Abstract
Background
Recent advances in endovascular therapy (EVT) have improved safety and efficacy, increasing the popularity of EVT for lower-extremity artery disease (LEAD).1,2 Additionally, EVT provides high technical success and shorter hospitalization. Notably, a substantial proportion of patients with chronic limb-threatening ischemia (CLTI) may be considered at high surgical risk, rendering them unsuitable for open bypass surgery. Under these conditions, EVT can be used as a nonsurgical therapeutic approach. 3 Although considerable progress has been made in the application of EVT, anatomically complex lesions in the affected vessel have remained technically challenging. 4 In order to improve the prognosis of patients undergoing EVT, efforts to reduce the risk of procedure-related complications are crucial for patients with LEAD. 5
Bleeding complications are among the most frequently encountered procedure-related adverse events following cardiovascular interventions. 6 Bleeding complications in patients with symptomatic LEAD could potentially induce subsequent cardiovascular and limb events resulting from hemoglobin drop and discontinuation of antiplatelet therapy. 7 Thus, identifying individuals at higher risk of bleeding complications during EVT and subsequently addressing modifiable risk factors among this population may further improve both health and economic outcomes.
The purpose of this study was to assess procedural and anatomical characteristics that affect in-hospital bleeding complications using data from the nationwide registry of EVT for symptomatic LEAD.
Methods
Study population
The present study analyzed the database of a prospective nationwide registry of EVT cases in Japan (J-EVT) organized by the Japanese Association of Cardiovascular Intervention and Therapeutics. This association mandates that interventionalists and their affiliated cardiovascular centers register all EVT cases in the registry for broad certification and renewal. A data manager in each hospital is responsible for collecting data and submitting them to the dedicated online database. The registered data included clinical information such as baseline patient characteristics, treated vessel territories, lesion severity, and in-hospital outcomes. The present study extracted cases undergoing EVT for symptomatic LEAD (aortoiliac, femoropopliteal, and/or below-knee EVT for CLTI or aortoiliac and/or femoropopliteal EVT for intermittent claudication) between January 2021 and December 2022.
The study was performed in accordance with the Declaration of Helsinki and approved by the Ethics Committee of the Clinical Research Promotion Network, Japan. Because the data were anonymous, the requirement for informed consent was waived.
Variable definitions
Symptomatic LEAD is defined as intermittent claudication and CLTI. CLTI includes a broader and more heterogeneous group of patients with varying degrees of ischemia that may delay wound healing and increase amputation risk. 8 Patients who underwent EVT for acute limb ischemia were excluded from this study. Treated lesions were categorized as aortoiliac, femoropopliteal, and infrapopliteal segments. Multilevel intervention was defined as the number of lesions treated in more than one segment. Peripheral arterial calcium was classified based on fluoroscopic or nonsubtracted images obtained during the endovascular procedure (no visible calcification, unilateral arterial calcification, or bilateral arterial calcification at the target lesion). The presence of one-sided versus circumferential vessel wall calcification at the target lesion was reviewed using multiple projections, if available. When the treated lesions involved more than one segment, the most advanced category of arterial calcification was assigned. The presence of a chronic occlusion was defined as a complete occlusion of any length. The nonfemoral access sites used for EVT in this registry included radial, brachial, distal superficial femoral, popliteal, posterior tibial, and dorsalis pedis access sites. Multiple approach sites were defined as sites that required two or more approaches during the index procedure. Procedural urgency is deemed emergent if the intervention is performed within hours of presentation. 9
Smoking was defined as a history of smoking within the past year. Hypertension was defined as a blood pressure ≥ 140/90 mmHg with antihypertensive medication. Dyslipidemia was defined as a serum total cholesterol concentration ≥ 220 mg/dL, a low-density lipoprotein-cholesterol concentration ≥ 140 mg/dL, or currently requiring lipid-lowering therapy. Diabetes mellitus was defined as hemoglobin A1c ≥ 6.5% or treatment with insulin or hypoglycemic agents. Chronic kidney disease (CKD) was defined as the presence of proteinuria, serum creatinine ≥ 1.3 mg/dL, or an estimated glomerular filtration rate ≤ 60 mL/min/1.73 m2. 10 Patients with coronary artery disease were defined as previously treated with percutaneous coronary intervention, coronary artery bypass grafting, or with a history of myocardial infarction. Chronic obstructive pulmonary disease was diagnosed based on the medical record, the use of drugs to treat this disease, or the results of spirometry at each site. Patients were diagnosed with impaired mobility if they were in a wheelchair or bedridden.
