Abstract
Introduction:
Although renal stenting is the standard revascularization method for atherosclerotic renal artery stenosis (RAS) (FMD-RAS), stenting in fibromuscular dysplasia (FMD) RAS is usually limited to periprocedural complications of angioplasty and primary arterial dissection. The main aim of the study was to retrospectively analyze the immediate and long-term results of renal stenting versus angioplasty in patients with FMD.
Methods:
Of 343 patients in the ARCADIA-POL registry, 58 patients underwent percutaneous treatment due to FMD-RAS (in 70 arteries). Percutaneous transluminal renal angioplasty (PTRA) was performed as an initial treatment in 61 arteries (PTRA-group), whereas primary stenting was undertaken in nine arteries (stent-group). Stent-related complications were defined as: in-stent restenosis > 50% (ISR); stent fracture; under-expansion; or migration.
Results:
In the PTRA-group, the initial restenosis rate was 50.8%. A second procedure was then performed in 22 arteries: re-PTRA (12 arteries) or stenting (10 arteries). The incidence of recurrent restenosis after re-PTRA was 41.7%. Complications occurred in seven of 10 (70%) arteries secondarily treated by stenting: two with under-expansion and five with ISR. In the stent-group, stent under-expansion occurred in one case (11.1%) and ISR in three of nine stents (33.3%). In combined analysis of stented arteries, either primarily or secondarily, stent-related complications occurred in 11/19 stenting procedures (57.9%): three due to under-expansion and eight due to ISRs. Finally, despite several revascularization attempts, four of 19 (21%) stented arteries were totally occluded and one was significantly stenosed at follow-up imaging.
Conclusion:
Our study indicates that renal stenting in FMD-RAS may carry a high risk of late complications, including stent occlusion. Further observational data from large-scale registries are required.
Keywords
Introduction
Fibromuscular dysplasia (FMD) is an idiopathic, nonatherosclerotic, and noninflammatory disease of the arterial wall of medium-sized arteries. It may affect all vascular beds including, most frequently, renal, extracranial carotid, and vertebral arteries. 1 Typically, the morphological presentation of FMD includes multifocal (string-of-beads) or focal lesions, but recently phenotypic expressions of FMD have been expanded to include arterial dissection, aneurysm, and tortuosity. 2 Multivessel involvement is common. 2 Renal FMD is the most frequent nonatherosclerotic cause of renal artery stenosis (RAS) and in 5.6–13.8% of cases may be also associated with renal artery aneurysm (RAA).3–6 Renal artery intervention, predominantly percutaneous transluminal renal angioplasty (PTRA), may be indicated for hemodynamically significant FMD-related RAS (FMD-RAS). The indication for RAA embolization, either with or without stenting, is based on the size and morphology of the aneurysm. 1
Recommendations for the interventional management of FMD-RAS and FMD-related RAA (FMD-RAA) are based upon expert consensus derived from limited published data. 2 Previous case reports or small case series describing the outcomes of FMD-RAS stenting have provided equivocal results.7 –13 Better outcomes have been reported for FMD-RAA stenting used to support aneurysm closure but these data are currently limited to two small studies.14,15 Hence, although renal artery stenting has become the standard method of invasive treatment for atherosclerotic RAS and is used as an adjunct for selected cases of RAA embolization, primary stenting in FMD-RAS is generally not recommended. Stenting in this context is currently considered to have a limited role as a bail-out strategy for complications arising during angioplasty and for some cases of primary renal artery dissection. 2
The Polish ARCADIA-POL study has reported the clinical characterization of patients with FMD and showed that systematic multimodal imaging of all vascular beds had an impact on patient management. 5 The registry also included patients with a history of renal vascular interventions (angioplasty with or without stenting for FMD-RAS and stenting for FMD-RAA). The first aim of the present study was to retrospectively analyze subjects from the ARCADIA-POL cohort who underwent renal artery stenting for FMD-RAS and to compare the immediate and long-term results of stenting with those undergoing PTRA without stenting. The second aim was to evaluate the results of renal artery stenting as an adjunct of FMD-RAA closure.
Methods
The study was conducted in accordance with the 1975 Declaration of Helsinki. The protocol was approved by the ethics committee of the National Institute of Cardiology in Warsaw (Poland) and all participants provided written informed consent. The ARCADIA-POL study is a Polish multicenter national observational registry. 16 At the time of the study, it included 343 patients, aged over 18 years with newly diagnosed, known or suspected FMD or spontaneous artery dissection, out of which 203 had confirmed renal artery FMD (see Fig. 1). All patients were invited to the National Institute of Cardiology (Warsaw) where they underwent detailed clinical evaluation, including blood tests, 24-hour ambulatory blood pressure monitoring (ABPM) and computed tomography angiography (CTA) arterial imaging (from head to pelvis) as previously described. 16 Pretreatment CTA scans were analyzed with lesions classified into two types: multifocal (multiple stenosis or string-of-beads appearance) and focal (stenosis < 1 cm in length or tubular stenosis > 1 cm in length). 17 Multivessel FMD was defined as the presence of FMD in two or more vascular beds or by the presence of FMD in one vascular bed accompanied by either aneurysm or dissection in another one. 2 A renal artery aneurysm was defined as a dilated segment of the renal artery that exceeded twice the diameter of the normal renal artery. 18 For patients with renal FMD and a history of percutaneous treatment for either FMD-RAS or FMD-RAA, detailed data on invasive procedures including indications for intervention, patients’ medical history, plasma creatinine level, and procedural reports (including the use of stents) were retrospectively gathered based on a uniform questionnaire completed by the participating centers. Clinical data and available follow-up imaging results were also obtained from the centers.
