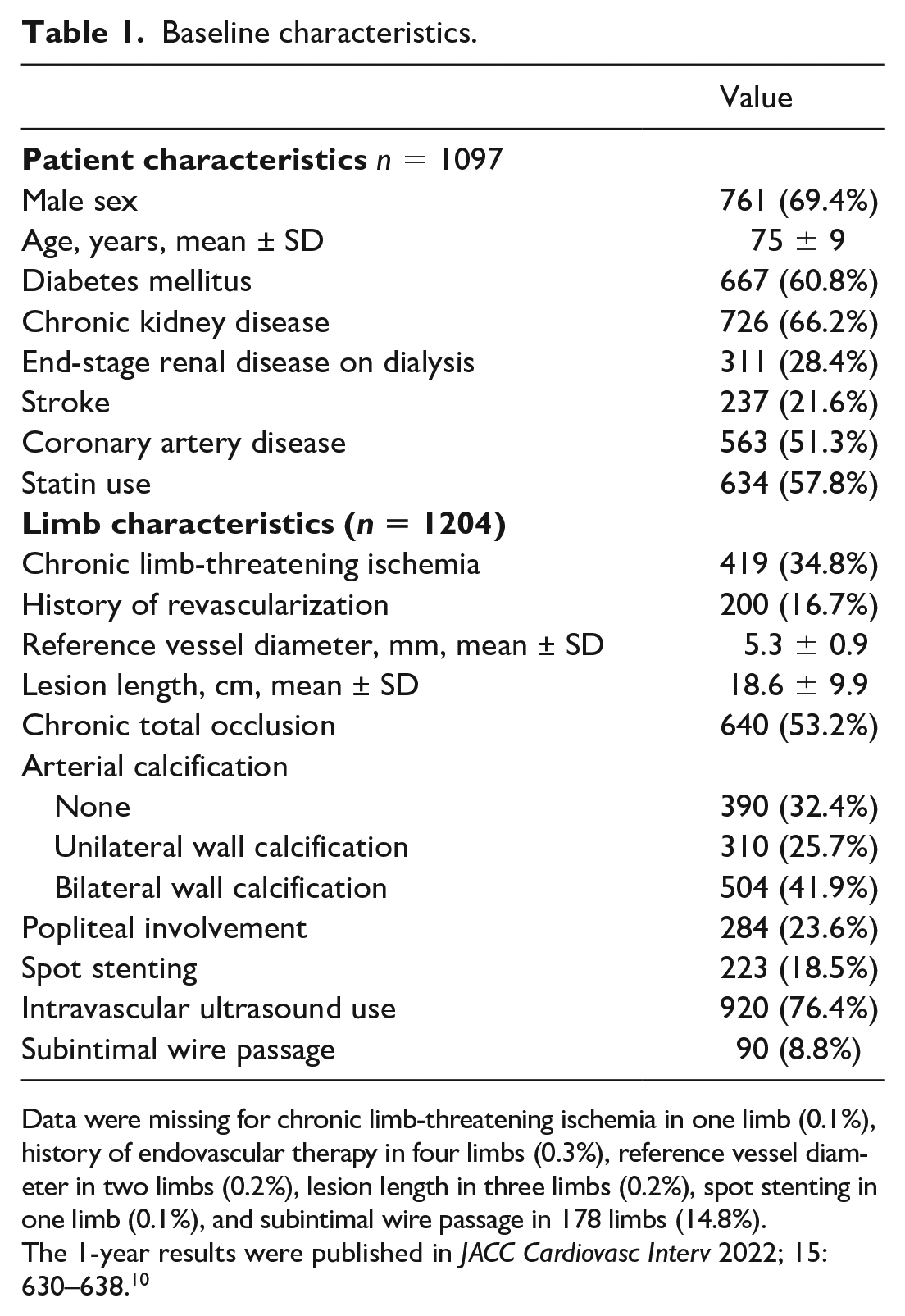
Data were missing for chronic limb-threatening ischemia in one limb (0.1%), history of endovascular therapy in four limbs (0.3%), reference vessel diameter in two limbs (0.2%), lesion length in three limbs (0.2%), spot stenting in one limb (0.1%), and subintimal wire passage in 178 limbs (14.8%).