Procedures
EVT procedures were performed using standard techniques.5,11,12 All patients were recommended dual antiplatelet therapy (aspirin 81–100 mg/day and clopidogrel 75 mg/day) on the day before EVT or earlier. After insertion of a 4-, 5-, 6-, or 7-Fr sheath, bolus administration of 70- to 100-IU/kg unfractionated heparin was generally recommended and the activated clotting time during EVT was controlled between 250 and 400 seconds. A stent with subsequent postdilatation was placed for aortoiliac lesions. In femoropopliteal lesions, a stent was implanted in cases of flow-limiting dissection or > 30% residual stenosis. At the time of registration, atherectomy devices were not commercially available in Japan; therefore, they were not used in this study. Below-knee lesions were treated with optimal balloon angioplasty; however, stenting was not approved for this location in Japan.
Outcomes
The primary outcome measure was in-hospital bleeding complications, including bleeding at access and nonaccess sites and hemorrhagic stroke. Bleeding complications at the access and nonaccess sites were defined as perioperative and/or postoperative bleeding requiring intervention (surgical or endoscopic) or blood transfusion. All transfusions were counted irrespective of whether an intraoperative puncture site or a gastrointestinal hemorrhage prompted the transfusion. The diagnosis of hemorrhagic stroke was based on the focal collection of blood within the brain parenchyma, ventricular system, or subarachnoid space. 13 The secondary outcome measure was all-cause in-hospital mortality.
Statistical analysis
Data on baseline characteristics were presented as means ± SD for continuous variables and as frequencies (percentages) for discrete variables. Statistical significance was defined as a two-sided p-value of less than 0.05, and 95% CIs were reported where appropriate. Predictive factors for bleeding complications were determined by logistic regression analysis. Adjusted odds ratios (ORs) were derived from a multivariable model wherein the following factors were included as explanatory variables: sex, age, mobility, smoking, hypertension, dyslipidemia, diabetes mellitus, CKD, dialysis dependence, coronary artery disease, cerebrovascular disease, chronic obstructive pulmonary disease, antiplatelet agent, cilostazol, oral anticoagulant, emergent revascularization, CLTI, multilevel intervention, multiple approach sites, calcification severity, and chronic occlusion. In addition to the variables used in the model mentioned above, bleeding complications were entered into a separate model to identify predictors of in-hospital mortality. All statistical analyses were performed using R version 4.1.1 (R Foundation for Statistical Computing, Vienna, Austria).
Results
Clinical profiles of the 73,990 cases included in the study are summarized in Table 1. The mean patient age was 75 ± 9 years, and 69% of the patients were men. In-hospital bleeding complications were observed in 613 (0.8%) patients. Patients with bleeding were older and had comorbidities, such as impaired mobility, CKD, and cerebrovascular disease, more often. Patients who were men, smokers, or had diabetes mellitus had lower bleeding rates. There was a significant difference in the use of antiplatelet agent between both groups. The use of oral anticoagulants was significantly higher among patients who developed bleeding complications. For procedural and anatomical characteristics, the prevalence of emergent revascularization, CLTI, target lesion location in the femoropopliteal segment, multilevel intervention, multiple approach sites, and treatment of chronic occlusion were significantly higher in patients with bleeding than in those without. Notably, there was a significant difference in the severity of calcification between the groups. In contrast, patients with bleeding were less likely to have a target lesion below the knee segment. The incidence of bleeding complications according to different access sites is shown in the Supplemental Table.
Baseline characteristics.