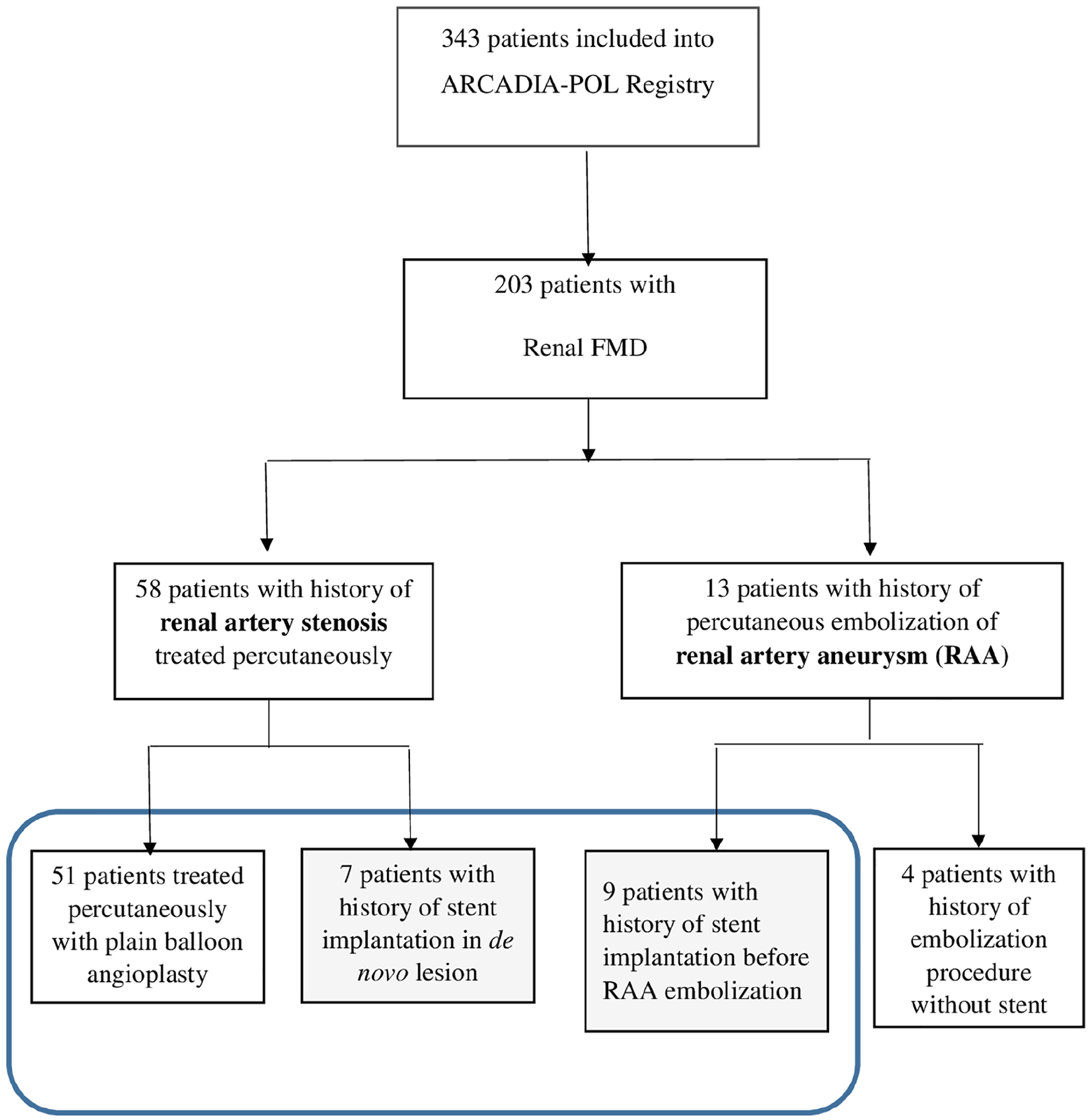
Study flow chart. Blue box represents the final study group.