The 1-year results were published in JACC Cardiovasc Interv 2022; 15: 630–638. 10
Data on 3-year restenosis and aneurysmal degeneration were available for 975 (81.0%) and 624 limbs (51.8%), respectively. As shown in Table 2, the 3-year cumulative incidence rate of restenosis was 27.3% [24.6–30.0%], indicating that the primary patency rate (i.e., freedom from restenosis) was 72.7% [70.0–75.4%]. The corresponding proportions of occlusive restenosis, stent thrombosis, and TLR were 16.1% [13.8–18.5%], 7.3% [5.8–8.9%], and 19.6% [15.3–24.0%], respectively. The annual occurrence of restenosis significantly decreased over the years (12.0% [10.1–13.9%] in the first year, 9.5% [7.5–11.4%] in the second year, and 5.8% [4.4–7.2%] in the third year; p < 0.001) (Figure 1). So was that of occlusive restenosis (7.9% [6.1–9.7%] at the first year, 5.5% [4.0–7.0%] at the second year, and 2.7% [1.6–3.8%] at the third year; p < 0.001) and stent thrombosis (3.4% [2.4–4.4%] at the first year, 2.6% [1.6–3.6%] at the second year, and 1.4% [0.7–2.0%] at the third year; p < 0.001) (Table 2). The proportions of occlusive restenosis and stent thrombosis to the overall restenosis are shown in Figure 3. The annual occurrence of TLR did not significantly differ between the years (7.7% [6.1–9.4%] in the first year, 5.8% [4.3–7.4%] in the second year, and 6.1% [2.7–9.5%] in the third year; p = 0.15) (Table 2).
Occurrence of adverse stent outcomes over 3 years.
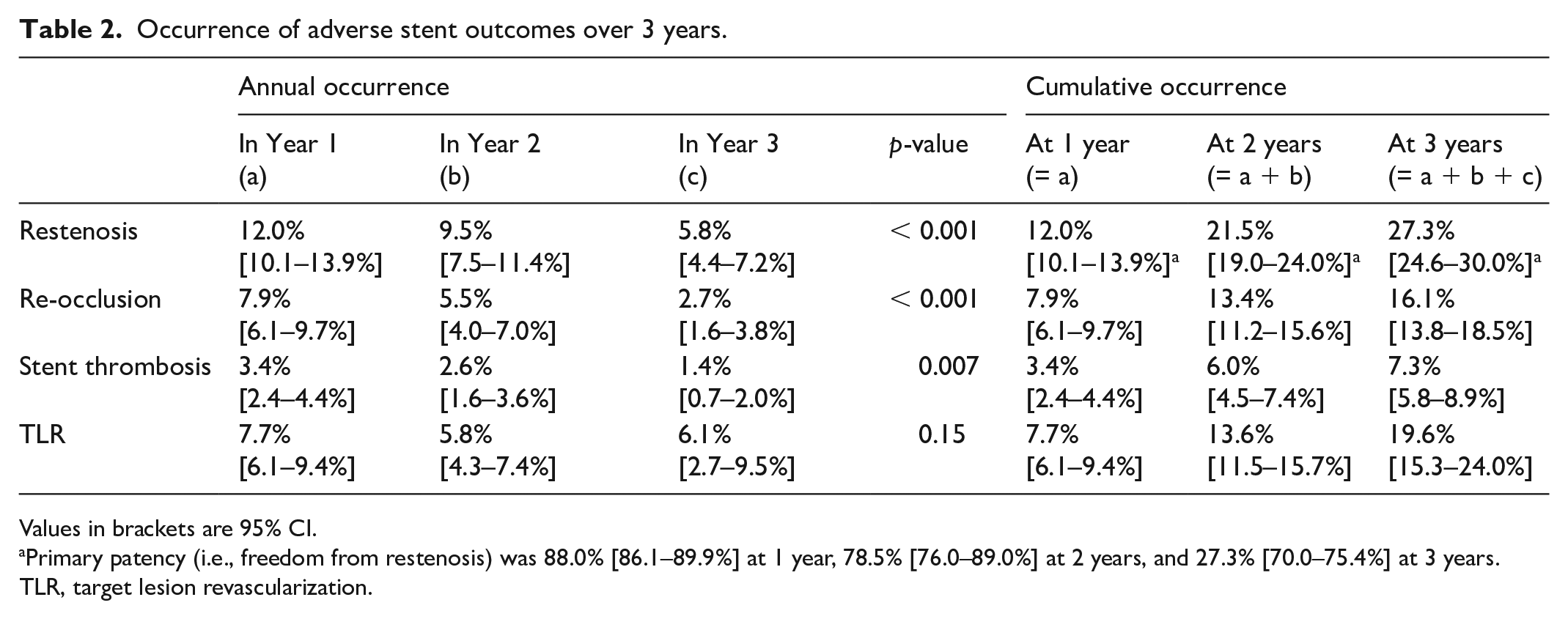
Values in brackets are 95% CI.