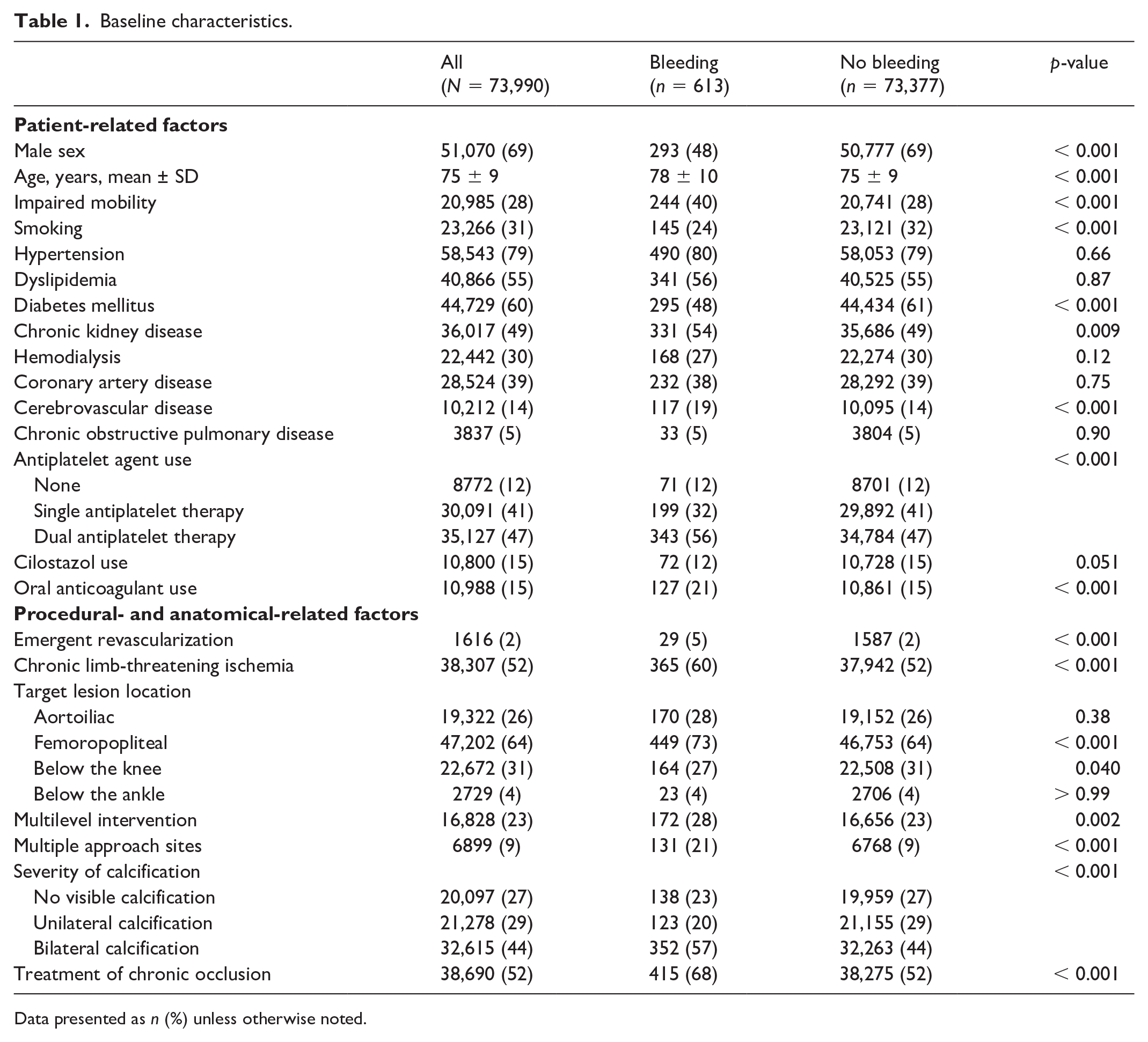
Data presented as n (%) unless otherwise noted.