Outcomes were assessed from both clinical and imaging data (either CTA or conventional angiography) undertaken at any timepoint. Restenosis after PTRA was defined as at least 50% diameter stenosis. Clinical restenosis was diagnosed if it was accompanied by deterioration of blood pressure control or a decline in kidney function. The presence and nature of stent-related complications was categorized into: (1) in-stent restenosis (ISR) more than 50% of diameter; (2) stent fracture defined as the complete or partial separation of struts 19 ; (3) stent under-expansion defined as the presence of an inadequately expanded stent with a lower diameter when compared with the adjacent reference segment; and (4) stent migration assessed as previously described, 20 that is, as proximal or distal movement of the device of at least 4 mm relative to a vascular landmark (renal stenosis or aneurysm) – found on CTA or conventional angiography (CA) at any time point after stent implantation.
Results
Out of 343 participants, 203 had confirmed renal artery FMD. Percutaneous revascularization for FMD-RAS was performed in 58 patients (70 arteries). Whereas in 61 out of 70 arteries primary treatment was limited to PTRA, nine arteries were treated by primary stenting. A further 13 patients had FMD-RAA treated by embolization, out of which nine (with 10 aneurysms) had stenting before embolization (Figure 1). The characteristics of patients treated primarily by PTRA (PTRA-group), by stenting (stent-group), or those with stented FMD-RAA are reported in Table 1 (median time between first procedure and evaluation in the registry: 72 [IQR 39–119] months). Patients with FMD-RAS were much younger than those with FMD-RAA (31.7 ± 12.0 vs 54.1 ± 7.7 years, p < 0.05) and trended towards a higher frequency of focal FMD (21 vs 11%, p = 0.14).
Baseline characteristics of patients according to the initial invasive treatment for FMD-RAS and FMD-RAA.
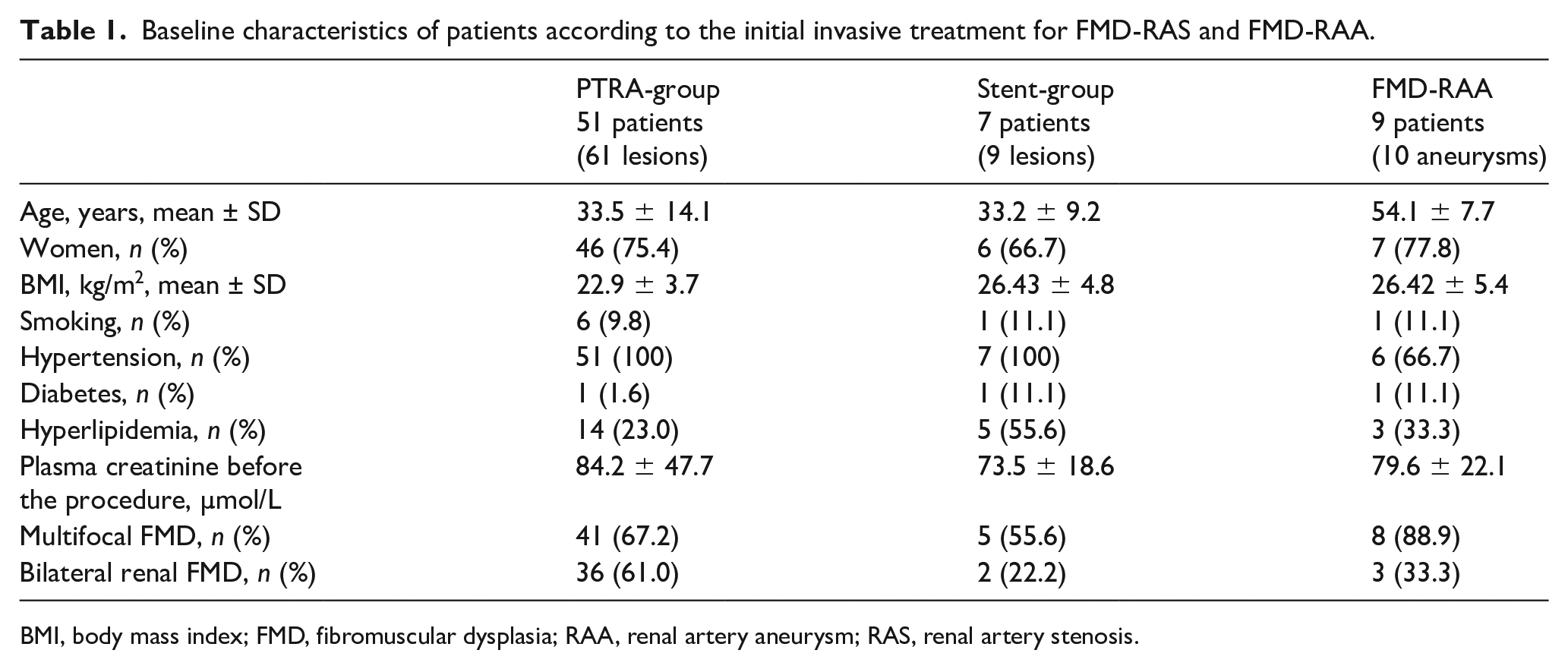
BMI, body mass index; FMD, fibromuscular dysplasia; RAA, renal artery aneurysm; RAS, renal artery stenosis.