Primary patency (i.e., freedom from restenosis) was 88.0% [86.1–89.9%] at 1 year, 78.5% [76.0–89.0%] at 2 years, and 27.3% [70.0–75.4%] at 3 years.
TLR, target lesion revascularization.
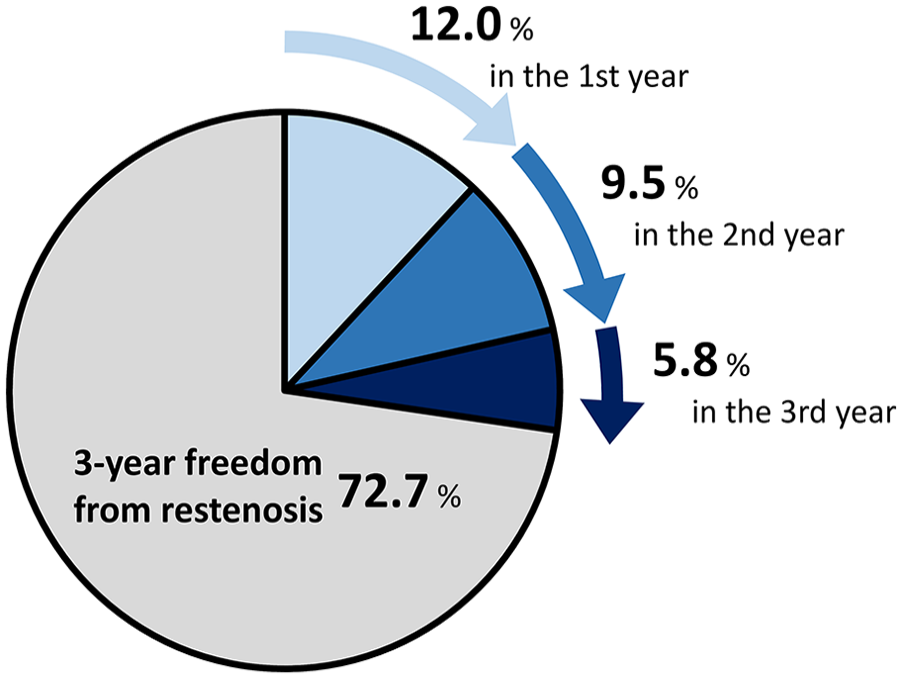
Three-year annual occurrence of restenosis after fluoropolymer-based drug-eluting stent implantation. The annual occurrence of restenosis significantly decreased over the years (12.0% [10.1–13.9%] in the first year, 9.5% [7.5–11.4%] in the second year, and 5.8% [4.4–7.2%] in the third year; p < 0.001).