Figure 1 shows the patient, procedural, and anatomical characteristics, after multivariable adjustments, independently associated with bleeding. Parameters independently associated with bleeding included age (OR: 1.01, 95% CI: 1.00–1.02, p = 0.005), impaired mobility (OR: 1.36, 95% CI: 1.11–1.66, p = 0.003), CKD (OR: 1.52, 95% CI: 1.24–1.85, p < 0.001), cerebrovascular disease (OR: 1.35, 95% CI: 1.10–1.66, p = 0.004), dual antiplatelet therapy (OR: 1.70, 95% CI: 1.27–2.28, p < 0.001), and use of an oral anticoagulant (OR: 1.63, 95% CI: 1.31–2.03, p < 0.001). Male sex (OR: 0.46, 95% CI: 0.39–0.55, p < 0.001), presence of diabetes mellitus (OR: 0.64, 95% CI: 0.54–0.76, p < 0.001), and hemodialysis (OR: 0.72, 95% CI: 0.57–0.92, p = 0.008) were independently associated with fewer bleeding complications. For procedural- and anatomical-related factors, emergent revascularization (OR: 1.90, 95% CI: 1.29–2.79, p = 0.001), multiple approach sites (OR: 2.46, 95% CI: 2.00–3.01, p < 0.001), bilateral arterial calcification (OR: 1.46, 95% CI: 1.19–1.79, p < 0.001 vs no visible calcification), and treatment of chronic occlusion (OR: 1.53, 95% CI: 1.28–1.83, p < 0.001) were independently associated with in-hospital bleeding complications. Additionally, we tested the association between the accumulation of these identified procedural and anatomical factors (emergent revascularization, multiple approach sites, bilateral arterial calcification, and chronic occlusion) and the incidence of in-hospital bleeding. Notably, the adjusted incidence