The immediate and long-term results of revascularization in the PTRA-group and stent-group are presented in Figure 2 and described below.
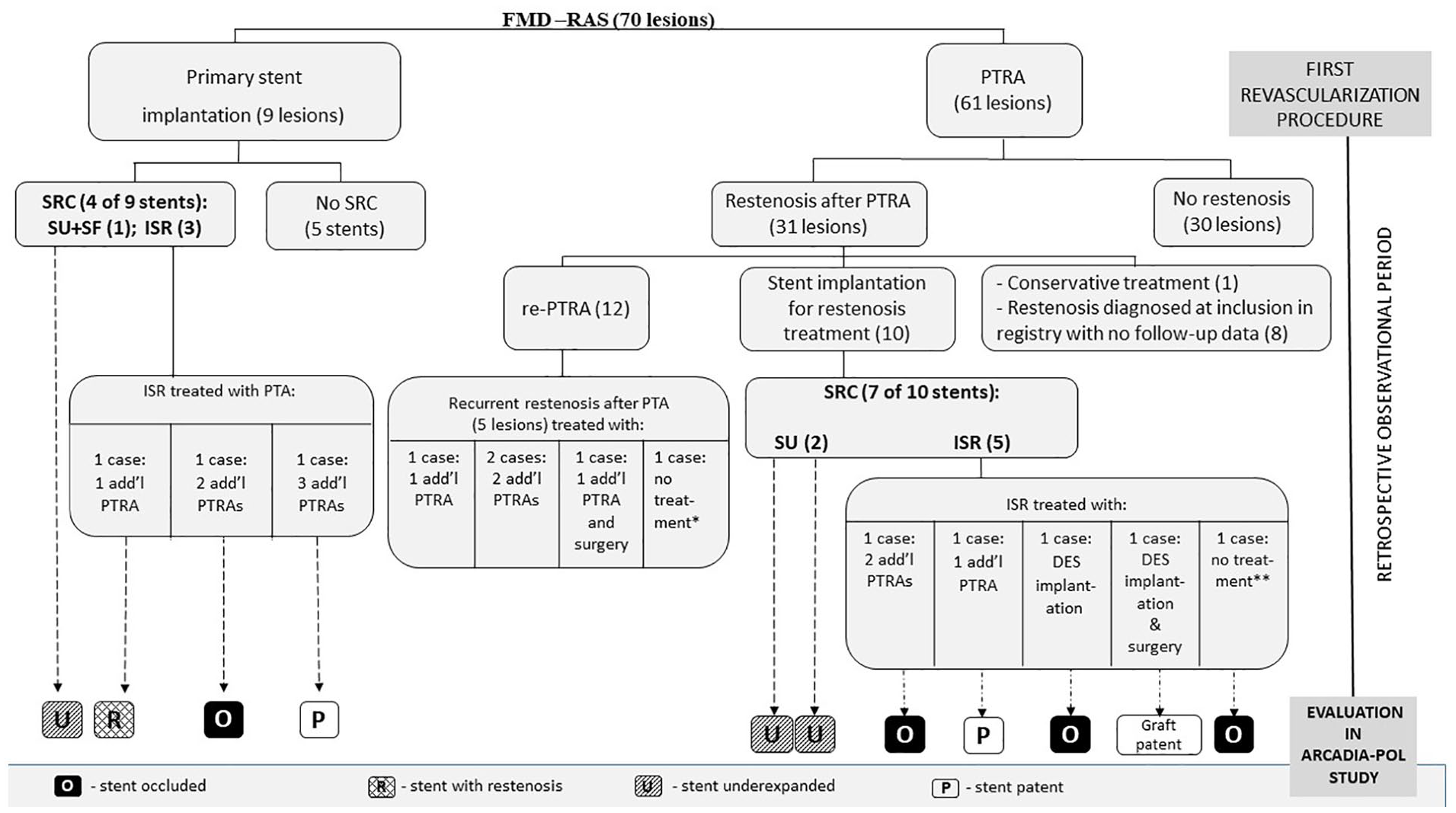
Immediate and long-term results of renal artery revascularization.
Outcomes of percutaneous transluminal renal angioplasty (PTRA) in FMD-related renal artery stenosis (FMD-RAS)
In the PTRA-group, 61 arteries were treated with PTRA as an initial treatment, with no immediate complications recorded. Of these, angiographic restenosis occurred in 31 arteries (50.8%) after a median of 5 months [IR 3–28]. The per patient restenosis rate was 54.9% (28 of 51 patients). Clinical restenosis occurred in 47.0% (24 of 51 patients). Of the 31 arteries with restenosis after PTRA, 22 underwent a second procedure with either stent implantation (10 arteries) or re-PTRA (12 arteries) because of deterioration of blood pressure (BP) control. One asymptomatic patient was treated conservatively. The remaining eight arteries were diagnosed as restenotic only at registry imaging so no prospective follow-up is available. Among the 12 arteries treated by re-PTRA, the incidence of recurrent restenosis was 41.7% (five arteries). One artery was totally occluded at the time of registry evaluation.