The presence of aneurysmal degeneration was 16.2% [13.5–18.8%] at 1 year, 15.2% [10.5–20.0%] at 2 years, and 15.7% [12.3–19.1%] at 3 years, with no significant differences among the years (all p > 0.05) (Figure 2). The presence of aneurysmal degeneration was not associated with the 3-year risk of restenosis, with odds ratios of 1.10 [0.74–1.64] (p = 0.64), 1.35 [0.79–2.32] (p = 0.26), and 1.42 [0.87–2.31] (p = 0.16) for aneurysmal degeneration at the first, second, and third year, respectively.
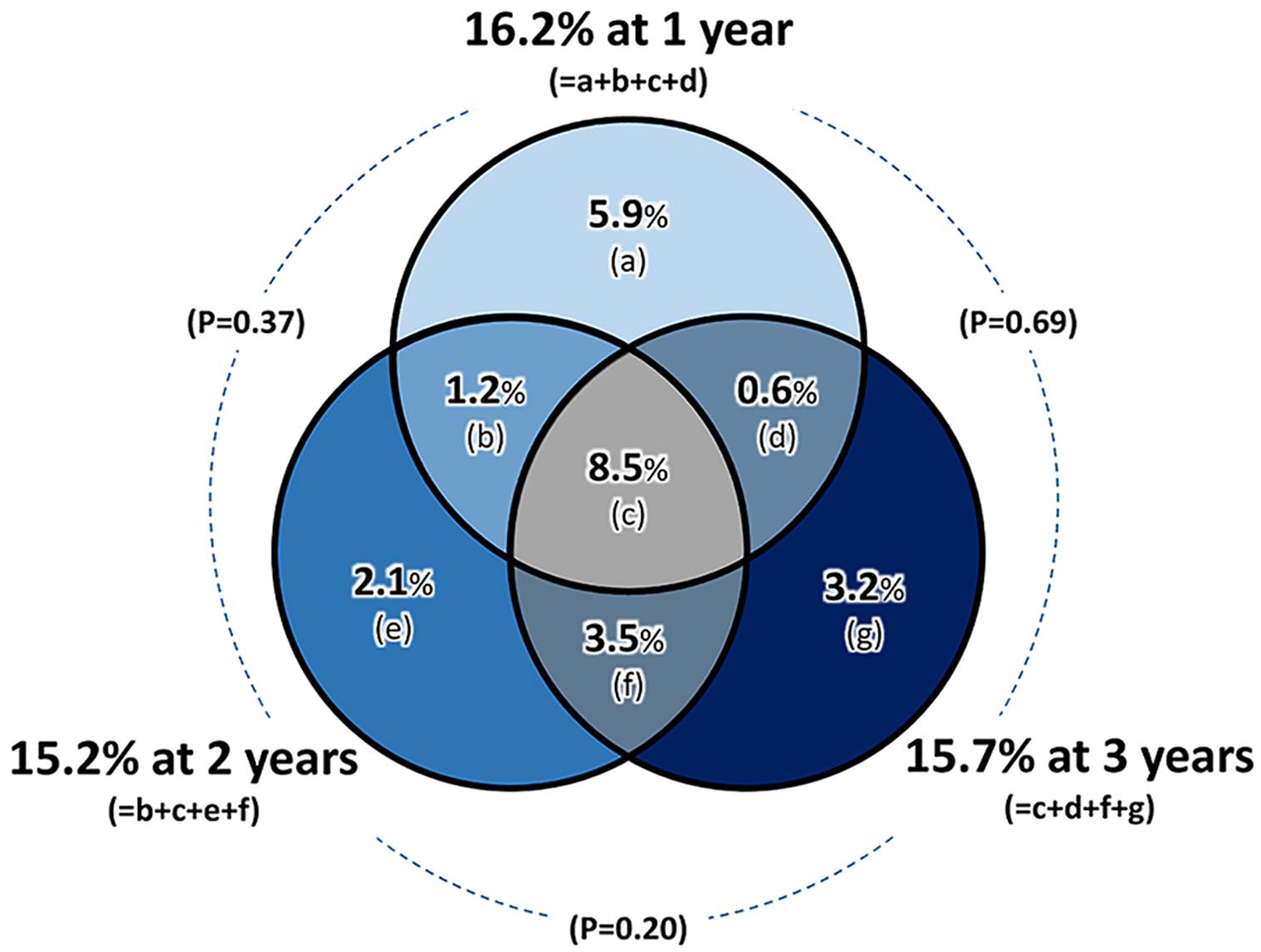
The presence of aneurysmal degeneration at 1, 2, and 3 years after fluoropolymer-based drug-eluting stent implantation. The presence of aneurysmal degeneration was 16.2% [13.5–18.8%] at 1 year, 15.2% [10.5–20.0%] at 2 years, and 15.7% [12.3–19.1%] at 3 years, with no significant differences among the years (all p > 0.05).