of in-hospital bleeding complications was estimated to be 0.59% (95% CI: 0.51–0.68%) in patients with one of the identified procedural and anatomical risk factors, 0.96% (95% CI: 0.82–1.13%) in patients with two factors, and 2.40% (95% CI: 1.88–3.05%, p < 0.001) in patients with three or four factors (Figure 2).
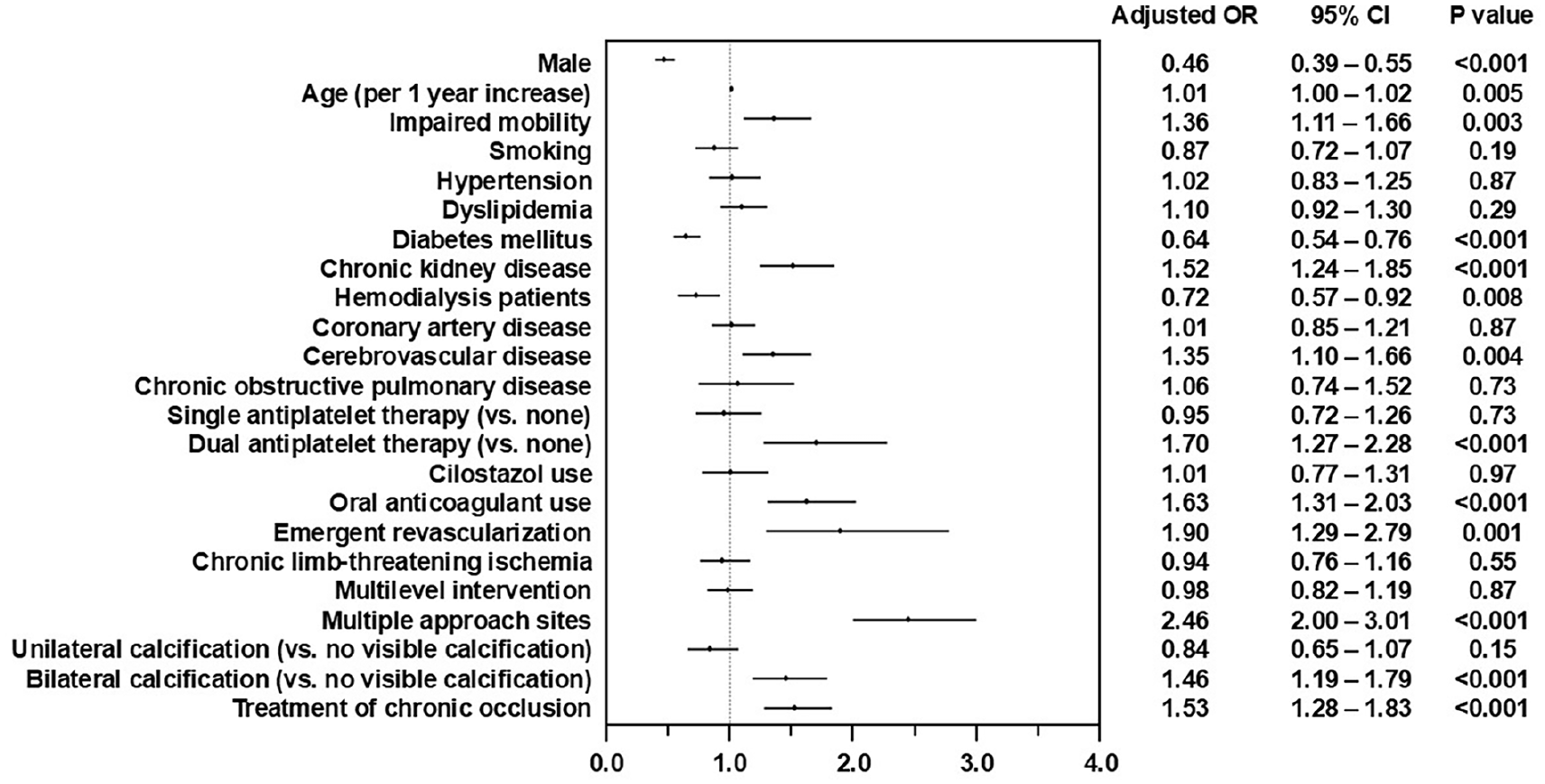
Forest plot of independent predictors of in-hospital bleeding complications showing odds ratio (OR) and 95% CI. The characteristics associated with a higher risk of bleeding complications are on the right side of the axis (vertial line at 1.0), and factors associated with a lower risk of bleeding are on the left.
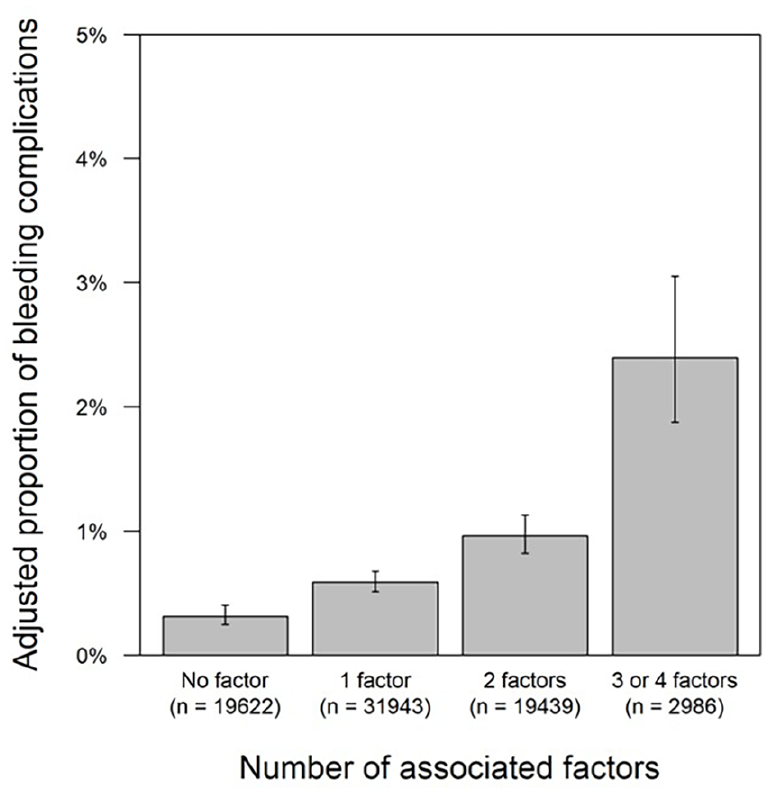
Adjusted incidence of in-hospital bleeding complications according to the number of associated factors. The adjusted incidence of in-hospital bleeding is 0.32% (95% CI: 0.25–0.40%) in patients with no factors, 0.59% (95% CI: 0.51–0.68%) in patients with one factor, 0.96% (95% CI: 0.82–1.13%) in patients with two factors, and 2.40% (95% CI: 1.88–3.05%, p < 0.001) in patients with three or four factors. The number of bleeding complications in each category were 76, 232, 220, and 85, respectively. The proportions are adjusted for the following patient characteristics: age, sex, mobility, smoking status, hypertension, dyslipidemia, diabetes, chronic kidney disease, hemodialysis dependence, coronary artery disease, cerebrovascular disease, chronic obstructive pulmonary disease, antiplatelet agents, cilostazol, and anticoagulants.