Of the 10 arteries treated with stenting due to restenosis after PTRA, stent under-expansion at deployment occurred in two cases (20%). Additionally, ISR occurred in five of 10 stents (50%), leading to a combined rate of complications of 70% (seven of 10 stents).
One patient with stent under-expansion underwent two additional unsuccessful angioplasties. In four out of five cases with ISR, further attempts at percutaneous treatment were made because of clinical indications (deterioration of BP) and one patient with an occluded stent was managed conservatively (Figure 2). One patient with recurrent ISR was finally treated surgically with a good outcome. At the time of registry evaluation, total in-stent occlusion was demonstrated in three stents.
No correlation between the age at the procedure or the type of FMD and clinical results were observed (online Supplementary Tables 1 and 2).
Outcomes of stenting in FMD-RAS
In the stent-group, nine renal artery stenoses were treated with stent implantation as an initial treatment. Of these, stenting was the primary elective treatment strategy in three cases. In four arteries, stenting was used during the PTRA procedure due to significant residual stenosis after the initial balloon predilatation. In two cases, the indication for stent insertion is not provided in the procedural report, but no complications such as dissection or rupture were recorded as a potential justification for the use of stenting. Among complications, stent under-expansion occurred in one case (11.1%). This stent was subsequently found to be fractured at the time of registry CTA evaluation. The incidence of ISR was 33.3% (three out of nine stents). In all three cases, additional percutaneous treatment was performed due to clinical restenosis (deterioration of hypertension in two cases and renal function decline in one case) (Figure 2). Despite this, recurrent ISR (including total in-stent occlusion in one case) was recorded in two of these three cases.
In an additional analysis, we combined the patients from the PTRA-group, who subsequently received a stent for restenosis treatment, with patients from the stent-group, primarily treated with stenting. In this merged group of 16 patients (19 arteries treated), the overall incidence of stent-related complications was 57.9%. This consisted of three (15.8%) immediate complications (stent under-expansion) and eight (42.1%) late complications (ISR). The median time between stent implantation and ISR diagnosis was 6 months [IR 2–16]. Of note, ISR was confirmed by CTA or conventional angiography in all cases. In one patient with early restenosis, additional intravascular ultrasound imaging allowed exclusion of a thrombotic etiology.
One patient with total in-stent occlusion was managed conservatively. Seven ISR cases had clinical restenosis (six patients with deterioration of hypertension, one with worsening renal function). After further percutaneous treatment, the incidence of recurrent ISR was 71.4% (five out of seven treated cases). Finally, at the time of follow-up registry imaging, four of 19 (21%) stented arteries were found to be totally occluded and one remained with significant restenosis (Figure 2).
At the time of evaluation in the registry, patients with stent-related complications had a significantly higher 24-hour systolic BP than those without (134.5 ± 10.8 vs 120.5 ± 7.7 mmHg;p = 0.004). Of note, patients with stent-related complications were more often men (50% vs 25%) and more frequently had focal FMD (75% vs 50%) compared to patients without stent-related complications. The comparison of patients with and without stent-related complications is presented in online Supplementary Table 3.
Outcomes of stenting in FMD-related renal artery aneurysm (FMD-RAA)
In nine patients with FMD-RAA, 10 aneurysms were present, including eight of type III and one of type II, according to the Rundback classification (in one the type of aneurysm was unknown). 21 Clinical data are shown in Table 1. Hypertension was diagnosed in 66.7% of cases. In all cases renal FMD was diagnosed before the embolization procedure, with multifocal FMD present in 90% of cases. Nine FMD-RAA patients underwent 10 stent implantation procedures (one patient with bilateral aneurysms underwent two separate procedures). Nine aneurysms were treated with stent-supported coil embolization. RAA closure was confirmed in all cases after treatment. In one artery, emergency stent graft insertion was required due to artery rupture when removing a migrating coil. Thus, nine (90%) self-expanding stents and one graft stent (10%) were implanted.
One immediate complication of stenting occurred during 10 procedures due to distal edge stent migration into the sack of the aneurysm without rupture.
There were no late stent-related complications in patients with RAA. However, at the time of follow-up registry imaging, one renal artery with a stent inserted in the middle part of the trunk was proximally occluded with subsequent kidney atrophy. Based on retrospective analysis of the initial angiography, this was not considered to be directly related to stent implantation, as the renal artery was found to be occluded in the proximal segment, where a nonsignificant stenosis had been present at the time of the procedure. It is therefore postulated that renal artery occlusion resulted from the progression of this stenosis.
Discussion
The present retrospective analysis reports the clinical characteristics and results of the percutaneous treatment of 58 patients with FMD-RAS and nine patients with FMD-RAA, derived from 343 patients with FMD systematically evaluated in a national referral center. This cohort, the largest reported so far, includes 16 patients with renal artery stenting in FMD-RAS. In previous stenting studies, the number of reported patients ranged from four to nine subjects.7–15
In the FMD-RAS group, the overall initial intraprocedural success of stenting was 84.2%, with all periprocedural failures due to resistant stent under-expansion (15.8%). However, the rate of late stent-related complications was very high, reaching 57.9%.