Discussion
The current study investigated the 3-year clinical course after FP-DES implantation in real-world practice. The cumulative occurrence of restenosis at 3 years was 27.3% [24.6–30.0%], indicating that the primary patency at 3 years was 72.7% [70.0–75.4%]. The annual incidence of restenosis has also decreased. In contrast, the frequency of aneurysmal degeneration at 3 years was 15.7% [12.3–19.1%] and was unchanged from that at 1 and 2 years (p > 0.05).
Three-year primary patency
The primary patency rate of FP-DES at 3 years was estimated to be 72.3% [70.0–75.4%]. The lower limit of the 95% CI was still higher than 66%, which is in compliance with the VIVA Physicians’ objective performance goal (OPG) of 1-year primary patency for bare nitinol stents. 14 Furthermore, the 3-year cumulative incidence of TLR was as low as 19.6% [15.3–24.0%]. Our findings suggest that FP-DES has acceptable long-term performance. It should be noted that we demonstrated acceptable performance of the FP-DES using real-world data rather than a clinical trial. Clinical trials aim to prove the safety and effectiveness of new devices and are often conducted in the ‘trial-based’ population (i.e., mainly consisting of lesions with mild-to-moderate severity in patients with mild-to-moderate comorbidities presenting with claudication or rest pain). By contrast, the real-world population often includes more severe and complex lesions in patients with severe comorbidities who present with tissue loss. Previous clinical studies on conventional endovascular devices, including DCB, polymer-free drug-coated stents (PF-DCS), and stent grafts, demonstrated that primary patency in real-world practice was generally lower than that reported in clinical trials.1,3,8,13,15,16 Our data suggest that the performance of FP-DES in real-world practice is equivalent to that demonstrated in clinical trials; the IMPERIAL trial reported that the 2-year primary patency and TLR rates of FP-DES were 83.0% and 12.7%, respectively, 17 whereas the corresponding 2-year rates in our study were 78.5% [76.0–89.0%] and 13.6% [11.5–15.7%], respectively.
The structural features of the FP-DES indicate that the release of antirestenotic drugs from the stent lasted for 12–15 months. In other words, drug release ceases 12–15 months after the implantation. Clinicians are concerned that the depletion of antirestenotic drugs and the exposure to uncoated polymers may increase the risk of restenosis in subsequent years. Contrary to this concern, the present study demonstrated that the occurrence of restenosis did not increase, but rather significantly decreased each year. This restenotic pattern seemed different from that of another drug-delivering device, DCB. 18 Several real-world studies on DCB have reported that its primary patency linearly decreases over the years, suggesting that drug delivery strategies themselves would be effective in delaying but not inhibiting restenosis in complex FP lesions. 19 By contrast, DES can decelerate incident restenosis, which can be attributed to its scaffolding structure.
Pattern of restenosis
The morphology of in-stent restenosis (ISR) is a clinically important determinant of prognosis after reintervention, including recurrent ISR and major adverse limb events. In particular, totally occlusive ISR has a dire prognosis after reintervention, with an extremely high incidence of repeated intervention and surgical bypass conversion.20–22 In the present study, occlusive restenosis and stent thrombosis occurred less frequently in the second and third years than in the first year, which may have been due to the progression of arterial repair at the stented segment after drug release. An angioscopic study reported that a significantly higher dominant neointimal coverage was observed in the late phase compared with the early phase, 23 which supports our findings. Concomitantly, angioscopy revealed that thr-ombus adhesion was still observed in the late phase, suggesting that arterial repair was still incomplete and that the risk of occlusive restenosis and stent thrombosis could not be completely eliminated. We found that the annual occurrence of occlusive restenosis and stent thrombosis decreased annually (Table 2), but at the same time, the proportion of occlusive restenosis and stent thrombosis to overall restenosis remained unchanged over the years at approximat-ely 60% and 30%, respectively (Figure 3). Notably, occlusive restenosis and stent thrombosis remain significant safety concerns, even after the drug release is depleted. Thus, careful follow-up is warranted years after FP-DES implantation.