In-hospital deaths were observed in 1642 cases in the current study. The adjusted incidence was 4.57% (95% CI: 3.52–5.91%) and 0.84% (95% CI: 0.76–0.94%) in cases with and without in-hospital bleeding complications, respectively. The adjusted OR of in-hospital bleeding complications for in-hospital death was 5.62 (95% CI: 4.33–7.31, p < 0.001) (Figure 3). With respect to in-hospital death, the effect of in-hospital bleeding complications appeared to be consistent, irrespective of concomitant coronary artery disease (data not shown).

Forest plot of independent predictors of in-hospital mortality showing odds ratio (OR) and 95% CI. Variables associated with higher in-hospital mortality are on the right side of the axis (vertical line at 1.0), and those associated with lower in-hospital mortality are on the left.
Discussion
In the present study, using nationwide registry data on patients with symptomatic LEAD undergoing EVT, we demonstrated that bleeding complications occurred more commonly in patients with specific procedural and anatomical characteristics. These characteristics included emergent revascularization, multiple approach sites, bilateral arterial calcification, and chronic occlusion. Furthermore, the accumulation of these factors synergistically increased the incidence of complications. These results suggest that certain procedural and anatomical characteristics, which are easily assessed in clinical practice, could serve as reliable predictors of in-hospital bleeding complications in patients with LEAD who undergo EVT.
The incidence of in-hospital bleeding complications was 0.8% in this study, which was relatively low compared with previous data from patients with LEAD undergoing EVT.14 –16 This discrepancy may be due to differences in the study population, EVT procedures, and definition of bleeding. Specifically, a previous report included the assessment of dynamic changes in hemoglobin levels as the definition of bleeding, suggesting more sensitive criteria than the current study. 15 Moreover, our study focused on in-hospital bleeding, and less attention was given to postdischarge bleeding. These differences may be attributed to the lower incidence of bleeding complications observed in this study.
Our data also demonstrated that most patients undergoing EVT were older adults and had CKD, which are important criteria for high bleeding risk.17,18 This analysis identified several patient factors, including advanced age, female sex, and comorbidities that were independently associated with bleeding complications. These results are consistent with those of previous studies. However, our study showed that patients with diabetes mellitus and hemodialysis had less frequent bleeding complications, which is consistent with another study. 14 Although the precise mechanisms of the inverse association between these comorbidities and the rate of bleeding complications are not entirely understood, a significant percentage of patients with diabetes mellitus and hemodialysis might have isolated infrapopliteal disease, thereby predisposing them toward a lower incidence of periprocedural bleeding complications. Meanwhile, bleeding complications were strongly associated with increased in-hospital mortality in the current study. Recent studies have demonstrated that the prevalence of high bleeding risk in patients undergoing EVT is much higher than that in patients following percutaneous coronary intervention. 19 Therefore, patients with LEAD might have a higher potential risk for bleeding and subsequent mortality. Our study indicated that clinicians might assess bleeding risk to mitigate observed disparities in mortality among patients with LEAD who underwent EVT.
Regarding procedural-related factors, emergent revascularization and multiple approach sites were associated with bleeding complications in the current study. The high rate of bleeding among patients requiring emergent revascularization was concurrent with the results from prior EVT registries.14,15 Our study also revealed that multiple approach sites were independent predictors of bleeding complications, which has not been documented in prior studies. Indeed, EVT strategies using multiple approach sites can contribute to difficulties in achieving precise puncture and effective hemostasis. The relationship between multiple puncture sites and an increased risk of bleeding complications has already been reported in patients undergoing percutaneous coronary intervention; 20 the current study extends these insights to patients with LEAD who underwent EVT.