The most frequent complication was ISR. In the current cohort, the restenosis rate after stenting of a de novo lesion was high, although less frequent than after PTRA (33.3% vs 50.8%). This is a comparable or even slightly higher rate than that reported in other PTRA studies. For example, Jenkins et al. reported that more than one angioplasty procedure was required in 15 of 42 (32.6%) patients with FMD-RAS. 22 The largest meta-analysis of 36 studies including 1916 procedures showed an increasing restenosis rate during the follow-up period, from 13.2% at 1 year to 29.1% at 5 years. 23
It should also be emphasized that, in this study, stenting for restenosis after PTRA was not associated with a lower rate of recurrent restenosis as compared to repeat PTRA without stenting. Indeed, ISR occurred in 50% of stented cases as compared to 42% of arteries treated with re-do angioplasty. Moreover, the stenting procedure was associated with an increased risk of additional complications, such as stent under-expansion or fracture and, even more importantly, with an unacceptably high risk of artery occlusion over a long-term follow-up (21%). This compares to only one out of 70 arteries occluded at long-term follow-up after treatment with balloon angioplasty alone (whether one or several procedures). Similar favorable outcomes of patency after PTRA were demonstrated by Mousa et al, who reported that all 43 arteries studied in their series remained patent 9 years after PTRA. 24
Long-term observational studies of patients with FMD-RAS treated with renal stenting are scant. Only five original papers with at least four patients included were published between 2007 and 20227 –10,13 (one study also included a previously reported group). The reported ISR rates ranged from 22.2% to 50%. In the study of Barrier et al., the rate of complications was similar to our observations, with long-term patency achieved in only 43% cases. Moreover, stent fracture occurred in two of seven cases. 9 The results of previously published studies are summarized in Table 2.
Summary of published studies evaluating renal artery stenting in renal fibromuscular dysplasia.
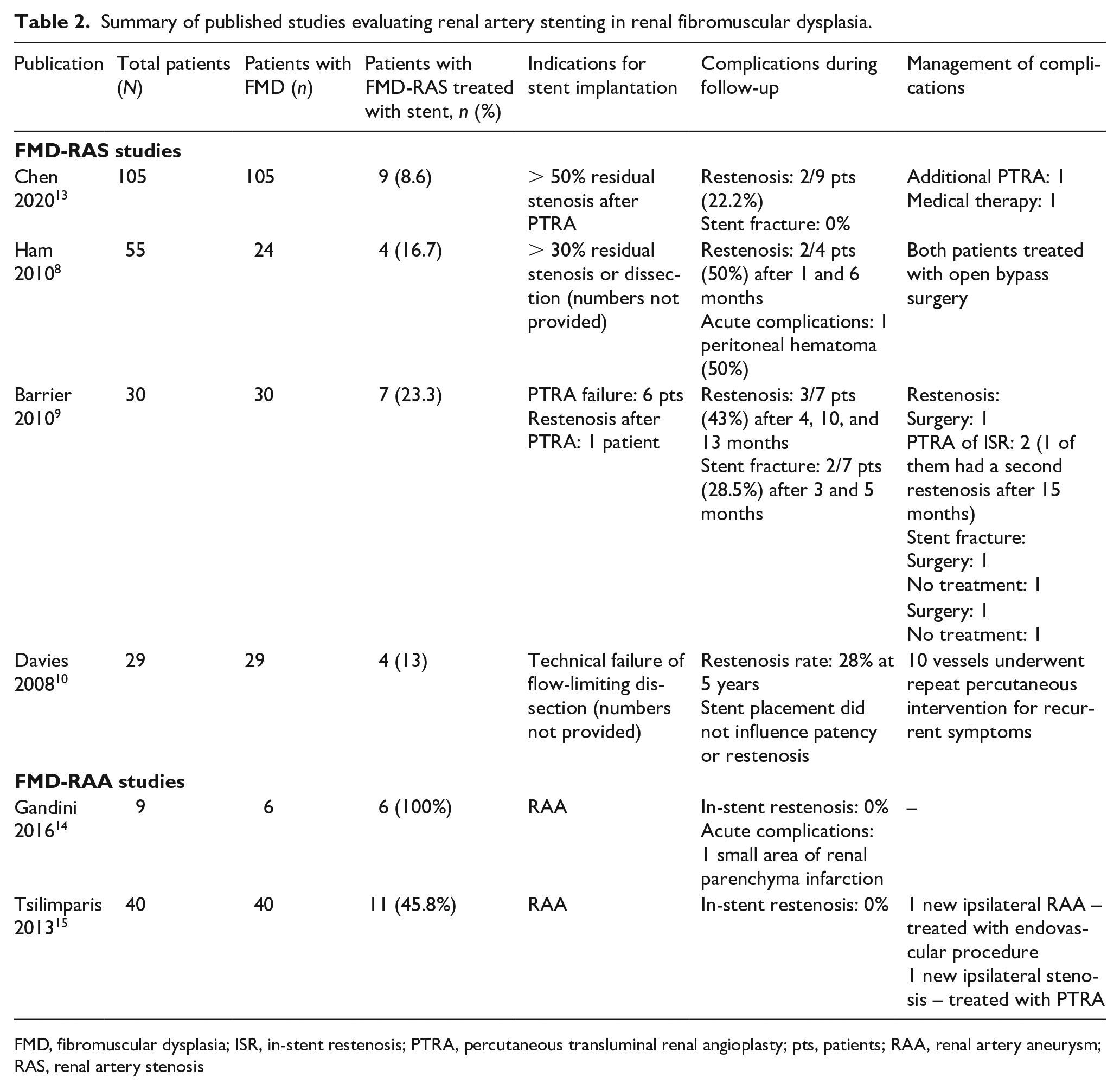
FMD, fibromuscular dysplasia; ISR, in-stent restenosis; PTRA, percutaneous transluminal renal angioplasty; pts, patients; RAA, renal artery aneurysm; RAS, renal artery stenosis
The prevalence of ISR in our study was also much higher than that observed in studies with atherosclerotic renal disease (< 20% after bare metal stent implantation), suggesting that there may be different mechanisms driving restenosis in FMD. 25 Potential factors promoting in-stent tissue proliferation in FMD are unknown. Similar to atherosclerosis, the length of the stent used may impact the restenosis rate. However, several other mechanisms may be relevant. First, renal artery hypermobility may have an impact on stent kinking, provoking local intimal damage and stimulating tissue proliferation. Second, the vessel wall architecture in FMD, particularly in some focal lesions, seems to be more resistant to dilatation and may promote restenosis by stent malapposition. Stent malapposition may also occur more frequently in long multifocal lesions. Also, the predominant involvement of the middle artery segments with multifocal lesions may be associated with the ‘geographic miss’ phenomenon (i.e., that a stent does not fully cover the predilated segment of the artery). Given these putative explanations for restenosis, the use of intravascular imaging during stenting might impact the outcomes.
Of note, a recent study showed that the use of pressure wire to guide renal artery procedures may result in better long-term outcomes. 26 In our group, most procedures were angiographically guided, with the support of pressure measurements reported in only six PTRA procedures.