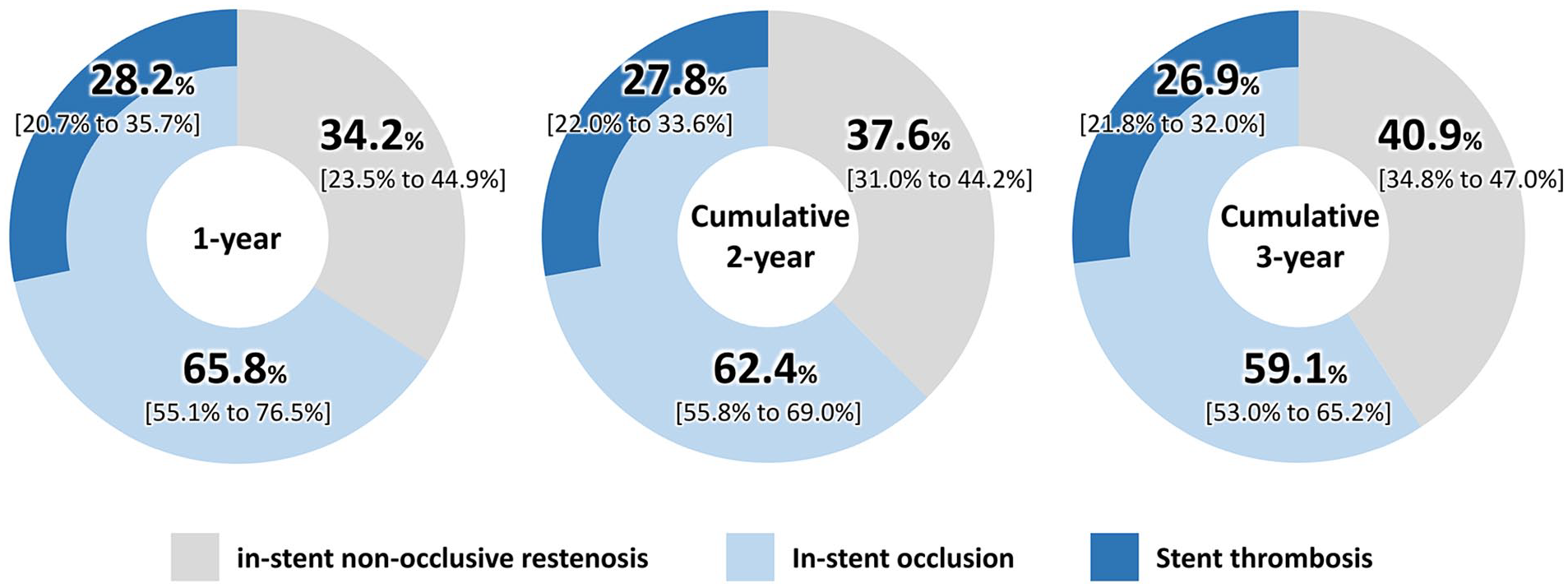
Proportions of in-stent occlusion and stent thrombosis to overall in-stent restenosis after fluoropolymer-based drug-eluting stent implantation.