In this study, we found that complex anatomical lesions, including chronic occlusion and bilateral arterial calcification, were associated with bleeding complications. The treatment of chronic occlusion requires more aggressive treatment as well as complex procedures, resulting in an increased likelihood of iatrogenic hemorrhagic events. 21 Additionally, several mechanisms may underlie the association between severe calcification and a greater risk of bleeding complications in patients with LEAD. Patients with medial calcification have various comorbidities such as advanced age, high prevalence of CKD, and CLTI, which overlap with the risk of bleeding. 22 In this regard, patients with vascular calcification are believed to be associated with oxidative stress and inflammation, predisposing them to a higher incidence of bleeding events. 23 A recent study in the field of coronary artery disease demonstrated a significant association between a high calcium score and the risk of bleeding. 24 Thus, the presence of arterial calcification during EVT might offer useful information beyond known risk factors of bleeding that are clustered in patients with LEAD.
Limitations
Our study had several limitations. First, the predictors of access and nonaccess site bleeding complications were not explored separately in the present study. Thus, it could not be clarified whether these complications share similar predisposing factors. Moreover, our study only included in-hospital bleeding events; therefore, our results may have underestimated the rates of nonaccess site bleeding complications which are commonly observed during the postdischarge-phase. 25 Consequently, future studies with more vigorous protocols for antithrombotic strategies and an extended duration of the bleeding assessment are warranted. Second, serial hemoglobin data were not collected during admission in this registry. Thus, we could not assess bleeding events using an alternative definition. 26 However, the presence of blood transfusion, a criterion of the validated bleeding definition, was considered in the present study. The definition of bleeding was derived from patients with coronary artery disease and varies greatly depending on the previous literature, therefore a gold standard definition of bleeding in patients with LEAD undergoing EVT is desirable. Third, this study did not include the calculation of previously reported bleeding scores; therefore, comparing the identified procedural and anatomical factors with traditional risk scores is not possible. Fourth, the absence of data regarding particular access site locations that could provide potential modifiers of bleeding risk is another limitation of this study. Finally, because the number of variables was limited in this registry, unevaluated procedural variables, including sheath size and closure devices, may have affected the outcome.14,27 Additionally, we noted that data related to ultrasound guidance, which could reduce access site bleeding complications, were not available in the present study. Therefore, future investigations examining the effects of ultrasound-guided arterial access on bleeding complications are essential. Despite these limitations, the strengths of the current study include its large sample size and the amount of data collected for each procedure in the current era.
Conclusion
The results of this study suggest that certain procedural and anatomical characteristics as well as antithrombotic strategies, including emergent revascularization, multiple approach sites, bilateral arterial calcification, chronic occlusion, dual antiplatelet therapy, and oral anticoagulant, were independently predictive of in-hospital bleeding complications in patients with LEAD who underwent EVT. In addition, these identified procedural and anatomical factors demonstrated a cumulative additive effect.
Supplemental Material
sj-pdf-1-vmj-10.1177_1358863X251320113 – Supplemental material for Cumulative impact of procedural and anatomical factors on in-hospital bleeding complications in endovascular therapy for lower-extremity artery disease: A nationwide registry study in Japan
Supplemental material, sj-pdf-1-vmj-10.1177_1358863X251320113 for Cumulative impact of procedural and anatomical factors on in-hospital bleeding complications in endovascular therapy for lower-extremity artery disease: A nationwide registry study in Japan by Takuya Nakahashi, Mitsuyoshi Takahara, Osamu Iida, Shun Kohsaka, Yoshimitsu Soga, Kazunori Horie, Kenji Sakata, Masayuki Takamura, Tetsuya Amano and Ken Kozuma in Vascular Medicine
Footnotes
Acknowledgements
The authors appreciate the contributions of the investigators, staff who managed the J-EVT registry, and members of the Japanese Association of Cardiovascular Intervention and Therapeutics and its secretariat.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