The results of our study may suggest that management of such difficult patients should potentially favor surgery, at least after the initial failure of angioplasty, in order to avoid unnecessary additional invasive procedures and an unacceptable low rate of patency. From the literature it is known that the results of surgical treatment are favorable, with an early postoperative occlusion or restenosis rate after elective procedure reported in 3.8–13% of cases (more often with venous grafts than with arterial autografts).27–29 Our findings support the current consensus recommending restriction of renal artery stenting in the management of acute periprocedural complications of renal angioplasty. 2
The median time between stent implantation and ISR diagnosis was 6 months. Hence, our study supports a strategy of early follow-up imaging (during the first 3–6 months) in patients after renal stenting to detect restenosis and initiate further treatment if indicated.
In comparison to patients with FMD-RAS, the overall outcomes in patients with FMD-RAA treated with stenting were more favorable. Only one of 10 intraprocedural and no late stent-related complications were reported. Neither in-stent restenosis nor stent under-expansion or fracture occurred in patients with FMD-RAA. Considering the higher incidence of stent-related complications found in patients with FMD-RAS, particularly with focal FMD, the low incidence of complications in patients with FMD-RAA may be attributed to a predominance of the multifocal FMD subtype in this subgroup. Thus, it might be hypothesized that focal and multifocal lesions reflect two different disease entities or disease processes. To the best of our knowledge, only two other studies have reported the long-term outcomes after FMD-RAA stenting, including relatively small numbers of patients and showing favorable long-term results (presented in Table 2).14,15
Study limitations
We presented the incidence of complications after renal stenting in patients with renal fibromuscular dysplasia. Despite the fact that our study is the largest published so far, the numbers are inevitably still small and the conclusions should be interpreted with caution. Our retrospective observational study has several potential limitations. First, despite meticulous gathering of retrospective data using a uniform questionnaire, some data were missing and irretrievable, including the indication for stent implantation in a few cases, the long-term antiplatelet therapy recommended after stenting, and some procedural details. Second, there was no systematic measurement of the pressure gradient allowing assessment of stenosis severity. This might have had an impact on long-term results. Third, ISR was diagnosed based only on angiographic appearance without routine confirmation with intravascular imaging. Hence, we cannot exclude the possibility that some cases were of thrombotic etiology. However, only one of eight stents with ISR was found totally occluded at the time of the first follow-up. This suggests neointimal proliferation rather than a thrombotic etiology as the cause of most late complications. Owing to the retrospective nature of the study, we were unable to provide complete data on blood pressure and renal function before stenting and to document whether stent treatment had a beneficial effect on these clinical outcomes. Finally, the limited sample size did not allow us to investigate potential predictors of stent-related complications. For the same reasons, we cannot exclude a selection bias – for example, that patients with a favorable outcome after stenting were less often referred for inclusion in the ARCADIA-POL study.