Aneurysmal degeneration
The Munster registry reported that the incidence of aneurysmal degeneration at 2 years was 20% [27/137], which was higher than that at 1 year (8% [5/62]). 11 However, the sample size was limited; therefore, the time-course was inconclusive. The present study showed that the presence of aneurysmal degeneration did not significantly change over 3 years. Notably, aneurysmal degeneration may not be specific to FP-DES implantation. A recent study comparing FP-DES with bare nitinol stents, namely the EMINENT trial, demonstrated that both groups had a comparable incidence of aneurysmal degeneration without a statistically significant difference. 24 A case of aneurysmal degeneration after PF-DCS implantation has also been reported. 11 Whe-ther aneurysmal degradation adversely affects the clinic-al outcomes remains controversial. Although the present study did not find any statistically significant association between aneurysmal degradation and restenosis, its impact on long-term clinical outcomes remains unclear. Furth-ermore, as Figure 2 shows, some patients presenting with aneurysmal degeneration at 1 year were free from aneurysmal degeneration in subsequent years, whereas others free from aneurysmal degeneration at 1 and 2 years presented with aneurysmal degeneration at 3 years. This suggests that aneurysmal degeneration may be a transient response to intervention. Therefore, long-term monitoring of aneurysmal desecration is required.
Clinical implication
The present study showed that the FP-DES had a low 3-year incidence of restenosis and TLR. However, a small but substantial number of patients experienced stent thrombosis even after 1 year. Aneurysmal degeneration was observed for more than 3 years. Although it was not significantly related to the risk of restenosis, it remains a mysterious phenomenon. Safety concerns cannot be completely eliminated and long-term follow-up should be conducted.
Study limitations
First, it was an observational study that enrolled only Japanese patients with PAD who presented with femoropopliteal lesions. Therefore, a selection bias could not be eliminated. Second, this was a single-arm study that could only evaluate the performance of 3-year FP-DES. Therefore, we cannot conclusively determine whether the clinical outcomes of FP-DES were superior to those of other devices, including DCB. Third, restenosis and aneurysmal degeneration were not evaluated in a core laboratory, which may have affected reliability. However, the participating centers had experience in performing clinical studies on femoropopliteal endovascular devices, which would complement the data reliability. Fourth, data on aneurysmal degeneration at 3 years were available for only approximately half of the population. This was complemented by multiple imputations using the chained equations method. Finally, 3-year data on medication use, which might affect outcomes, were not available.
Conclusion
The 3-year incidence of restenosis after FP-DES implantation is low in real-world practice. However, stent thrombosis and aneurysmal degeneration were observed even after 1 year and remain an unsolved issue.
Footnotes
Acknowledgements
The authors thank the cardiac catheterization laboratory medical staff and clinical research coordinators at the participating centers.
Declaration of conflicting interests
Drs Iida and Soga are consultants who received honoraria from Boston Scientific. Drs Yamaoka, Fujihara, Kawasaki, and Ichihashi received remuneration from Boston Scientific Japan KK. Dr Mano received a research grant from Abbott Vascular Japan. Dr Sakata received an honorarium from Otsuka Pharmaceutical, Daiichi Sankyo, Takeda Pharmaceutical, Mitsubishi Tanabe Pharma, Medtronic Japan, and Boehringer Ingelheim Japan; a research grant from Edwards Lifesciences, FUJIFILM RI Pharma, REGiMMUNE, and Roche Diagnostics; and a scholarship (educational) grant/endowed chair from Otsuka Pharmaceutical, Johnson & Johnson, St Jude Medical Japan, Daiichi Sankyo, Takeda Pharmaceutical, Mitsubishi Tanabe Pharma, Teijin Pharma Limited, Boehringer Ingelheim Japan, Bayer Yakuhin, BIOTRONIK Japan, Boston Scientific, and Medtronic Japan. The remaining authors have no conflicts of interest to declare.
Funding
The CAPSICUM study was supported by the Research Association for Lower Limb Artery Revascularization (LIBERAL) sponsored by the following companies (in alphabetical order): Boston Scientific Japan KK, OrbusNeich Foundation, Terumo Corp, and Kaneka Medix Corporation. The funding companies played no role in the design of the study; selection of the enrolled patients; treatment strategy; revascularization procedures, or equipment; collection, analysis, or interpretation of the data; or manuscript preparation.