Conclusions
Although available data remain limited, this study confirms on a larger scale existing reports suggesting that renal artery stenting may have limited utility for either the primary treatment of FMD-RAS or the treatment of restenosis following PTRA in patients with FMD, and may be associated with late complications including renal artery occlusion. As such, it strengthens the current consensus 2 that stenting should be used only as a bail-out strategy for the management of intraprocedural complications of percutaneous angioplasty. In case of restenosis after PTRA, stent implantation is not associated with a lower risk of recurrent restenosis and surgical treatment should be considered. In contrast, this small series provides supportive data for renal stenting used to support FMD-RAA closure, which appeared to be associated with very favorable outcomes. As the number of patients included in our study is relatively small, these conclusions should be interpreted with caution. Further observational data from large-scale international registries are required to confirm the wider applicability of these findings.
Supplemental Material
sj-pdf-1-vmj-10.1177_1358863X231210523 – Supplemental material for Risks and benefits of renal artery stenting in fibromuscular dysplasia: Lessons from the ARCADIA-POL study
Supplemental material, sj-pdf-1-vmj-10.1177_1358863X231210523 for Risks and benefits of renal artery stenting in fibromuscular dysplasia: Lessons from the ARCADIA-POL study by Jacek Kądziela, Katarzyna Jóźwik-Plebanek, Marco Pappaccogli, Patricia van der Niepen, Aleksander Prejbisz, Piotr Dobrowolski, Ilona Michałowska, Paulina Talarowska, Ewa Warchoł-Celińska, Łukasz Stryczyński, Jan Krekora, Piotr Andziak, Małgorzata Szczerbo-Trojanowska, Rafał Maciąg, Ignacy Sterliński, Adam Witkowski, Andrzej Januszewicz, David Adlam, Magdalena Januszewicz and Alexandre Persu in Vascular Medicine
Supplemental Material
sj-pdf-2-vmj-10.1177_1358863X231210523 – Supplemental material for Risks and benefits of renal artery stenting in fibromuscular dysplasia: Lessons from the ARCADIA-POL study
Supplemental material, sj-pdf-2-vmj-10.1177_1358863X231210523 for Risks and benefits of renal artery stenting in fibromuscular dysplasia: Lessons from the ARCADIA-POL study by Jacek Kądziela, Katarzyna Jóźwik-Plebanek, Marco Pappaccogli, Patricia van der Niepen, Aleksander Prejbisz, Piotr Dobrowolski, Ilona Michałowska, Paulina Talarowska, Ewa Warchoł-Celińska, Łukasz Stryczyński, Jan Krekora, Piotr Andziak, Małgorzata Szczerbo-Trojanowska, Rafał Maciąg, Ignacy Sterliński, Adam Witkowski, Andrzej Januszewicz, David Adlam, Magdalena Januszewicz and Alexandre Persu in Vascular Medicine
Supplemental Material
sj-pdf-3-vmj-10.1177_1358863X231210523 – Supplemental material for Risks and benefits of renal artery stenting in fibromuscular dysplasia: Lessons from the ARCADIA-POL study
Supplemental material, sj-pdf-3-vmj-10.1177_1358863X231210523 for Risks and benefits of renal artery stenting in fibromuscular dysplasia: Lessons from the ARCADIA-POL study by Jacek Kądziela, Katarzyna Jó;źwik-Plebanek, Marco Pappaccogli, Patricia van der Niepen, Aleksander Prejbisz, Piotr Dobrowolski, Ilona Michałowska, Paulina Talarowska, Ewa Warchoł-Celińska, Łukasz Stryczyński, Jan Krekora, Piotr Andziak, Maałgorzata Szczerbo-Trojanowska, Rafał Maciąg, Ignacy Sterliński, Adam Witkowski, Andrzej Januszewicz, David Adlam, Magdalena Januszewicz and Alexandre Persu in Vascular Medicine
Footnotes
Acknowledgements
The authors thank Professor Pierre-François Plouin (Université de Paris, Hôpital Européen Georges-Pompidou, Paris, France) for his expertise, suggestions, and help in establishing the Polish FMD registry ARCADIA-POL. The authors also thank Elżbieta Florczak and Marek Kabat (National Institute of Cardiology, Warsaw, Poland), Krzysztof Madej, Sławomir Nazarewski, and Olgierd Rowiński (Medical University of Warsaw, Warsaw, Poland), students Krzysztof Pieluszczak and Aleksandra Soplińska (Medical University of Warsaw, Warsaw, Poland), Łukasz Światłowski (Medical University of Lublin, Lublin, Poland), and Andrzej Tykarski (Poznan University of Medical Sciences, Poznań, Poland) for their contributions to the manuscript.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The study was supported by the National Institute of Cardiology, Warsaw, Poland.
ORCID iDs
Supplementary material
The supplementary material is available online with the article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
